N-Propyl Acetate: A Deep Dive Into Its Story, Science, and Significance
Historical Development
Curiosity for how simple molecules shape the world runs deep in chemistry. N-Propyl Acetate’s story goes back to the early years of industrial chemistry, when demand for solvents and esters began to rise in the 19th century. Researchers explored how acetic acid and alcohols combined to create molecules with unique physical and sensory properties. Early chemical engineers and perfumers experimented with these new substances to give flavorings their punch and paints their spread. Factories that once struggled to find safer replacements for turpentine started using esters like n-Propyl Acetate because they were less volatile, less pungent, and carried fewer immediate health risks. This switch not only shifted industrial practices but later prompted a need for better understanding of long-term exposure and chemical interactions.
Product Overview
Ask most people about n-Propyl Acetate and the response is likely a blank stare, but walk through any paint shop, perfume lab, or food plant, and its presence becomes obvious. This clear, sweet-smelling liquid gets used as a solvent in coatings, printing inks, adhesives, and flavor formulations. Its ability to dissolve tough resins and leave without a trace after evaporation often puts it at the top of a manufacturer’s list. Its fruity odor means it also slips into artificial flavors for baked goods and candies. Because it brings both utility and a pleasant scent, decision-makers in many industries view it as both a workhorse and a finishing touch.
Physical & Chemical Properties
In the lab, n-Propyl Acetate appears as a colorless liquid with a flashing, slightly fruity odor. It weighs about 0.89 grams per cubic centimeter at room temperature—noticeably lighter than water. Its boiling point hovers around 101°C. Compared to other solvents, its evaporation rate falls into a moderate range. That matters in real-world applications like paints or inks, where drying time and product consistency can swing costs and quality. Its molecular formula is C5H10O2, and its structure gives it just the right balance between solubility and volatility. It mixes easily with alcohols, ketones, and esters, although water barely dissolves it. Storage calls for tightly sealed containers, away from sparks or heat sources, because it can catch fire under the wrong conditions.
Technical Specifications & Labeling
Accuracy in labeling and technical data gives users the information they need to manage risks and extract value. Suppliers list CAS Number 109-60-4 for n-Propyl Acetate, along with UN 1276 for shipping. Purity levels often reach 99% or higher for industrial applications, with water and acidity content measured in parts per million. Industry standards push for transparency on residue after evaporation, color numbers, and distillation range. Labels warn about flammability, possible health risks, and the need for ventilation. Companies handling large volumes often rely on standardized batch certifications and safety data sheets for traceability and compliance.
Preparation Method
Getting n-Propyl Acetate on the shelf means combining acetic acid and n-propanol, usually in the presence of a catalyst like sulfuric acid. Factories either run this reaction in batches or use continuous methods, depending on demand and purity needs. Once the esterification process wraps, the mixture gets separated by distillation, and the product passes through drying and purification stages. By-products, generally water and some unreacted ingredients, get recycled or disposed of according to local regulations. Improved distillation columns and closed-loop processing have pushed yields and purity higher, cutting costs and limiting waste.
Chemical Reactions & Modifications
Though stable under normal conditions, n-Propyl Acetate offers some interesting chemistry. Exposed to strong bases or acids, it can break back down into n-propanol and acetic acid, a process called hydrolysis. This makes it less suitable in certain alkaline or acidic environments, yet ideal for temporary roles such as solvents. Industrial chemists have tweaked its structure to produce higher or branched esters for specialty applications, though straight-chain n-Propyl Acetate keeps the widest appeal because it’s easier and cheaper to make. Some researchers keep sniffing at ways to lower impurities or boost performance for green chemistry goals, aiming for alternatives to petroleum-based feedstocks.
Synonyms & Product Names
N-Propyl Acetate wears many hats, with names like 1-propyl acetate or propyl ethanoate cropping up on international shipping invoices. Chemists reach for abbreviations such as NPA or use product codes unique to each supplier. In the fragrance industry, it sometimes gets branded under custom blends. The United States, Europe, and Asia sometimes use slightly differentiated nomenclature due to historical quirks in chemical registration, but the basic identification is consistent enough that confusion seldom slows business.
Safety & Operational Standards
Once opened, a drum of n-Propyl Acetate sends out a flammable vapor. Regulations strictly limit how much vapor can linger in a workspace, capping exposure where possible. Long-term exposure can cause headaches, drowsiness, or skin irritation, especially when ventilation drops or protective gear runs short. Workers in printing shops or warehouses learn quickly to respect the rules: eye protection, gloves, and flame arrestors become second nature. Facilities often keep spill kits and emergency showers on hand to respond quickly to leaks. Insurance companies, inspectors, and safety officers rely on audits and continuous training. In my experience, nobody wants a close call with airborne esters or an unexpected flash fire, so safety becomes part of the routine.
Application Area
Step inside any plastics or lacquers plant and n-Propyl Acetate almost always turns up on the ingredient list. Its moderate solvency finds a home in nitrocellulose and acrylic resins—materials needed for automotive coatings and industrial finishes. Print shops use it in quick-drying inks for magazines and packaging. Food and flavor companies blend it in tiny amounts to build fruit or rum notes in candies, baked goods, or beverages. Regulators limit its use in foods, but testing confirms safety at low concentrations. Even labs lean on it as a medium for extractions or as a reference compound. Its versatility explains the steady demand, unlike more exotic esters which only fit into niche formulation recipes.
Research & Development
Current research focuses on more sustainable feedstock options and improved process efficiency. Many labs look for ways to swap out petrochemicals for bio-based acids and alcohols. Catalysts that work under milder conditions or regionally available materials could shrink costs and environmental impacts. Analytical chemists refine detection techniques, making trace analyses faster and more reliable to ensure product purity. Some universities dig deeper into how n-Propyl Acetate interacts with other solvents or resins at the molecular level, hoping to boost compatibility for next-generation materials. Not every research effort delivers a breakthrough, but progress often stacks up quietly until a new benchmark is set.
Toxicity Research
Public health agencies rely on animal studies and controlled exposure data to set workplace limits. Large-scale surveys find that short-term inhalation or skin exposure triggers irritation but rarely creates long-term health problems at controlled levels. Chronic exposure in poorly ventilated spaces raises risks—dizziness, nausea, respiratory distress—so workplace monitoring makes sense. Environmental impact studies test its breakdown in soil and water, finding moderate persistence but low bioaccumulation. The science says acute toxicity is relatively low, but the solvent shouldn’t be brushed off as harmless—occupational safety still takes priority.
Future Prospects
Growth in manufacturing, paints, and coatings means the outlook for n-Propyl Acetate stays bright. Pushes for sustainable chemistry drive effort toward greener production routes and lower-emission applications. New regulations and market trends—especially in Asia and emerging economies—create both challenge and opportunity. Whether shifting to biomass-derived propanol or optimizing processes to squeeze every drop of yield, the industry keeps evolving. Investment in safety and automation aims to limit losses and downtime. Researchers keep one eye on shifting consumer tastes, which may, in turn, drive new uses for this old but indispensable solvent.
What is N-Propyl Acetate used for?
The Workhorse in Paints and Coatings
N-Propyl acetate plays a big part in the world of paints and coatings. Step into most paint factories and you'll find stacks of drums filled with this clear, fruity-smelling solvent. It speeds up drying times for spray paints and lacquers. A painter working in humid weather always looks for something that helps paint form a smooth finish, free of bubbles or streaks. N-Propyl acetate delivers on that front. It mixes well with pigments and resins, producing gloss and helping the paint stick to surfaces like wood, metal, or plastic.
Role in Printing Inks
Printing presses run fast, and inks must dry quickly to avoid smudging. This runs especially true for packaging plants, where customers want labels with crisp graphics. N-Propyl acetate has been a go-to ingredient in flexographic and gravure inks. It helps printers avoid the headache of sticky prints and ghosting on consecutive sheets—a common complaint. The quick evaporation rate means machinery can work non-stop and customers get neat, high-quality prints.
Use in Adhesives and Sealants
Manufacturers of adhesives turn to N-Propyl acetate for its good “bite.” Walk into a carpentry workshop or a flooring company; find industrial glue with this solvent. It thins glue just the right amount, allowing even spread and strong hold. Construction workers rely on that formula on floors, panels, and siding—work must stand up to both weight and weather.
Cleaning and Degreasing Applications
I’ve spent time in auto shops where engines come in covered in grease. In cleaning jobs that need a bit of punch but don’t call for aggressive chemicals, N-Propyl acetate fits the bill. Mechanics count on it to clean engine parts before they put them back together. It removes tough oil and grease, but evaporates fast enough so the next step isn’t delayed. This quality matters not just for speed, but also safety—no one wants residue that might cause slips or other hazards.
Perfume and Fragrance Production
Anyone who’s dabbled in blending essential oils or worked in the fragrance business knows about the role of solvents. N-Propyl acetate appears in certain perfumes and personal care products because it blends fragrance oils evenly. It doesn’t add a strong scent of its own, which sometimes happens with solvents. Manufacturers prefer it when they’re aiming for delicate floral or citrus notes without chemical overtones.
Safety and Environmental Concerns
There’s always a tradeoff with chemicals. Workers and manufacturers value N-Propyl acetate for its effectiveness. But health and safety guidelines can’t get ignored. Repeated exposure in closed spaces leads to headaches or irritation. Good ventilation matters—nobody should leave windows shut when using large amounts. Regulations in the EU, US, and other places rank it as lower risk than many strong solvents, but users still require gloves and proper handling. In my own work, even just the pleasant smell can be misleading. Just because something doesn’t smell harsh doesn't mean you can get careless.
Paths Toward Safer Alternatives
The green chemistry movement is gaining steam in every sector. Chemical engineers look for alternatives with fewer health concerns and less environmental impact. Water-based products keep improving, but for now, N-Propyl acetate strikes a practical balance in industrial applications. As technology evolves, manufacturers and chemists must stay aware and ready to switch when safer solutions come to market.
What are the safety precautions when handling N-Propyl Acetate?
Understanding the Basics
N-Propyl Acetate pops up in tons of places: paints, coatings, even some flavorings. Most people run into it at work far more than at home. It smells fruity and sweet, which tricks folks into underestimating it. In the shop or lab, pretty much any chemical with that potent aroma deserves real respect. The first time I opened a drum of the stuff in a paint shop, it hit my nose fast. I learned quick that some chemicals want you to drop your guard. That’s the wrong move here.
Keeping Your Space Safe
Fumes sneak up on you if you get careless. Good air flow matters. Cracking a window won’t cut it in a closed room. Fans, exhaust hoods, and open bays make the difference. Folks tend to wear just a dust mask if they’re spraying finishes. That won’t protect your lungs from these vapors. A proper respirator with organic vapor cartridges blocks those chemical fumes. If you’re inside the shop all day, skipping the right mask turns into headaches, nausea, and feeling off for hours.
Propyl acetate catches fire much faster than water evaporates. One strike of a spark in a half-shut room, and suddenly flames break out. Old timers toss cigarette butts in a can, but one bad moment with this solvent brings trouble. Keep all containers sealed tight. Only open what you need, then close it back up right away. Even static electricity needs attention. Grounding metal cans honestly stops surprise sparks.
Protecting Hands and Eyes
It feels strange to slap on thick gloves for a liquid that doesn’t sting like acid, but that’s honest risk management. Latex gloves together with safety glasses block those dime-size sprays from getting into cuts or your eyes. Splashing thinner on bare hands all shift leaves them dry and itchy—after a whole summer, skin starts cracking, and the hospital bills pile up.
If you clean brushes or wipe down gear, use gloves that hold up—nitrile lasts longer than vinyl for this kind of job. With any spill near your face, goggles help more than regular glasses. Accidents don’t make appointments. Every splash, every time.
Dealing with Spills and Storage
Spills always find a way in hectic shops—mop up with absorbent sand or pads. Never just let it go down the drain where it ends up in the water or air. Scrap rags soaked in the solvent need their own metal bin. Left in a plastic trash barrel, they heat up, dry out, and can self-ignite—more than one shop burned down from a pile of rags nobody thought twice about.
Store all this solvent at a distance from anything hot or sparky. That means heaters, light bulbs, power tools. Locking up large drums keeps curious hands—especially teens—away.
Building Safer Habits
Every time somebody skips the protective gear, they roll the dice on their own health. Headaches and nausea just signal the start—long-term exposure can affect liver and nerves over the years, too. It’s easy to think, “Just this once, I don’t need all this stuff.” Most of the real trouble I’ve heard about started with shortcuts that only saved a few minutes. Having the right equipment nearby, labeling all bottles, and taking time for cleanup goes a long way to making sure everyone clocks out safe.
The bottom line: respect any chemical enough to read the data sheet and get the right equipment ready up front. Mistakes stick with you, but safe habits keep shops running and families healthy.
What is the chemical formula and structure of N-Propyl Acetate?
Understanding the Compound
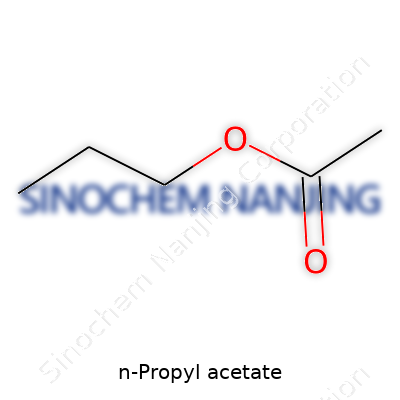
N-Propyl acetate shows up in conversations about solvents more often than folks might expect. The chemical formula for n-propyl acetate comes down to C5H10O2. In plain terms, it brings together five carbon atoms, ten hydrogens, and two oxygens. The compound forms by putting acetic acid and n-propyl alcohol together, which creates an ester bond. This isn’t some abstract chemical talk—this synthesis opens the door for something with real-world usefulness and safety.
Diving into the Structure
The structure of n-propyl acetate isn’t a maze of complexity. The molecule starts off with an acetic acid backbone, where a methyl group (CH3–) connects to a carbonyl group (C=O). Then, an oxygen atom acts as a bridge—it links this backbone to a straight-chain propyl group (–CH2CH2CH3). Chemists write the structure as CH3COOCH2CH2CH3. For anyone who dealt with paint thinners or printing inks, this arrangement isn't just an academic point—it makes n-propyl acetate flexible and effective as a solvent.
Recognizing Its Natural Connection
Trace amounts of n-propyl acetate show up naturally in pears. That sweet, fruity smell in certain spirits or food flavorings? N-propyl acetate often contributes to that. The World Health Organization confirms that n-propyl acetate appears in natural and manmade settings, and exposure through foods usually doesn’t cause harm. This isn’t just chemical trivia—the natural prevalence serves as one reason for its strong regulatory track-record when added to foods or fragrances.
Why Structure Matters in Its Use
The straight-chain nature of the propyl group means n-propyl acetate evaporates at a steady, moderate pace. That’s critical in factories churning out coatings and adhesives. Quick-evaporating solvents throw off timing, but n-propyl acetate lets workers control drying times, leading to fewer defects in finished products. That practical benefit ties directly back to bond arrangements and molecular shape, so understanding its structure leads to safer, more predictable workspaces.
Health and Safety Realities
OSHA sheets warn workers to handle n-propyl acetate with care. Even if natural sources pose little threat, the pure chemical straight from a barrel can irritate eyes, noses, and lungs and—at high enough concentrations—cause dizziness. Good ventilation and protective gear turn out to be simple fixes. Training teams to read labels and recognize exposure symptoms makes workplaces safer, reinforcing the need to bridge chemistry knowledge and boots-on-the-ground action.
Better Use Through Awareness
Companies working with n-propyl acetate benefit from regular audits and refreshers on storage, labeling, and spill cleanup. Responsible stewardship means weighing solvent needs against safer alternatives wherever possible. Research keeps pushing for greener options, reducing emissions and waste. Sustainable choices don’t hinge on a single molecule, but understanding why n-propyl acetate works the way it does is part of smarter decision-making—and a step toward cleaner, safer industry practice.
Is N-Propyl Acetate flammable or hazardous?
Getting to Know N-Propyl Acetate
N-Propyl Acetate shows up often in products like paints, coatings, inks, and nail polish remover. Walk into any auto shop or print room, and you can catch the hint of its fruity scent. What really stands out, though, isn’t the smell—it’s how easily this chemical can catch fire. Thanks to a low flash point, even a spark from static electricity can turn an open container into a hazard.
Fire Risk Isn’t Just a Lab Problem
I spent a few weeks cleaning up warehouses during college breaks. Pallets of metal drums would roll in, and labels didn’t always tell the whole story. We had fire-resistant gloves and extinguishers on hand, but few people knew why. With N-Propyl Acetate in the mix, all it takes is an open flame, a faulty power tool, or even a lightbulb breaking to start an emergency. Data from the European Chemicals Agency ranks N-Propyl Acetate in the highest category for flammability, confirming what experienced workers already know. Stories circulate about shed fires that spread in seconds, all tied back to careless handling of flammable solvents.
Breathe Easy? Not Quite
Jobs that involve regular exposure to solvents can wear people out—not just through spills or burns. The vapors from N-Propyl Acetate irritate the nose, throat, and sometimes make people dizzy or tired. Factories and workshops try to use ventilation fans, but some spaces just trap fumes. Chronic exposure adds up, risking headaches, skin irritation, and in rare cases, damage to the nervous system. The American Conference of Governmental Industrial Hygienists set a threshold limit value to keep workplace air safe, but small businesses sometimes ignore or misread these rules, putting workers at risk.
Taking Hazards Seriously
Accidents in small garages often go unreported, yet they hit hard. A friend who ran a print shop used to ignore gloves and would work all day with solvents until his hands cracked and burned. Time and again, the link between workplace safety and health comes down to information and habits. Using chemical-resistant gloves, goggles, and ensuring cans are tightly sealed goes further than any single safety poster. Keeping chemicals away from open flames or heat sources, even from something as simple as a space heater, means workers make it home without injury.
Moving Toward Safer Solutions
Customers and workers ask for cleaner air, fewer hazards, and better outcomes. Manufacturers look for alternative solvents that don’t catch fire so easily or that break down quicker in the environment. Some water-based products already shift away from N-Propyl Acetate entirely. In places where switching isn’t possible, real training—actual walk-throughs, not just paperwork—helps people spot risks early and handle emergencies. Regulators and professional groups push for better labeling, smarter storage, and fire prevention systems. None of these steps requires huge investment, just a commitment to caring for the people on the floor and the spaces they work in.
How should N-Propyl Acetate be stored and disposed of?
Everyday Understanding for Safer Rooms and Workspaces
N-Propyl Acetate doesn’t show up on most shopping lists, but it’s familiar to anyone who’s worked in a lab, print shop, or paint factory. This solvent carries a sweet, fruity scent, making it easy to mistake for something harmless. Used to dissolve inks, paints, and adhesives, it brings a valuable punch where good solvents matter. But with that usefulness comes responsibility. Handling it wrong can put health and safety on the line. Proper storage and disposal need attention to detail—real people depend on getting both steps right.
Practical Pointers on Storage
Anyone storing N-Propyl Acetate should focus on keeping it cool, dry, and away from anything that sparks or overheats. I remember a rush job at a print shop, reaching for a container that felt warm after hours next to a window in midsummer. Heat builds up risks: higher temperatures can cause pressure inside containers and, in some cases, small leaks, which feed fumes into the workplace air. Good ventilation keeps everyone breathing easy, but safe storage goes way beyond air circulation.
Flammable liquids demand containers designed to resist corrosion and keep contents locked away from the open air. Steel cabinets rated for flammables stand up to rough handling, accidental bumps, or the rare splash. Labels stay visible. Even in the most organized shop, mistakes happen. Labels fade, get covered, or peeled off—but clear, persistent labeling cuts confusion and keeps emergency responders from guessing what’s inside.
Food and drink never belong near N-Propyl Acetate. Pairing solvents with break rooms or food storage brings cross-contamination worries. Locks on storage rooms make a difference, too. Inexperienced hands and curious kids can create hazards quicker than most folks realize. Securing chemicals adds a layer of real-world protection nobody regrets.
Thoughtful, Lawful Disposal
Throwing N-Propyl Acetate down the drain might sound convenient, but it creates risks for water treatment systems, aquatic life, and public health. It’s disappointing to see shortcuts in waste rooms—a couple of unsupervised students or hurried staff thinking a flush will do the trick. Local, state, and federal rules exist because even a small slip-up has consequences far downstream.
Recycling and reclaiming spent solvents often makes the best sense, both environmentally and financially. Working with licensed hazardous waste handlers removes chemicals safely, preventing accidental spills and uncontrolled mixing of incompatible substances. At a university I worked with, dedicated collection drums stood ready. Every drop went through professional channels—students learned that shortcuts just weren’t worth the risk.
Anyone still confused about disposal options can always lean on curated fact sheets from local waste authorities or the Environmental Protection Agency. Most communities recognize the challenge and offer specific pick-up points or directions for hazardous waste. Companies and organizations should dedicate time each year to review protocols and retrain team members, as guidance evolves and new risks emerge.
Why It Matters for Communities and the Planet
Responsible storage and disposal of industrial chemicals like N-Propyl Acetate keep workplaces, homes, and the wider environment in better health. It’s not just about obeying rules on paper. It’s about protecting neighbors, colleagues, and the waterways and air we all share. Thoughtful handling goes beyond compliance; it’s an everyday sign of respect for the people and places around us.
| Names | |
| Preferred IUPAC name | propyl ethanoate |
| Other names |
Propyl acetate 1-Acetoxypropane Acetic acid n-propyl ester Acetic acid propyl ester n-Propyl ethanoate |
| Pronunciation | /ɛn ˈproʊpɪl əˈsiːteɪt/ |
| Identifiers | |
| CAS Number | 109-60-4 |
| Beilstein Reference | 635740 |
| ChEBI | CHEBI:8773 |
| ChEMBL | CHEMBL1492348 |
| ChemSpider | 7335 |
| DrugBank | DB02590 |
| ECHA InfoCard | 03c0630b-5a40-46e6-a3c3-ecd38c6c5a1a |
| EC Number | 123-86-4 |
| Gmelin Reference | 877 |
| KEGG | C02448 |
| MeSH | D017348 |
| PubChem CID | 8005 |
| RTECS number | AJ3675000 |
| UNII | YNGRLJGVLJQFHK-UHFFFAOYSA-N |
| UN number | UN1276 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | 102.13 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | fruity |
| Density | 0.888 g/mL at 25 °C |
| Solubility in water | 6.3 g/100 mL (25 °C) |
| log P | 0.97 |
| Vapor pressure | 18 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | pKb ≈ 15.5 |
| Magnetic susceptibility (χ) | -48.3×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 1.0 mPa·s (20 °C) |
| Dipole moment | 1.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 236.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -425.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2211 kJ/mol |
| Pharmacology | |
| ATC code | V03AB36 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H336, H319 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P264, P271, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | The flash point of N-Propyl Acetate is **"15°C (59°F)"**. |
| Autoignition temperature | 421 °C (790 °F; 694 K) |
| Explosive limits | 1.7–8% |
| Lethal dose or concentration | LD50 oral rat 8700 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 8700 mg/kg |
| NIOSH | NA 1231 |
| PEL (Permissible) | 200 ppm (TWA) |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 1700 ppm |
| Related compounds | |
| Related compounds |
Ethyl acetate Isopropyl acetate Propyl formate Propyl propionate Butyl acetate Methyl acetate |

