N-Phenylacetamide: A Chemical With Roots, Roles, and Responsibilities
Historical Development
N-Phenylacetamide, often called acetanilide, brings to mind the early days of modern organic chemistry when people approached science with a healthy mixture of curiosity and uncertainty. This chemical entered the public eye in the 19th century, during a period known for bold experiments and accidental discoveries. Researchers hoped to harness its properties for medicines and dyes, not foreseeing every consequence. Early fans touted it as a fever reducer, but missteps soon revealed dangers lurking behind its seemingly simple formula. Stepping through its timeline, priorities shifted: medicine turned cautious, industries diversified its applications, and governments began paying attention to the risks. The lessons pulled from N-Phenylacetamide's past echo across chemical development—a reminder that popularity never guarantees long-term benefit, and curiosity must walk hand-in-hand with care.
Product Overview
Taking a direct look at N-Phenylacetamide, the name doesn’t draw much attention to its role as a bridge between the aromatic and the practical. In its heyday, this compound carved out a space in pharmaceuticals and the textile world. When chemistry labs introduced it as a mild painkiller and fever reducer, people thought they had struck gold, but the dark clouds of side effects soon changed that view. The chemical's shelf life, ability to dissolve in many solvents, and general stability attracted makers of dyes and rubber. In more recent years, demands shifted toward synthesis feedstock and research material. This change reflects the pull between old uses and new opportunities, against the backdrop of shifting regulations and consumer priorities.
Physical & Chemical Properties
Solid at room temperature, white to off-white crystals, faintly reminiscent of the aroma left by certain childhood art supplies—these quick cues usually announce the presence of N-Phenylacetamide. The melting point sits high enough to pose no issue at room temperature, and a moderate boiling point ensures manageable handling. This compound dissolves fairly well in organic solvents like ethanol and ether, but doesn’t throw itself readily into water. Its chemical stability, especially the strong amide linkage tying aniline and acetic acid together, tempts chemists who need a base to start tinkering. The molecule’s aromatic ring stays relatively unreactive under mild conditions, yet gives way under strong attack, making selective modification possible.
Technical Specifications & Labeling
Trying to buy or research N-Phenylacetamide unearths a list of technical details meant for safe, informed use. Purity often runs above 99% in lab-appropriate batches. Most suppliers lean toward powder or crystalline formats, typically sealed tight to limit exposure to dust or moisture. The label usually shouts warnings louder than claims—possible hazards for skin and eyes, and advice against inhalation or ingestion. Regulations focus on trace impurities, detection of related anilines, and careful storage separate from oxidizers. Responsible handlers won’t skip over labeling, not just for their own health but for anyone downstream. Professional circles push for barcodes and QR codes to trace the product’s history through supply chains, reflecting a culture that remembers the cost of ignoring what’s on the bottle.
Preparation Method
Chemists favor straight routes. Reacting aniline with acetic anhydride, either in the lab or in factories, yields N-Phenylacetamide quickly and in high purity, without creating unnecessary headaches. Some still use glacial acetic acid as a slower, less efficient route, but the anhydride path wins for speed and clean separation. Temperature control makes a difference—excess heat can break the delicate amide bond or scorch the product. The process doesn’t use many exotic reagents, making it approachable for teaching labs and scaled production alike. Waste management deserves as much attention as yield, since runoff and leftover reactants hold risks for the environment.
Chemical Reactions & Modifications
Synthetically, N-Phenylacetamide opens doors but won’t give up all its secrets. Its amide bond stays put under most gentle conditions, but a determined chemist, armed with acids or bases and heat, can break it back into roots—aniline and acetate. Substitutions on the aromatic ring open more possibilities for dyes and intermediates. Sulfonation and nitration, for example, stretch the uses into domains from pigments to chemical sensors. Researchers routinely use N-Phenylacetamide as a platform for teaching basic organic transformations: hydrolysis, oxidation, and other lively classroom experiments. Tinkering with this molecule sharpens skills for bigger challenges in drug discovery and material science.
Synonyms & Product Names
Spending time in chemical catalogs or regulatory paperwork, it doesn’t take long before familiar names start blurring. Acetanilide, N-phenylacetamide, and even antifebrin—each title carries echoes of previous uses, language conventions, and market spin. The older “antifebrin” hints at medicinal times now ended. In trade, synonyms like acetanilid and N-acetylaniline show up, so cross-checking registry numbers and chemical drawings is routine practice to avoid costly confusion.
Safety & Operational Standards
Years of handling have taught experienced chemists that complacency breeds disaster. Careful storage requires cool, dry spots away from acids and oxidizers, with containers that reduce dust. While basic gloves and goggles often suffice for benchwork, more robust protocols kick in for industrial settings. Respiratory protection steps in where dust clouds threaten. Product safety sheets warn of methemoglobinemia—a blood disorder—if the compound slips past gloves and into the bloodstream. Authorities in Europe and North America classify N-Phenylacetamide as harmful, drawing a line between historical naivete and modern prudence. Many educators insist on stringent waste protocols. Disposal skips the sink, heading for high-temperature incineration under close watch, leaving as little trace as possible.
Application Area
N-Phenylacetamide once set the tone in the medical world, promising easier relief from fever and pain before overtaking safer alternatives exited the lab. Aniline dye makers kept the demand steady, spinning out vivid colors and stable products for the textile trade. Rubber and plastic manufacturers still tap its stabilizing power. Researchers, always on the hunt for reliable model compounds, rely on its predictability during reactions or as a marker substance in analytical checks. The chemical even crops up in studies of environmental fate—its presence helps trace contamination from workshops, leaching sites, or old landfill records. Each use stands as a chapter in the longer story of shifting priorities and technical progress.
Research & Development
Scientific progress rarely rests. This molecule keeps popping up in textbooks and grant proposals, teaching students the ropes in organic synthesis. Medicinal chemists probe its skeleton for safer derivatives, hunting methods to sidestep the blood complications noted a century ago. Environmental chemists follow its decay in soil and water, evaluating risks and cleanup options. Patent records reveal a steady trickle of process improvements for cleaner, less wasteful synthesis. Academic journals track tweaks to preparation—microwave methods, catalysis, greener solvents—underscoring a discipline that measures its worth not by the novelty but by the care for safety and sustainability.
Toxicity Research
The lessons from its medicinal phase left deep tracks. Toxicologists learned the hard way about blood changes stemming from regular contact, as acetanilide’s breakdown product—aniline—interferes with hemoglobin. Doses effective against fever proved dangerous for repeated use, spurring the birth of newer, less risky painkillers like paracetamol. Animal studies and cell cultures continue to map the risk profile, alert for chronic impacts and subtle metabolic changes. The compound’s persistence in the environment sets off alarms among developmental toxicology experts, who track accumulations near chemical factories or discarded stockpiles. Regulations for workplace air and water discharges draw sharp lines on acceptable exposure, reflecting a body of lessons paid for in health and lawsuits.
Future Prospects
Looking ahead, N-Phenylacetamide won’t reclaim the limelight as a miracle medicine, but it will stick around in labs, classrooms, and chemical plants. Green chemistry trends push for safer synthetic routes, better waste capture, and alternatives that share the old benefits without the baggage. Continued research focuses on biodegradability and the search for structurally similar, less hazardous alternatives. Some predict a future where modern analytical tools can sweep up any trace released to the environment, cutting risks for workers and communities. In the right hands, history’s mistakes become teaching moments, and the molecule’s journey serves as a living archive for anyone dedicated to learning, safety, and better science.
What is N-Phenylacetamide used for?
A Closer Look at What N-Phenylacetamide Does
Back pain, headaches, fevers—half the time, folks reach for a small white pill and trust it to do its job. That pill may contain a compound called N-Phenylacetamide, better known in pharmacies as paracetamol or acetaminophen. For generations, people haven’t really asked questions about what’s inside their pain relievers or fever reducers. Most just care that the pain fades and the fever drops.
Why People Trust It
N-Phenylacetamide makes a difference in daily life because it takes care of mild to moderate pain and brings down a fever. Hospitals and clinics across the world rely on it because it works without the stomach issues that come with some painkillers. Aspirin and ibuprofen can bother the lining of your stomach, but N-Phenylacetamide doesn’t pick that fight. It becomes the go-to for parents treating sick kids, patients with sensitive stomachs, and those who need something gentle and reliable for a cold or a toothache.
Digging Into Its Role Outside Pain Relief
Beyond easing pain and fever, there is more to N-Phenylacetamide than most folks imagine. In manufacturing, companies put it to use as an intermediate for producing other medicines and dyes. Schools and research labs turn to it in chemistry lessons, since its structure and reactions teach students about organic chemistry and synthesis. As a writer who has spent hours trying to chase down facts about raw materials in pharmaceuticals, I’ve seen first-hand that this compound shapes more than just pills. Its ripple affects other fields by acting as a building block for products the public often doesn’t recognize by name.
The Flip Side: Safe Use Matters
No compound comes without risks. Paracetamol works at low doses, but the line between helping and harming the body isn’t as wide as most expect. I’ve listened to clinicians explain the dangers of taking too much. The liver processes N-Phenylacetamide, but if overloaded, it can lead to liver failure. The problem doesn’t go away just because the medicine is sold over-the-counter. It’s tempting to believe that the medicine cabinet can’t hide danger, but doctors see cases each year where things go wrong. Alcohol, underlying liver disease, or just ignoring dosage instructions can tip the scales from safe use to serious harm.
What Should Change?
Labeling and patient education fall behind science sometimes. Lots of packages list complex instructions, but people skim the fine print, or forget that multiple medicines can contain the same ingredient. Community pharmacists have told me that one of their biggest worries is “double-dosing”—a patient taking two cold remedies or combining painkillers without realizing both contain N-Phenylacetamide. Focusing on clearer labels and public campaigns can save lives. Putting dose charts in big, bold print, and making sure healthcare workers take a minute to point out risks with each prescription, goes a long way. The tools already exist—the missing piece is real conversations at the pharmacy counter and in the doctor’s office.
Low-Cost Relief, High Responsibility
This compound fills a gap that more expensive or harsher drugs can’t touch, but it works best when folks treat it with the respect it’s earned. There’s wisdom in knowing what something as familiar as paracetamol can do and what it can harm. Keeping public awareness up and encouraging careful use helps this popular medicine remain a dependable part of basic healthcare for years to come.
Is N-Phenylacetamide safe for human use?
Looking Closer at N-Phenylacetamide
N-Phenylacetamide, widely known as acetanilide, first showed up in cough medicines and fever reducers in the late 1800s. People back then didn’t know much about side effects, but the belief held that once a doctor gave out a prescription, the medicine worked safely. Over time, researchers learned more about what this chemical does inside the body. To this day, N-Phenylacetamide’s story raises questions: Should anyone trust it around humans?
Evidence Speaks Louder Than Hype
Doctors stopped using it for pain and fever relief for a good reason. Back in the day, N-Phenylacetamide often triggered dangerous conditions like methemoglobinemia—a blood disorder where oxygen delivery dips dangerously low. Kids were especially at risk. Even the best intentions went sideways, as doctors watched patients report blue lips and shortness of breath due to low oxygen. The data from those years can’t be brushed aside.
Modern toxicology research backs up what older physicians found. After breaking down in the liver, N-Phenylacetamide produces substances that hurt the body’s red blood cells. Manufacturers switched to safer acetaminophen or paracetamol, since both handle pain and fever without carrying the same risks.
Not Just a Drug Ingredient
Today, N-Phenylacetamide shows up in other places—laboratories, rubber, dyes, and plastics. Manufacturers don’t push it as a medicine anymore. Workers who spend time around large amounts of it still face health hazards. The Centers for Disease Control and Prevention label it as a potential occupational toxin. Short-term exposure can cause headache, weakness, and cyanosis. Some animal studies point to issues with kidney and liver function over extended periods.
These risks call for good handling during manufacturing. Strict safety training and engineering controls matter, especially where workers or the public face possible exposure.
Transparency and Trust
One lesson I’ve learned researching dangerous chemicals is that trust dries up fast once manufacturers or health experts hide bad news. People want to know what goes into their bodies or their environment. That’s one reason regulatory agencies, such as the U.S. Food and Drug Administration and the Occupational Safety and Health Administration, post clear warnings about N-Phenylacetamide. These warnings go beyond medical use. They touch on everything from accidental contact to food packaging concerns.
I’ve met chemists who wear gloves and masks every time they handle it. That shows more than caution—it shows respect for scientific evidence. No shortcuts. Their message seems clear: Once a substance hurts people in the past, it deserves scrutiny every step of the way.
Solutions Worth Considering
Substitution has already cut out most of the medical risk. Keeping this chemical far from medicine cabinets meant people now have better alternatives such as acetaminophen. In industrial settings, more ventilation, protective gear, and real-time exposure monitoring help protect workers. Regulators keep tracking new research as fresh evidence turns up.
For people worried about exposure, asking for product safety reports makes sense. Parents and consumers ought to keep pressing for honesty about what’s in products. The lessons from acetanilide’s past tell us this: safety doesn’t just belong in textbooks—it’s personal, tied up with family health and worker well-being. Evidence drives decisions, not reputation or history.
What are the storage conditions for N-Phenylacetamide?
Understanding the Basics
N-Phenylacetamide, also known as acetanilide, crops up often in research labs and manufacturing spaces, pulled in for its usefulness as an intermediate or a pharmaceutical ingredient. Unlike many of the volatile chemicals researchers encounter, this compound actually shows some stability under normal conditions. That sounds reassuring, but storing any chemical, even the more cooperative ones, calls for smart choices to avoid costly mistakes, health hazards, or product loss.
Keeping It Dry and Cool
Every seasoned lab worker knows moisture can ruin a perfectly good sample. If N-Phenylacetamide picks up water from humid air, not only does its appearance change—it might clump, cake, or turn slightly discolored—but it can also lose its effectiveness. Mold and impurities creep in over time, even changing how the substance performs in critical reactions. Best practice means placing it in a tightly sealed container right after opening, away from any sinks or spots known for dampness. In my years around research benches, the jars kept in low cabinets under fume hoods always ended up messier than those on upper shelves, simply because of the occasional splash.
Temperature swings bring another challenge. This compound handles room temperature well, but that means a steady 20-25°C, not sitting in a windowsill for afternoon sun or that spot near rusty radiators. Direct heat shortens shelf life. I’ve seen containers warp ever so slightly, which may not look like much, but even a small opening welcomes moisture and air. For anyone who’s relied on a trusted reagent only to find it ruined, a little discipline—cool, dry, and dark corners—goes a long way.
Labeling and Segregation
Shopping for chemicals brings expiration dates and batch numbers front and center. The same routine should continue in storage. Clear labeling prevents confusion, especially since some powders can appear almost identical. Too often, accidents or surprises happen because a tech grabbed the wrong bottle, thinking it was another white crystalline solid. In multi-user labs, a clear date and initials on the container go a long way for traceability.
Incompatibility sneaks up on those who stack bottles thoughtlessly. As a non-volatile, relatively nonreactive compound, N-Phenylacetamide gets along with most neighbors, but it shouldn’t share space with concentrated acids or strong oxidizers. Segregated shelving, with flammable solvents in one section and solid organic compounds like this in another, keeps everything safe. A single spill from a broken acid bottle can destroy more than one bottle if stored carelessly.
Personal Experience and Safety Habits
I remember one project that got held up because our main bottle caked so badly, we couldn’t weigh it without chiseling. The lesson: the best storage is not just about following safety codes, but thinking ahead. Proper storage avoids product loss and wasted time. Resealing containers, minimizing air exposure, and inspecting bottles periodically—these habits don’t take long, but prevent plenty of headaches.
Beyond that, safety data sheets deserve more than a glance. Chemical burns and unexpected exposures come from misjudging a product’s hazards. Even with N-Phenylacetamide’s fairly mild profile, gloves and goggles always make sense. Not everyone in a shared lab knows every chemical’s risks, so minimizing direct contact protects both the experienced worker and the new hire. Emphasizing these habits in every department helps build trust and consistency.
Better Storage for Reliable Results
Solid storage practices do more than protect inventory—they protect people and research. With N-Phenylacetamide, a little attention to dryness, temperature, labeling, and basic safety saves a lot of frustration. Consistency pays off, safeguarding both the budget and everyone’s well-being.
What is the chemical structure of N-Phenylacetamide?
Why Understanding Structure Matters
The smallest tweak in a molecule can flip a whole industry or make the difference between a painkiller and a poison. Take N-Phenylacetamide, known to folks in the pharmacy as acetanilide. Chemists and doctors pay attention to this compound for good reason. It was one of the first modern fever reducers, and its chemistry became the foundation of acetaminophen (paracetamol). If you’re curious why studying the chemical makeup of simple compounds can still matter, look at how a century-old molecule still shapes pain relief today.
The Backbone: Atoms and Bonds
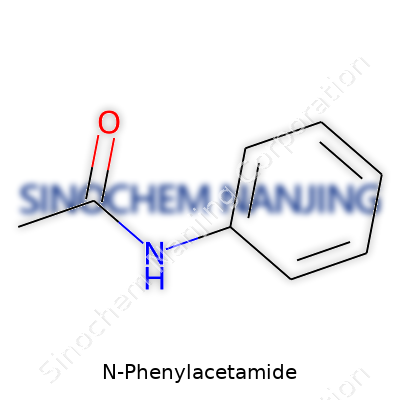
N-Phenylacetamide brings together two parts. First, there’s an acetamide group. That’s a carbon atom double-bonded to an oxygen (a carbonyl group), and that carbon also links to a nitrogen. On the other side, its nitrogen forms a bond with a phenyl ring, which is just a fancy name for a six-carbon benzene ring. The formula comes out to C8H9NO. So, you’ve got this flat, stable benzene ring glued to a two-carbon tail with an amide stuck in between.
Lay it out on paper, and you’d draw the phenyl ring (hexagon) with a line pointing from one carbon out to the nitrogen. The nitrogen holds tight to a carbon that's double-bonded to oxygen, and that same carbon has a single bond to a methyl group (a simple -CH3). It’s a common arrangement found in drugs and dyes. The direct link between the nitrogen of the amide group and the benzene ring puts this compound into a lot of textbooks and patents.
The Impact of Minor Tweaks
Swap a tiny group on this structure, you end up with something else. Switch out the acetamide's methyl group for a hydroxyl group, and you’re looking at paracetamol—one of the safest painkillers in your medicine cabinet. This makes it clear that chemical structure isn’t just an academic exercise. It shapes safety, metabolism, and how the drug works inside the body. Acetanilide fell out of favor when folks realized it could damage red blood cells and sometimes harm the liver. Still, the core skeleton carried through to better alternatives.
History and Lessons
In the late 1800s, chemists made acetanilide by mistake while looking for aniline dyes. They noticed it brought down fevers fast. That accidental discovery kicked off a whole class of medicines and dyes that depend on the amide and benzene ring combination. Over time, the industry learned the importance of both safety and innovation. The tweaks chemists made to this core structure led not just to safer drugs but gave rise to a whole sweep of chemical industries—including those that create the colors in everything from denim jeans to printer ink.
Future and Solutions
Today’s labs use synthetic routes that keep waste and hazards in check. Chemists lean on green chemistry principles, searching for better catalysts and cleaner pathways to build compounds like N-Phenylacetamide without producing dangerous byproducts. Advanced analytical equipment, like NMR and high-res mass spectrometry, lets researchers double-check products and spot minute contaminants that could sneak past old-school quality control. These details matter when you’re talking about drugs or colors ending up in the food chain. As research tools get sharper, it’s possible to build on historic molecules while aiming for health and safety rather than just chasing profit.
The structure of N-Phenylacetamide might look simple, but its journey through history shows that even basic chemistry can change lives—if scientists watch the details, keep digging for better options, and remember the risks and rewards baked into every bond.
Where can I purchase N-Phenylacetamide?
Background on N-Phenylacetamide
N-Phenylacetamide, known to many researchers as acetanilide, pops up in textbooks as a precursor, a stabilizer, and sometimes an intermediate compound. Labs and some industrial sectors use it for antiseptics, dyes, and even as a component in rubber processing. It’s a workhorse, but it also straddles the line between routine and strictly regulated, depending on what country you call home.
Why You Can’t Just Walk Into a Store and Buy It
Chemicals like N-Phenylacetamide come with baggage. Their connection to pharmaceuticals and potential illicit uses brings scrutiny from regulators. In my years working behind the counter in a chemical supply business, we saw requests for all kinds of materials, but anything tied to drug fabrication or high-risk industrial applications drew paperwork, background checks, and clear intent statements.
If you’re trying to buy N-Phenylacetamide, most reputable suppliers will want a solid reason up front. Academic labs, pharmaceutical companies, and established manufacturers stand the best chance by showing business licenses and explaining their project or research. Everyday folks or hobbyists run into a wall—not out of spite, but because the potential dangers call for strict controls.
Where Scientists and Companies Can Buy It—Legally
Industrial suppliers like Sigma-Aldrich, Thermo Fisher Scientific, and TCI America stock N-Phenylacetamide. They cater to licensed buyers: universities, labs, manufacturing outfits with traceable histories, and other organizations with a real use for the chemical. Retailers avoid the liability, leaving business-to-business portals as the primary route.
Online, platforms like Alibaba or ChemSpider list global sellers, but scams, outdated listings, and legality issues muddy the water. Before going down this road, consider local rules. Countries in the EU, the US, and many Asian nations tie chemical access to stricter controls than ever. Failure to comply can mean hefty fines or even criminal charges.
The Danger of Skirting Regulations
A few years back, I saw would-be entrepreneurs skirt the law by buying from unvetted overseas vendors. Shipments got snagged by customs, goods never arrived, or worse—unidentified substances arrived in bland packaging, sometimes contaminated and useless. That’s more than lost money. Exposure to impurities or toxins poses real risks in both home and commercial settings.
Strict regulation isn’t just red tape. Bad actors can use chemicals like N-Phenylacetamide to manufacture harmful products, feeding the illicit drug trade or putting unsafe products into the supply chain. Stores and suppliers keep up compliance, not only to avoid trouble but also to protect end users and broader communities.
Seeking Solutions: Transparency and Guidance
If you’re pursuing N-Phenylacetamide for legitimate research or manufacturing, reaching out directly to established chemical suppliers is step one. Get documentation in order—a purchase intent letter, proof of lab affiliation or business activity, and proper storage plans. Suppliers help guide you through the regulatory process, and in my experience, talking directly to the compliance department helps smooth things over.
Clear communication and honesty work best. As technology and algorithms improve, authorities can spot sketchy orders faster, and trusted buyers find life a little easier. For students and new researchers, connecting with faculty or compliance officers makes the process clearer. For companies, investing in transparent policies and strong supplier relationships keeps operations running, and everyone stays on the right side of the law.
| Names | |
| Preferred IUPAC name | N-phenylacetamide |
| Other names |
Acetanilide N-Phenylacetamide Acetanilid Phenylacetamide N-Acetylaniline |
| Pronunciation | /ɛn-fɪˌnɪl-əˈsiː.tə.maɪd/ |
| Identifiers | |
| CAS Number | 103-84-4 |
| Beilstein Reference | 605984 |
| ChEBI | CHEBI:17219 |
| ChEMBL | CHEMBL1442 |
| ChemSpider | 5791 |
| DrugBank | DB00316 |
| ECHA InfoCard | 03e8e8faa4a3-48e3-bd3b-9171e1b39462 |
| EC Number | 200-431-6 |
| Gmelin Reference | 6076 |
| KEGG | C06527 |
| MeSH | D010551 |
| PubChem CID | 7518 |
| RTECS number | AK4375000 |
| UNII | 49UMM0F7PY |
| UN number | 2810 |
| CompTox Dashboard (EPA) | DTXSID0031575 |
| Properties | |
| Chemical formula | C8H9NO |
| Molar mass | 135.16 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 1.17 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 0.90 |
| Vapor pressure | 1 mmHg (at 218°C) |
| Acidity (pKa) | 15.0 |
| Basicity (pKb) | pKb ≈ 9.4 |
| Magnetic susceptibility (χ) | -69.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.571 |
| Viscosity | 33.5 mPa·s (50 °C) |
| Dipole moment | 3.85 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 202.0 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -103.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3227 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N02BE01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | P264, P270, P301+P312, P330, P501 |
| Flash point | 146°C |
| Autoignition temperature | 300°C |
| Lethal dose or concentration | LD50 oral rat 1790 mg/kg |
| LD50 (median dose) | LD50 (median dose): 500 mg/kg (oral, rat) |
| NIOSH | RN3675001 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 10 mg/kg |
| Related compounds | |
| Related compounds |
Acetanilide Phenacetin Paracetamol Aniline Acetanisole |

