N-Phenyl-2-Naphthylamine: A Closer Look Beyond the Data
Tracing the Path: Historical Development of N-Phenyl-2-Naphthylamine
N-Phenyl-2-Naphthylamine has roots running over a century deep into chemical innovation. Chemists exploring alternatives to natural rubber preservatives latched onto this compound as synthetic polymers flooded markets. In an industry chasing longer tire life and better stability, this substance—often shortened to P2NA or PANNA—found a home behind the scenes in manufacturing plants. Throughout its history, the compound wasn’t just a formula penciled into textbooks; it gathered stories from workers, manufacturers, and regulators grappling with the balance of industrial progress and safety. As tires and technical rubber goods became common, so did research to look at not just how P2NA helped with aging resistance but also what costs trailed its use.
Lifting the Lid: What Sets N-Phenyl-2-Naphthylamine Apart
People who spend time working with or studying industrial chemicals know each has its own quirks. P2NA stands out for its antioxidant properties. That means in rubber and plastics, it acts like an invisible shield, stretching out the life of these materials against the effects of oxygen and heat. The story doesn’t end there, though. This molecule doesn’t just hide in the shadows—its chemical structure brings challenges in storage and handling, reminders that not every useful thing comes without a cost. If you’ve worked in a lab or on a shop floor, you’ve heard stories where a bit of carelessness with such substances led to more than just a failed batch.
Physical & Chemical Properties: More Than Just Numbers
Physical properties often get brushed off as textbook trivia until they show up as odd behavior during handling. P2NA presents itself as a pale or yellowish powder. Once you’ve seen enough of these industrial powders, you start picking up on how their texture and color provide clues about purity and stability. P2NA does not dissolve easily in water, but mixes with organic solvents such as benzene and acetone—both old friends and new hazards in chemical workplaces. Its melting point and moderate volatility mean it has practical storage considerations, especially in hotter environments where risks of leakage or dusting get real fast. Its chemical makeup—naphthylamine bound to a phenyl group—sets the stage for both useful reactions and unwanted byproducts.
Technical Specs and Getting the Label Right
Reading a chemical’s specification label goes beyond legal checkboxes; it’s about safety and clarity for those who work with it daily. Labels on P2NA sources tend to feature purity thresholds, expected by-products, and warnings that echo past incidents involving skin contact or inhalation hazards. If you’ve ever been part of an audit or safety drill, you know regulatory compliance circles around clear specification and labeling. It’s not about ticking boxes; it’s about ensuring that every shift worker, technician, and driver understands what they’re handling. The way these specs and warning symbols are drafted reflects years of accumulated knowledge—sometimes acquired after things have gone wrong somewhere else.
How It’s Made: The Human Element in P2NA Preparation
Ask an experienced chemist and you’ll hear about the practical side of P2NA synthesis. Common routes involve condensation of aniline with 2-naphthylamine, often in the presence of acids or catalysts. The process doesn’t just call for technical know-how but also vigilance regarding waste and contaminant management. Having spent time around synthesis labs, I’ve seen how the reality of daily production pushes people to tweak conditions, chase higher yields, and occasionally troubleshoot equipment that’s seen better days. Efforts to reduce unwanted emissions or hazardous byproducts have changed how these processes look compared to a generation ago, driven by both regulation and a growing safety culture.
Reactivity and Modifications: More Than Just Lab Curiosity
On paper, P2NA doesn’t rush into reactions, which is part of why it works as an antioxidant. Yet, with strong oxidizers or reactive chemicals, transformation can still happen, leading to substances that could be either more useful or significantly more hazardous. In my experience, controlled modifications aim to tailor the molecule for specific industry needs—though such efforts often must weigh added benefit against risk and new unknowns. Researchers sometimes try to tweak P2NA’s structure in pursuit of less toxic, more stable alternatives, but the balance is tough. Forging ahead with modifications brings the responsibility of re-evaluating every downstream effect.
What’s In a Name? Synonyms and Product Aliases
Walk down an aisle in any chemical warehouse and confusing labels aren’t just a nuisance—they can be dangerous. P2NA has collected a shelf-load of synonyms: N-phenyl-2-naphthylamine, phenyl-beta-naphthylamine, and lesser-known brand names from eras when marketing battled with technical jargon. Mixing up similar-sounding compounds isn’t rare; staffers sometimes recount close calls when shipments arrived mislabeled or documents used outdated trade names. Ensuring everyone means the same thing, from procurement to waste handling, matters more than anyone new to chemical logistics might guess. The right name saves time, prevents mistakes, and underlines a commitment to safety.
Working Safe: Standards and the Realities of Handling
Talk to older workers and you’ll hear stories of less strict days, where gloves and respirators were optional and training lasted minutes. These days, operational standards are stricter—often shaped by both tragic lessons and regulatory push. P2NA isn’t to be handled lightly. It demands protective equipment, well-managed ventilation, and robust storage away from direct heat or incompatible chemicals. Regular health monitoring has become the norm for those handling substances flagged by occupational safety groups. Detailed standards back up this vigilance, but, as those of us who’ve worked in hazardous environments know, standards are only as good as the culture on the floor. Ensuring processes work isn’t just about paperwork; it’s about lived daily practice and respect for the risk.
Where It Gets Used: Real-World Application
Most synthetic rubber parts—especially tires and conveyor belts—owe some of their resilience to antioxidants like P2NA. Engineers and managers focus on extending product life, fighting loss from oxidation or heat. Rubber goods, electrical insulation, and even some greases pull in this compound for the edge it brings in resisting aging. People working in production or maintenance often take for granted what keeps their machines running or tires rolling longer; behind that reliability sits the unseen work of chemical innovation. Sometimes, innovators try to swap out P2NA for less controversial alternatives, but in many legacy applications, it persists because of established performance records.
Research & Development: The Ongoing Conversation
Years ago, research on P2NA turned up questions about its longevity in environmental and occupational settings. Studies compared it to newer antioxidants, leading some companies to seek alternatives with better safety profiles. I’ve watched R&D teams wrestle with finding replacements that match P2NA’s performance while losing some of its hazards, often coming up short on cost or effectiveness. Current work focuses not only on performance tweaks but also on reducing formation of harmful byproducts during manufacture or use. Cross-disciplinary teams—combining toxicologists, engineers, and process chemists—continue to push for solutions, spurred by both ethics and tightening regulations.
Learning From the Evidence: Toxicity and Human Risk
The dark side of industrial chemistry rarely gets enough attention until headlines appear. Toxicity research on P2NA has cast a long shadow, fueled by studies linking it to possible carcinogenic effects in lab animals, and increased health monitoring for workers. Regulatory agencies have responded with stricter exposure limits and calls for substitution where feasible. In my own professional circles, the reminder comes often: substances with a track record of health risks deserve extra care and honest debate about their role moving forward. Protecting worker health, especially in industries slow to turn over legacy formulas, means pushing for transparent data, updating safety sheets, and encouraging rapid reporting of adverse effects.
Looking Ahead: Where Does N-Phenyl-2-Naphthylamine Go From Here?
The future of P2NA rides on the line pulled between technical necessity and health priorities. The search for safer, equally effective antioxidants in rubber and plastics accelerates as concerns about long-term toxicity grow louder. Regulations in many regions press plants to rethink continued reliance on established chemicals, pushing innovation across scientific and engineering disciplines. Some alternatives, both new compounds and improved processes, promise less risk—but require industry to invest in adaptation. Those invested in chemical safety urge ongoing transparency in research and continued funding for studies of both replacements and legacy risks. The industry’s story with P2NA isn’t over, but the next chapters will reflect choices shaped by hard lessons, technical ingenuity, and a growing insistence on putting worker and environmental health ahead of short-term convenience.
What is N-Phenyl-2-Naphthylamine used for?
What N-Phenyl-2-Naphthylamine Does in Everyday Industry
N-Phenyl-2-naphthylamine might not turn heads in the local hardware store, but its presence runs deep in many of the things people rely on. I’ve spent time in an old tire warehouse where stacks of aging rubber tires didn’t crack, split, or dry out. Looking closer, I learned about the unglamorous crew of chemicals, including N-Phenyl-2-naphthylamine, lending a real hand behind the scenes.
In the rubber industry, this chemical acts as an antioxidant. Tires, belts, and seals all work under tough conditions—exposed to heat and sunlight day in, day out. Without antioxidant chemicals, the rubber in these products would harden, turn brittle, and break far too soon. N-Phenyl-2-naphthylamine slows down the process, stopping those cracks and splits that send a tire to an early grave. It soaks up the nasty free radicals outdoor life brings, protecting the rubber from wearing out before its time.
Pushing Beyond Tires: More Uses in Industrial Life
Factories don’t just shape rubber for tires. Conveyor belts, gaskets, and hoses run nonstop in a factory setting, and many of these parts depend on antioxidants for their life. N-Phenyl-2-naphthylamine gets mixed into the rubber during production, and it stays within the product throughout its life, holding off chemical decay caused by heat and oxygen. This support cuts down on breakdowns, shrinkage, and the failure that can shut down entire production lines.
Synthetic lubricants also benefit from this chemical. In engines and machines that run hot, oils and greases break down quickly. With N-Phenyl-2-naphthylamine around, oils last longer, resist sludge buildup, and keep motors turning smoothly. This not only saves on oil changes but also protects moving parts from wearing down before their time.
Why Safety Keeps Coming Up
I’ve seen industry workers pull on gloves and respirators, knowing that not all helpful substances are safe to handle. The same is true with N-Phenyl-2-naphthylamine. While it adds value to products, its risks to human health remain a real concern. Studies link long-term exposure to this chemical with health issues, including potential cancer risk. Regulatory agencies—from OSHA in the United States to REACH in Europe—require strong safeguards, limiting worker exposure and tracking chemical handling from plant gate to final product. This attention matters, because lives don’t refill as easily as machine oil.
Thinking About Better Solutions
The chemical industry keeps exploring safer and greener substitutes. Rubber manufacturers test alternative antioxidants, aiming to cut back on risk without sacrificing the strength or resilience the old chemicals provide. Researchers dig deep into the science of degradation, hunting for new formulas that get rid of the aging problem with fewer health hazards. Some tire companies now use blends of natural antioxidants or seek chemicals that break down into harmless byproducts.
For me, seeing how a small tweak in chemistry can lengthen the life of a tire—or the downtime of a machine—shows how important it is to balance durability with safety. Nobody wants to trade a longer-lasting product for a greater risk to the people making or handling it. That balancing act still challenges scientists and workers alike, pushing them to ask: How can we keep the rubber on the road without trouble down the line?
What are the safety precautions when handling N-Phenyl-2-Naphthylamine?
Why Handle It Carefully?
N-Phenyl-2-Naphthylamine gets used in rubber manufacturing. It stops tires from breaking down too fast. Still, working with this chemical often comes with risk. Many people don’t realize health problems may come just from skin contact or breathing dust over years. Cancer researchers have seen a pattern: workers who spent decades around this chemical sometimes develop bladder cancer. That fact pushes safety far beyond just company checklists. Protecting yourself and your coworkers makes sense because no tire or belt is worth ruining your health.
Physical Risks On the Job
If you’ve handled N-Phenyl-2-Naphthylamine, you know it doesn’t smell much. That makes spills or dust clouds easy to underestimate. Fine powder can land on skin or clothes. Wiping your brow or grabbing lunch before washing up may seem harmless, but those small acts let toxins enter the body. In warm plants, sweat mixes with powder, so hands and face become risky delivery routes for chemicals. Eyes can feel itchy if powder blows around, and skin may get red or dry after washing with too little soap.
Safe Steps That Actually Work
The real defense always comes from personal habits and a solid plant culture. Goggles, gloves, and lab coats made of materials that don’t soak up chemical dust keep skin covered. Avoiding shorts and t-shirts on a hot day sounds tough, but covering up matters most. Respirators and local exhaust hoods pull dust away from your face when pouring or mixing. Scrubbing up before eating or smoking goes beyond following rules: it reduces what your body absorbs. After my first month in a chemical plant, my hands started cracking from all the washing; switching to a gentle pH-balanced soap made a difference. It reminded me that “protection” isn’t only about gear—it’s about taking care of skin and eyes, too.
Monitoring Air and Surfaces
Any space with fine powders should have regular air sampling. Too much dust floating around means fans or exhausts aren’t strong enough. If monitors catch levels above safe limits, supervisors can’t just ignore it. I once saw a coworker keep working through the warning “just to get the job done.” He felt fine for a week, but got sick later. That day stuck with me: air monitors matter only if people respect their warnings. Wiping down benches, door handles, and especially common spaces with the right cleaners prevents dust from moving around the plant.
Long-Term Health Watch
Some companies offer bladder screenings to longtime workers who handled this chemical. Early detection improves survival and is not just a box to tick. If you’ve ever handled N-Phenyl-2-Naphthylamine, regular checkups should include honest talk with your doctor. Mention your chemical exposure—many don’t connect plant work with symptoms until years later. If a friend or coworker avoids the doctor or skips tests, reach out and talk about it. Personal stories sometimes convince more than any rulebook.
Shared Responsibility and Future Actions
It helps to build a workplace culture where no one looks down on double-checking a glove, re-washing hands, or reporting dust leaks. Quick fixes like more signage and briefings go further when experienced workers lead by example. Technology changes, so companies should keep reviewing which respirators work best and what glove material actually holds up in real use. Pressure to cut corners hurts everyone. If a plant team stands together—everyone looking out for each other—safety improves beyond any single rule or tool.
Is N-Phenyl-2-Naphthylamine hazardous to health or the environment?
Understanding N-Phenyl-2-Naphthylamine
N-Phenyl-2-naphthylamine, often called P2NA, shows up in the chemical industry as an antioxidant. Rubber manufacturers use it to make tires last longer. It can slow down deterioration and keep products on the road or in use for years. Growing up in an industrial region, I saw warehouses full of drums marked with chemical names, and I’ve often wondered—what happens when chemicals spill outside the factory fence?
Risks to Health
P2NA does not grab headlines like asbestos or lead, but science points to real health concerns. Evidence suggests it may be toxic if inhaled or if it touches skin over long periods. The European Chemicals Agency and other regulators classify it as “suspected of causing cancer.” Inhalation can irritate airways and eyes. Skin contact sometimes leads to redness or allergic reactions. In most labs and workshops, gloves and masks are not suggestions—they are necessities. My experience in a tire recycling plant proves that workers handle dust, fumes, and residues daily, placing their health at risk if safety takes a back seat.
Recent studies reviewed by the International Agency for Research on Cancer link P2NA to tumors in animal studies. These risks led several countries to restrict or phase out its use. Product labels rarely warn end-users, but people working where P2NA is made or processed face higher exposure. Chronic inhalation raises the stakes, especially in older facilities lacking proper ventilation and modern equipment.
Environmental Worries
N-Phenyl-2-naphthylamine also poses questions for the environment. Chemicals used in factories do not always stay put. Over time, P2NA can escape through wastewater or air emissions. Soil and water near dumping sites sometimes show traces of this compound years after it left the factory. As a former volunteer at river cleanup events, I’ve seen firsthand how persistent chemicals linger in muddy banks long after industrial use winds down.
Wildlife runs risks too. Accidental releases or improper disposal can threaten aquatic life, since P2NA resists breakdown and can build up in the food chain. According to reports from the US Environmental Protection Agency, fish exposed to high levels show problems with growth and reproduction. Long before anyone starts fishing or swimming, contamination quietly does its work.
Facing Up to the Problem
Manufacturing communities do not want to trade livelihoods for poisoned air and rivers. Companies can switch to safer alternative additives. Regulators have already forced change in Europe and parts of North America, setting up strict controls, regular monitoring, and disposal guidelines. In other countries, black-market sales and limited oversight drag progress down. Industry leaders who want to stay ahead have begun collaborating with independent researchers to test newer, less hazardous compounds that fill the same role in rubber and plastics.
People living near chemical facilities deserve transparency. Sharing monitoring results openly can build trust and awareness. Training workers to handle chemicals safely remains essential. My time spent conducting safety drills in factories showed me how a single careless spill can result from lack of knowledge, not bad intent. Community engagement, smart substitution, tougher standards, and extra vigilance offer a path toward cleaner factories and safer homes.
What is the chemical structure and formula of N-Phenyl-2-Naphthylamine?
What N-Phenyl-2-Naphthylamine Actually Looks Like
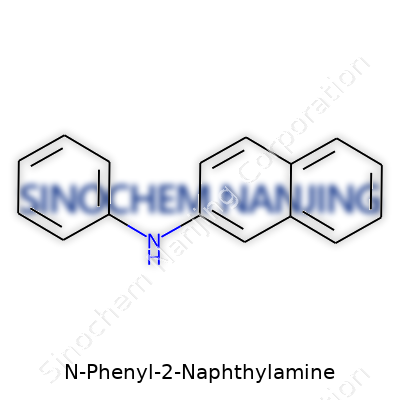
Working in research labs, I’ve learned that the face of a chemical says a lot about its story and purpose. N-Phenyl-2-Naphthylamine, which often pops up in conversations about antioxidants in rubber, carries a somewhat hefty structure. The base is a naphthalene ring—a two-fused benzene ring—that grows from the second position into a spot where a phenyl group (a simple benzene ring) attaches to a nitrogen atom. Put simply, you’ve got a naphthalene backbone at the core, with an aniline arm hugging on as the nitrogen-phenyl bridge.
You won’t get much out of just picturing it; the real value comes from seeing how each atom lines up. The chemical formula is C16H13N. Counting carbons and hydrogens hints at possible use-cases—dense frameworks like this one usually resist heat and chemical breakdown, which is what makes it attractive to manufacturers.
Drawing from Real-World Uses
Old-school tire technicians would recognize this molecule, often shortened to P2NA, because it functions as a tire preservative. Heat and oxygen hammer at rubber compounds over time, but P2NA stands in the way and takes one for the team. In effect, its rings disperse attacking oxygen species before they can gnaw away at the material. This comes straight from the layout of its atoms: the naphthalene and phenyl groups distribute electrons in a way that soaks up reactive threats.
On the downside, scientists found links between compounds like this and certain cancer risks. European regulators flagged P2NA after studies highlighted how its aromatic rings—strong as they are for chemistry—may produce toxic metabolites inside the human body. As a chemist working among safer antioxidant alternatives, I understand why the search for replacements has grown stronger over the years.
Why Structure Shapes Significance
The specifics of the N-Phenyl-2-Naphthylamine molecule—those two aromatic systems and the nitrogen bridge—hand over chemical stability. This stability pushes its antioxidant activity, as these robust rings intercept radicals that break rubber bonds. Manufacturers, especially those producing heavy machinery tires, appreciated this endurance.
With better technology and more public awareness, industries seek less toxic replacements for P2NA. Researchers and regulatory bodies ask: can we get the same benefit without the risks? Modern alternatives use hindered phenols and amine blends, designed to break down cleaner and offer transparency in their by-products. Chemists focus effort on balancing chemical redox activity with lower toxicity, aiming for substances that give protective benefits but clear faster from biological systems.
Solutions Rooted in Responsibility
From my experience, sustainable chemistry comes from matching creativity with caution. Following the science—screening compounds for both performance and biological impacts—keeps our products and environment on safer ground. It prompts innovators to design next-generation antioxidants that mimic the strength of P2NA’s framework, yet skip the harmful legacy. Regulatory clarity and industry leadership continue pushing for molecular blueprints that respect both function and wellbeing.
How should N-Phenyl-2-Naphthylamine be stored and disposed of?
Understanding the Risks and Necessity of Care
Every chemical tells its own story, and N-Phenyl-2-naphthylamine—let’s just call it P2NA—has a reputation that deserves respect. Folks working in rubber manufacturing or industrial labs often cross paths with P2NA. Years ago, I watched a chemist handle a tiny bottle like he was cradling a newborn. That stuck with me. This compound doesn’t just stain skin; real concerns surround its health effects. Being cautious isn’t sterile paranoia; it’s survival wisdom.
Sensible Storage Practices Make a Difference
P2NA demands a home that’s cool, dry, and away from sunlight. I remember an incident where heat warped containers, and fumes drifted into nearby spaces. The panic on the floor was enough to convince anyone that direct sunlight isn’t just a storage faux pas—it’s a recipe for a hazardous spill. Airtight containers made from materials that resist chemical corrosion serve better than cheap plastics, since leaks put people and surrounding areas at risk.
Labeling plays just as vital a role. Workers should never squint and guess whether a bottle holds P2NA or sugar—one mistake can bring an entire shift to the emergency room. Labels with bold print and hazard symbols stop accidents before they start. Keeping incompatible chemicals far apart eliminates opportunities for unexpected reactions. Let’s not stack containers like Tetris pieces; good spacing keeps ventilation up and the chance of cross-contamination down. Locking cabinets behind secure doors keeps staff and the night crew safe.
Personal Responsibility and the Right Gear
Access control sounds strict, but really, it’s common sense. No reason for unauthorized folks to poke around in the chemical storage room. Protective gloves, long-sleeved coats, face shields—these don’t just look official. They serve as a simple line of defense for all skin, eyes, and lungs. The right training ensures that people know what gear to grab and how to check for cracked containers that leak invisible but harmful fumes. Ventilation plays the silent hero, whisking risky vapors away. In my own work, a busted fan meant a day of itching eyes—proof that cutting corners turns small problems into health crises.
Disposal: More than a Technicality
Dumping P2NA down the drain might seem quick. It’s also illegal and dangerous. Municipal waste plants can’t handle the toxin load, and ground water contamination poisons more than just one unlucky worker. One lab I visited collected used or spilled P2NA in designated drum barrels, clearly marked for hazardous waste pick-up. You want a specialty disposal firm—folks trained to process P2NA waste through incineration or other controlled destruction methods. This costs money, sure, but the alternative brings fines and environmental disaster.
Spillage calls for action right away. Not mops—sorbent pads meant for chemicals. Any contaminated material goes straight into hazardous waste bags, sealed and labeled. No shortcuts. Every worker deserves a workplace where safety trumps convenience. If handling instructions seem tedious, that nuisance keeps you upright, healthy, and penalty-free.
Fact-Based Safety Means Fewer Regrets
P2NA is not a “set it and forget it” chemical. Long-term exposure links straight to cancers and severe tissue damage. The law doesn’t just suggest caution—it demands documentation, from storage logs to waste disposal receipts. Building a culture of chemical responsibility ensures that nobody learns the hard way. Treat P2NA with care, stick to the guidelines, and you cut risks down to size. That’s how you make sure everyone gets home safe—and with clean hands.
| Names | |
| Preferred IUPAC name | N-phenylnaphthalen-2-amine |
| Other names |
Antioxidant PAN Fenamine Neozon D PBNA Phenyl-α-naphthylamine |
| Pronunciation | /ɛn-ˈfiːnɪl-tuː-næfˈθɪl-əˌmiːn/ |
| Identifiers | |
| CAS Number | 135-88-6 |
| Beilstein Reference | 135873 |
| ChEBI | CHEBI:34951 |
| ChEMBL | CHEMBL14122 |
| ChemSpider | 20419 |
| DrugBank | DB14008 |
| ECHA InfoCard | 100.012.379 |
| EC Number | 205-355-7 |
| Gmelin Reference | 97179 |
| KEGG | C06518 |
| MeSH | D010376 |
| PubChem CID | 8465 |
| RTECS number | RN3675000 |
| UNII | 8X848O210P |
| UN number | UN1334 |
| CompTox Dashboard (EPA) | DTXSID7020391 |
| Properties | |
| Chemical formula | C16H13N |
| Molar mass | 269.35 g/mol |
| Appearance | White to grayish purple crystals |
| Odor | Odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | Insoluble |
| log P | 3.94 |
| Vapor pressure | 0.000001 mmHg (25°C) |
| Acidity (pKa) | 5.0 |
| Basicity (pKb) | 6.10 |
| Magnetic susceptibility (χ) | -98.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.692 |
| Viscosity | 450 cP (25°C) |
| Dipole moment | 2.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -5649 kJ/mol |
| Pharmacology | |
| ATC code | D27AA02 |
| Hazards | |
| Main hazards | May cause cancer. Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H351, H410 |
| Precautionary statements | P261, P264, P270, P272, P280, P302+P352, P308+P313, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: — |
| Flash point | Flash point: 224 °C |
| Autoignition temperature | 500°C |
| Lethal dose or concentration | LD50 oral rat 9000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >10 gm/kg (oral, rat) |
| NIOSH | NJ7800000 |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 2 mg/m³ |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
p-Aminodiphenylamine Diphenylamine Phenyl-α-naphthylamine |

