N-Nitrosodimethylamine: Risks, History, and the Lessons We Still Learn
Historical Development
Long before its name alarmed toxicologists, N-Nitrosodimethylamine (NDMA) quietly emerged from basic chemical curiosity. Back in the mid-20th century, researchers studying nitrosamines noticed some odd connections between industrial processes and clusters of illness. Early on, the compound found accidental production in anything with dimethylamine and nitrite under acidic conditions. Before long, evidence began piling up from contamination events in water supplies, foods, and pharmaceuticals. Government agencies and independent labs started tracking NDMA around the 1970s. By then, cancer links in lab animals raised red flags, forcing regulators and chemists to rethink manufacturing processes and monitoring systems.
Product Overview
NDMA doesn’t walk into the spotlight often, but its unsettling reputation makes it a household name in regulatory circles. In its pure form, it appears as a yellowish, oily liquid. Most people outside of laboratory settings never see NDMA directly. Instead, they hear about it when it shows up in tainted medication, factory waste streams, or chlorinated water. Because the compound shows up in such a wide range of places, keeping tabs on it requires coordination among food safety officials, pharmaceutical watchdogs, and water treatment engineers. Pharmaceutical recalls and drinking water warnings often trace their origins back to this one little molecule.
Physical & Chemical Properties
NDMA boils at 151 °C, and it dissolves easily in water and many organic solvents. It gives off a faint, fishy odor. Chemically, NDMA belongs to the nitrosamine class, a group not known for stability or long shelf life, especially when exposed to light and air. The molecule’s structure—short, simple, yet reactive—lets it form as a byproduct wherever nitrites and methylamines meet under the right conditions. Because of its volatility and water solubility, NDMA contaminates groundwater with alarming ease. Handling it requires good ventilation and tight controls to prevent inhalation or spills.
Technical Specifications & Labeling
Laboratories and chemical suppliers provide NDMA as a reagent-grade material, with purity levels above 98%. Packaging usually involves amber glass containers to block light, and labeling reflects the compound’s carcinogenicity with hazard codes under GHS and OSHA. The instructions warn about avoiding sources of heat, ignition, or direct sunlight. Suppliers need to track quantities tightly, since regulatory agencies monitor every shipment closely and demand detailed records on every gram produced, stored, or shipped.
Preparation Method
Chemists usually synthesize NDMA by reacting dimethylamine with sodium nitrite in an acidic environment. In a reaction flask cooled to near 0 °C, the dimethylamine reacts quickly with nitrous acid, formed in situ from the addition of the nitrite to the acid. The product separates as an oily layer, which gets washed, dried over sodium sulfate, and further purified by distillation. Even small changes in temperature, concentration, or timing can swing the yield or, worse, increase the risk of dangerous byproducts. This lab-scale synthesis doesn’t scale up safely, and industrial settings avoid deliberate NDMA formation whenever possible, preferring to prevent it rather than manage it.
Chemical Reactions & Modifications
NDMA’s main story focuses not on its use, but on its fate in the environment and living bodies. Sunlight, particularly UV, breaks the molecule into more benign fragments, and some advanced water treatment systems rely on photolysis to destroy NDMA. In biological systems, the liver metabolizes it through cytochrome P450 enzymes, but the process creates reactive intermediates that damage DNA. Fire, heat, and strong acids don’t easily decompose the molecule—the resilience that helps it persist in water supplies also makes it challenging to eliminate from contaminated sources.
Synonyms & Product Names
NDMA wears a few different names, popping up in the literature as Dimethylnitrosamine, N,N-dimethylnitrosamine, and sometimes simply DMN. In regulatory filings and safety documents, the full IUPAC name, N-Nitrosodimethylamine, appears alongside chemical abstracts numbers and hazard codes. Occasionally, trade documents reference synonymous terms, but regulators insist on clear language.
Safety & Operational Standards
Few chemicals prompt sharper protocols than NDMA. Workplaces that handle it must provide fume hoods, personal protective equipment, and continuous air monitoring. Storage happens far from any source of food or drinks, and employees train on spill response, decontamination, and medical monitoring. Regulatory bodies like the EPA and OSHA set allowable exposure limits below one part per billion for water and air. Laboratories frequently audit their procedures because one lapse can mean months of sensitive remedial work. NDMA earns its spot on the list of substances that demand both technical expertise and a healthy respect for its hazards.
Application Area
NDMA earns little favor as an intentional ingredient. Instead, its presence haunts the margins: as a trace contaminant in food, tobacco smoke, pharmaceuticals, rubber manufacturing, and industrial solvents. Brewers have to check malted barley for nitrosamines, and municipal water plants constantly tweak disinfection to keep precursors from reacting. Pharmacies discovered the molecule in blood pressure drugs and antacids, sparking massive recalls. Every industry that encounters amines and nitrites, whether food processing or plastics, faces the ongoing challenge of limiting NDMA generation.
Research & Development
Research on NDMA splits between environmental monitoring and toxicology. Analytical chemists refine methods like gas chromatography-mass spectrometry to measure traces even below a nanogram per liter. Wastewater engineers experiment with UV-based processes and novel adsorbents to scrub NDMA from water. In pharmacology, detection methods get ever more sensitive, looking to intercept contaminated drugs before they reach dispensaries. Toxicologists continue to probe the molecule’s effects in test systems and animal models, hoping for clues that translate to real-world disease prevention. The work never slows, with funding tied to every new contamination event or breakthrough in detection.
Toxicity Research
NDMA’s carcinogenicity stands among the most studied properties. Even low doses cause DNA methylation and mutations in mammals; rats and mice develop liver, lung, and kidney tumors after chronic exposure. The International Agency for Research on Cancer lists it as a probable human carcinogen. Acute exposure can cause nausea, abdominal cramps, liver damage, and ultimately death at high enough concentrations. Most human data comes from accidental exposure, tracing increased rates of certain cancers following contaminated drinking water or drugs. As detection improves, researchers unearth smaller risks that matter over a lifetime, especially for communities with vulnerable water supplies.
Future Prospects
NDMA’s legacy challenges science with the ongoing need for vigilance, not just technical innovation. Eliminating the compound calls for multidisciplinary teamwork, connecting organic chemists, engineers, manufacturers, public health experts, and regulators. New catalysts, detection tools, and water treatments show promise for removing NDMA before it ends up in the environment or products. Advances in green chemistry take the source problem seriously, redesigning industrial reactions and food processing to keep precursors away from nitrosating agents. Public awareness drives change, as more people demand testing, transparency, and accountability. Even with modern advances, NDMA’s lesson endures: invisible dangers require both scientific insight and practical, everyday care.
What is N-Nitrosodimethylamine used for?
Tracing Its Roots
N-Nitrosodimethylamine, known to chemists as NDMA, shows up in more places than most people realize. Decades ago, folks used it in the production of rocket fuel and lubricants. Scientists once counted on it as a reference chemical when they wanted to understand how dangerous certain substances could get in lab tests. Anyone who has studied environmental chemistry will have heard of NDMA because of its notorious toxicity.
NDMA and Everyday Risks
NDMA doesn’t have any benefit in medicine or consumer products. People became aware of it mainly as a pollutant—one that slips into drinking water, processed foods, and, in rare cases, medications. Water treatment plants face headaches trying to get rid of it. Contaminated wells can force small towns to rely on trucked-in water or expensive filtration systems. In my neighborhood a few years back, the local water authority spent months explaining why they had to issue boil notices.
Meat processing sometimes leads to NDMA formation, especially during high-temperature cooking. I noticed the concern rise when Canadian regulators found it in some batches of ranitidine, a common heartburn medicine. Drug recalls rattled people’s trust in familiar treatments and pushed the industry to examine its manufacturing methods more closely.
Why It Matters
NDMA doesn’t escape scrutiny for no reason. Researchers have shown that it causes cancer in animals, even in low amounts. The US Environmental Protection Agency and the World Health Organization both warn against it. The concept of “acceptable risk” isn’t friendly to NDMA; it takes incredibly tiny concentrations to trigger concern—think nanograms per liter in water.
The trouble lies in how NDMA forms. Chlorination or disinfection, two common steps in water treatment, can turn seemingly harmless materials into this toxic substance. Regulators look puzzled when contamination pops up, because no one is adding NDMA on purpose. Instead, it sneaks in after chemical reactions between other ingredients.
Solutions That Deserve Attention
Better monitoring plays a big role. Labs and factories can use advanced testing to catch NDMA at trace levels, more sensitive than the techniques used in the past. Pharmaceuticals now face stricter guidance on solvent handling and temperature control. For water, processes like activated carbon filtering and advanced oxidation help reduce its concentration. In my work with municipal water advocacy, I saw first-hand how community pushback led to upgrades in local water plants.
Regulators carve tighter limits with each passing year; the US Food and Drug Administration and Health Canada both demand more rigorous inspection for drugs likely to contain or produce NDMA. Public awareness also helps. If people ask about what’s in their pills or food, companies feel more pressure to answer with transparency and improvements.
NDMA stands as a reminder that attention to detail, from factory floors to kitchen tables, really does matter in public health. Strong oversight and investment in cleaner technology keep this invisible threat in check—one small test at a time.
What are the health risks associated with N-Nitrosodimethylamine exposure?
What is N-Nitrosodimethylamine?
N-Nitrosodimethylamine, commonly called NDMA, turns up in places most people wouldn’t expect. Water, cured meats, some medicines, and even some personal care products can all have a link to NDMA. This chemical forms during certain industrial processes, like rubber or dye manufacturing, and it can creep into the water we drink through treatment plants and wastewater. Because of the ways it gets into different environments, people face exposure in ways that often aren’t obvious.
Why NDMA Raises Health Concerns
NDMA’s danger comes mainly from what it does inside the body. The chemical belongs to a group called nitrosamines, which scientists have studied for decades. Research highlights that NDMA can damage DNA once inside cells, which plants the seeds for cancer down the road. This risk pushed the World Health Organization and the U.S. Environmental Protection Agency to label NDMA as a probable human carcinogen. When people take in enough NDMA over a long stretch, the body’s natural repair processes struggle to keep up with the harm done to liver cells and other tissues.
Short-Term vs. Long-Term Exposure
Short-term contact with NDMA rarely causes symptoms right away, but at very high levels, a person can suffer headaches, vomiting, or an upset stomach. The real concern starts when small amounts slip into the system day after day from sources like water and food. The liver takes most of the hit because it filters toxins, including NDMA. Over time, animal studies show that continual exposure causes scarring, tumors, and even total liver failure in some cases. This work lines up with cases in people who developed liver problems after years of consistent low-level NDMA intake.
NDMA in Medicines
In recent years, NDMA showed up in certain medications like ranitidine and some blood pressure drugs. The U.S. Food and Drug Administration pulled several of these off shelves right away once testing revealed NDMA above accepted limits. These recalls came after routine analysis using advanced methods, which pierced the illusion that modern pharmaceutical processes fully protect the public from chemical impurities. The fact that NDMA could sneak into products designed to heal highlights the need for tough oversight and constant improvement in production checks.
How to Lower Risk and Stay Informed
Water treatment changes have played a big role in reducing how much NDMA people take in at home. Plants now use alternative disinfectants and tighter controls to stop NDMA from forming during the process. Consumers help themselves by using home water filters certified for nitrosamine reduction. On the medical side, reading up on FDA announcements and speaking to healthcare providers if recalled medications surface can prevent harm long before symptoms start. Choosing foods with fewer processed meats and relying less on smoked and cured items also makes a difference. Education goes a long way—staying aware and making small choices every day can limit exposure from several sources at once.
Future Steps for Public Safety
Agencies continue to monitor NDMA and set strict limits in drinking water and pharmaceuticals. Regular updates in regulation, tougher inspection routines, and public reporting all play a part in limiting exposure. Real change starts with transparent data and a system that flags problems as soon as they surface. Scientists keep searching for better testing devices and smarter ways to screen food, water, and medicine. These efforts matter, not just for those already dealing with liver concerns, but for anyone wanting to live in a world where hidden chemicals have less chance to do long-term harm.
How is N-Nitrosodimethylamine detected in water and food?
The Shadow of NDMA in Daily Life
N-Nitrosodimethylamine, better known as NDMA, sends up red flags for anyone who pays attention to public health. It was not always on people’s minds, though. Back in the day, folks drank tap water and ate lunchmeat without ever wondering about hidden contaminants. That changed once NDMA’s cancer-causing potential became known. It can show up in water from industrial runoff, make its way into cured meats, and even sneak into some medications. With risks like this, reliable ways to catch NDMA before it lands on the dinner table or in a glass of water matter more than ever.
How Laboratories Spot NDMA in Water
All the talk about NDMA isn’t just noise. Testing facilities use some hefty tools to sniff out even tiny traces. Gas chromatography paired with mass spectrometry (GC-MS) stands as the gold standard. Over years of following public drinking water issues, I’ve seen how cities try to stay one step ahead of the problem. They often rely on these methods because NDMA turns up at such low concentrations that older testing couldn’t spot. Because NDMA breaks down in the presence of sunlight but persists in soil and water, the push for strong, current methods keeps growing.
A solid test run starts off with extracting a water sample, cleaning it up, concentrating it, then running it through GC-MS. The technique detects NDMA levels down to nanograms per liter. The precision gives health officials valuable information for making sure regulations keep people safe. These steps cost time and money, but there’s no way around them if we want honest answers about what’s flowing from the tap.
NDMA in Food—Tricky to Track, Hard to Ignore
It’s not just about water. Processed meats, cheese, beer, and some vegetables can carry NDMA. Not everyone realizes that certain cooking processes—like smoking or grilling—let the compound form. Spotting NDMA in food means cracking open more than just a soda. Right now, food safety labs use similar advanced detection systems as those for water, but the samples get a bit messier. Food needs breaking down and special prep steps to separate NDMA from fats and proteins.
Monitoring programs play catch-up as new sources get discovered almost every year. During outbreaks, like those involving tainted drinking water in California or pharmaceutical recalls across the US, regular labs struggled to meet demand. Better training and funding would get more labs ready for sudden surges.
Real-World Solutions for Real Problems
Regulators now keep a closer eye on NDMA by setting strict limits. The Environmental Protection Agency suggests no more than 0.7 nanograms per liter in drinking water. Countries take different approaches—some impose stricter limits, others lag behind. I’ve seen how contamination scares prompt upgrades in both detection technology and everyday water treatment. Techniques like advanced carbon filtration and UV oxidation chop NDMA down to safe levels. Municipalities with newer equipment handle the threat much better.
In the food industry, things move slower. Manufacturers can reduce NDMA by tweaking curing agents and refining industrial processes. Pressure from watchdog groups has nudged some brands to clean up their act, but more transparency builds public trust. More routine testing and better communication help everyone make safer choices.
NDMA’s not going away. As people wake up to the risks, science and policy keep moving forward. The more accurately we can test, the safer our plates and our environment become. That’s a story worth following—and demanding action on.
What safety precautions should be taken when handling N-Nitrosodimethylamine?
Understanding the Risks
I've seen plenty of hazardous chemicals in my years working around labs and industrial environments, but N-Nitrosodimethylamine (NDMA) always demands extra respect. This stuff carries a reputation for being both highly toxic and a likely cancer-causing agent. Just a whiff or a small spill can spell real trouble, so there’s no room for shortcuts. Think of a chemical that once snuck into drinking water or contaminated certain blood pressure medicines—NDMA's name doesn’t pop up by accident in those stories. The federal government classifies it as a probable human carcinogen, and for good reason. Small doses over time add up, and even brief exposures can harm organs like the liver.
Prioritizing Engineering Controls
Working with NDMA means you start with engineering controls before anything else. I’ve learned that the fume hood is your best friend. In a properly functioning hood, vapors stay away from your face and lungs. Ventilation keeps airborne concentrations in check, cutting down inhalation risks. Investing in reliable local exhaust systems prevents small mistakes from turning into big accidents. Never settle for a faulty or over-crowded hood, and check the filters before you even think about uncapping a vial.
Personal Protective Equipment: No Room for Shortcuts
Nobody grows up wanting to suit up like a hazmat character, but with NDMA, this level of protection turns into common sense. Double up on gloves: nitrile over latex has held up nicely for me, especially when spills threaten. Splash goggles keep vapor and droplets from reaching your eyes. Wear a lab coat—preferably disposable—to prevent tracking any residue beyond your work zone. Don’t take gear out of the lab. Dedicated shoes or boot covers cut down on accidental spread.
Strict Handling and Storage Routines
NDMA doesn’t travel alone in a workspace. One messy bottle can expose a whole bench. Use sealed containers designed for strong, volatile chemicals. Store them away from sunlight and heat, locked up and labeled in bold lettering. I’d always keep an updated inventory—running out of a good label or missing a date triggers confusion that nobody needs when dealing with poison. Transportation deserves equal care. Move NDMA in enclosed carriers and never by hand or in your pocket.
Training and Emergency Preparedness
You can’t fake preparation. Every person near NDMA must know what they’re doing. I still remember a co-worker grabbing a wrong container by mistake—quick training prevented a disaster. Everyone should understand the route to the eyewash and shower, spill kit locations, and the proper number to call if something goes wrong. Have written emergency procedures on the wall. Run drills a couple of times a year, just as you might for a fire. Spills need to be cleaned right away using dedicated kits, never with regular towels or household cleaners, since those spread the risk.
Waste Management and Environmental Care
You can’t pour NDMA down the drain. Special waste containers lined with absorbent material take every drop and solid left over. Coordinate pickup promptly through a licensed hazardous waste contractor with a clear paper trail. Never assume that a rinse or wipe has finished the job. A little residue can linger and contaminate surfaces or even wastewater.
Why All These Steps Are Worth It
Safety habits with NDMA don’t reflect paranoia—they stem from real-life lessons and documented harm. Protecting yourself and your team means following proven protocols each time, not just when the supervisor watches. Ultimately, every step shields both people and the surrounding environment from a chemical that gives no second chances.
Is N-Nitrosodimethylamine regulated by government agencies?
What Draws Regulators’ Eyes to N-Nitrosodimethylamine?
N-Nitrosodimethylamine, or NDMA, doesn’t show up in everyday conversations, but it’s hidden in places that touch lives all over the world. Take a moment to look at where it turns up: drinking water, medicines, cured meats, and even some cosmetics. The reason so many people keep a close watch on NDMA is clear—it’s a known carcinogen. Decades of research show animals exposed to this chemical face a higher risk of developing cancer, and human studies echo that warning.
Government Oversight—Not Just Red Tape
Agencies have stepped in because no one wants toxic contaminants sneaking into what they eat, drink, or take as medicine. In the United States, the Environmental Protection Agency (EPA) has set a lifetime health advisory level for NDMA in drinking water at 0.00002 milligrams per liter. That’s so tiny, you’d need to line up about a million gallons of water before finding a gram of it. The FDA launched investigations into NDMA contamination in several medications like ranitidine (Zantac) and certain blood pressure drugs after laboratory testing found unacceptable levels.
Globally, health authorities from Canada, Europe, and Asia have tracked NDMA in food and pharmaceuticals, setting limits and recalling products. The World Health Organization and International Agency for Research on Cancer each reviewed the science and listed NDMA as a probable human carcinogen. No one thinks “probable human carcinogen” is something to sweep under the rug, so countries use that science as a reason to clamp down.
Roots of the Problem
NDMA forms in the environment and in manufacturing plants where certain chemicals or conditions combine. For example, water treatment sometimes leads to trace NDMA formation when disinfectants meet organic compounds. That’s not just a water issue—pharmaceutical manufacturing, too, creates conditions where NDMA can slip in unnoticed, especially if raw materials carry traces of dimethylamine or nitrites.
From my own experience as a science writer, the biggest surprises show up after a routine check or new testing method. Medicines pulled from shelves in 2019 made headlines, but many people only learned the term “NDMA” after that point. News like this shakes public trust. If NDMA can hide in stuff that should improve health, regulators need ways to check, re-check, and improve their systems.
How Can Agencies Do Better?
Stopping a contaminant like NDMA calls for more than strict rules. Government offices need funding for advanced labs. They benefit from research partnerships that track new pathways for NDMA to form and ways to catch it before mass production. Auditing supply chains more often makes sense. Companies should have recipes and processes checked before sending out ingredients, not after problems reach patients or supermarket shelves.
Real fixes also depend on clear communication. When a recall happens, agencies move faster by talking plainly about what’s at risk and how people should respond. There’s no room for jargon when trust is at stake. Community testing programs and open data access help close the loop between science and the public, letting everyone see not just what the rules require, but how well those rules keep food, water, and medicine safe.
NDMA might not turn heads at the dinner table, but fighting it brings together scientists, regulators, and the everyday people who just want to live without worrying what’s in their water or on their plate.
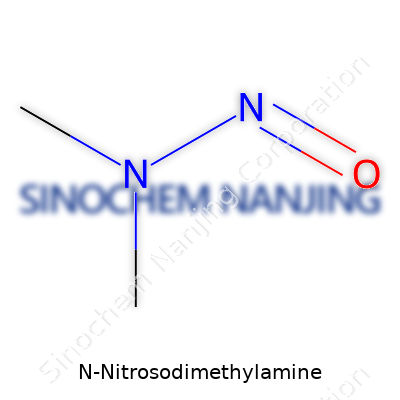
| Names | |
| Preferred IUPAC name | N-nitroso-N-methylmethanamine |
| Other names |
NDMA Dimethylnitrosamine DMN N-Nitroso-N,N-dimethylamine |
| Pronunciation | /naɪˌnɪtrəʊsəʊˌdaɪˈmɛθ.əl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 62-75-9 |
| Beilstein Reference | RT1984030 |
| ChEBI | CHEBI:35807 |
| ChEMBL | CHEMBL43036 |
| ChemSpider | 6827 |
| DrugBank | DB00636 |
| ECHA InfoCard | ECHA InfoCard: 100.005.587 |
| EC Number | 206-455-4 |
| Gmelin Reference | 87837 |
| KEGG | C00474 |
| MeSH | D009635 |
| PubChem CID | 6124 |
| RTECS number | JZ0525000 |
| UNII | W8U8U8V0QM |
| UN number | UN2227 |
| CompTox Dashboard (EPA) | N-Nitrosodimethylamine CompTox Dashboard (EPA) string: **DTXSID3020405** |
| Properties | |
| Chemical formula | C2H6N2O |
| Molar mass | 74.08 g/mol |
| Appearance | Pale yellow liquid |
| Odor | odorless |
| Density | 1.004 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.57 |
| Vapor pressure | 2.7 kPa (at 20 °C) |
| Acidity (pKa) | pKa = 3.54 |
| Basicity (pKb) | 3.45 |
| Magnetic susceptibility (χ) | -15.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.445 |
| Viscosity | 0.605 cP (20 °C) |
| Dipole moment | 3.57 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 277.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -11.56 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -390.6 kJ/mol |
| Hazards | |
| Main hazards | Suspected of causing cancer; causes damage to organs; toxic if swallowed, inhaled, or in contact with skin; may cause genetic defects; harmful to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H350, H341, H301, H311, H331, H370, H372, H412 |
| Precautionary statements | P201, P202, P260, P264, P270, P280, P308+P313, P405, P501 |
| NFPA 704 (fire diamond) | 4-3-2 Carc |
| Flash point | 50 °F (10 °C) |
| Autoignition temperature | 430 °C |
| Explosive limits | Explosive limits: 0.7–12.0% |
| Lethal dose or concentration | LD50 (oral, rat): 30 mg/kg |
| LD50 (median dose) | DMN: 0.02 mg/kg (oral, human) |
| NIOSH | NIOSH#: QQ9625000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of N-Nitrosodimethylamine is "0.0005 ppm (1 μg/m³)". |
| REL (Recommended) | 0.00005 |
| IDLH (Immediate danger) | 5 ppm |
| Related compounds | |
| Related compounds |
Dimethylamine Trimethylnitrosamine N-Nitrosodiethylamine |

