Rethinking N,N'-Dinitroso-N,N'-Dimethylterephthalamide: Progress, Pitfalls, and Possibility
Charting the Journey: Historical Development
Chemicals often slip under the radar, their impact tucked away in lab reports and technical papers. N,N'-Dinitroso-N,N'-Dimethylterephthalamide, or DNDMTA as many in the field call it, tells one of these hidden stories. This compound didn’t spring from a vacuum—it traces its roots to academic curiosity in the mid-20th century, when researchers scoured aromatic amides for fresh functional groups. Its foundation rests on modifications of terephthalic acid derivatives, a class bustling with activity since polyesters and high-performance fibers became staples in industrial chemistry. DNDMTA stood out thanks to its dinitroso structure, opening doors in nitroso chemistry, a space that’s often overshadowed by better-known nitro or amino compounds. Over time, the nitroso motif attracted attention for its reactivity and its knack for spawning novel materials or serving as intermediates in advanced synthetic routes. If you work in synthetic labs, you’ll recall how the initial literature hardly offered practical syntheses—makers had to improvise and refine conditions through sheer stubbornness and bench-top tinkering.
Getting into the Details: Product Overview
DNDMTA’s chemical signature gives it a certain notoriety among specialists. With two nitroso groups attached to a dimethyl terephthalamide core, this molecule straddles the fence between stable and reactive. The structure means it doesn’t play well with high heat or strong illumination for long durations. Still, the backbone ensures it lasting enough to ship and store for use in research and niche applications. Looking at the powder, most folks note its fine particle size and distinct color—a clue to its nitroso content. This isn’t something you toss around lightly; familiarity with hazardous organics becomes an unspoken requirement.
Physical and Chemical Properties
People often focus on numbers—melting point, solubility, decomposition temperature—yet there’s more to a compound like this than lab stats. As a solid, DNDMTA picks up moisture if left out, but it resists breakdown unless exposed to careless handling or incompatible chemicals. Water isn’t its favorite partner. It shows more affinity for organic solvents, sometimes dissolving sluggishly, which frustrates those of us attempting chromatographic purification. A compound’s color gives hints about its electronic structure, and DNDMTA’s hue stands as a badge for its nitroso groups. Reactivity-wise, the molecule dances a fine line: direct exposure to reducing agents spells quick changes, while exposure to oxidizers could set off unwanted reactions. The material’s sensitivity lays hidden until a moment’s inattention—seasoned chemists respect that risk.
Technical Specifications and Labeling
Specialty chemicals like DNDMTA rarely show up with fancy branding or splashy labels. Packaging does the job of keeping light and air out, usually in amber-glass containers or tightly capped vials. Labels skip the marketing fluff, focusing on concentration and purity figures, sometimes purity by elemental analysis, other times by HPLC or melting point. The product’s nitroso functionality means regulatory filings put it under the lens of chemical safety and transport standards, with proper hazard pictograms showing up on any legitimate academic or industrial shipment.
Preparation Method: Lessons from the Lab
Making DNDMTA is not for the faint-hearted. The synthesis draws on nitrosation of dimethylterephthalamide, often done with nitrosating agents or nitrite sources in acidic media. Experience in the lab tells me that temperature and acidity can swing the yield from miserable to respectable in a heartbeat. Excess acid or carelessness with reagent concentration leads to side products or washed-out yields. Post-reaction workups challenge your patience, with chromatography and recrystallization steps taking longer than anyone would like. This is not a “just add water” reaction, but the result rewards the careful hand. Tinkering with conditions lets some teams nudge the process towards greener chemistry, using less hazardous reagents or experimenting with solvent systems.
Chemical Reactions and Modifications
DNDMTA doesn’t exist in isolation. Its nitroso groups serve as sites for further transformation—think reduction to amines, oxidative coupling, or even diazo chemistry under the right conditions. Each reaction draws on the molecule’s balance between aromatic stability and nitroso lability. In practice, most modifications seek to wring new functional materials or probe mechanisms relevant to polymer science. In my time working with related compounds, it’s clear that keeping a close eye on reaction exotherms and byproduct gaseous emissions keeps things safe and productive.
Synonyms and Product Names
Depending on which paper you pick up, DNDMTA goes by several handles, usually slight shifts on its true name: Dinitrosodimethylterephthalamide, DNDT, or even combinations that confuse all but the most attentive researchers. Commercial sources may use shorthand codes, more out of convenience than any concerted effort at branding. Anyone reading across patents or journals learns quickly to check CAS numbers or best yet, structure diagrams.
Safety and Operational Standards
Working with DNDMTA, no one cuts corners. Safety sheets drill in the hazards: skin and respiratory irritation, risk of unknown chronic toxicity, and the potential for environmental persistence. Labs enforce glove and eyewear rules, and fume hoods become mandatory working space anytime this material leaves its jar. Waste handling doesn’t mean down the drain—proper storage for organic lab waste prevents accidents and regulatory headaches. From my experience, smart labs track their inventory closely, never stockpiling more than what the next experiments require.
Application Area: Where Does It Matter?
In the broader sweep of chemical manufacturing, niche compounds like DNDMTA rarely headline industrial processes. Yet, they carve out stakes in specialty polymer synthesis, organic electronics, or as test cases for developing new nitroso chemistry. Research teams push boundaries in material science by evaluating DNDMTA’s backbone as a scaffold or intermediate, hoping for new traits like improved conductivity or selective reactivity. The compound might not flood the market, but its influence drifts quietly into patents or academic theses focused on next-generation materials.
Research and Development: On the Shoulders of Determined Minds
DNDMTA’s story ties directly to ongoing research in aromatic nitroso chemistry. Publications from independent and corporate labs dig into its reactivity patterns and physical characteristics. R&D shines brightest in attempts to coax greater yields or safer preparation methods from classic protocols. Environmental considerations push some chemists to hunt for more sustainable nitrosation techniques, less reliant on crude acids or hazardous byproducts. Collaboration between academia and industry sometimes emerges around this molecule, as teams seek to apply freshly gained knowledge to functional coatings, adhesives, or molecular sensors. It’s an uphill trek—progress depends less on splashy breakthroughs, more on small, persistent advances layered year by year.
Toxicity Research: Avoiding Unseen Hazards
DNDMTA’s safety profile remains clouded by limited toxicity data. Early animal studies hint at possible toxic effects with repeated exposure, but peer-reviewed toxicology rounds out the picture only partially. For those of us focused on worker safety, the uncertainty feeds caution: even minor residues stay out of reach from general lab populations, and cleanup routines double as mini-safety drills. Environmental agencies raise concerns about possible persistence should spills occur outside containment. These worries don’t generate alarming headlines, but they color every conversation about scaling up or commercializing any compound with a nitroso core. New regulations could tighten restrictions as more hazard data surfaces in the literature. In practice, trust and verify remains the guiding motto—handle as though the risks are greater than proven, until time and data say otherwise.
Future Prospects: Where Trouble Meets Promise
DNDMTA stands at a crossroads many specialty chemicals face: promising enough for targeted research, dogged by safety worries and uncertain broad impact. Continued investment in greener synthesis, improved waste management, and comprehensive health research could transform the compound’s risk profile. The real gains may come from derivatives—structures that tweak the parent compound’s properties towards improved stability or lower toxicity—offering safer building blocks for material science or microelectronics. Regulatory trends now steer toward greater transparency and chemical stewardship, turning the spotlight on boutique molecules like this. For chemistry researchers, the challenge never shrinks: balancing pursuit of new function against a deep responsibility for safety and environmental impact. The lessons drawn from DNDMTA’s journey could serve as a blueprint for how today’s specialty chemicals face growing demands for responsibility, without giving up the spark of discovery.
What is N,N'-Dinitroso-N,N'-Dimethylterephthalamide used for?
What Does This Chemical Do?
N,N'-Dinitroso-N,N'-Dimethylterephthalamide looks like a mouthful, but in the world of rubber manufacturing, it’s a dependable tool that gets overlooked outside of specialty labs and factories. It goes by the trade name DPT, and it’s used as a blowing agent, which means it helps make holes—millions of tiny, precise ones—in rubber and plastics. Think about the last time you picked up a yoga mat or a car tire with a soft, foamy texture inside. That springiness and cushioning didn’t just happen by accident.
Chemically, DPT releases nitrogen gas when heated. That gas gets trapped inside rubber or plastic as it solidifies, making a lightweight, airy material that’s tough at its core. I remember walking through a tire plant once, seeing conveyor belts full of black rubber. Most people see a tire and notice the tread, but people in the industry know the secret work that chemicals like DPT do under the surface.
Why Does It Matter?
Foamed rubber pops up everywhere—from shoe soles to gaskets, insulation, floor mats, and soundproofing panels. The right chemical recipe inside these products often dictates whether they end up as soft as a pool noodle or as tough as a track surface at a sports stadium. Years of trial and error have made DPT a go-to choice because it releases gas smoothly and doesn’t break down into anything that would weaken the finished product.
Some blowing agents make foam with big, uneven bubbles. That causes weak spots and wasted material. DPT’s fine-grained bubbles keep everything consistent, which means safer tires and longer-lasting products. Cars, trains, and even playgrounds count on that kind of reliability. Manufacturers learn quickly if a blowing agent starts producing off-gas that smells strong or leaves toxic traces behind. In that sense, DPT offers a sweet spot: it’s effective at the right temperature and doesn’t leave behind high-risk residue when processed correctly.
What About Health and Safety?
Safety in the workplace always looms around discussions of industrial chemicals. DPT works well at lower concentrations, so factories limit dust and vapor that can be inhaled. Nobody wants residual nitrosamines—recognized as risky in some chemical cousins—lingering in a warehouse. Regulatory agencies focus on monitoring production lines, personal protection, and proper storage. The chemical should stay far from open flames and heavily trafficked spaces that might see a spill.
Some people in the industry push for greener options, hoping to phase out anything that hints at long-term environmental effects. Recyclers still face challenges pulling apart foamed rubber, so future improvements might come from new formulations or better end-of-life planning for rubber products.
Looking Forward
Researchers chase innovations that match DPT’s performance with fewer health and environmental concerns. The push for safer manufacturing and closed-loop recycling drives a lot of the new work in this corner of the chemical world. Transparency about the chemical processes behind everyday objects remains important for both manufacturers and consumers. The more people understand what’s inside the materials we step on, bounce on, or drive with, the smarter choices we can make about how they’re made—and what happens after we’re done with them.
Is N,N'-Dinitroso-N,N'-Dimethylterephthalamide hazardous to health?
Understanding What We’re Dealing With
N,N'-Dinitroso-N,N'-Dimethylterephthalamide usually goes by a much shorter name in industrial circles: DNDMTA. It works as a blowing agent, helping plastics and rubber form the foamy shapes we use in all sorts of products. To most people, the name doesn’t ring any bells, but anyone who’s handled chemical substances at work knows that every unfamiliar label deserves a closer look.
Assessing the Health Hazards
DNDMTA falls into the group of nitrosamines and related compounds. Several nitrosamines already have a reputation for being toxic. They’ve made their way onto lists of possible human carcinogens, with studies linking some of them to serious health risks. DNDMTA, by its structure, shares some characteristics with notorious chemicals like NDMA, which has caused huge recalls for everything from heart medicine to food products. DNDMTA hasn’t been studied as much, but that’s not a reason to brush off concerns.
Regulatory bodies in North America and Europe look closely at blowing agents like DNDMTA. Tests on similar chemicals revealed that long-term exposure can increase the risk for cancer, liver damage, and other chronic issues, mostly by way of inhalation or skin contact. DNDMTA on its own doesn’t appear in consumer goods—it gets used during the manufacturing process, with finished items cleaned up before reaching customers. That said, people who work at plants making rubber soles, foamed seals, or insulation get much closer to the substance. Here, exposure becomes a real concern.
The Real-World Risks
Having worked for years in facilities where chemicals like DNDMTA come into play, I’ve watched the same hazards play out across different plants. Sometimes, a lack of proper ventilation lets dust and fumes build up; sometimes, gloves and masks go unworn because production moves faster when nobody gears up. In those cases, it’s not hard to see skin rashes or even more severe symptoms in long-term workers. Some folks shrug off the worry, but years later, health issues start cropping up—then the questions turn pointed: “Was it the stuff we used to handle?”
Scientific reviews by agencies like the European Chemicals Agency consider DNDMTA a substance of concern. Animal studies, while not a direct stand-in for humans, suggest a link to tumors after chronic exposure. Direct evidence for people might still be thin, but the structure and family of DNDMTA alone push for caution. Manufacturers in the US have begun weighing alternatives because of mounting evidence. Data published over the last decade urges extra safeguards for those who come in contact regularly.
Steps Toward Safer Work Environments
Plant managers and safety teams benefit from treating every nitrosamine-like compound with respect, not just the ones already causing headline recalls. Good air circulation, protective equipment, and regular health screenings make a concrete difference for those on the ground. In my experience, some shops excel because workers actually feel empowered to speak up about chemical hazards, while others lag far behind because concerns get ignored in favor of keeping machines running.
Substituting DNDMTA with less risky additives remains a work in progress—switches sometimes cost more or create extra hurdles in production. Even so, some sectors committed to phasing out the highest risk chemicals after enough studies pointed toward chronic harm. Tracking exposure in real time, improving training on chemical handling, and prioritizing the search for safer alternatives—these actions protect real people on the job, sometimes decades before new laws catch up.
What is the molecular formula of N,N'-Dinitroso-N,N'-Dimethylterephthalamide?
Digging Into the Structure
Chemical structures might look intimidating, but learning to see the story behind a name can clear some of the fog. Here, N,N'-Dinitroso-N,N'-Dimethylterephthalamide stands as a mouthful with a precise personality. This molecule contains two nitroso groups, two methyl groups, and the backbone of terephthalamide. The molecular formula—C10H10N4O2—lays out its carbon, hydrogen, nitrogen, and oxygen framework.
Understanding Its Shape and Usage
To most, even just hearing the full name sounds like something out of a lab manual. The value lies in the details. The nitroso groups offer reactivity that chemists hunt for in synthetic routes or explosives research. The dimethyl groups provide stability and, in many cases, prevent breakdown or rapid reaction with air and water.
The backbone—terephthalamide—links to processes that build more complex structures. Chemists use this class for applications like creating polymers or adding special functions to materials. So, C10H10N4O2 doesn’t just exist as a loose curiosity. A few decades back, researchers explored its role as a blowing agent in plastics, releasing nitrogen gas when heated to give materials new shapes and properties. The environmental impact of traditional alternatives pushed scientists to explore other options, and compounds like this one became part of the solution.
Molecular Formula in Health and Safety
It’s easy to focus just on the theory, but handling these chemicals means taking real safety steps. Nitroso compounds often come with red flags due to their biological activity. Some members of this family link to carcinogenicity—it’s not something for backyard experiments. Lab protocols emphasize gloves, hoods, and careful disposal. Mismanagement of such compounds can create harm, both immediate and long-term.
In industry, regulated handling isn't just about avoiding spills. The breakdown products require proper containment. Responsible chemical use cuts down on the risk of harm to water sources, workers, and residents nearby. The formula C10H10N4O2 means more than atoms; it stands for a responsibility chain.
Broad Lessons from a Specific Case
Studying specific formulas teaches lessons that reach beyond the lab. Every molecule’s story reminds me that progress doesn't happen in a vacuum. As a student, handling complex compounds developed respect for the power inside these small arrangements of atoms. Precision matters—one swapped group changes use and hazard completely.
The broader chemistry community faces the challenge of finding safer, leaner, and more sustainable ways to harness such molecules. Replacing hazardous intermediates or cutting down on waste drives innovation. Collaboration between academia and industry gives momentum—new research often lands on my reading list months before the public hears about it.
So, understanding C10H10N4O2 isn’t about memorizing letters and numbers. It means knowing where a single molecule fits in our technology, our environment, and our safety net. Each formula crack open a part of our modern world—one question about a formula, one answer about how society moves forward.
How should N,N'-Dinitroso-N,N'-Dimethylterephthalamide be stored?
Why Proper Storage of Chemicals Matters in Everyday Work
Anyone who's ever spent time in a lab knows the mix of anxiety and routine that comes with storing chemicals. I’ve seen accidents come from things as simple as leaving a lid loose or putting a sensitive compound on the wrong shelf. For something like N,N'-Dinitroso-N,N'-Dimethylterephthalamide, the lesson comes in sharper focus. This compound has ties to the rubber industry and often shows up as part of rubber vulcanization. Most people outside chemical manufacturing have never heard its name, but mishandling it can ripple out in ways nobody wants to see.
Safe Storage Requires More Than a Locked Cabinet
Chemical storage isn’t just about finding any empty spot. I remember once hearing a tech say, “If it fits, it ships.” That phrase might work at the post office, but not here. Experience tells me solid protocols matter, especially with nitroso compounds that carry safety risks. For this chemical, start with a cool, dry, well-ventilated room to stop moisture and heat from doing their worst. Excessive heat increases the odds of decomposition, which sometimes means releasing fumes nobody wants to breathe. Keep it away from light, since photodecomposition can create trouble without warning.
It pays to use containers that resist corrosion. Glass or certain plastics work well, not those metal cans that react or rust. In any lab I’ve worked in, the best practice involves clearly labeling everything, right down to a date stamp and the full chemical name. Confusing this compound with something less risky can get people hurt.
Why Personal Respect for Safety Makes a Difference
Complacency is a tough enemy. Over years of handling thousands of samples, I can pinpoint moments when I figured nothing bad could happen. That’s just not how chemistry works. For example, N,N'-Dinitroso-N,N'-Dimethylterephthalamide remains stable if sealed and kept under the right conditions, but oxygen and moisture quietly degrade it. This breakdown can lead to the formation of nitric oxides, which are hazardous. The right storage solution blocks out both air and water, reducing spoilage and health risks for everyone in the facility.
I always double-check that chemicals like this keep their original manufacturer's seal intact. The habit seems small, but once a container cracks or a lid warps, waste and danger aren’t far behind. In shared spaces, I’ve seen accidents arise from putting one chemical with incompatible mates. Clear segregation from acids, peroxides, or bases means fewer fire risks and unpredictable reactions.
Fixing Common Storage Problems
One quick way to avoid issues: schedule regular storage checks. In my own experience, catching a leaking bottle early prevented an embarrassing call to the fire department. Employees should get routine training on storage practices too. Relying on serious-sounding safety posters doesn’t cut it—nothing replaces hands-on instruction. If your workplace handles chemicals, someone on staff should own the responsibility for routine inspections and keep an updated inventory. Emergency spill kits and good ventilation offer a first line of defense should something go wrong.
Creating a Culture That Looks Out for Each Other
When people take ownership of proper storage, the odds of injury or product loss shrink fast. Simple actions—like writing up a near-miss report or giving a quiet heads-up to a coworker who left a bottle out—build a workplace where safety sticks. Chemicals bring value, but only to teams that show them the respect they call for every single day.
What are the handling precautions for N,N'-Dinitroso-N,N'-Dimethylterephthalamide?
Understanding the Compound
N,N'-Dinitroso-N,N'-Dimethylterephthalamide shows up in workplaces because of its use in polymer industries and rubber manufacturing. The big issue with compounds like this: they have heavy safety baggage. Years in chemistry taught me, anything with nitroso groups deserves respect—these substances react, decompose with heat, and sometimes release gases you don’t want to breathe. Industrial accidents rarely come from dramatic failures, but from ignoring simple rules day after day. Getting the basics right matters.
Direct Skin and Eye Contact Risks
Gloves are not optional. I tell colleagues that even in quick transfer jobs, nitroso compounds can sneak up on you. Skin absorbs chemicals through micro-cuts, and some organic nitroso compounds cause delayed irritation or worse. I recommend nitrile gloves rather than latex, as they're more resistant. Eye protection means goggles that seal on the sides—not just basic safety glasses. Splash accidents never announce themselves in advance.
Inhalation and Ventilation
Dust from this family of substances triggers strong warnings in safety bulletins. I never trust an open bench with anything aromatic and nitroso. Working in a well-functioning fume hood reduces risk. Not every lab keeps hoods pristine and uncluttered—make it a habit to check air flow before starting. Respirator masks with proper filters remain a last line of defense for emergencies. Colleagues who spent time in rubber compounding tell me even small amounts of these chemicals in the air lead to headaches and nausea fast.
Heat, Storage, and Decomposition Issues
Storing nitroso derivatives where sunlight or excessive heat sneaks in is asking for trouble. The decomposition risk jumps as temperature rises, so I always label and tuck these bottles away in dark, cool cabinets. Metal shelving stays away because corrosion or sparks from scraping cause unwanted surprises. All containers must close tightly, with an extra seal if possible. It doesn’t matter how short the storage interval: leaks and contamination get expensive and dangerous quickly.
Spill and Waste Management
Universities and work sites both make the mistake of downplaying small spills, yet trace residue builds up over time. Sweeping dry with a broom spreads dust—using disposable wet wipes followed by immediate bagging in chemical-resistant bins offers better control. Trained staff ought to oversee hazardous waste disposal, and records should log every disposal event. Pouring down the drain isn’t just lazy, it's illegal. I’ve read reports of wastewater systems getting contaminated from this type of oversight, costing millions to remediate.
Emergency Response and Training
A lot of workplace injuries link back to shortcuts or lack of training. Routine drills on spill containment, eyewash use, and quick-access to shower stations help save vision and skin. Printed SDS folders belong at eye level, not hidden. I suggest supervisors run through spill scenarios, see where panic or hesitation kicks in, and correct the gaps. No one wishes to need an emergency plan, but regretting its absence costs too much.
Supporting a Culture of Safety
Knowledge and vigilance turn risky chemicals into reliable tools. Young workers especially need hands-on demos, not just lectures. The stories people swap about near-misses and lessons learned last far longer than any dry manual. Industries using chemicals like N,N'-Dinitroso-N,N'-Dimethylterephthalamide have the responsibility to equip everyone, from the newest intern to the seasoned tech, with what really keeps them safe.
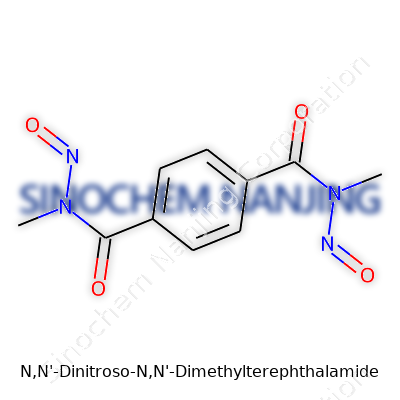
| Names | |
| Preferred IUPAC name | N,N'-dimethyl-1,4-benzenedicarboxamide dinitroso |
| Other names |
AI3-24051 Dimethylterephthalamide dinitroso NSC 15162 |
| Pronunciation | /ˌdiːˌɛnˌdaɪˈnɪtrəˌsoʊˌdiːˈɛmˌdaɪˈmɛθəlˌtɛrəfˈθæl.əˌmaɪd/ |
| Identifiers | |
| CAS Number | [940-69-2] |
| Beilstein Reference | 3846923 |
| ChEBI | CHEBI:51372 |
| ChEMBL | CHEMBL2011301 |
| ChemSpider | 26666621 |
| DrugBank | DB08347 |
| ECHA InfoCard | 03f78f3e-5c17-4170-ae6e-ba462ad2a1e7 |
| EC Number | 616-145-0 |
| Gmelin Reference | C10/C10:2 |
| KEGG | C18601 |
| MeSH | D013739 |
| PubChem CID | 13938 |
| RTECS number | IP9450000 |
| UNII | YG8B1P1G59 |
| UN number | 3261 |
| CompTox Dashboard (EPA) | WQ94E3Z363 |
| Properties | |
| Chemical formula | C10H10N4O4 |
| Molar mass | 252.20 g/mol |
| Appearance | Yellow solid |
| Odor | odorless |
| Density | 1.38 g/cm3 |
| Solubility in water | Insoluble |
| log P | 1.01 |
| Vapor pressure | 3.1E-4 mmHg at 25°C |
| Acidity (pKa) | 10.38 |
| Basicity (pKb) | 12.34 |
| Magnetic susceptibility (χ) | -35.4×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.603 |
| Viscosity | 1.185 mPa·s (25 °C) |
| Dipole moment | 4.83 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 392.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -34.61 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -366.7 kJ·mol⁻¹ |
| Hazards | |
| Main hazards | Suspected of causing cancer. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Danger |
| Hazard statements | H302 + H332: Harmful if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P321, P362+P364, P333+P313, P304+P340, P312, P501 |
| NFPA 704 (fire diamond) | 2-0-2 |
| Flash point | Flash point: 186°C |
| Lethal dose or concentration | LD50 oral (rat) 519 mg/kg |
| LD50 (median dose) | LD50 (median dose): 860 mg/kg (oral, rat) |
| NIOSH | RS2475000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 mg/m³ |
| Related compounds | |
| Related compounds |
Nitrosamine Dimethylnitrosamine |

