Looking Closer at N,N-Dimethylcyclohexylamine: More Than Just a Chemical Name
Tracing Its Roots Back Through Chemical History
Chemistry moves fast, but some compounds quietly shape whole corners of industry. N,N-Dimethylcyclohexylamine traces its lineage not to splashy discoveries, but to decades of incremental tinkering. The amine group, familiar to anyone who spent time in a lab, laid groundwork back in the early twentieth century, nudging organic synthesis in new directions. Cyclohexylamines carved out a steady, if unglamorous, place in manufacturing. Today, looking at N,N-Dimethylcyclohexylamine, you see echoes of history in its chemical design: a formula spoken in the language of utility and adaptability. I remember old chemistry texts describing amines as “workhorses”—not the show ponies, but dependable problem-solvers. This reputation didn’t spring from nowhere; it grew from real, day-to-day value in production.
Getting Acquainted With the Product
Spend any time reading up on this compound, and it’s clear why chemists and manufacturers reach for it. N,N-Dimethylcyclohexylamine—sometimes shortened to DMCHA—is a tertiary amine, most commonly showing up as a colorless to pale yellow liquid with a noticeable, fishy odor. On paper, it sounds unremarkable, but anyone handling it in the lab knows the importance of a substance that delivers reliable amine functionality without unexpected reactivity or degradation. I’ve seen its name slipped into polyurethane production or as a catalyst in curing reactions, always somewhere in the technical notes where the demands for consistency outweigh any desire for flashiness.
The Turning Gears: Physical and Chemical Life
People who work with chemicals appreciate the shorthand of boiling points, miscibility, and structural stability. DMCHA boils at a moderate range, making it manageable across standard application temperatures, and dissolves well in most organic solvents—a point that saves a lot of hassle when you’re moving from benchtop to factory. Unlike some more finicky amine derivatives, it doesn’t break down or discolor readily under normal conditions. That stability, in my experience, reassures process engineers aiming to avoid downtime. It’s less about extraordinary capability, more about showing up the same way batch after batch, year after year. This is a property you notice most when it’s missing from other compounds.
Technical Labels Tell a Story
Labeling for DMCHA usually lines up with industrial chemical standards. Labels specify a minimum assay—say, more than 99 percent purity for high-performance uses. Water and other residue levels stay low, flagged by tight spec sheets, because even small impurities throw a wrench into large-scale manufacturing. I once saw a polyurethane batch scrapped because a trace contaminant from another amine shifted the reaction window, stalling a whole week of work. Anyone relying on DMCHA expects full lot traceability and clear hazard flags, and that’s what technical labels focus on: composition, purity, and safety.
From Reactants to Results: The Preparation Method
Synthesizing DMCHA usually means starting with cyclohexylamine, blending it with excess methylating agents under conditions that favor full substitution. In real terms, this translates to reactors built for temperature and pressure control, careful monitoring, and a set of cleanup steps to haul away unreacted raw materials. Over time, this process matured, lessening byproducts and boosting overall yield. Those changes matter beyond efficiency; they shape the economics and accessibility of the chemical. I’ve spoken to chemists who remember old batch methods cranking out messy, impure product. Modern methods close the loop, containing emissions and leftover reagents.
What Chemists Do With It: Common Reactions and Modifications
DMCHA tends to stay on the sidelines in terms of further modification; its tertiary amine structure resists casual alkylation or acylation. The amine acts as a solid base or nucleophile, lending itself to catalytic or auxiliary roles in organic synthesis. Sometimes researchers attach protecting groups or tweak the ring to form functionalized derivatives, but stability trumps versatility for this molecule. Where it shines is in catalysis—polyurethane chemists count on it for balanced reaction rates, while in other sectors, it can direct selectivity in more specialized pathways.
What’s in a Name? Synonyms and Aliases
Few things cause more headaches than mixed-up chemical names on supply orders. DMCHA shows up under a handful of synonyms, each tied to different vendor or regulatory contexts: N,N-Dimethylcyclohexanamine, N,N-Dimethylaminocyclohexane, and even straightforward abbreviations like DMCHA. Earlier career, I watched a shipment held up over a discrepancy in paperwork—a reminder that a clear, universal label saves time and money. Global trade in chemicals thrives on clarity, and aliases only work when paired with robust cross-referencing.
Living Up to Safety and Operational Standards
People who work in labs or plants take workplace safety seriously. DMCHA can cause strong irritation or acute toxicity if handled carelessly, so proper gear, training, and ventilation keep teams and communities safer. Regulations like REACH in Europe or EPA controls in the US have teeth. There’s no room for shortcuts here, and I’ve seen audits force even the most established operators to tighten up. Spills or leaks, though rare, draw immediate response—neutralizers at the ready, personnel trained for fast action. The chemical’s volatility and odor bring quick attention if something goes wrong, adding to its practical safety profile.
The Landscape of Application
If polyurethane production ever had a favorite catalyst, DMCHA would land near the top of the list. This amine slips easily into reaction profiles, controlling foaming and crosslinking in large foam blocks for furniture, insulation, and even automotive parts. The first time I watched a slab of foam rise on the plant floor, the role of the catalyst became crystal clear: too little or poorly chosen, and the result is weak, uneven, or riddled with defects. Beyond that, DMCHA plays a part in rubber vulcanization, coatings, and some specialty chemical synthesis. These uses add up; global demand reflects the need not just for the end products, but for reliable process chemistry.
Peering Into Research & Development
Researchers explore tweaks to DMCHA’s structure, hunting for amines with better selectivity, lower toxicity, or greener synthesis paths. Academic and industrial labs chase incremental improvements, seeking catalysts that cut cycle times or lower waste. I remember a project focused on streamlining foam chemistry—each new amine blend needed testing for both performance and regulatory compliance. Innovation often shows up as small process gains rather than sweeping breakthroughs. The slow shift towards bio-based feedstocks and sustainable amines stems from these ongoing R&D efforts, not grand gestures.
Untangling the Knot of Toxicity Research
Toxicologists look closely at amines like DMCHA, examining everything from short-term exposure to chronic impacts. Its volatility means inhalation risks top the list, and studies on skin or eye exposure yield guidelines for workplace safety. Regulatory agencies require robust data sets, pressuring manufacturers to fund animal studies and environmental tests. Beyond acute effects, long-term data help guide safe disposal practices and emissions monitoring. I’ve read reports warning about cumulative impacts near processing plants, underlining the need for environmental controls and responsible waste management.
Scanning The Horizon: Future Outlook
Looking ahead, the story of DMCHA isn’t about being replaced by some exotic molecule, but about evolving within a landscape that prizes both efficiency and sustainability. The push toward greener chemistry challenges old habits, nudging suppliers and users to rethink sourcing, energy use, and waste. Emerging polyurethane alternatives, stricter standards for air and water quality, and pressures for non-toxic catalysts point to slow but steady reinvention. My experience with process engineers and R&D teams suggests optimism is justified—incremental gains add up. DMCHA will likely stick around, though in forms or methods shaped by modern demands. And like so many chemicals before it, its value rests on the balance between robust utility and responsible stewardship.
What is N,N-Dimethylcyclohexylamine used for?
Behind Everyday Products: The Role of N,N-Dimethylcyclohexylamine
Stepping into the world of specialty chemicals often means running into names that look more at home in a science lab than in day-to-day life. N,N-Dimethylcyclohexylamine might sound technical, but this liquid finds its way into plenty of things we all use. Walk through a home improvement store, and the paints and foams lining the aisles likely owe part of their punch to this amine.
Polyurethane Foams: Insulation and Comfort
The stuff that softens couch cushions, props up car seats, and provides insulation in houses calls for some careful chemistry. Polyurethane foam production uses catalysts to kick reactions into gear. N,N-Dimethylcyclohexylamine steps in as one of those key players. Manufacturers value it for its ability to manage how fast the foam forms and to influence its texture. Get that wrong and you end up with either mushy or brittle foam, or just a plain mess on the factory floor. Reliable catalysts mean better quality for foams in pillows or insulation boards.
Adhesive Industry Applications
Water-based adhesives show up everywhere from construction projects to craft tables. The glue holding together particleboard or keeping a kitchen laminate countertop in place often uses a little help from N,N-Dimethylcyclohexylamine. By controlling how these adhesives set and how much time workers have to apply them, this compound makes large-scale assembly work easier. That time flexibility means jobs get done without the rush or the waste that comes from adhesives drying too soon.
Paints and Coatings: Cleaner Reactions, Fewer Issues
Nobody likes paint that peels early or coatings that disappoint. N,N-Dimethylcyclohexylamine helps create the right chemical environment in paints, speeding up curing and keeping finishes smooth. The coatings industry always looks for tweaks that offer better coverage and greater durability without driving up costs or loading the formula with extra chemicals. Using this amine fits the bill for many producers — better results, less trouble.
Industrial Cleaners and Gas Treating
Factories and refineries don’t stay productive if their metal surfaces and pipelines gum up with residue. N,N-Dimethylcyclohexylamine can boost the cleaning power of industrial degreasers. Refineries and power plants sometimes turn to this same chemical to help ‘scrub’ unwanted gases like hydrogen sulfide out of process streams, cutting pollution and wear-and-tear on equipment.
Safety, Stewardship, and Future Choices
Like with many chemicals, using N,N-Dimethylcyclohexylamine calls for paying attention to safety data. Direct exposure brings health risks — strong odors, irritation, toxicity at the wrong levels. Producers and industrial users carry the responsibility of training workers and keeping emissions under control. Regulations in the US and EU call for labeling, handling protocols, and proper waste treatment to protect both public health and the environment.
Experience on manufacturing floors highlights that safety steps do not just protect workers; they also keep product quality up. Routine monitoring means fewer production hiccups and less downtime due to accidental releases. The industries using N,N-Dimethylcyclohexylamine have learned to weigh the benefits with the baggage, especially as safer alternatives, cleaner processes, and greener chemistry become a louder part of the conversation.
Shifts toward sustainability echo the bigger story. As demand grows for products with fewer environmental downsides, chemical companies work on formulating replacements with lower toxicity or improved biodegradability. For now, N,N-Dimethylcyclohexylamine remains a workhorse across several sectors, its presence often unseen but vital for the quality and comfort so many expect from everyday goods.
What is the chemical formula and structure of N,N-Dimethylcyclohexylamine?
Why Chemical Structure Shapes Performance
Countless industries rely on the right blend of chemistry to move projects forward—everything from rubber detergents to pharmaceuticals. N,N-Dimethylcyclohexylamine, often shortened to DMCHA, stands out as a go-to compound for a mix of challenging tasks. Its story starts with the basics: the molecular formula and the way its atoms fit together.
Chemical Formula and the Features That Matter
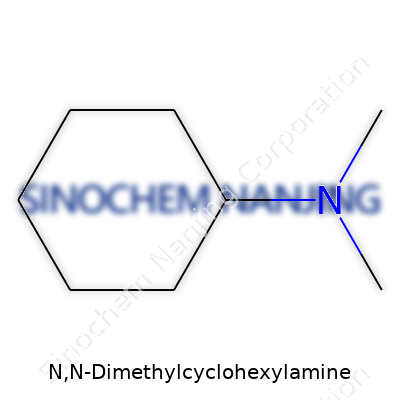
DMCHA carries the chemical formula C8H17N. That reveals a cyclohexane ring, which brings stability and backbone to the molecule. By swapping two hydrogen atoms for two methyl groups attached to a nitrogen atom, the structure gains effciency and reactivity. The basic skeleton places a nitrogen attached directly to a cyclohexane ring, with two methyl groups sticking out from the nitrogen. Looking at the two-dimensional structure, it appears as:
Cyclohexyl group (a six-carbon ring)—linked to a nitrogen atom—carrying two methyl (CH3) side chains. In chemical terms, you’ll see this written out as (CH3)2NC6H11. If you like visualizing molecules, imagine the ring as a sturdy wheel with a small propeller: the nitrogen as the hub, the two methyl "blades" pointing off, influencing the molecule's behavior in reactions.
The Role of Structure in Real Work
Structure often guides what a compound can actually do. For DMCHA, the cyclohexyl ring doesn’t just offer bulk. It brings a serum of hydrophobic character, meaning it sheds water easily and likes to nestle in with organic materials. This property helps in making foams or acting as a catalyst where controlling moisture is key. The two methyl groups stuck onto the nitrogen aren’t just hanging around, either. In many reactions, they block out acids or unwanted byproducts, giving chemists better control over how fast— and how cleanly— a reaction goes.
Applications Driven by Chemical Identity
DMCHA finds its place in polyurethane foam production, industrial catalysts, and corrosion inhibitors. Its molecular design means it can persist under tough conditions, like those found in high temperature or aggressive chemical environments. These features are not just chemical trivia—they direct everything from the shelf stability of a cleaning product to the way a foam expands. I’ve seen teams in the field rely on DMCHA to keep operations smooth in casting resins, thanks to its ability to adjust curing times without unpleasant odors or tricky residue.
Addressing Safety and Environmental Points
The chemistry behind DMCHA brings up real-world concerns about exposure and environmental persistence. Since this amine acts as a strong base, it causes irritation if handled casually. Factories handling DMCHA often invest in targeted extraction ventilation and glove protocols to protect workers. Disposal practices call for care—catching leaks quickly and ensuring local water sources don’t get contaminated. Research over recent decades highlighted risks tied to uncontrolled amine release into the environment, which triggered tighter workplace standards and better monitoring.
How Industry Responds
Companies take steps to reduce risks around DMCHA by switching to closed-loop systems and improved personal protective equipment. On the environmental side, efforts include working with specialized waste handlers and designing less persistent substitutes that break down more easily. It still gets used for its reliability and effectiveness, but awareness around safe practice continues to shape how it's stored and transported.
Is N,N-Dimethylcyclohexylamine hazardous or toxic?
Understanding the Chemical
N,N-Dimethylcyclohexylamine is a mouthful to pronounce, but it turns up in way more places than most might think. This chemical serves as a catalyst in polyurethane foam production, which is common in furniture, mattresses, car seats, and insulation. From the start, it carries an ammonia-like odor, and a small spill can clear out a room pretty fast. Folks handling it have learned through experience that you never want to get careless around this stuff. Honest mistakes can lead to headaches, nausea, and stinging eyes. Signs like that set off alarm bells for anyone who works with chemicals on a regular basis.
Exposure Brings Real Risks
Most who’ve spent time in labs or factories learn that direct contact with N,N-Dimethylcyclohexylamine irritates skin and eyes. Even light exposure can cause a burning sensation in the throat or coughing fits, so it’s tough to ignore if safety gear slips for even a second. The fumes alone can overwhelm, especially in rooms with poor ventilation. There’s a temptation to assume all workplace smells are harmless, but this chemical proves that taking shortcuts leads to trouble down the line.
Looking at what scientists already know, this chemical can harm the liver and kidneys in animals exposed to higher concentrations for just a short time. Over periods lasting weeks, studies have shown organ weight changes and enzyme shifts, both signs that the body’s defenses take a beating. There isn’t strong evidence linking this chemical to cancer in people. Still, the fact it can make someone seriously sick in small amounts speaks volumes about just how cautious workers must act.
Protecting Workers
Precautions shape daily life in environments filled with N,N-Dimethylcyclohexylamine. Gloves, splash-proof goggles, face shields, and effective fume hoods aren’t optional; they’re essential. The best managers I’ve ever seen make sure workers not only have the right gear but also know how to use it. Training that sticks in your head after 10-hour days does more good than lectures or pamphlets. There’s also no substitute for regular air testing to keep an eye on levels in the factory or lab. High readings mean fans need to run harder, or activities need to pause until the air clears out.
How Companies Limit the Damage
Emergency plans must be more than a binder on a shelf. Spills get neutralized and cleaned quickly, with fast evacuation if vapor fills the air. Personal stories from seasoned workers show that ignoring a leak, even for a few minutes, carries real cost. Managers who enforce rules and actually walk the floor have a bigger impact than endless reminders posted near the time clock. Companies switching to safer alternatives wherever possible not only protect staff but also avoid the fines and lawsuits that come when shortcuts backfire.
The Push for Safer Substitutes
Green chemistry has come a long way in recent years, opening the door for less toxic catalysts in foam production. Many groups research and develop substitutes that break down more easily and demand less personal protection. Whenever leaders move the industry toward safer compounds, that change trickles down—it means less risk for the newest trainee, the long-timer ready to retire, even the customer who never sets foot in a factory.
Everyday Awareness Matters
People outside the industry rarely hear about N,N-Dimethylcyclohexylamine, yet millions trust objects that once depended on it. Clean air and healthy organs aren't negotiable costs for the sake of a comfortable couch. Thorough training, strict handling, and a constant search for safer solutions all count as real investment. Lessons learned by those who sweat through chemical workdays serve as a reminder: behind every manufactured comfort, safety rules draw the line between convenience and harm.
How should N,N-Dimethylcyclohexylamine be stored and handled safely?
Why This Chemical Draws Attention
Anyone working around chemicals learns quickly that some demand more respect than others. N,N-Dimethylcyclohexylamine falls into that group. This amine offers industry and research labs some particularly useful properties, but its strong smell, volatility, and ability to irritate skin, eyes, and lungs means a casual approach can backfire. Countless labs have gotten burned—sometimes literally—by cutting corners with storage or handling.
The Risks Speak Louder Than Labels
It helps to call things what they are. Splashing this liquid on bare skin leads to inflammation—redness, itching, sometimes even blistering. Breathing its vapor too long triggers headaches, dizziness, or worse. From fires to lung injury, one mistake can ripple through a lab, shutting down work for days. After seeing colleagues rushed to the eyewash station or flushing their faces after a spill, nobody forgets to check their goggles again.
Storage: Thinking Beyond the Safety Sheet
A shelf in direct sunlight or next to a heat source turns N,N-Dimethylcyclohexylamine into a ticking clock. Its low flash point and tendency to create vapors mean a locked flammables cabinet with clear, undamaged labeling wins every time. Choosing containers out of glass or metal, with caps that seal tightly, helps keep the material from leaking odors into a workspace and limits evaporation. Any sign that a bottle has warped or the lid doesn’t fit spells trouble. Swapping out aging gear for new pays off.
Anyone who’s dealt with leaky shelves or overflowing cabinets knows that simplicity beats overcrowded storage. Grouping this amine with other non-reactive organics—never with acids or oxidizers—keeps accidents rare. I’ve seen entire benches cleared because a minor bottle spill mixed with acid storage close by. Nothing brings home the importance of chemical compatibility like a near miss.
Handling: Building Good Habits
Labs run best when everyone sticks to their routine: gloves, goggles, lab coat, and, in tight spaces, splash aprons. That thick chemical-odor mask isn’t an accessory—it’s the line between a regular shift and a trip to the clinic. I remember one project where an unplanned cleanup sent three of us home early because someone grabbed the wrong gloves. Nitrile or neoprene holds up to this amine, standard latex never does.
Pouring and pipetting go over spill trays—not over open benches. Even small drops stain surfaces or drip onto sleeves. Squeeze bottles with controlled flow work better here than wide-mouth containers. Everyone who’s had to scrub crusted amine off glassware learns to double-check lids and pressure before starting a task.
Good ventilation counts. I’ve worked in spaces with and without working fume hoods, the difference is night and day. With this chemical, air flow should sweep vapors away from breathing zones. It’s worth checking that vents and fans run regularly, not just sitting as part of the décor.
What Actually Solves Problems
I’ve learned the hard way that written checklists—hanging right next to the cabinet—cut down on mistakes. Regular training and unambiguous labeling go further than quarterly safety talks. Highlighting spill kits and emergency washes in clear view means even new staff find them without delay. Labs that keep a tight log of chemical use see fewer incidents. Real responsibility—more than policies on paper—forms the backbone of safe work.
This is more than just box-ticking. It’s about seeing safety walk hand in hand with productivity. Every story of averted disaster or smooth clean-up adds up, shaping experienced chemists and careful students alike.
What are the typical applications or industries for N,N-Dimethylcyclohexylamine?
Everyday Items Rely on Chemical Building Blocks
Many people rarely think about the chemicals shaping daily life, yet some ingredients work quietly behind the scenes. N,N-Dimethylcyclohexylamine doesn’t ring a bell for most, but it plays a big part in products used and trusted across homes, workplaces, and entire industries.
The Role in Polyurethane Foams
Walk through any furniture store or auto showroom, and chances are you’ll bump into a couch or a car seat made with polyurethane foam. This chemical acts as a strong catalyst during foam production. It helps the process move along fast, giving cushion makers flexibility and control over how the material turns out. From mattresses and insulation panels to the headrests in cars, the feel and durability people enjoy trace back to this small but vital ingredient.
After working in a warehouse that shipped mattresses, I noticed how quality and comfort shifted even when suppliers tuned their formulas just a little. Consistency depends on strong catalysts, and N,N-Dimethylcyclohexylamine often sets the standard.
Industrial Cleaners & Electronics
Offices, factories, hospitals: all need greasy parts and smudged electronics cleaned safely. Many industrial and institutional cleaners use this amine in their formulas because it tackles tough residues without harsh side effects like rapid corrosion. Tech firms keep electronics humming with circuit boards scrubbed clean by solvents containing it. Its stability matters, especially when cleaning tasks can’t tolerate residue or static.
Techs in electronics repair know that leftover grime or flux can cause shorts and failures. Reliable cleaning agents make sure devices don’t break down before their time. Trusted chemicals keep things simple and safe.
Pesticides, Rubber, and Dye Making
Working behind the fields and factories, N,N-Dimethylcyclohexylamine supports global agriculture. Crop protectant makers use it as an intermediate for building pesticides. That helps fight insects and fungus, defending yields so farmers feed more families.
In the rubber world, this ingredient helps speed up vulcanization, the process that gives car tires and conveyor belts their bounce and strength. Dye producers turn to it as a step in synthesizing complex colors found in paints and textiles. From bright clothing to truck tires, few realize how base chemicals lay the foundation for everyday innovation.
Health, Environment, and Smarter Choices
No chemical comes without responsibility. Exposure to N,N-Dimethylcyclohexylamine can bring eye and skin irritation, so workers need gloves and goggles. Tough regulations protect workers and keep pollution in check. Facilities must track and manage emissions, making sure the material stays contained.
Safer alternatives keep popping up as companies look for greener solutions. Some labs now research catalysts with lower toxicity or better environmental breakdown. Sharing these findings helps protect both people and the planet.
Looking Forward
Cleaner production in the foam and chemical sectors depends on smart oversight, tighter supplier checks, and investments in training. I’ve seen improvements in safety with better ventilation and digital tracking systems. Open dialogue, sharing best practices, and taking the initiative all help build trust in the products customers bring into their homes.
| Names | |
| Preferred IUPAC name | N,N-dimethylcyclohexanamine |
| Other names |
N,N-Dimethylcyclohexylamine N,N-Dimethylcyclohexanamine N-Methylcyclohexylamine Dimethylcyclohexylamine N,N-Dimethyl-1-cyclohexanamine DMCHA |
| Pronunciation | /ˌdiːˈmɛθɪlˌsaɪ.kloʊˈhɛk.sɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 98-94-2 |
| Beilstein Reference | 1209249 |
| ChEBI | CHEBI:51904 |
| ChEMBL | CHEMBL15909 |
| ChemSpider | 7401 |
| DrugBank | DB11290 |
| ECHA InfoCard | 03b7b412-028d-4d1e-b97f-203315b506d9 |
| EC Number | 203-679-0 |
| Gmelin Reference | 8227 |
| KEGG | C06368 |
| MeSH | D03.438.505.519.425 |
| PubChem CID | 11815 |
| RTECS number | GU6475000 |
| UNII | X4W7ZR7023 |
| UN number | UN2264 |
| CompTox Dashboard (EPA) | urn:uuid:06f02d20-2745-42c9-9a6c-65fdc5709c87 |
| Properties | |
| Chemical formula | C8H17N |
| Molar mass | 155.28 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Aminelike |
| Density | 0.86 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 1.87 |
| Vapor pressure | 3.8 mmHg (20°C) |
| Acidity (pKa) | 10.68 |
| Basicity (pKb) | pKb = 3.27 |
| Magnetic susceptibility (χ) | -7.27 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.443 |
| Viscosity | 0.837 cP (25°C) |
| Dipole moment | 1.25 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −138.1 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4046.6 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H311, H314, H331 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P304+P340, P305+P351+P338, P312, P330, P337+P313, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-0 |
| Flash point | 58°C |
| Autoignition temperature | 215°C |
| Explosive limits | 1.1% - 8.4% |
| Lethal dose or concentration | LD50 (oral, rat): 323 mg/kg |
| LD50 (median dose) | LD50 (median dose): 279 mg/kg (oral, rat) |
| NIOSH | SKC40250 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.6 mg/m3 |
| IDLH (Immediate danger) | 60 ppm |
| Related compounds | |
| Related compounds |
N-Methylcyclohexylamine Cyclohexylamine N,N-Dimethylaniline Dimethylamine |

