The Many Sides of N,N-Dimethylbenzylamine
Tracing the Roots: Past to Present
N,N-Dimethylbenzylamine, found its way into the chemical industry during the big push for specialty amines in the early 20th century. Chemists realized that swapping out hydrogen atoms on an amine for methyl groups gave a boost to volatility, solubility in organic solvents, and sometimes reactivity in synthesis. Folks who spent long months in bench labs, like myself, remember prepping derivatives of this compound to unlock new applications or improve old ones. If you ever thought about how certain resins cure faster or why paints from decades ago started lasting longer outdoors, you bumped into the legacy of materials that contained or depended on amines like this.
What Are We Really Working With?
At first glance, N,N-Dimethylbenzylamine looks simple—a colorless to pale yellow liquid with a faint amine smell. It slips easily into organic liquids and doesn’t hang around in water for long. That characteristic alone paved its way into countless formulations. Its boiling point hovers around where a stovetop simmers, making it easy for folks in factories to distill and handle at scale. From a reactivity standpoint, the molecule balances basicity and nucleophilicity, which my colleagues in organic synthesis loved. We used to joke about its ‘just right’ power: strong enough to shift reactions forward, mild enough to avoid runaway conditions or off-target messes.
Technical Details and the Story Their Numbers Tell
Manufacturers who ship N,N-Dimethylbenzylamine in drums take care with purity, moisture level, and color. Purity can swing the entire output of a batch process off course, especially in fine chemicals, so labs keep a watchful eye with NMR, GC, and other quality tests. If you ever endured the ritual of checking technical sheets for residual acid or amine numbers, you’ll know why technical standards matter. Chemical companies print CAS numbers and other tags on labels, but those only scratch the surface. Behind every label is a silent agreement among suppliers, buyers, and regulators—keep it pure, keep it safe, and the rest will take care of itself.
How the Chemistry Gets Done
There’s nothing magical about the process for making this compound, but sometimes chemistry just works. Putting benzyl chloride and dimethylamine together under the right conditions reliably churns out N,N-Dimethylbenzylamine. Side products lurk in the shadows, so folks in plant operations spend endless hours fine-tuning temperatures, timer settings, and workups to chase unwanted byproducts out of the final mix. After the main reaction, it’s a matter of stripping out excess reactants and unwanted water, so the finished material meets the demands of the next chemist in line.
What It Gets Called and Why That Matters
Walk through a catalog and you’ll find synonyms by the handful: Benzyl dimethylamine, BDMA, or even N,N-DMBA. If you’ve ever trudged through regulatory paperwork or tried to match an order number from a customer with your own inventory, you’ll know the headache. Having three or four names for the same material sounds harmless until you accidentally get the wrong substance, which every chemist dreads. Names make a difference—not just for clarity, but for legal and safety reasons.
Guidelines: Getting Safety Right and Building Trust
Safety in chemical handling evolves and hard lessons from real-world incidents have shaped the standards around N,N-Dimethylbenzylamine. It wasn’t always this way. There were decades when gloves, glasses, and even ventilation were optional or left to individual judgment. These days, proper procedures and training help everyone from seasoned plant staff to new lab techs avoid inhalation or skin contact problems. For an old hand who spent years with amine vapors in the air, reminders about working with a fume hood or changing gloves on time matter. I’ll never forget a mentor’s motto: “Respect the label, or regret the spill.”
From Industry Floors to the Modern Lab
This amine keeps showing up in familiar and unexpected places. Right now, it plays a big part in making polyurethane foams for furniture, car seats, and insulation. It helps resins cure faster during coatings production, and it acts as a gentle but reliable base in many organic syntheses, letting pharma chemists and materials scientists push the limits of their fields. I’ve watched as environmental chemists track where it might go after use and how it breaks down—questions that stay front of mind as regulations tighten and the push for greener chemistry gains steam.
Curiosity Drives the Industry Forward
Researchers don’t stand still on a compound once they understand its basic properties. Over the last decade, labs dug into ways to tweak the structure, hoping to get new catalysts, better curing agents, and even smart materials with self-healing properties. Graduate students in universities and lab techs in R&D centers test how changing the amine’s backbone or adding groups can turn a well-known workhorse into a specialty tool. Anyone who’s worked on process innovation knows the frustration—and the excitement—of pulling something new out of a familiar molecule. Progress here depends on curiosity, close partnerships between industry and academia, and grit.
Keeping an Eye on Toxicity and Environmental Impact
Science runs on evidence, so health and safety assessments never rest. Decades ago, little was known about the dangers that N,N-Dimethylbenzylamine posed to workers, let alone to the outside environment. Modern toxicology has made headway, with studies on inhalation and skin exposure, acute and chronic effects, and environmental breakdown. From my own time reviewing safety data sheets and following incident reports, I learned not to take amine safety lightly. Regulatory reviews turn up possible long-term complications, urging users to adopt proper storage, ventilation, and waste disposal protocols. With tightening requirements around chemical safety in the workplace and the environment, companies must adapt quicker and report more often than ever.
Where Things Could Head Next
I see the conversation about N,N-Dimethylbenzylamine shifting toward sustainability. As industries search for lower-impact chemistries, the spotlight is turning to biodegradable alternatives, more selective catalysts, and solventless preparation methods. Researchers have their work cut out for them to keep performance high while chipping away at toxicity and waste. At the same time, regulatory agencies are pressing for transparency, lifecycle assessments, and open reporting—trends that any supplier or user ignores at their peril. Those of us in the field realize that the basic science of the molecule isn’t done evolving yet, and with time, new applications and risks will become clear. Until then, experience, vigilance, and a willingness to learn from mistakes will shape how the industry handles this reliable but ever-changing chemical.
What is N,N-Dimethylbenzylamine used for?
Understanding the Chemical
N,N-Dimethylbenzylamine doesn’t turn heads unless you’re deep into chemistry or industrial manufacturing. This chemical acts like a workhorse behind the scenes. Its clear, liquid form packs a sharp smell, reminding anyone who’s worked near it about the potency tucked away inside those simple molecules.
Real-Life Uses in Manufacturing
This isn’t a chemical people store under their kitchen sink. Factories use N,N-Dimethylbenzylamine mainly for making polyurethane foams. These foams fill cushions, car seats, insulation—things most of us use every single day without thinking about their chemical origins. The reason is simple: this amine speeds reactions and helps control exactly how those foams set and expand. Fast curing matters on an assembly line, where time means money, and small tweaks in chemistry make the difference between stiff, lumpy foam and the smooth, bouncy type we expect in a new mattress or office chair.
Why It Matters for Product Quality
Poorly cured foams bring real problems. Maybe you’ve sat in a car seat that feels uneven, or a couch cushion that sags too quickly. Behind those letdowns sits a production issue, often tied to catalysts. N,N-Dimethylbenzylamine helps manufacturers get ahead of those issues by offering fine control during the mixing stage. With just the right dose, foam becomes sturdier, more supportive, and longer-lasting. Attention to these details doesn’t just save money—it shapes the way people experience everyday products.
Not Just Polyurethanes
The journey doesn’t stop with foam. Paint makers also turn to this amine when they want coatings to dry at just the right speed. Ink and adhesive producers value it for similar reasons. Artists rarely think about chemicals like this, but their tools and supplies often depend on such ingredients to deliver predictable results. I’ve spoken with builders and renovation workers who swear by fast-drying coatings, especially when schedules run tight. N,N-Dimethylbenzylamine makes their lives easier—speeding up drying, toughening up surfaces, shrinking downtime between coats or repairs.
Points of Caution
The stuff shouldn’t be handled carelessly. Anyone involved in production needs proper training and protective gear. Long-term exposure carries health risks, especially for workers on the floor mixing these chemicals daily. I’ve seen industrial hygiene teams spend hours planning safe storage and air handling just to keep everyone healthy. It’s worth remembering, chemicals that drive progress should be partnered with solid safety protocols. Good gloves, goggles, fume hoods—absolutely essential. Factory managers who cut corners only invite trouble.
The Road Ahead: Finding Alternatives?
While performance matters, there’s growing interest in greener, safer alternatives. Regulations around industrial solvents tighten every year, nudging companies toward innovation. Labs keep looking for catalysts that give the same reliable curing with fewer environmental or health concerns. Transition isn’t easy or cheap, but I’ve seen how consumer demand for safer products can drive real change. If new formulations arrive that match the effectiveness of N,N-Dimethylbenzylamine without the headaches, industry will adopt them quickly. Until then, this chemical remains part of the backbone in manufacturing many daily goods.
What is the chemical formula of N,N-Dimethylbenzylamine?
Peeling Back the Science
N,N-Dimethylbenzylamine pops up in more chemistry conversations than most folks expect. This compound has the chemical formula C9H13N. It stands out in both labs and industry due to its mix of practicality and versatility. You can spot its building blocks just from the name: a benzyl group tacked onto a nitrogen that carries two methyl groups. In short, it blends the backbone of a benzene ring with a bit of amine flair. If you draw it all out, you get a six-carbon aromatic ring connected to a CH2 group, which then links up to a nitrogen that’s holding two CH3 groups.
A Closer Look at the Structure
The formula C9H13N isn’t just a string of numbers and letters—it tells the story of each atom’s place in the bigger picture. Walk through a typical undergraduate organic lab, and you might see N,N-Dimethylbenzylamine used as a solvent or as a base that helps reactions run smoother. It owes its power to the nitrogen atom, lending a touch of electron-rich character. That nitrogen doesn’t just sit quietly; in fact, the two methyl groups make it more basic and sterically protected than simple benzylamine. This chemistry echoes out in other fields where catalysts and intermediate compounds drive innovation.
Economic and Practical Impact
It matters quite a bit to recognize what C9H13N represents. In manufacturing—think rubber accelerators, pharmaceuticals, and resins—N,N-Dimethylbenzylamine avoids unnecessary drama in processing. Its chemical shape brings stability, so fewer unwanted side products form, which leads to less waste and less money spent cleaning up afterwards. Those savings stretch beyond just factories; they can make everyday items like coatings, foams, and adhesives more reliable.
Where Safety and Knowledge Intersect
Handling any amine, including this one, means a strong focus on safety. Exposure to N,N-Dimethylbenzylamine demands that folks know what they’re working with. Lab safety training becomes more than a routine; material safety data sheets guide the right ventilation and handling, since vapor can irritate eyes, skin, and the respiratory system. People—workers, teachers, students—need straightforward information. That’s more than just looking up a formula in a textbook; it’s about building habits that respect both the science and the risks.
Building a Smarter Approach
Full understanding of the chemical formula serves more than curiosity. Teachers break down these formulas in school labs, showing students how aromatic rings and substituents change reactivity and shape outcomes. Researchers map out how these tweaks affect catalyst performance or pharmaceutical effectiveness. Industry uses this information to make decisions about sourcing, scaling production, and minimizing environmental impact.
Paving the Way for Better Practices
Paying close attention to the formula, structure, and properties of N,N-Dimethylbenzylamine helps ensure better choices throughout its lifecycle. Safer handling, smarter purchasing, and clearer communication about its chemical behavior can limit the reach of accidents, reduce costs, and even spark new uses. My own experience working on a team tasked with reducing harmful emissions taught me that these details—down to each atom in a formula—carry real weight. It’s not just abstract chemistry; it’s part of daily life for anyone in the industry, all the way down to the consumer using a finished product.
What are the storage recommendations for N,N-Dimethylbenzylamine?
Why Safe Storage Matters
N,N-Dimethylbenzylamine pops up in many chemical processes, from making rubbers to helping cure epoxy resins. Years working near this compound taught me that storage isn’t just a checklist item. It’s about keeping everyone in the building healthy and projects on track. Even small leaks or sloppy practices lead to headaches—literally, if you breathe it in—and piles of paperwork if spills reach drains or open air. Regulations like OSHA and REACH spell out a lot, but working with this amine every day made me realize that guidelines stick when the logic behind them is clear.
Practical Conditions for Storage
N,N-Dimethylbenzylamine stays stable in cool, dry places. High heat speeds up reactions. Humidity introduces water, which doesn’t mix well with this compound. In one summer, I saw a bottle sweating on a warehouse shelf during a heat wave—it gave off fumes that set off the ventilation alarms. After that, our crew agreed on a few non-negotiables. Storage rooms should stay below 25°C. Humidity stays under 60%. This keeps the chemical from breaking down and stops rust on metal containers.
Direct sunlight breaks down amines faster. We moved inventory away from windows and used solid doors. Storing raw materials in opaque drums or brown bottles helped control light exposure. Extra expense up front saved more in wasted reagents.
Handling Compatibility
Mixing incompatible chemicals caused big trouble in my early years on the job. N,N-Dimethylbenzylamine reacts with acids, making harsh fumes. We kept acids and oxidizers far away on dedicated racks. Spill trays underneath containers caught drips, since even drops gave off strong odors that lingered in the warehouse. Using clear color codes on storage areas—blue for bases and amines, red for acids—reduced mistakes, especially for new staff.
Ventilation and Containment
This amine smells strong at low concentrations. Unventilated rooms made opening a drum harsh on the lungs even through an N95. I once saw a coworker ignore fume hoods, and the next hour was spent recovering from a pounding headache. Key lesson: good air flow matters as much as temperature. Industrial fans, vented shelves, and regular air monitoring keep concentrations low.
Spills or leaks threaten both workers and building structure. We switched from makeshift shelving to chemical-resistant shelving with raised lips. It sounds like nitpicking, but it played a huge role during one big leak—drips stayed contained instead of running down along the floor. Polyethylene secondary containers provided a good backup if a glass or metal main bottle failed.
Container Choice and Labeling
We switched to HDPE and stainless steel after problems with corrosion in older cans. New containers resisted swelling or breaking down. Clear hazard labels and up-to-date inventory sheets helped responders during a fire drill or incident. In a real emergency, quick access to safety info convinced first responders to stay calm and act fast.
Personal protective gear completed the system—chemical-resistant gloves, goggles, and lab coats stopped splashes from becoming chemical burns.
Routine Checks and Continuous Improvement
Success with chemical storage doesn’t rest on initial setup. We checked room temperature, container integrity, and ventilation systems each month. In one case, a cracked cap went unnoticed until a routine check caught it; fixing the small problem early averted a major spill. Training refreshers every six months made rules stick even as teams shifted or new products entered storage.
Storing N,N-Dimethylbenzylamine isn’t just about what the shelf looks like—it's about vigilance, good habits, and knowing why every safety step matters. Clear procedures, thoughtful setups, and shared experience keep the risks low and the workflow smooth.
Is N,N-Dimethylbenzylamine hazardous or toxic?
Understanding What We’re Dealing With
N,N-Dimethylbenzylamine might sound like something any of us could skip right over on a product label or safety sheet. Most folks working in labs, factories, or even small workshops have come across chemicals with unfamiliar names, and this one fits the bill. Used as a catalyst in making polyurethane foams or as an intermediate in various industrial processes, its presence might feel routine. But before grabbing that bottle or firing up production, it’s essential to look closer at what this substance can do to a person's health, coworkers, and the environment.
Health Concerns Deserve Attention
Skimming the safety data on N,N-Dimethylbenzylamine, the first thing that jumps out is the risk with direct contact. Skin rashes, eye irritation, and that unmistakable chemical smell should give anyone pause. Breathing in its vapors can irritate the respiratory system — not just a sneeze and move on, but coughing, wheezing, even headaches. Getting this chemical in your eyes burns and can damage vision if not washed out quickly.Some folks shrug off these warnings, saying, “It’s just a little exposure.” That mindset gets people into trouble, whether they’re in the lab or out on the shop floor. Repeated contact and poor ventilation set the stage for more severe health problems over time. Studies show prolonged inhalation in animals triggers liver and kidney damage. While direct proof in humans may not always be clear, it’s not worth rolling the dice when safer handling practices exist.
Environmental Footprint
Letting N,N-Dimethylbenzylamine escape into the drain or air does more than clear out a lunchroom. Studies demonstrate that it’s not friendly to aquatic life. Fish, insects, even algae can suffer if this chemical shows up in waterways. That means cleaning up is about more than just keeping inspectors happy — it protects local ecosystems where people fish, swim, or just take their kids to see frogs. I’ve seen how tiny leaks in one part of a process eventually show up miles downstream, so it pays to take cleanup and disposal seriously.
No Room for Complacency
I’ve worked in places where the “it won’t happen to me” attitude lingers until an accident shakes up the whole team. Gloves and goggles gather dust, warnings become background noise, and chemicals get handled with bare hands. That’s the dangerous part about familiarity; it tricks us into thinking that because something’s always around, it’s safe. Regulations from agencies like OSHA and the EPA don’t exist for paperwork’s sake. They’re there because people paid with their health, sometimes their lives, for shortcuts that never paid off.Proper storage, working fume hoods, clear labeling, and regular training save headaches and stop emergencies before they start. It’s not just about ticking boxes. Every worker, manager, or even small business owner dealing with this stuff should remember: there’s no substitute for caution.
Finding a Better Way Forward
Engineering teams and safety officers focus on substitutions wherever possible. Swapping N,N-Dimethylbenzylamine out for less toxic alternatives cuts the risk before it even makes it onto the shop floor. If switching isn’t possible, using closed systems and automating where possible brings exposure down. At a grassroots level, open conversations between employees and management about concerns mean problems get solved together instead of staying hidden.With the right respect for the hazards of N,N-Dimethylbenzylamine and a little common sense, it’s possible to keep the workplace — and the wider world — safer for everyone.
What are the common applications of N,N-Dimethylbenzylamine in industry?
The Quiet Catalyst in Polyurethane Foam
Growing up near a mattress factory, the smell of chemical curing filled the air on summer mornings. Back then, I had no clue that part of what helped those foams set up so reliably was N,N-Dimethylbenzylamine. Countless beds, car seats, and insulation boards depend on this substance. In polyurethane production, this amine acts as a catalyst, speeding up the reaction between isocyanates and polyols. Factories want speedy, reproducible cures, and just a small amount of N,N-Dimethylbenzylamine keeps lines moving at pace. With production output depending on every minute, companies trust it for consistency in foam properties and ease of scaling up batches.
Essential for Epoxy Resins and Adhesives
Anyone who’s ever repaired a tabletop with an epoxy kit has encountered this compound, whether they realize it or not. In adhesives and epoxy coatings, N,N-Dimethylbenzylamine helps jump-start the hardening process. Commercial manufacturers rely on this timing control. Imagine projects stalling in construction, or large-scale flooring never setting up right. Adding the amine ensures faster turnaround and strong bonds. It’s not about using more, but about using the right ingredient at the right time for a reliable outcome.
Fine Chemicals and Pharma: Steering Reactions
Specialty chemicals fall under the radar compared to big batches of foam or glue. In labs and plants, N,N-Dimethylbenzylamine plays a role as a base or promoter. With pharmaceuticals, small details in quality and yield turn into huge differences in supply chains. Professionals in this field want predictable, clean chemical reactions, and this amine makes the recipe work for fine chemical synthesis and some drugs. It brings out the best results not just by itself, but as part of a broader formula that raises output and lowers unwanted byproducts.
Making Synthetic Rubber and Coatings Work
On the street, tires and shoe soles seem simple, but their resilience owes a debt to chemicals like N,N-Dimethylbenzylamine. Rubber processing uses it to control polymerization. Manufacturers change mixing windows with surprising precision, so racetrack tires and kids’ rain boots both hit the right feel. In paints and coatings, the same amine gives fast cure rates and a strong finish. Vehicle bodies, metal beams, and even wind turbines often rely on coatings prepped with catalysts like this for long-term toughness.
Safety and Sustainability Considerations
Safe handling counts for more than efficiency. N,N-Dimethylbenzylamine has a strong odor, and exposure calls for good ventilation and proper protection. Companies monitor use under strict standards and turn to closed systems to cut risk. As industries look for greener options, there’s growing research into alternative catalysts. Still, efficiency gains and lower overall consumption mean many manufacturers stick with this amine until drop-in replacements can offer the same blend of speed, safety, and cost.
Looking Ahead: Room for Improvement
I’ve talked to people behind factory doors who want cleaner air, fewer emissions, and less hazardous waste. Pushing for safer processes—like using “intelligent” dosing systems or choosing less toxic co-catalysts—promises better workplaces and products. While N,N-Dimethylbenzylamine keeps industry running today, pressure mounts to find options that blend speed with sustainability. That’s the real test: delivering the reliability the market expects without sacrificing the health of workers or the environment.
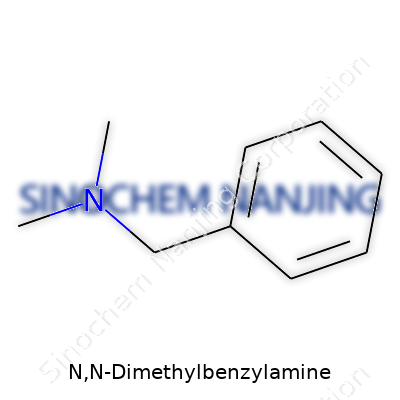
| Names | |
| Preferred IUPAC name | N,N-dimethyl-1-phenylmethanamine |
| Other names |
Benzyldimethylamine N-Benzyl-N,N-dimethylamine BDMA |
| Pronunciation | /ˌdiːˈmɛθɪlˈbɛn.zɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 121-72-2 |
| Beilstein Reference | 1322313 |
| ChEBI | CHEBI:52084 |
| ChEMBL | CHEMBL1439 |
| ChemSpider | 8456 |
| DrugBank | DB03755 |
| ECHA InfoCard | 100.011.326 |
| EC Number | 204-709-8 |
| Gmelin Reference | 8187 |
| KEGG | C06216 |
| MeSH | D003911 |
| PubChem CID | 7499 |
| RTECS number | PY7175000 |
| UNII | EC065L1MPI |
| UN number | UN2619 |
| CompTox Dashboard (EPA) | DTXSID2020397 |
| Properties | |
| Chemical formula | C9H13N |
| Molar mass | 135.21 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | ammonia-like |
| Density | 0.88 g/cm3 |
| Solubility in water | soluble |
| log P | 1.95 |
| Vapor pressure | 0.5 mmHg (20°C) |
| Acidity (pKa) | 9.26 |
| Basicity (pKb) | pKb = 4.19 |
| Magnetic susceptibility (χ) | -62.58·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.530 |
| Viscosity | 2.04 mPa·s (25 °C) |
| Dipole moment | 1.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 352.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -10.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4147.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02, GHS07 |
| Signal word | Warning |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P312 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | 58 °C |
| Autoignition temperature | 234 °C (453 °F; 507 K) |
| Explosive limits | Explosive limits: 1.0–7% |
| Lethal dose or concentration | LD50 (oral, rat): 1230 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 528 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Benzylamine N-Methylbenzylamine N,N-Diethylbenzylamine N,N-Dimethylaniline |

