N,N-Diisopropylethanolamine: More Than a Mouthful—A Deep Dive Into a Quiet Workhorse of Modern Chemistry
Historical Development
If someone asked me to name unsung heroes in the chemical world, N,N-Diisopropylethanolamine would make that list. It was first introduced to industrial chemistry circles several decades ago, emerging from a wave of interest in specialized amines. This period saw researchers looking beyond basic ethanolamines, striving for molecules with tweaked properties that could fill gaps in manufacturing and research. N,N-Diisopropylethanolamine stepped onto the scene, not with fanfare, but with a quiet usefulness. Back then, companies and labs dug deep into the structure-activity relationships of ethanolamine derivatives, and this particular compound provided new options for chemical engineers who wanted selective reactivity and tamper-resistant formulations in their processes.
Product Overview & Application Area
What keeps bringing N,N-Diisopropylethanolamine up in technical discussions isn’t flashy marketing but a set of characteristics that matter where practicality and repeatability count. It bridges organic and industrial chemistry, serving as a building block, a neutralizing agent, and a corrosion inhibitor. People have used it in fields ranging from gas sweetening (removing acidic gases in natural gas processing) to polyurethane catalysts and basic chemical syntheses. The compound’s tertiary amine structure has drawn attention for selectivity in alkylation reactions and as a masking agent during synthesis, which is something bench chemists appreciate when they want tight control over their reaction conditions. Researchers working in fine chemicals, resins, and even in some pharmaceutical intermediates will tell stories of how this one molecule solved problems that simpler amines could not touch.
Physical & Chemical Properties
This chemical manages to be both intriguing and predictable: a clear, colorless-to-light yellow liquid at room temperature, with a mild amine odor that is not as harsh as some related substances. Its boiling point can stretch above 200°C, which brings stability in high-temperature applications, and its miscibility with water opens doors for aqueous processing. N,N-Diisopropylethanolamine’s molecular weight and branching create a unique blend of alkaline strength and steric hindrance. That combination gets it chosen where less hindered amines falter due to over-reactivity or instability. With a basicity higher than monoethanolamine but lower than diethanolamine, it proves adaptable to tasks where pH control or selective reaction targeting matters.
Technical Specifications & Labeling
Production-grade material is delivered with attention to purity, color, water content, and amine number. Most suppliers focus on these factors, because a shift in purity brings risks during downstream synthesis. Storage instructions lean toward cool, dry environments, since prolonged exposure to air can result in degradation. My experience in the lab has shown that spilled N,N-Diisopropylethanolamine reeks more sharply when left open, and improper handling leads to coloration and off-odors. Clear hazard labeling, including skin and eye irritancy, signals that professionals should treat this chemical with diligent care. Responsible distributors also emphasize traceability, in line with regulatory changes that demand tighter oversight on specialty amines.
Preparation Method
Synthesizing N,N-Diisopropylethanolamine is not a kitchen-table project. The main route starts with isopropylamine and ethylene oxide in a controlled, catalyzed environment. Precision is everything, since accidental overexposure to ethylene oxide leads to unwanted byproducts and brings safety hazards. The production process taps into decades of experience, balancing throughput with rigor to suppress impurities that can derail fine chemical uses. Some manufacturers have adopted continuous flow reactors, believing that tighter reaction control contributes not only to safety but also to product consistency, which research labs deeply appreciate when scaling up runs.
Chemical Reactions & Modifications
Ask any synthetic chemist about N,N-Diisopropylethanolamine and stories about versatile reactivity will pop up. This molecule doesn’t just sit there. It forms quaternary ammonium salts, acts as a phase transfer catalyst in halide reactions, and takes on protective group roles through its tertiary nitrogen. Chemical engineers introduce specific modifications by alkylating the nitrogen or converting it into quaternary ammonium salts for use in water treatment. Its branched side arms grant resistance to acid-catalyzed ring formation, so it stays intact during reactions where many secondary amines degrade or become contaminated.
Synonyms & Product Names
Anyone in chemical purchasing or regulatory compliance knows the tangle of names circling a substance. N,N-Diisopropylethanolamine can also come labeled as DIPEA, 2-(Diisopropylamino)ethanol, or even under trade names that industry folks recognize differently across continents. This lack of consistency causes confusion on shipping manifests and in procurement, but old hands in chemical management keep close lists of common synonyms for safety data integration and shipment tracking. It’s proof why keeping up with regulatory synonyms reduces risk of compliance errors.
Safety & Operational Standards
Every time I walk past a bottle of N,N-Diisopropylethanolamine, its safety label stands out. Handling protocols call for gloves and eye protection, and adequate ventilation. Contact with skin leaves a sensation similar to astringent soap, and lab incidents—thankfully rare—usually result from forgetting just how effective the chemical can be at removing oils from surfaces, including skin. The compound’s relatively low vapor pressure keeps inhalation risks on the modest side, but splashes matter. Regulatory agencies have flagged prolonged contact as a concern, so safety sheets now push thorough washing and environmental controls, especially because waste streams can leak trace amine residues.
Application Area
Chemistry-heavy industries look toward N,N-Diisopropylethanolamine for its blending of steric and electronic properties. Beyond oil and gas sweetening, it finds repeated use in synthetic lubricants, rubber processing, and even cement additives—tasks where the compound’s base strength brings measurable process improvements. In dye and pigment production, it stabilizes color and prevents unwanted side reactions from trace acids. Pharmaceutical manufacturers sometimes reach for N,N-Diisopropylethanolamine in preparing active pharmaceutical ingredients, largely because its byproduct profile is well characterized and easy to handle in purification workflows. Each field keeps adding layers to the molecule’s reputation, demonstrating its value in settings where reliability trumps novelty.
Research & Development
Active R&D explores N,N-Diisopropylethanolamine’s use as a ligand for catalytic reactions, since bulky amines can control selectivity in metal-catalyzed transformations. University labs also study derivatives for novel surfactants and biocidal formulations, expanding what started as a niche molecule into territory once reserved for more expensive or less stable chemicals. There’s growing curiosity in green chemistry circles to substitute fossil-feedstock amines with bio-derived equivalents—an effort still in its infancy for this compound, given the complexity of its branched backbone. Rapid advances in computational modeling have made characterization faster, but real-world testing remains crucial to ensure modifications don’t open new safety or environmental headaches.
Toxicity Research
Traditionally, safety research on amines focused mostly on acute risks, and N,N-Diisopropylethanolamine has passed many industrial health reviews with warnings centered on skin and eye irritation. Chronic exposure studies sit on a thinner evidence base, which keeps some research groups busy with toxicology assays and long-term ecological tracking. Fish and aquatic invertebrates seem particularly sensitive, so regulatory teams often monitor waste management systems to keep concentrations in check. Calls for expanded animal-free toxicity testing have found support, considering rising concerns about persistent chemicals. My experience with chemical safety teams highlights the importance of pre-emptive hazard mitigation, not only for worker protection but also for wider communities downstream from manufacturing plants.
Future Prospects
Looking ahead, the story of N,N-Diisopropylethanolamine will hinge on balancing capability with sustainability. Legislative changes in chemical regulation suggest tougher scrutiny, especially on amines with potential for persistence and bioaccumulation. Green chemistry advocates are pushing for smart process redesign, with greater recycling of spent streams and the search for renewable sources. At the application end, demand for controlled basicity and customized side-chain chemistry drives ongoing interest from pharma, polymers, and specialty chemicals. Where innovations in catalysis and clean energy intersect, researchers look to molecules like this for modular building blocks that have already demonstrated both resilience and versatility in complex setups.
What are the main uses of N,N-Diisopropylethanolamine?
Introduction
N,N-Diisopropylethanolamine sounds like a mouthful, but it pops up in a bunch of places most folks never notice. Industry counts on it for its chemical flexibility. My journey researching industrial chemistry showed how these basic chemicals end up shaping daily life in quiet but critical ways. Let’s dig into where this compound really matters, why it gets chosen for tough jobs, and what could improve its use.
Role in Chemical Manufacturing
This compound finds its biggest stage creating surfactants and corrosion inhibitors. Companies often use it as an intermediate, especially when making products that break down grease or keep industrial pipes from rusting out. Surfactants coming out of N,N-Diisopropylethanolamine don’t just help soap foam up, they land in more places than most expect—detergents, textile treatment, even coatings for paper. Several big detergent makers built production lines around the stability and solubility this raw material gives their formulas. The truth is, without boring chemical intermediates like this one, everyday cleaning products wouldn’t perform nearly as well.
Buffering Power in Water Treatment
N,N-Diisopropylethanolamine works as a buffer in water treatment and gas purification. In water systems, pH swings can mess everything up—from pipes scaling over to bacteria growing where it shouldn’t. Plants sometime lean on this compound to keep pH steady, especially in high-demand industrial water loops. It’s not glamorous, but hundreds of cities indirectly rely on chemicals like this to keep the water running clean. In my conversations with plant managers, the biggest praise always goes to products that make their jobs less stressful. Consistent pH means fewer surprises that cost time or bring fines.
Strength in Gas Treatment Solutions
This chemical often lands in natural gas plants and refineries, too. Gas needs cleaning before it enters homes or factories, and removal of acid gases such as carbon dioxide and hydrogen sulfide is key. N,N-Diisopropylethanolamine blends into amine solutions used to scrub out these unwanted gases. The design advantage comes from its ability to reduce the formation of heat-stable salts, which can otherwise eat away at process equipment. Overhauling systems for fewer shutdowns and maintenance bills has real economic impact, especially at scale.
Pharmaceutical Applications
Though not a household medicine, this compound sometimes gets into active pharmaceutical ingredient synthesis. In pharma, precision counts. Small changes in chemical structure can mean the difference between an effective drug and wasted batches. Chemists have picked N,N-Diisopropylethanolamine because its profile offers certain advantages in controlling reaction rates and product purity. I’ve read several papers showing its role in tweaking reactions that require careful pH control and mild base strength.
Looking Forward: Balancing Safety and Performance
The safety records of N,N-Diisopropylethanolamine matter. Responsible handlers stick with strict protocols—good labeling, solid ventilation, and solid personal protection. Improving workplace safety isn’t about inventing new rules, but about making sure companies listen to evidence and act on lessons from past accidents. Some research labs explore greener alternatives but haven't hit on replacements with matched versatility at comparable cost. Real progress comes from tighter process controls, staff training, and greener handling without cutting performance.
From keeping pipes clean to supporting chemical reactions, the varied uses of N,N-Diisopropylethanolamine remind us how much modern life leans on behind-the-scenes science. Staying curious about these connections pushes both safety and inventiveness ahead.
Is N,N-Diisopropylethanolamine hazardous or toxic?
Getting Familiar With the Chemical
N,N-Diisopropylethanolamine plays a steady role in manufacturing, surfactant production, and certain specialty chemical processes. Workers might handle it during synthesis of detergents or in some types of textile and oil formulations. Those who’ve spent time walking factory lines or handling raw materials for industrial cleaning agents may have come across its sharp, fish-like odor or needed to work a little extra on ventilation to keep indoor air clear.
Real Risks in Handling
This compound doesn’t carry the same kind of danger as strong acids or solvents used for stripping paint. Yet, it’s never as harmless as water. Its safety data sheets warn about irritation. Skin contact leads to dryness or even a rash after extended exposure — especially if gloves are missing or thin. Breathing in vapor for too long dries out the throat and may bring a burning feeling to eyes or nose, something you remember after working near a leaky storage drum or a splash on sweaty arms during summer shifts. Eye contact turns into stinging and redness fast, and even a small spill splashing near the eye can mean a quick trip to the eyewash station. Swallowing small amounts causes discomfort, and anyone working near food or drink without proper hygiene standards could risk this, though it’s rare in professional environments.
Long-Term Health Concerns
No strong scientific evidence suggests it builds up in the body or causes cancer with typical industrial contact. Most research so far points to irritation instead of systemic toxicity. Still, the mere fact that it can cause acute symptoms tells manufacturers to keep controls in place. Chronic exposure to even mild irritants over several years, based on observations in some chemical plants, increases the chance of dermatitis or sensitization, especially in warm climates where safety gear sticks and sweat pools inside gloves.
The Importance of Good Practice
Anyone who’s done a shift in chemical formulation plants knows that personal protection keeps the work safe. Chemical-resistant gloves, goggles, boots, and hand-washing routines make the difference between an ordinary day and a scramble after a spill. Regular health checks on team members underline a company's commitment to worker safety. Some plants rely on local exhaust systems above mixing vats and enforce training on splashing risks during drum transfers. Factories where corners get cut might see more reports of dry, cracked skin, eye irritation, or breathing complaints. That kind of workplace culture often ends up costing more in lost time and medical bills.
Preventing Problems at the Source
I’ve found that an open safety culture goes a long way. Encouraging workers to report symptoms early, double-checking drum transfer procedures, and switching to closed transfer systems cuts down accidental spills and exposure. Suppliers can offer diluted forms to minimize risk or smaller packaging to make calibrating safer. No one wants to work next to an anonymous liquid that causes discomfort after a ten-minute spill. Knowledge makes routine interaction safer, not scarier.
Conclusion
N,N-Diisopropylethanolamine doesn’t top the list of the most dangerous chemicals, but treating it with respect is non-negotiable. Eyes, skin, and the lungs all react to careless handling. Simple changes — gloves, ventilation, early reporting — keep the workplace moving smoothly and workers healthy. In any industrial setting, a little vigilance beats complacency every time.
What is the chemical structure of N,N-Diisopropylethanolamine?
Unpacking the Skeleton: What’s Under the Name?
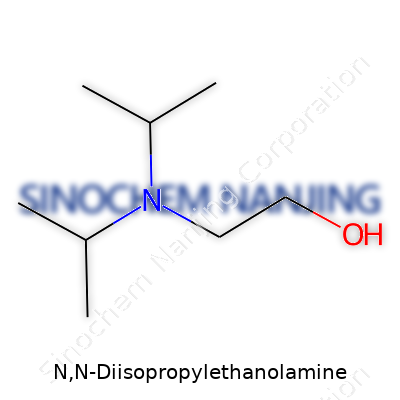
N,N-Diisopropylethanolamine shows up in plenty of chemical conversations, especially for folks working in pharmaceuticals or specialty chemicals. Its backbone carries an ethanol group attached to a nitrogen, which also holds two isopropyl arms. The structure shapes how it behaves and interacts during real-world tasks.
Let’s break that down a bit. Starting from a simple ethanol (think of a two-carbon alcohol), you get a chain like CH2CH2OH. Chemists replace one of the terminal hydrogens with a nitrogen atom. That nitrogen doesn’t like to be lonely; both its free hands clasp onto isopropyl groups—each structured like (CH(CH3)2).
Why Does This Structure Matter?
Small tweaks in a molecule’s shape steer its uses and safety. The isopropyl groups bulking up around the nitrogen prevent the molecule from forming tight hydrogen bonds with water or other similar molecules. As a result, you get a molecule that mixes just enough with water to play nicely in laboratories but won’t vanish in a bucket of it. This quirk helps chemists distance themselves from harsh, caustic amines and makes handling safer without giving up much effectiveness.
The ethanol portion gives a reactive spot for further chemical tinkering. That’s key for anyone synthesizing fine chemicals or pharmaceuticals. Modifying that little alcohol group turns N,N-Diisopropylethanolamine into a launch pad for designing more complex compounds.
Daily Touchpoints: Where Does It Show Up?
In my time developing specialty solvents for industrial labs, I ran across N,N-Diisopropylethanolamine in a suite of buffer systems. The tertiary amine (nitrogen with three alkyl groups) made it less likely to trigger an odor punch like the harsher, more basic amines. That means lab workers appreciate not having their nostrils stung mid-shift.
This structure also pops up in corrosion inhibitors and polymer production. The bulky isopropyl arms help the molecule fit into spaces where you want to stop metal from rusting or help synthetic resins shape up with better physical traits. This isn’t a “one-size-fits-all” story. Chemistry rewards carefully built skeletons, and this molecule owes its niche job to the way its parts fit together.
Safety and Environmental Concerns
With all the talk about the structure, it pays to glance at safety data. Because of the extra carbon branches, N,N-Diisopropylethanolamine doesn’t break down as easily as some simple amines. There’s a growing worry about how such molecules travel through water systems if spilled. Workers need gloves, good ventilation, and an understanding that just because something smells less offensive doesn’t make it harmless.
Better labeling and clear exposure limits from reliable sources like OSHA or the European Chemicals Agency help keep risks manageable. In my experience, consistent training and routine reviews of handling procedures make the difference—not just the rules on paper.
Solutions for Smarter Use
Anyone handling this chemical can do better by leaning on trusted sources and supplier guidance, not just a quick read of the safety sheet. A deep dive into storage, disposal, and containment makes labs safer and stops unnecessary waste. If someone must substitute or tweak the compound for a greener process, a closer look at the core structure helps. Alternatives might shave off one isopropyl group or choose a different alcohol chain, keeping function but shrinking environmental impact.
In chemical work, the story often follows the structure. N,N-Diisopropylethanolamine teaches us that details down to the smallest branch can turn a chemical into a valuable tool or a long-term headache. Learning from each molecule doesn’t just advance the sciences—it builds a safer, more responsible industry for everyone involved.
How should N,N-Diisopropylethanolamine be stored?
Understanding N,N-Diisopropylethanolamine
N,N-Diisopropylethanolamine stands out as one of those laboratory chemicals with a long, tricky name and a punchy responsibility. I remember stepping into old chemistry stockrooms, catching the faint whiff of amines from plastic drums tucked behind eye-watering labels. If you’ve handled this compound, you know careless storage doesn’t just waste money—it can invite serious safety hazards. Even a small oversight brings risks, both short-term and long-term.
Why Storage Methods Matter
From my days working with specialty chemicals, I learned that amines, especially those with ethanol chains like this one, tend to attract moisture from the air. Leave a cap loose or overlook a tiny crack, and you might find a sticky residue later. That’s not just messy—it’s a sign you’ve got contamination. Water in the drum lowers quality, throws off your research, and sometimes forms unstable byproducts. Moisture isn’t your friend here, so an airtight seal makes a difference.
A cool, dry spot far removed from direct sunlight always benefits this class of chemicals. Direct UV can degrade even stable products over months. My old mentor used to say, “Give your reagents the basement treatment, but without the mildew.” Aim for temperatures under 25°C, since higher temps gradually alter purity. Seek out chemical-grade cabinets with good airflow. Still air traps heat, so shelving near a vent or HVAC outflow pays off in the long run.
Compatibility: Keep It Separated
Cramming shelves won’t save space when safety’s on the line. N,N-Diisopropylethanolamine behaves unpredictably near acids; a splash or leak leads straight to noxious fumes. Store it in its own tray—plastic works better than metal—and slide that tray onto a shelf with similar low-reactivity amines, away from acids, oxidizers, and halogenated compounds. Even seasoned chemists sometimes get complacent and put “just this once” bottle here or there. That can turn into a headache quick.
Container Quality and Labeling
Don’t reuse old water bottles or soda jugs, even for a quick transfer. Use only thick, sealed HDPE containers built for organic bases. I once had a glass bottle crack from slow, unnoticed corrosion; plastic tends to hold up much better over years of storage. Re-label everything the instant it moves shelves. Legible, current dates, hazard symbols, and owner initials help newcomers or shifting staff keep the whole lab in sync. I always kept a roll of neon tape and a good marker close by because faded or peeling labels lead to mix-ups—you don’t want a rookie mistaking this for something benign.
Spill Kits and Precautions
Realistically, spills happen. You need more than just paper towels on deck. Absorbent pads for organic bases, thick gloves, and a nearby eyewash station build trust that safety isn’t just a checklist. I once witnessed a rookie skip gloves during a clean-up; no one repeats that mistake after feeling an amine on the skin. Make the rule clear: goggles, gloves, and thorough ventilation, every single time.
Beyond the Stockroom: Respect the Risks
Whether running a small startup lab or a university storeroom, safeguarding chemicals protects people and results. Too many focus only on immediate hazards. The damage from improper storage compounds over weeks and months—compromising research, putting colleagues at risk, and racking up regulatory fines nobody wants. Approaching it with discipline and practical structure signals real commitment to quality and safety, and those habits scale beyond this one compound.
What are the physical properties of N,N-Diisopropylethanolamine?
Chemical Backbone and What Sets it Apart
N,N-Diisopropylethanolamine lands as a colorless to pale yellow liquid. I've worked around a lot of amines in the lab, but this one stands out because of its faint amine-like odor that never fails to remind you to put the cap back on tight. It carries a chemical formula of C8H19NO. On the molecular scale, you get a combination of an ethanol backbone and two bulky isopropyl groups sticking out, which give it the kind of steric hindrance that keeps things from getting too reactive.
Boiling and Melting Points: Real-World Relevance
N,N-Diisopropylethanolamine boils at around 195-197°C—a temperature you can't mistake for water, yet practical for a lot of distillation setups. That higher boiling point means you can work with it at elevated temperatures without losing the compound too quickly to evaporation. If you've got a project that involves heating, this amine tends to stay right where you need it. Its melting point sits well below freezing at -60°C, but most people never see it solid unless they're at a cryogenic facility. This kind of fluidity helps when mixing, measuring, or transferring, since the substance keeps a consistent liquid form in almost any day-to-day situation.
Solubility and Compatibility
This amine blends well with water, pulling off full miscibility. In practice, that means it's easy to dilute, rinse away, or formulate into a range of liquid mixtures without worrying about stubborn layers. I remember adding it to an aqueous buffer and it mixed instantly, which doesn’t always happen with similar compounds. While it loves water, it also dissolves in organic solvents like methanol, ethanol, and acetone. That sort of crossover compatibility opens doors in both analytical chemistry and industrial formulation, where switching between solvents can be routine.
Viscosity, Density, and Handling
You don’t need special tricks to handle N,N-Diisopropylethanolamine. It has a viscosity that’s moderate—noticeable thicker than water, but nowhere near syrup. The density pins down at about 0.86 g/cm³ at 20°C, making it lighter than water, which matters if you’re dealing with layers or extractions. In my experience, ingredients with this physical profile are less messy, pour easily, and let you work efficiently, especially in larger scale applications.
Why These Properties Matter
Any time a chemist works with a material like N,N-Diisopropylethanolamine, these physical properties don’t just stay on the paper—they shape what happens at the bench. The moderate boiling point means less risk of accidental vapor inhalation during heating. Water miscibility means storage vessels clean up with ordinary rinse steps, so there’s less waste and lower clean-up costs. Even the subtle odor, though not hazardous at low concentrations, cues you into spills before they evaporate unnoticed. These simple realities impact safety and efficiency for people every day.
Practical Advice and Safer Approaches
Working safely with N,N-Diisopropylethanolamine means taking ventilation seriously if there’s a leak or spill, even if the boiling point seems high enough to keep vapors low. Always check the material safety data sheet—manufacturers update physical property details often, and that number changes with purity or supplier. For any chemical that blends with water and organics, plan storage in sealed containers away from reactive acids. If you plan to heat the material, use local extraction and gloves, because amines can irritate skin. For the sake of labs and scale-up sites, knowing these properties before opening a bottle leads to fewer surprises and a smoother process.
| Names | |
| Preferred IUPAC name | 2-[Di(propan-2-yl)amino]ethan-1-ol |
| Other names |
DIPEA Diisopropylethanolamine Ethanol, 2-(diisopropylamino)- 2-(Diisopropylamino)ethanol |
| Pronunciation | /ˌdiː.aɪ.aɪˈsɒ.prəʊ.pɪl.iːˈθæn.ə.ləˌmiːn/ |
| Identifiers | |
| CAS Number | 96-80-0 |
| Beilstein Reference | 1468586 |
| ChEBI | CHEBI:84973 |
| ChEMBL | CHEMBL1230507 |
| ChemSpider | 15712 |
| DrugBank | DB14150 |
| ECHA InfoCard | 03c153005d42-48ce-8057-047fac161fb7 |
| EC Number | 202-306-7 |
| Gmelin Reference | 67783 |
| KEGG | C19547 |
| MeSH | D004092 |
| PubChem CID | 12561 |
| RTECS number | KL7000000 |
| UNII | 6B82H96I9V |
| UN number | UN number: "3439 |
| CompTox Dashboard (EPA) | DTXSID1066742 |
| Properties | |
| Chemical formula | C8H19NO |
| Molar mass | 133.23 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Amine-like |
| Density | 0.856 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | 0.99 |
| Vapor pressure | <0.01 mmHg (25°C) |
| Acidity (pKa) | 8.9 |
| Basicity (pKb) | 5.44 |
| Magnetic susceptibility (χ) | -59.6×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.422 |
| Viscosity | 58.9 mPa·s at 25 °C |
| Dipole moment | 2.50 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 358.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −467.8 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4827.7 kJ/mol |
| Pharmacology | |
| ATC code | N07AB05 |
| Hazards | |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: --- |
| Flash point | 94 °C |
| Autoignition temperature | 320°C |
| Explosive limits | Explosive limits: 1.1–6.4% (V) |
| Lethal dose or concentration | LD50 (oral, rat): 3,280 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 4,730 mg/kg |
| NIOSH | SAF87630 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 10 ppm (47 mg/m3) |
| Related compounds | |
| Related compounds |
Diisopropylamine N,N-Diisopropylmethylamine Triethanolamine Diisopropylethylamine N,N-Diisopropylacetamide |

