N,N-Diethylethylenediamine: Examining Its Paths and Prospects
Tracing Back the Origins
Chemistry often carries stories as tangled as the molecules it describes, and N,N-Diethylethylenediamine is no exception. Its origins link back to foundational work in amine synthesis during the early 20th century, long before anyone fretted over safety data sheets or intricate regulatory maps. Researchers at the time weren’t running massive pilot plants—they tinkered at the lab bench, heating, stirring, and charting new territory in nitrogen-based compounds. The arrival of versatile alkyldiethylenediamines was more than technical progress; it gave chemical makers a route into ever-more-complex synthesis of pharmaceuticals, surfactants, and dyes. The knowledge grew, inching forward with each batch and mishap, until processes became better understood and controlled. Decades on, every bottle and drum of N,N-Diethylethylenediamine carries the weight of that persistent curiosity and the gradual loss of trial-and-error uncertainty.
Understanding the Compound
This diamine looks unremarkable—clear, with that sharp smell a chemist learns to respect—but on a molecular scale, it’s a two-faced workhorse. N,N-Diethylethylenediamine can easily act as either base or nucleophile, opening doors across organic synthesis. Commercial batches arrive with technical documentation because processes want predictability. This compound, made from combining ethylenediamine with ethyl halides or similar agents, ends up sporting two ethyl groups attached to the nitrogen atoms. It’s more than an academic puzzle; this structure shapes how it behaves across temperatures, pressures, and solubilities. Its moderate boiling point and ready miscibility with water and many organics make it useful in industrial labs where shelf-life, safe handling, and storage matter for both cost and peace of mind.
Labeling, Specs, and Handling Realities
Technical sheets often feature purity percentages, color, odor, and sometimes additional contaminants that might show up if the reaction isn’t cleaned up just right. Behind all the graphs and tables, people working with N,N-Diethylethylenediamine watch for the headaches caused by inhalation or skin exposure—the standards for gloves, goggles, and hoods come from hard-learned lessons, not just theory. While much of the chemical industry leans on safety catchphrases, it’s the collective awareness and memory of past accidents that really guides lab practice. Chemical plant workers and bench chemists do not forget leaks, spills, or the biting fumes after a mishandled transfer of a basic amine. Over the years, these experiences shape rules, from ventilation to labeling and all the way to emergency wash stations.
Preparation and Tinkering
Producing N,N-Diethylethylenediamine doesn’t involve futuristic reactors; it relies on controlled alkylation techniques that started as small-scale glassware experiments. Chemists aim to attach just the right number of ethyl groups so the molecule keeps its balance—too much or too little ruins the batch. Purifying the product matters as much as making it. Column chromatography, distillation, and repeated washing help remove unwanted side-reactions. Each synthesis run teaches another incremental lesson—how to save yield, prevent byproduct buildup, streamline waste handling. Over time, this collective knowledge sets the bar. Depending on its end use, further modifications shape the molecule for diverse chemical reactions, sometimes to anchor more complex materials, sometimes to build advanced pharmaceuticals.
Names, Nicknames, and Context
Like many specialty chemicals, N,N-Diethylethylenediamine hides behind a list of synonyms. Chemists know it as DEEDA, sometimes as N,N-Diethyl-1,2-ethanediamine. This naming flexibility reflects both the global nature of fine chemical trade and the tendency of research communities to develop their own shorthand. These different names can cause confusion for newcomers in procurement or compliance, particularly if they’re scanning old research papers or technical reports where the terminology varies regionally or historically. The trick remains in always matching the structure, not just the label—a mistake in this area can halt a production line or turn an experiment into wasted time.
Why Safety Standards Stick
Nobody working with N,N-Diethylethylenediamine gets to ignore its toxic bite. Like many amines, it brings health risks—most notably respiratory and skin irritation. GHS symbols and pictograms aren’t just for show; they summarize years of toxicological data. Reports have shown that accidental inhalation or skin contact requires quick action. Workshops and labs across the world carry ingrained routines: check the hoods, label containers, double-glove during transfers. Where compliance lapses, injury can follow. These lessons build a stubborn kind of safety culture. Having seen a fire or chemical burn firsthand, workers don’t need further convincing to respect the material. Mistakes do happen but become less frequent as stories, not just protocols, circulate through the ranks.
Applications: Where It All Ends Up
The uses for N,N-Diethylethylenediamine keep it relevant. It features heavily as a building block for epoxy and polyurethane catalysts, providing chemical reactivity that larger companies rely on for plastic and resin synthesis. Biomedical research teams have used it for modifying biomolecules, showing just how well this flexible molecule adapts. Its role in surfactant production and specialty cleaners ties into everything from textiles to electronics. These practical applications, observed in the workshop and the lab, create value beyond academic discussions about structure or reactivity. Companies choose this amine because it works reliably, not just because it looks good on paper.
Frontiers in Research and Pushing the Boundaries
Scientific curiosity never sits still. Researchers continue testing N,N-Diethylethylenediamine’s range in ligand development, complexation chemistry, and as a precursor to functionalized materials. Patents suggest new uses for selective extraction, advanced coatings, and high-performance polymers. University labs, running on limited budgets, often choose this compound for its versatility—one bottle fuels dozens of different projects. Each new finding spins off questions about stability, reactivity, and potential side effects. Researchers share both their triumphs and failures, gradually adding detail to the compound’s story. Real progress shows up slowly, as review articles, citations, and eventually, new products.
Toxicity and the Value of Vigilance
Conversations around toxicity never end with the Material Safety Data Sheet. Chronic exposure risks and questions about the breaking down of amine compounds in the environment linger. Interdisciplinary studies consider both worker safety and environmental persistence; some reports suggest breakdown products can carry their own hazards. Over the decades, workers have seen how inconsistent handling practices can impact both individual health and long-term site safety. Responsible companies keep monitoring exposure levels and update training as more data appears. This approach doesn’t draw headlines, but it keeps operations safe and sustainable, and sets a model for smaller or newer producers who lack historical knowledge.
Looking Ahead: Questions and Possibilities
Industry and academia both see further possibilities for N,N-Diethylethylenediamine, whether in next-generation battery electrolytes or in greener solvent systems. Some challenges remain: as regulations around hazardous substances tighten, producers must look for better containment, lower emission routes, and perhaps alternatives where risk can’t be justified. Meanwhile, the sheer flexibility of the molecule keeps it featured in both research catalogs and price lists. Real progress will likely depend on collaborative efforts—industry sharing incident data, universities exploring untapped reactivity, and regulators adapting standards to fit new realities. In the end, the continued use of N,N-Diethylethylenediamine will rest on its safety, performance, and adaptability across a changing landscape, shaped as much by stories and hard-won lessons as by molecular formulas.
What is N,N-Diethylethylenediamine used for?
The Chemistry Behind N,N-Diethylethylenediamine
N,N-Diethylethylenediamine belongs to the family of ethylenediamines. It looks simple on paper—just a couple of ethyl groups attached to the base structure. Chemists see it as a versatile building block. In my own work, tweaking molecules with small changes like these always gives different properties, which can be both a blessing and a headache. In this case, its structure makes it a popular choice where selectivity and reactivity need a little push.
Applications in Industry
Companies use N,N-Diethylethylenediamine mostly as an intermediate. Think of it as a tool in the kit for producing chemicals you find in everyday products. Organic synthesis draws on compounds like this to unlock reactions that otherwise stall. In the land of pharmaceuticals, it sometimes helps shape the skeleton of bigger and more complex drugs. The textile industry leans on it for certain dye-making processes. The world of crop science also taps into its potential, working it into formulations for agrochemicals.
One area I saw it put to work was in the creation of corrosion inhibitors. These are the behind-the-scenes heroes in cooling systems and boilers, giving metal parts a longer lease on life. Researchers found that by using N,N-Diethylethylenediamine as a starting point, they could tweak molecules to bind tighter to metal surfaces. This costs less down the road, reducing the need for repairs or replacements—something any facility manager can appreciate.
Benefits and Challenges
N,N-Diethylethylenediamine offers a unique balance. It’s reactive but not so touchy that it explodes at the drop of a hat. Workers like this predictability in a chemical. Handling this compound takes some care, though. Skin contact can cause burns, and vapors bring headaches. I have always stressed training and ventilation among teams working with such chemicals. A strong safety culture does more than follow regulations—it protects lives and reputations.
Some also worry about waste. Disposing of spent chemicals incorrectly can poison water or soil. Facilities must install the right measures, like on-site neutralization beds or access to trusted hazardous waste handlers. In places where rules go ignored, people and wildlife pay the price. We stand to learn a lesson here: watch the stewardship, not just the profit sheet.
Looking at Innovation and the Future
With the world shifting to greener approaches, scientists continue searching for alternatives and better processes. Companies now trace the supply chain for every raw material, weighing cost, safety, and sustainability. Laboratories explore better catalysts based on N,N-Diethylethylenediamine, which might someday replace older, more toxic players.
The key comes down to responsible use. I’ve seen the good that can come from open sharing between companies about safer handling and recycling practices. Industry roundtables allow direct talks—one company might have solved a problem that another is still wrestling with. Often, it’s this kind of knowledge sharing that moves the dial faster than big new discoveries.
Final Thoughts
N,N-Diethylethylenediamine plays a quiet, steady role in chemical manufacturing. It helps make better products, but only when handled with respect for health, safety, and the earth under our feet.
What are the safety precautions when handling N,N-Diethylethylenediamine?
Understanding the Risks
N,N-Diethylethylenediamine sounds like a mouthful, but to someone with time behind a lab bench or running plant operations, it means a job with its share of hazards. This chemical shows up in organic synthesis and metalworking, among other places. Anyone who’s worked around aliphatic amines comes to respect what a spill or a careless whiff can do. We’re not talking about something harmless. Short-term exposure triggers throat irritation, coughing, headaches, and eye pain. Let it touch your skin and you’re in for a burn that stings for hours.
PPE isn’t Optional
No matter how comfortable you might get around chemicals, no one shrugs off a compound like this. Gloves do the heavy lifting—nitrile or butyl rubber keeps it from soaking through. I once saw a coworker carelessly use cheap latex gloves while transferring a similar amine. The look on his face when the glove started dissolving still sits at the back of my mind. Splash goggles shield from surprise sprays, and a face shield tops it off during tasks with bigger risks, such as pouring or mixing. A decent chemical apron does more than save shirts; it saves skin. These steps slow nothing down and save plenty of regret.
Keep the Air Moving and Safe
One lesson I took to heart: open benches invite trouble. Work inside a certified chemical fume hood if at all possible. Fume hoods mean you breathe air far cleaner than what hovers over a beaker out in the open. N,N-Diethylethylenediamine’s vapors irritate the respiratory system fast and stick around. Just last year, a friend ignored a ductless hood's warning light and spent the next hour nursing a splitting headache. Maintenance keeps ventilation alive; ignore filter changeouts and you might as well work outside.
Safe Storage: No Surprises
Chemicals like this don’t forgive messy shelves or mystery containers. Store in tightly sealed, correctly labeled glass or HDPE bottles. Keep them cool and well ventilated. I cringe every time I see drums stacked near acids, as this sort of mixing can take a simple day and turn it into a call to emergency services. Store amines away from sources of ignition—these vapors catch fire more easily than most expect, and static discharges do the rest.
Cleanup and Waste: Practice Makes Perfect
Spills happen more often than anyone admits. Absorbent pads and neutralizing agents stay close, and used supplies go into labeled chemical waste containers right away. Letting someone else stumble on an unlabeled bag creates more work and bigger risks. A proper chemical waste program, with periodic training, takes the guesswork out of disposal and keeps environmental authorities off your back.
Training and Vigilance
No fancy fume hood or high-tech glove does any good if folks don’t understand why the routine matters. Safety training needs to go beyond checking off boxes. Techs who have real stories to tell—near-misses, burns, close calls—make the rules stick. Reviewing safety data sheets before touching a bottle keeps everyone grounded in real risks, not just imagined ones. Rehearse response drills, know exactly where eyewash and safety showers sit, and don’t treat shortcuts as badges of honor.
Safer Work through Shared Experience
Experience changes how you run a lab or a plant. It’s not just about memorizing precautions; it’s about building safer habits through repetition, shared mistakes, and constant reminders. Every warning label earned its spot through someone’s hard day. Handling N,N-Diethylethylenediamine safely means trusting the process, leaning on your team, and never underestimating what a bit of carelessness can cost.
What is the chemical structure of N,N-Diethylethylenediamine?
Getting Down to the Atoms
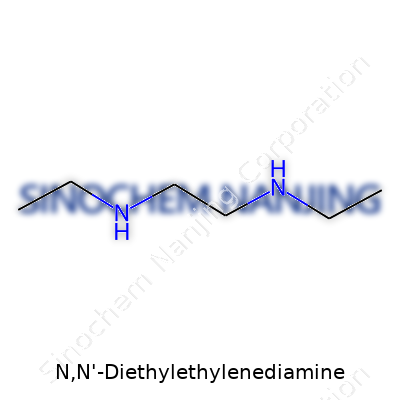
N,N-Diethylethylenediamine might sound complicated, but the story is pretty direct. This molecule comes from the family of ethylenediamines, known for holding two amine groups connected by an ethylene, or –CH2CH2–, bridge. In everyday lab work, this basic backbone pops up in everything from coatings to making pharmaceuticals. The “N,N-diethyl” part simply means both nitrogens have an ethyl group stuck to them.
Drawing the Skeleton
Chemical structure means how those atoms hook up. In this case, the formula looks like C6H16N2. On paper, you’ll find a two-carbon link with a nitrogen at each end. Each nitrogen takes on two ethyl arms, which are –CH2CH3 sections. Picture it as:
CH3CH2–N–CH2CH2–N–CH2CH3
If you’ve ever messed around with models in chemistry class, you build an ethylenediamine stick, then trade out the hydrogens for ethyl pieces on each nitrogen.
Why Structure Shapes Behavior
The way these groups hang off the nitrogens actually changes everything. That structure means it acts less basic, compared to plain ethylenediamine, since ethyl groups block water from reaching the nitrogens to grab protons as easily. In basic chemistry, the more stuff hanging off a nitrogen, the less it tends to gobble up free hydrogen ions. That’s real information for folks working in synthesis or looking to make stable chemical solutions.
I’ve used ethylenediamine derivatives to scrub up gas streams or as building blocks when chasing new molecules. N,N-Diethylethylenediamine breaks away from its simpler cousin by being more oily and not as eager to mix with water, which often helps in separating chemicals in real-world processing. That comes from the bigger ethyl groups. Chemists lean on these details for reactions where you want certain atoms protected or less reactive, like when making drugs or special coatings.
Practical Uses and Health Concerns
You won’t see N,N-Diethylethylenediamine in your medicine cabinet, but it plays a supporting role behind the scenes. Industrial and research labs tap it for making complex molecules. Its chemical quirks also turn it into a handy chelator — meaning it can snatch up metals that might wreck a reaction. In the manufacturing world, reliable access to such materials keeps supply chains steady for chemicals that end up in everything from plastics to dye fixes.
No chemical story stays complete without touching on safety. Exposure can bring skin or eye irritation, so anyone handling it in school or work labs needs basic protective gear. Good ventilation and gloves go a long way. The U.S. Environmental Protection Agency keeps tabs on compounds like this for workplace safety.
Learning and Moving Forward
Taking the time to break down structures like N,N-Diethylethylenediamine helps everyone from students to seasoned scientists pick the right tool for the job. Open-access chemical databases let you check the details before jumping into any synthesis. Universities and companies alike need to keep sharing these insights, making sure the basics don’t get lost as chemistry moves faster each year.
How should N,N-Diethylethylenediamine be stored?
Why Storage Matters for N,N-Diethylethylenediamine
The story with N,N-Diethylethylenediamine starts way before the bottle makes it to your lab. It pops up in the world of specialty chemicals pretty often, known for both its usefulness and the trouble it can cause if left unchecked. Reading the safety sheet, people usually spot warnings about flammability, skin irritation, and reactivity. That isn’t some regulatory overreaction—chemicals like this don’t forgive mistakes.
People sometimes forget how storage forms the backbone of safe chemical handling. One poorly kept drum can endanger an entire lab or warehouse. I've seen what happens after a leak: ruined supplies, hours of cleanup, and a building that smells like burnt fish for weeks. Everyone remembers the near-misses more than another production report.
Conditions That Cut Risk
This stuff demands respect for a good reason. Store it out of sunlight. Heat speeds up unwanted reactions and boosts vapor release, so a cooler spot works better. In a building I worked at, we followed the same routine for every volatile amine: keep it at 20°C or lower, in a spot with steady air flow, far from heaters and radiators.
A dry room helps. Too much moisture, and the chemical might start breaking down or corroding its container. Humidity sometimes sneaks up on you, so a few silica gel packs, regular checks, and a well-sealed bottle save a lot of grief later.
Choosing the Right Container
N,N-Diethylethylenediamine eats through soft plastics and reacts with some metals. I never trust generic containers. Stick with tightly sealed, corrosion-resistant bottles—high-density polyethylene or glass with a proper cap win every time. I met a team that used cheap lids, only to find their stocks evaporated faster, leaving behind sticky residue and a chemical stink impossible to wash off skin. Cut corners, and the material does the talking.
Keep It Away from Trouble
This isn’t a substance you want near acids, oxidizers, or halogenated chemicals. I’ve seen one accident where incompatible storage led to a runaway reaction, with enough fumes that a whole lab section had to be cleared out. Designate a room or, at the very least, a few shelves just for amines and organics. Keep a fire extinguisher close by, and line shelves with trays so spills won’t spread. Mark the whole area with clear labels—nobody appreciates unlabeled bottles in the dark corners of storerooms.
PPE: Not Optional, Not Just a Rule
Grabbing N,N-Diethylethylenediamine with bare hands or splashing it on shoes launches a world of pain—skin blisters, nasty headaches, ruined jeans. I’ve always made it a rule: anyone opening one of these bottles wears goggles, gloves, and an apron. A nearby eyewash station stands ready in the labs I’ve worked in, and it gets checked weekly because the odds catch up to everyone.
Accountability Builds Good Habits
Logging every container in and out, checking expiration dates, and rotating stock isn’t busywork. It helps catch leaks early and stops old or broken bottles from sitting for months. I've helped labs switch to digital logs after we found three half-empty bottles way past their prime in a back cupboard. Organization saves resources, health, and budgets alike.
Smart storage of N,N-Diethylethylenediamine means planning, regular checks, and the discipline to follow through every week, not just after near disasters. In the chemical world, safety starts with where—and how—you keep your bottle.
What are the physical properties of N,N-Diethylethylenediamine?
Understanding the Core: What Sets It Apart
N,N-Diethylethylenediamine rarely pops up in casual conversation, but this compound quietly makes a difference in research labs and industry. With the chemical formula C6H16N2, it shows up as a colorless to pale yellow liquid. Most folks would miss its faint amine odor if they walked past an open container. Its appearance might seem simple, but physical properties shape how chemical plants and research spaces handle and work with it.
Boiling Point and Volatility: Handling With Care
The boiling point clocks in near 146 to 149 degrees Celsius (about 295 to 300 degrees Fahrenheit). In my experience working in college chemistry labs, a boiling point in this range signals a liquid that costs you some energy to drive off as a vapor, but it still evaporates faster than water when stored at room temperature. This means you won’t see rapid vapor loss at typical lab or warehouse temperatures, but if you heat it up, you’ll see it head into the air long before most household liquids.
That same volatility points to a need for good ventilation. This is true whether you're transferring it between containers or weighing out for a reaction. My colleagues in industrial settings often talk about how amines like this can escape into the air and cause strong odors. Fans, sealed containers, and chemical fume hoods earn their keep here.
Density and Mixing: More Than Just Numbers
Density makes a real difference during mixing and measurement. N,N-Diethylethylenediamine has a density near 0.81 grams per milliliter at room temperature. That’s lighter than water. Whenever I’ve measured liquids with lower densities, they float on top of water-based solutions and oils, and sometimes require careful stirring to get uniform mixtures. Pouring too quickly invites splashing.
Solubility: Distributed By Water and Solvents
One standout feature: high solubility in water and a bunch of organic solvents. You can pour a fair amount of N,N-Diethylethylenediamine into water, stir, and see the liquid dissolve completely. This property pushes it to the front of the line for processes in dyes, resins, and even pharmaceuticals. If it didn’t dissolve so well, chemical engineers would need to put a lot more effort into getting it to react efficiently.
This ease of mixing comes in handy for cleanup and spill management too. Spill some on a bench and water usually does a solid job lifting off the residue. But because it dissolves so readily, it can spread into drains and sewers without much trouble—a flag for anyone concerned with environmental compliance.
Tackling Odor, Storage, and Safety
There’s a mild but distinctive amine smell. Even from a closed bottle, you pick up hints of its presence. Experienced chemists know this means storing containers with secure caps and away from acids—mixing with the wrong chemicals can lead to pressure build-up or even dangerous reactions. I’ve watched a well-sealed glass bottle fog up from vapor pressure on a warm day, hinting at why temperature-controlled storage matters even for something that doesn’t boil until nearly 150°C.
Flammability shouldn’t be ignored. N,N-Diethylethylenediamine will catch fire in the right conditions, so keeping it away from open flames and sources of sparks matters. Labels and safety data sheets keep everyone alert. Even in small-scale labs, it’s safer to work with spill trays and gloves, since amines can irritate the skin and eyes.
What Good Storage Looks Like
A well-organized chemical inventory can’t overlook details. Store this compound in a cool, dry place, label the containers, and keep compatible materials nearby for spill cleanup. Trained staff run quick checks for leaks and evaporation weekly, and ventilation systems keep odors under control.
N,N-Diethylethylenediamine may not draw much attention, but the way it behaves—how it boils, mixes, evaporates, and dissolves—shapes decisions every day in labs and factories. Safety improves as people understand physical properties beyond basic numbers on a sheet.
| Names | |
| Preferred IUPAC name | N,N-Diethyl-1,2-ethanediamine |
| Other names |
3-Diethylaminopropylamine N,N-Diethyl-1,2-ethanediamine N,N-Diethylethane-1,2-diamine |
| Pronunciation | /ˌɛnˌɛn.daɪˌɛθ.əl.ɪˈθɪl.iˈniːn.daɪ.əˌmiːn/ |
| Identifiers | |
| CAS Number | 100-36-7 |
| Beilstein Reference | 1718735 |
| ChEBI | CHEBI:38848 |
| ChEMBL | CHEMBL33341 |
| ChemSpider | 8808 |
| DrugBank | DB08727 |
| ECHA InfoCard | 100.007.698 |
| EC Number | 203-716-3 |
| Gmelin Reference | 10471 |
| KEGG | C06149 |
| MeSH | D004002 |
| PubChem CID | 8056 |
| RTECS number | KH6795000 |
| UNII | 5D0648F9KA |
| UN number | UN2370 |
| CompTox Dashboard (EPA) | DTXSID10889373 |
| Properties | |
| Chemical formula | C6H16N2 |
| Molar mass | 116.21 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Ammonia-like |
| Density | 0.801 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | -0.22 |
| Vapor pressure | 0.5 mmHg (20 °C) |
| Acidity (pKa) | 10.12 |
| Basicity (pKb) | 4.21 |
| Magnetic susceptibility (χ) | -59.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.427 |
| Viscosity | 1.9 mPa·s (at 20 °C) |
| Dipole moment | 4.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 290.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -107.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3933 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. May cause an allergic skin reaction. Toxic to aquatic life. |
| Precautionary statements | P261, P280, P302+P352, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 38 °C (100 °F) - closed cup |
| Autoignition temperature | 287 °C |
| Explosive limits | 3.2–14.2% |
| Lethal dose or concentration | LD₅₀ (oral, rat): 122 mg/kg |
| LD50 (median dose) | 370 mg/kg (rat, oral) |
| NIOSH | MI7700000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL: 10 ppm (40 mg/m³) |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
N,N-Dimethylethylenediamine N-Ethylethylenediamine Ethylenediamine Diethylenetriamine Triethylenetetramine |

