N,N-Diethyl-1-Naphthylamine: A Window Into Chemical Progress and Responsible Innovation
A Chemical With a Story
N,N-Diethyl-1-naphthylamine didn’t just show up in today’s world. Its roots dig deep into the history of aromatic amines, those compounds that fueled big leaps in color chemistry and materials science through the late 19th and early 20th centuries. Chemists in labs from Germany to England tinkered with naphthalene derivatives, trying to make deeper blues, more vibrant reds, and dyes that wouldn’t fade in the wash. N,N-Diethyl-1-naphthylamine caught attention less because of color and more for what its structure could do—its two ethyl arms on the nitrogen offered unusual solubility and reactivity. The old library catalogs and patent filings still tell a story of how competitive dye and pharmaceutical research made this molecule something more than a chemical curiosity.
Diving Into Structure and Properties
If you look at this molecule on paper, the core is the naphthalene ring: two benzene rings fused together, familiar to anyone who’s studied coal tar chemistry. Then comes the diethylamino group at the first carbon, turning what might have been a fairly standard aromatic hydrocarbon into something more reactive. This group doesn’t just give the molecule extra bulk; it affects how the compound dissolves in organic solvents, its boiling point, and even how it smells—often pungent, which anyone working in an organic chem lab will remember. In hands-on research, I’ve seen it coming out of a distillation setup as a clear, slightly yellow oil, dense on the walls of the flask and stubbornly slow to break down in the air, which makes storage easier but also calls for respect during handling.
Setting Benchmarks and Meeting Standards
Though not a headliner in everyday commerce, purity checks and technical specifications stand out as non-negotiables. Analytical chemists use gas chromatography and NMR to ensure the diethyl group sits where it should, not swapped by impure side products. Labels matter because a small contaminant in an amine can skew dyeing recipes or confuse a pharmacological study. In my lab work, the difference between a usable batch and a useless one often comes down to less than one percent impurity—or a faint hint of water. Correct labeling isn’t just a regulatory thing; it’s the best chance to build trust in the global chemistry community.
Preparing the Compound: Lab Craft and Know-How
Most preparation routes for N,N-Diethyl-1-naphthylamine build off classic strategies—alkylating 1-naphthylamine with ethyl halides under basic or catalytic conditions. This is bench chemistry, often smelly and messy, sometimes run beneath a hood to keep the vapors away. A strong base deprotonates the amine, then the diethylation shoots electrons straight through the molecule, often generating heat and unwanted byproducts. Getting the reaction to stop at the right point takes a steady hand: overshooting gives polyalkylated products, missing the step leaves a ragged mix. Purification is a slow trudge through distillation and extraction, usually ending in a stubbornly oily product that reminds you this isn’t textbook chemistry—it’s real life, with stained gloves and glassware to clean.
Watching Reactions Unfold
N,N-Diethyl-1-naphthylamine takes on a reactive life of its own. The lone pair on the nitrogen pushes electrons toward the aromatic rings, giving the compound a leg up in electrophilic substitutions. This behavior is key in dye and pigment chemistry and gives the molecule an edge for structural modifications. Add an acyl group in a Friedel-Crafts setup, or tweak the molecule through nitrosation—each step changes its character and opens new areas for research. In projects focused on molecular probes or chemical sensors, these reactions allow precise tailoring, and I’ve seen collaborators get excited when a particular derivatization finally pops up bright under a UV lamp.
Behind the Names: Synonyms and Context
N,N-Diethyl-1-naphthylamine has a handful of aliases. It appears in older research as “N,N-Diethyl-α-naphthylamine,” and some suppliers just call it “diethylnaphthylamine.” Getting familiar with these different names is not just a paperwork exercise—it’s critical for effective literature searches and avoiding mistakes when ordering reagents or interpreting old experiments. Chemical names can act as time markers, too, showing shifts in labeling standards and scientific conventions from decade to decade. This familiarity breeds a kind of chemical literacy that’s about more than just formulas; it’s about keeping up with the conversation in the field.
Putting Safety Into Perspective
Working with aromatic amines brings safety to the front of the mind, not just because of acute hazards, but due to the historical baggage of toxicity and cancer risk that comes from exposure to similar molecules. Gloves, goggles, a working fume hood, and clear protocols are non-negotiable here. Regulators and institutional safety committees set benchmarks for maximum exposure and disposal, learning from past incidents. Watching classmates and colleagues respect these rules during late-night syntheses drove home the point that no experiment is worth a long-term health risk. I’ve seen dozens of safety presentations reinforce the need for well-maintained records and reliable container labels—the boring stuff, but also the foundation for long careers in experimental science.
Why Application Areas Still Matter
N,N-Diethyl-1-naphthylamine has never matched more famous anilines or naphthylamines in commercial scale, but it finds its own importance in specialist dyes, analytical chemistry, and sometimes in pharmaceutical research as a probe or derivatization platform. Analytical chemists use it for colorimetric assays that detect trace metals or amines in water. Other labs synthesize molecular switches or look for small-molecule tools that interact with proteins in complex ways. During my own graduate research, assays using modified aromatic amines revealed subtle shifts in how enzymes work—giving a way to examine biological mysteries that genetics alone can’t explain.
Pursuing R&D: Innovation Onward
Research into compounds like N,N-Diethyl-1-naphthylamine doesn’t get major headlines, but that fundamental work often plants the seeds for bigger discoveries down the line. Investigators explore greener synthesis routes, try to cut down on byproducts, and constantly hunt for less toxic alternatives. Intellectual property filings build up around unusual modifications or application ideas that give new life to familiar structures. In academic settings, the molecule shows up in organic synthesis courses and as a building block in multistep problems—if you can master its transformations, you’re moving up the ranks as a synthetic chemist.
Toxicity and Its Real-World Impact
Researchers and regulators both know that aromatic amines sometimes pose tough questions for health and environment. N,N-Diethyl-1-naphthylamine deserves scrutiny. Animal studies and early tests point toward some level of toxicity, mainly if handled carelessly or disposed of improperly. Wastewater treatment faces a real challenge with persistent organic chemicals—once these compounds slip into rivers or soil, breakdown can be painfully slow. In-house safety data often doesn’t match real-world scenarios, so it’s up to industry and academia to back up claims with strong, replicable research. This work calls for modern analytical tools and honest reporting—a lesson that echoes across chemical safety debates every decade.
Looking Forward: What Comes Next?
The future of N,N-Diethyl-1-naphthylamine is tied to broader trends in green chemistry and regulatory scrutiny. As researchers push for higher performance materials, sensors, and pharmaceuticals, structural creativity often circles back to flexible aromatic amines and their derivatives. There’s strong motivation to create safer analogs, minimize environmental impact, and unlock new value by shifting to renewable feedstocks or bio-based processes. Many organizations explore catalytic or enzymatic strategies to replace old, wasteful methods. This spirit of continuous improvement drives the discipline—as new students learn from past mistakes, keep the best of old techniques, and use technology to open new molecular frontiers. The chemical world moves fast, but the lessons and opportunities in compounds like N,N-Diethyl-1-naphthylamine keep asking us to think clearly, act responsibly, and value both safety and discovery.
What is N,N-Diethyl-1-Naphthylamine used for?
Why Scientists Reach for N,N-Diethyl-1-Naphthylamine
Walk into a chemistry lab and you’ll spot all sorts of bottles with hard-to-pronounce names. N,N-Diethyl-1-naphthylamine stands out thanks to its usefulness, not just for research, but also for testing water quality. This compound has stepped in to give chemists a clearer way to measure substances like chlorine and nitrites—two things plenty of people should pay attention to, especially those worried about clean drinking water.
Water Testing and Public Health
Testing for chlorine in drinking water doesn’t just serve scientists; it protects neighborhoods. Chlorine keeps water safe from bacteria, but too much leaves problems of its own—bad taste, skin irritation, and even potential health issues. Standard water tests use N,N-Diethyl-1-naphthylamine to help spot and measure chlorine in clear, simple color-changing reactions. These tests rely on real results instead of guesswork, closing the gap between scientific jargon and something folks can trust.
Cities keep an eye on water with these tests for good reason. Water utilities use compounds like this to make sure chlorine levels sit right in the sweet spot, not too high and not too low. Homeowners and small labs use similar kits, built on old chemistry with N,N-Diethyl-1-naphthylamine at the core. These kits allow people outside of big institutions to watch their own water, giving peace of mind in places where water quality isn’t always guaranteed.
Beyond Water: A Place in Organic Chemistry
The usefulness of this molecule doesn’t end with water. N,N-Diethyl-1-naphthylamine slides into organic synthesis too. Research chemists often reach for it when they need a building block or a marker in chemical reactions. Its structure makes it a handy intermediate for dyes, pharmaceuticals, and other specialty chemicals. Once chemists figure out how different molecules fit together, whole industries evolve. That progress begins on the lab bench, sometimes with this very compound.
Some of the dyes we see today started as basic chemicals like this one. The path from a bottle in a lab to a shirt in a store might seem long, but every step counts. Researchers keep pushing for colors that stay bright, and to do that, they dig deep into chemical reactions where molecules like N,N-Diethyl-1-naphthylamine do the heavy lifting. I remember working on color-fastness in college chemistry labs. Those experiments used similar compounds to test how new dyes could hold up over months of sunlight and washing.
Looking at Safety and Best Practices
Every chemical has a flip side. N,N-Diethyl-1-naphthylamine calls for care because it can irritate skin and eyes, and long-term effects aren’t perfectly mapped yet. Safe handling matters much more than flashy technology. You’ll find guidelines everywhere a bottle gets opened: gloves, goggles, and proper ventilation. Good practice keeps people and the environment safer, which is more than just legal compliance—it builds trust in science and industry.
Building Better Solutions Together
Tools like N,N-Diethyl-1-naphthylamine keep chemists and water testers effective. For communities chasing safe drinking water, these small advances matter. By sticking with methods that balance safety, accuracy, and accessibility, science serves people in ways that actually show up in daily life—from the tap at home to new ideas in research labs.
What are the safety precautions for handling N,N-Diethyl-1-Naphthylamine?
Respecting Chemistry in Action
A bottle of N,N-Diethyl-1-Naphthylamine isn’t a household item you’ll find next to sugar and salt. This chemical shows up more in labs and research spaces, and it commands respect because of what it can do, both good and bad. A big part of safety comes down to treating the stuff like it deserves your attention at all times. I remember the strict protocols in my own university labs: no shortcuts, no complacency.
Good Gloves Go a Long Way
Let’s start with skin protection. Chemical-resistant gloves – nitrile stands out as a solid option for this compound – give your hands a barrier that means less worry about accidental contact. I’ve seen people try to get away with thin latex or just skip gloves, and more often than not, it ends with a quick trip to wash up or worse. It’s simple: suit up with decent gloves, then actually wash your hands after.
It Doesn’t Belong In Your Lungs
Inhaling chemical fumes doesn’t make you look tough, just unlucky. N,N-Diethyl-1-Naphthylamine has properties that release vapors. Use it in a fume hood or area with solid ventilation. If your workspace feels stuffy or smells odd, turn on the air system or let someone know. Even a brief lapse—just a few minutes outside the hood—can remind you why air flow matters. You’ll never want to learn that lesson by coughing for half a day.
Keep Eyes and Face in Mind
Even tiny splashes hit harder than you think. Safety goggles wrap your eyes up tight; face shields add another layer during pouring or preparing solutions. A moment’s distraction at my bench once sent a drop flying right onto my glasses instead of my face. Without eye protection, the story could’ve involved the campus medical clinic.
Don’t Eat, Don’t Drink, Don’t Touch Your Face
While it’s tempting to bring a coffee or snack into the lab, nothing good comes from chemical traces on your food. Eating, drinking, or chewing gum, even sneaking a sip of water, just raises your risk. Treat every surface as if it might have a little dust or droplet of something you don’t want inside your body.
Storage Matters
This chemical isn’t built to last on a sunny windowsill. Keep it in a tightly sealed container, away from light, heat, and incompatible substances. Clear labeling means no one grabs the wrong thing by mistake, whether you or a colleague. I’ve seen confusion over unlabeled bottles cause far too much hassle and wasted time.
Know What to Do if Something Goes Wrong
It’s easy to panic if N,N-Diethyl-1-Naphthylamine spills or splashes. Eye wash stations, safety showers, and spill kits should sit close by—never blocked or hidden behind boxes. Have emergency procedures memorized, not just printed on a dusty sheet stuck on the wall. Being prepared means speed and safety, not regret and confusion.
No Solo Experiments
Work with a buddy, especially if you’re new to this chemical. Even if you’re sure you know what you’re doing, a slip or mix-up needs another person around to help. It’s how countless accidents get caught before becoming real emergencies.
Final Thought
N,N-Diethyl-1-Naphthylamine can help you achieve a lot in the lab, but only if you respect the risks. Smart habits and solid preparation make for better science and safer days.
How should N,N-Diethyl-1-Naphthylamine be stored?
Understanding the Risks
Handling chemicals such as N,N-Diethyl-1-Naphthylamine just isn’t like tossing baking soda in a cupboard. Some people might think that once a bottle is sealed, it’s no big deal. In reality, the risks loom larger than many expect. This compound can irritate the skin, mess with your eyes, and it doesn’t take much inhalation of fumes to learn why good storage matters. Stories from old labs show how one lapse turns a quiet corner into a problem zone—maybe you don’t see a reaction right away, but the air feels sharp, your eyes water, and your skin starts to itch. That’s enough to convince anyone to set things up right from the start.
Choosing the Right Spot
Never store N,N-Diethyl-1-Naphthylamine near direct sunlight or in warm spots. Heat speeds up chemical breakdown, and light pushes those reactions along. If you’ve ever left a plastic bag near a sunny window, you’ve seen firsthand how light destroys some materials. It makes sense to store N,N-Diethyl-1-Naphthylamine in a cool, dark place. Keep it in a well-ventilated area to clear away any fumes. Ventilation plays a big role. Last year, someone forgot to cap a solvent can next to a storage cabinet, and the whole room turned uncomfortable. Smells lingered for hours. No one wants that with a trickier compound.
Separate the bottle from acids, oxidizers, and other chemicals that don’t mix well. Chemical compatibility charts aren’t just for textbook quizzes—they help stop real accidents. Some compounds release toxic or flammable vapors when they meet. Once saw a storage shelf with bleach stacked next to random bottles labeled only in marker. If an unlabeled container spills, the aftermath gets complicated fast.
Looking at the Container
Glass or high-quality plastic bottles usually hold up best. Cheap plastics crack or leach. Always double-check the seal: a crusty or loose cap lets fumes escape, and caps coated in old residue risk sticking or breaking off. Someone in our group made the mistake of storing their liquid in a stopper without a tight seal; the next month, what was left had thinned out, and the room picked up that strong chemical tang.
Clear labeling matters more than most think. Sloppy handwriting, peeling labels, or scribbles can make it hard to know what’s inside. Use tough labels with dates and full names, not just abbreviations. If emergency crews ever show up, they need answers at a glance.
People and Procedures Matter
No substitute exists for careful habits. Wear gloves and goggles every time. Don’t assume someone else wiped up drips on the counter or left things safe. Regular checks catch leaks, changing colors, or weakened seals. Leaving N,N-Diethyl-1-Naphthylamine unattended in the open increases risk, especially if children or pets wander near the area.
Keeping records doesn’t only cover your back during inspections. Tracking inventory stops folks from storing old or degraded chemicals out of laziness. One missed entry on a log sheet led to confusion during a safety drill; we found bottles that expired years before.
How to Do Better
Every person handling N,N-Diethyl-1-Naphthylamine should get training, not just the basics, but refreshers each year. Encourage a culture where people call out poor storage without fear of trouble. Tools like airflow monitors and secondary containment trays give extra peace of mind.
Strong safety practices come down to forming habits and paying attention to details. Not everyone sees the payoff until something nearly goes wrong, but in chemical work, it’s those daily choices that keep everyone safe.
What is the chemical structure of N,N-Diethyl-1-Naphthylamine?
A Closer Look at the Molecule
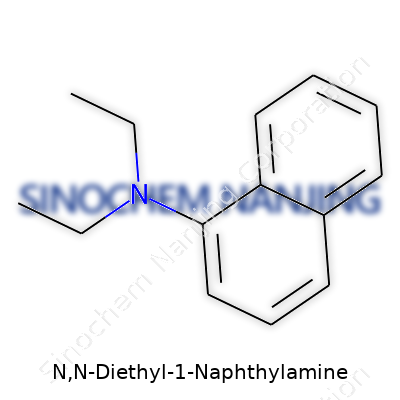
N,N-Diethyl-1-naphthylamine isn’t a compound most people talk about each day, yet chemists in the lab and manufacturers working with dyes and organic synthesis know its shape by heart. Its structure starts with a naphthalene backbone, which is basically two benzene rings fused together. On that naphthalene, at what chemists call the “1-position,” a nitrogen atom gets attached, holding two ethyl groups—making this an amine with both arms waving short chains of C2H5.
Every addition to a molecule’s skeleton changes its properties. The recipe for N,N-Diethyl-1-naphthylamine looks simple enough: take naphthalene, swap a hydrogen for an amine at the 1-position, and stick two ethyl chains on. The final layout: C14H17N. Knowing where each atom sits gives a hint at what this compound prefers to do, how it reacts, and what dangers or uses it may offer.
Why Structure Shapes Function
In my college lab days, we learned fast that the tiniest tweaks to an aromatic ring—adding an amine or tacking on alkyl groups—shift chemical behaviors. Tossing ethyl groups onto that nitrogen not only changes the size but also alters how this molecule dissolves, bonds, or even slips through a cell membrane. The naphthalene ring stays rigid and stable, perfect for stacking up with other flat molecules, which shows up in applications from dye production to sensors. The nitrogen’s ethyl attachments make it more “fat-loving,” less eager to mix into water, and likely to stick to oily substances or biological membranes.
Safety data points out that aromatic amines sometimes sneak past body defenses, causing toxic effects or, in worse cases, promoting cancers. A structure like N,N-Diethyl-1-naphthylamine demands checks before it winds up in commercial products or the environment. Accurate formula and clear diagrams help chemists track what’s safe, what’s not, stopping accidents before they start. Studies on molecules in this family show why strict controls matter when synthesizing, testing, and disposing of them.
Practical Solutions and Safer Chemistry
Making and using compounds like N,N-Diethyl-1-naphthylamine calls for deep respect for the molecule’s structure. Gloves, goggles, fume hoods—those are non-negotiables for lab work. Factories need closed handling systems and real-time monitoring, not just checklists on paper. Has a new molecule popped up with a sketchy aromatic amine core? It’s worth sending it through toxicity screens before rolling out production.
Green chemistry points to pathways for swapping hazardous amines for safer cousins, especially in dye manufacture or organic synthesis. Testing less toxic molecules and using tools like computational modeling can flag problem features—the same rigid ring, the same nitrogen placements—before chemists hit the bench. Researchers and industry teams can share data across borders so one lab’s lessons add up to safer practices for everyone.
Roughly sketched out, N,N-Diethyl-1-naphthylamine’s structure puts safety, reactivity, and application into a single frame. With every new aromatic amine, checking the blueprint—atom by atom—keeps research productive and workplaces secure.
Is N,N-Diethyl-1-Naphthylamine hazardous to health or the environment?
What Is N,N-Diethyl-1-Naphthylamine?
Chemical names with long strings of letters and numbers often signal that we’re not dealing with a household staple. N,N-Diethyl-1-naphthylamine fits that bill. It shows up in research labs and sometimes in manufacturing as a dye intermediate, or a building block in chemical synthesis. The compound got some attention because it’s not just hard to pronounce — it’s a synthetic chemical with potential impacts most of us never think about.
Health Hazards Tied to N,N-Diethyl-1-Naphthylamine
Direct contact with certain amines usually leads to skin irritation, eye damage, or acute poisoning in high enough doses. There’s limited concrete research on N,N-diethyl-1-naphthylamine specifically, but its structure suggests caution. Naphthylamines seem risky because their family already includes some members, like 2-naphthylamine, known to cause bladder cancer. N,N-Diethyl-1-naphthylamine lacks a wide safety review, though. Regulatory agencies like the EPA or OSHA haven't set full guidelines for daily or workplace exposure. That doesn't mean it's safe — just that nobody has thoroughly checked.
I remember visiting a chemistry department during my university days and seeing safety posters about chemicals that could burn, cause dizziness, or even linger for years in our bodies. Naphthylamine compounds often made those lists. The reality — and this shows up in scientific literature and safety data — is that long-term exposure to aromatic amines sometimes means a higher risk of cancer or chronic illnesses. This type of risk doesn’t always make headlines, but it’s real for workers who might handle the chemical day in and day out.
Environmental Concerns
The story doesn’t end with human health. Aromatic amines can hang around in soil and water for months or years. Some break down into smaller fragments that stick to aquatic life or make their way up the food chain. Nobody has done detailed studies tracking how N,N-diethyl-1-naphthylamine moves through the environment, but its relatives don’t paint a rosy picture. Amphibians and small fish take the brunt of contamination from similar compounds, sometimes showing stunted growth or strange mutations. The fact that this chemical resists easy breakdown only magnifies concern.
Safe Handling and Prevention
In labs and factories, a good rule is: treat every unknown chemical as if it could be harmful until proven otherwise. Lab workers wear gloves and goggles, rely on fume hoods, and use spill kits. Regulations exist for a reason. Companies can go a step further by swapping out risky compounds with safer ones when alternatives exist. Waste management matters too. Proper incineration of leftover chemicals keeps toxins out of the air and groundwater. It still surprises me how often businesses cut corners to save a buck, ignoring these precautions.
From a bigger-picture view, one answer lies with science. Funding more research can fill in the gaps around N,N-diethyl-1-naphthylamine’s long-term harm. Governments and industry leaders can push for tougher, smarter chemical policies — testing chemicals before they end up in streams or make daily contact with workers. Much like asbestos or lead paint, by the time a risk becomes obvious, the damage gets harder to undo.
Looking Ahead
We’ve learned enough about aromatic amines to know they deserve respect. Few people walk into a job expecting to face hidden dangers, nor should towns have to worry about unseen pollution from forgotten chemicals. Common sense tells us to take precautions and push for better research, not wait for disaster. The lessons from chemistry’s past urge us to act with care, especially with substances like N,N-diethyl-1-naphthylamine that sit just outside the spotlight.
| Names | |
| Preferred IUPAC name | N,N-Diethylnaphthalen-1-amine |
| Other names |
N,N-Diethyl-1-naphthalenamine N,N-Diethyl-1-aminonaphthalene N,N-Diethyl-1-naphthylamin 1-Naphthyl-N,N-diethylamine |
| Pronunciation | /ˌɛnˌɛn.daɪˈɛθəl.wʌn.næfˈθɪl.əˌmiːn/ |
| Identifiers | |
| CAS Number | 91-67-8 |
| Beilstein Reference | 2738576 |
| ChEBI | CHEBI:132747 |
| ChEMBL | CHEMBL371728 |
| ChemSpider | 15150 |
| DrugBank | DB08744 |
| ECHA InfoCard | 100.043.367 |
| EC Number | 202-980-7 |
| Gmelin Reference | 82290 |
| KEGG | C18819 |
| MeSH | D004220 |
| PubChem CID | 9837 |
| RTECS number | QL3675000 |
| UNII | 0LYK67N4HC |
| UN number | 2810 |
| CompTox Dashboard (EPA) | Q27294938 |
| Properties | |
| Chemical formula | C14H17N |
| Molar mass | 227.33 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | amine-like |
| Density | 1.006 g/mL at 25 °C |
| Solubility in water | insoluble |
| log P | 3.8 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | pKa = 4.87 |
| Basicity (pKb) | 5.19 |
| Magnetic susceptibility (χ) | -64.51·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6150 |
| Viscosity | 1.38 cP (25°C) |
| Dipole moment | 2.14 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 393.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -12.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3570 kJ/mol |
| Pharmacology | |
| ATC code | N02BB53 |
| Hazards | |
| Main hazards | Harmful if inhaled, swallowed, or absorbed through skin; causes skin and eye irritation; may cause respiratory irritation. |
| GHS labelling | GHS02,GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. |
| Precautionary statements | P261, P264, P271, P280, P304+P340, P312, P330, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 2, Instability: 0, Special: - |
| Flash point | 113 °C |
| Autoignition temperature | 410 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 1150 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1500 mg/kg (oral, rat) |
| NIOSH | SY8570000 |
| PEL (Permissible) | 10 mg/m3 |
| REL (Recommended) | 10 ppm (50 mg/m3) |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
1-Naphthylamine N,N-Dimethyl-1-naphthylamine N-Ethyl-1-naphthylamine 2-Naphthylamine N,N-Diethyl-2-naphthylamine |

