N-N-Butylaniline: A Closer Look at a Quiet Chemical Workhorse
Historical Development
Folks in the chemistry world started exploring aniline derivatives in the late 1800s. Synthetic chemistry made quick leaps, as researchers chased new dyes, rubber accelerators, and pharmaceuticals. N-N-Butylaniline didn’t land in the spotlight early on. Its cousins — aniline and N-methylaniline — saw more action in dye and rubber industries. But as chemical research grew, specialists realized that swapping in a butyl group changed properties and opened fresh avenues in manufacturing and research. Database records from as far back as the 1950s show N-N-Butylaniline quietly serving as an intermediate in making stabilizers and antioxidants. From lab benches to plant floors, its presence grew through decades of fine chemical expansion after World War II, driven by the hungry demands of plastics, coatings, and modern synthesis.
Product Overview
N-N-Butylaniline belongs to the aromatic amine family, built on an aniline backbone, swapped with a butyl group. Chemists work with both its liquid and solid forms, and it usually arrives clear to pale yellow, depending on age and storage. Commercial grades lean heavily on purity — research labs want the cleanest cuts for synthesis, while manufacturers worry about cost for bulk use. As a chemical intermediate, it moves quietly between different production lines. Factories often don’t list its name on finished goods, but it’s done its job in colorants, rubber chemicals, and pesticides. Not something people keep under the kitchen sink — it fits squarely into specialist circles, handled by those shaping compounds for technology, health, and daily life.
Physical & Chemical Properties
Grab a data sheet, and you’ll see N-N-Butylaniline has a melting point around -33°C, so it generally stays liquid at room temperature. Its boiling point sits at roughly 265°C, telling you it can take some heat but releases plenty of fumes if exposed. With a molecular weight of about 177.28 g/mol (C10H15N), it feels a bit heavier than some relatives. The molecule brings both hydrophobic and polar elements together, making it a good fit as a solvent for certain organic reactions. Its faint amine smell speaks to its roots, and it dissolves nicely in non-polar and polar organic solvents. Water won’t like it much; it needs coaxing to mix. Chemically, that aniline ring attracts electrophiles, and the butyl chain tweaks solubility and stability, letting researchers aim for different reaction pathways than plain aniline.
Technical Specifications & Labeling
Chemical suppliers get picky with labeling, for good reason. Material Safety Data Sheets (MSDS) should mark UN number 2735 for hazardous transport, and you’ll see hazard pictograms flagged for toxicity and environmental risk. Labels must have the chemical formula, batch number, and purity (often >99% for pharma or research sales). Storage guidelines call out keeping the drums or bottles cool, dry, and away from open air. Some regions treat it as a suspected hazardous substance under GHS standards. The chemical industry doesn’t leave much to guesswork — anyone working with this compound gets a clear roadmap for safe handling, spill response, and disposal.
Preparation Method
Synthesis usually starts with aniline and n-butyl chloride, using a basic N-alkylation approach. Reactivity depends a lot on the choice of base and solvent. Chemists might reach for sodium or potassium carbonate to mop up the acid byproducts, running reactions under reflux in solvents like ethanol or dimethylformamide. The process needs careful temperature control, since high heat can lead to over-alkylation or a sticky mess of unwanted side products. After the reaction, you’ll see purification steps with distillation and washing to reach pharmaceutical or reagent-grade quality. Bigger producers have engineered continuous-flow setups to push out large batches, shaving costs with closed systems to contain fumes and save energy.
Chemical Reactions & Modifications
N-N-Butylaniline doesn’t just play one role. Its structure opens it up for further reactions, making it a building block in additive and polymer chemistry. Chemists will often use it as a nucleophile in substitution reactions or as a ligand in complexation chemistry. Its nitrogen can accept more groups or get oxidized, and the molecule’s aromatic ring gets targeted for sulfonation, nitration, or halogenation, paving the way for dyes and pigments. In everyday work, N-N-Butylaniline earns its keep by allowing manufacturers to tailor-make antioxidants for rubber or add stability to coatings, offering tunable features that pure aniline just can’t match.
Synonyms & Product Names
Walk through catalogs or research papers and you’ll spot N-N-Butylaniline under a parade of names: N-Butylphenylamine, 4-Butylaniline, Butylaniline-N, and plenty of CAS numbers, most commonly 104-19-2. Commercial products might shy away from these technical terms, using house brands or application-specific tags. Researchers and purchasing teams need to double-check spectral data and manufacturer documentation to be sure they’re getting what the label promises, since mistakes here can spoil synthesis steps or compromise product testing.
Safety & Operational Standards
No one walks into a lab or factory expecting trouble — but N-N-Butylaniline deserves respect in the workplace. Prolonged exposure can bring on headaches, nausea, and in high doses, affects the central nervous system. The liquid irritates skin and eyes; it can even pass through gloves that aren’t chemical-rated. MSDS datasheets urge ventilation, nitrile gloves, and splash-proof goggles. Spills need neutralizing with activated carbon or inert absorbent, and labs are told to store it away from acids, oxidizers, or direct sunlight. If it catches fire, carbon dioxide and dry chemical extinguishers handle most situations. Companies keep safety drills regular, with waste treated according to hazardous material guidelines. Rules from OSHA, REACH, and local standards drive these efforts, making sure people on the frontlines aren’t taking unnecessary risks.
Application Area
Look for N-N-Butylaniline’s fingerprints in modern industry, and you’ll find them in places many folks overlook. Rubber cure accelerators rely on it for efficiency and durability. Dyes and colorants need its chemical stability and ability to lock bright colors into textiles and plastics. Agrochemicals, especially herbicides and fungicides, use derivatives for tough field conditions. Pharmacology and fine chemical research have tried it for synthesis routes where sterics and electronic effects demand a little more finesse than plain aniline offers. Some specialty coatings and adhesives depend on it for cross-linking power, and you may even find traces in lubricants and oil additives. Few end-users ever notice the compound, yet manufacturers lean on its performance to keep products lasting longer and doing their jobs better under stress.
Research & Development
Chemists keep digging into new uses for N-N-Butylaniline, especially in materials science. Polymer researchers are after new elastomers and heat-stable plastics that resist breakdown in harsh conditions — and aromatic amines like this one let them tune backbone properties for custom products. Analytical chemists use it as a standard for nitrogen quantification, and pharmaceutical scientists screen derivatives for antimicrobial and antifungal activity. As technology shifts toward renewable resources and more sustainable synthesis, researchers probe cleaner, greener pathways for making and modifying N-N-Butylaniline. Hydrogenation, catalytic amination, and even biocatalytic methods draw interest in slicing out petroleumbased reagents and toxic byproducts.
Toxicity Research
Any time a compound lands on chemists' benches, toxicologists want answers. Studies in rodents show N-N-Butylaniline can harm red blood cells, with methemoglobinemia as a known risk at higher exposures. Long-term studies still lag, but the structure tells us it should be handled with the same caution given to other substituted anilines, many of which show carcinogenic or mutagenic tendencies at certain doses. Fire fighters and emergency responders train for acute response; factory health surveys look for subtle trends among workers, monitoring for anemia or neurotoxic effects. Regulations keep shifting — if ongoing research points to higher risks, expect tighter controls. Safety always comes first, as direct human exposure isn’t worth the gamble.
Future Prospects
Industry keeps rolling, and N-N-Butylaniline looks set to stay relevant for years. Its market may not be flashy, but demand stands to climb as electronics and special polymers grow. Environmental rules will nudge producers toward cleaner synthesis, so process chemists will get busy minimizing waste and shrinking carbon footprints. With green chemistry at the forefront, labs look for catalysts and solvents that trim down hazards. Product specialists want more rigorous analytics, sniffing out impurities with sensitive HPLC and GC methods to meet higher regulatory standards. Sustainable manufacturing, stronger product safety, and new applications in nanomaterials and biocompatible coatings put this compound on the radar for researchers and manufacturers determined to build safer, smarter technologies, while keeping people and the planet protected.
What is the chemical formula of N-N-Butylaniline?
Getting to Know the Compound
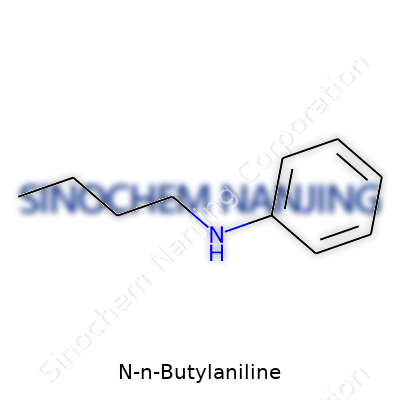
Chemistry sometimes gets tangled up in complicated names and tricky formulas. N-N-Butylaniline sounds like a mouthful, but the formula is simple: C10H15N. This compound forms by joining an aniline (that’s a benzene ring with an amine group) to a butyl group, both attached to the same nitrogen atom. Instead of shuffling through pages of textbooks, picture a benzene core with a straight string of four carbons and an amino group clipped to it. This combination may not make headlines, but the ways it connects to other molecules lets it carve out a quiet role in industry and science.
Practical Usefulness Beyond the Lab
N-N-Butylaniline is more than a drawing in a chemistry manual. The chemical shows up in applications that reach into everyday life. Some chemical manufacturers use it as a building block for dyes. Others use it when making antioxidants, especially those that protect rubber. My own experience in quality testing taught me how even “small” specialty chemicals like N-N-Butylaniline can help materials last longer. Factories making car tires and belts depend on chemicals like these to keep their products strong and flexible under heat, sun, and countless miles of use.
Safeguarding materials matters. Take colored fabrics. Without stable dyes built on compounds similar to N-N-Butylaniline, sun-faded clothes would show up much faster in stores. In chemical processes, using specific structures makes the difference between a dye that bleeds out on the first wash and one that stays vivid through many laundry cycles.
Industry Impact and Health Concerns
People sometimes overlook the toxicity risks tied to aromatic amines and their derivatives. Even if the specific risks of N-N-Butylaniline remain less studied than those of its well-known cousins, experience tells anyone working in a synthesis lab to protect skin and lungs. Aromatic amines have a track record of being hazardous in larger doses or after prolonged exposure. Old cases from dye and rubber factories showed how careless handling led to serious health problems, from anti-social headaches to far worse chronic diseases.
That history guided strict modern regulations. Gloves and proper fume hoods aren’t optional where this chemical gets used. Disposal also requires care, as dumping aromatic amines can contaminate groundwater and hurt aquatic life. Teams in chemical plants now track each step, from storage to waste, and many companies run neighboring monitoring programs to keep chemicals out of nearby environments. These safeguards build trust between companies and their communities, keeping chemicals from turning up in drinking water or the food chain.
Looking for Safer Alternatives
Growing attention on workplace safety and the environment drives researchers to develop friendlier compounds. Some new antioxidants and intermediates use plant-based chemistry, aiming for similar results with less risk. These “greener” chemicals sometimes cost more or need new processes for production, but support for green chemistry keeps growing. In my time consulting for a dye producer, we found ourselves phasing out some aromatic amines in favor of newly patented alternatives—partly to meet new market standards, partly to sleep better at night.
N-N-Butylaniline may not turn many heads, but its formula—C10H15N—reminds us how even plain-looking chemicals connect with bigger safety stories and real decisions in production labs and research departments. Each small change in materials can ripple outward, shaping safer and more sustainable futures, both in industry and beyond.
What are the primary applications or uses of N-N-Butylaniline?
Chemicals Shaping Everyday Life
N-N-Butylaniline often pops up where people least expect it. Its roots trace back to the family of aromatic amines, with a structure that helps it slip smoothly into reactions, especially those building new compounds for industry. Over the years, I've followed the growth of specialty chemicals and noticed how such substances quietly influence our daily routines—whether in the production plant or the products lining a drugstore shelf.
Crafting Dyes and Pigments
If you’ve ever wondered what brings intense color to certain plastics or textiles, you’ve seen the handiwork of N-N-Butylaniline. Dye and pigment manufacturers rely on this compound to make azo dyes, which color paper, synthetic fibers, and even some inks. These dyes stick well and offer vibrant tones that don’t fade with the first wash. Without chemical intermediates like N-N-Butylaniline, achieving that shelf-stable fuchsia or deep blue would get a lot trickier—and much more expensive.
Rubber Processing and Stability
Walk into any tire shop and you’ll see a world that depends on chemical durability. N-N-Butylaniline plays an underrated role in rubber processing. Compounds derived from it help slow down the breakdown of rubber, giving it a longer useful life. It’s easy to overlook this feature, but if car tires cracked after a few months or industrial belts degraded in weeks, costs would pile up both for businesses and drivers. Those antioxidants make a real-world impact.
Pharmaceutical Synthesis Backstage
The pharmaceutical sector leans on efficiency. Some drug intermediates trace their lineage to N-N-Butylaniline. It gets chosen for its reactivity and ability to create building blocks in complex molecule synthesis. A few modern antihistamines and specialty medications wouldn’t exist without it in the early stages of development. For someone who has watched supply chains struggle during global disruptions, I can say reliable sources for compounds like this can mean faster production and lower prices on crucial medicines.
Specialty Chemicals and Additives
N-N-Butylaniline forms part of a toolkit that chemists use in creating other specialty chemicals. These might function as stabilizers in oils, or as intermediates for more exotic uses in agriculture or electronics. The specificity of its chemical backbone allows for targeted reactions with minimal byproducts, so waste stays down and resource use makes more sense. Even at low concentrations, specialty applications like this can have a huge ripple effect across entire industries.
Safety, Environment, and the Push for Better Practices
Not every use story turns rosy. Aromatic amines raise red flags for both environmental persistence and toxicity. Over years in the chemical industry, I’ve seen that responsible handling and strict workplace limits help reduce those risks, but accidents and mishandling in informal sectors still happen. Proper training and tighter local oversight give workers better protection, and push manufacturers to recycle or treat chemical waste. New research looks for ways to phase out more hazardous amine derivatives without cutting off supply lines for key industrial goods.
The Path Forward
N-N-Butylaniline won’t pop up in household language, yet it underpins processes that touch modern living. Watching innovation in sustainable chemistry gives me hope that cleaner alternatives will continue to emerge, but for now, practical expertise, transparent manufacturing, and ongoing scientific review shape a safer future for those working with and affected by these industrial tools.
What are the safety precautions when handling N-N-Butylaniline?
Understanding Why Caution Matters
N-N-Butylaniline serves a useful purpose in a chemical lab or manufacturing setting, but it demands respect. Over the years working with chemicals, I’ve learned shortcuts don’t pay off. Safety is never about ticking boxes; it’s about protecting skin, lungs, and life itself. The stakes feel real to anyone who’s seen what exposure can do. This compound brings dangers both through the air and on contact. No one wants a hospital bill or damaged lungs just because a few steps seemed inconvenient.
Direct Exposure Means Real Consequences
Contact between N-N-Butylaniline and skin leads to redness, swelling, and more severe irritation with enough exposure. Splash in the eyes burns fast and could threaten vision. Inhalation isn’t much better; vapors irritate nasal passages and lungs, with headaches, nausea, or dizziness showing up after a short time. Larger spills leak fumes into the air, and light clothing isn’t enough to defend anyone. Every year workplace accidents prove that a simple mistake can leave a big mark. Safety data, including incident reports, support this. For instance, the National Institute for Occupational Safety and Health (NIOSH) outlines that aromatic amines like N-N-butylaniline lead to both acute and long-term toxicity, making small slip-ups costly.
Simple Precautions Save Careers
Using gloves and goggles isn’t optional. The goal is to give the chemical one less way to reach you. Choose gloves made from nitrile over latex or cloth; I’ve found nitrile holds up under exposure and resists penetration. Splash-resistant goggles prevent liquid from landing in sensitive eyes. Since n-n-butylaniline lets off fumes, a face mask or respirator rated for organic vapors keeps those vapors out of the lungs. I always double-check that hoods or fans work before starting anything—good ventilation means less chemical floating in the air. It takes only an extra minute, but it’s well worth it.
Clean-up Can’t Wait
Spills draw out mistakes. The best practice involves keeping absorbent material handy—granules or pads designed for chemicals. Small spills need to get mopped up fast and contained in a waste drum that never gets mixed with regular trash. Training everyone in the space to use this equipment keeps panic low and control high. Safety showers and eyewash stations need clear, easy paths. I’ve seen people run into trouble because a box or a cart blocked them when time mattered most. Keeping exits and emergency wash areas open is a simple habit that pays off in a crisis.
Training Prevents Guesswork
I’ve walked new technicians through the fine points of chemical handling, and the difference shows after a few months. People handle N-N-Butylaniline confidently, not carelessly. Understanding label warnings, reading the safety data sheet, and using the right containers for storage builds muscle memory. This isn’t just about avoiding trouble in the moment—chronic exposure builds up over time, so building strong habits from day one makes sense. It’s easier to remember the risks by talking about real events, not just what’s written on a page.
Securing Storage Cuts Risks
Don’t leave N-N-butylaniline sitting where sunlight or heat can reach it. Store it in tightly closed containers, away from acids, oxidizers, and food. I’ve seen what happens when incompatible chemicals get too close. Shelves should always have barriers so no bottle ever falls off by accident. Lock up anything not in immediate use to keep wandering hands—from coworkers to curious teens—away from it.
Final Thoughts: Respect the Tools
Working with strong chemicals like N-N-Butylaniline builds a healthy respect for what can happen if someone gets careless. Safety guidelines haven’t grown out of thin air. Each step—gloves, goggles, good ventilation, trained teams—exists because someone, somewhere, paid the price for missing them. Protecting yourself isn’t a hassle; it’s the difference between a safe shift and a serious accident.
What is the appearance and physical state of N-N-Butylaniline?
Down-To-Earth Insights on Its Appearance and State
N-N-Butylaniline lands in that group of chemicals you rarely notice in everyday life, yet it keeps popping up behind-the-scenes in science and industry. You might imagine something wild: lots of fumes, or bright colors, but the truth looks much simpler. If you poured some of it into a glass, you’d see a clear or pale yellow liquid. In good lighting, you can spot that faint yellow tint, almost like old cooking oil pulled from the back of a cabinet. This isn’t a flashy or mysterious substance—at room temperature, it stays liquid, almost syrupy when it gets cold.
That gentle, yellow shade comes from the way its molecules bend light, showing the impact even a single carbon chain can make when you tweak aniline with a butyl group. If you’ve ever handled other aniline derivatives, the weak, almost oily smell won’t surprise you. It feels slippery to the touch and clings stubbornly if spilled, leaving a reminder long after you’ve tried to clean it up. The chemical structure means it holds its liquid form in most lab or workroom conditions, only turning solid in the kind of cold not found outside well-stocked freezers.
Physical State and Everyday Relevance
Temperature shapes how this chemical behaves. Leave it in an average room and you get a liquid every time. Try putting it somewhere below zero, and it thickens but doesn’t freeze into a true solid until you hit much lower numbers—its freezing point sits well below what most people ever see. Typical for an organic compound like this, it boils below 300°C, which sounds high, but not unusual in the world of synthetic chemicals.
Handling N-N-Butylaniline for the first time reminded me of gloved hands and that nagging sense you ought to open a window just in case. It sticks to glassware and containers, and even with proper storage it leaves behind an oily residue. Chemistry textbooks list its density close to water, but in reality it feels thicker. Pour it from one bottle to another and you might spot the droplets hanging on the neck, refusing to let go.
Why Knowing the Physical State Matters
Ask any lab worker, and they’ll tell you: understanding what you’re really looking at—or smelling—isn’t trivia. Lab accidents often come from underestimating how a chemical spreads, settles or reacts. N-N-Butylaniline won’t explode or evaporate in the blink of an eye, but it’s persistent. Skin contact can bring irritation, even at low doses. Nobody wants to walk around all day with stained skin and that chemical scent trailing them.
Getting facts right isn’t just about ticking off safety boxes. In my early days working with anilines, small spills caught me off guard, and it took only a split second of distraction to realize gloves matter more than I’d thought. The oiliness of N-N-butylaniline makes cleanup tougher than with water-based substances. Proper ventilation and protective clothing go a long way, reducing the sting of minor mishaps.
Tackling the Challenges
Improved container designs can help limit leaks and stubborn residues. Labs adopting automatic pipetting systems often see fewer spills and less contamination. Researchers pushing for substituting less hazardous analogs—where possible—would benefit those handling larger volumes. For those still handling N-N-Butylaniline daily, simple, routine steps work best: labeling containers clearly, storing them away from heat, and prioritizing swift cleanup using absorbent pads designed for oily liquids.
Seeing this chemical for what it truly is—a persistent, oily, yellow-tinted liquid—grounds expectations and encourages respect. There’s no mystery once you know what you’re working with. That knowledge, built up from each experience, keeps the work predictable and safe.
How should N-N-Butylaniline be stored and disposed of?
Why N-N-Butylaniline Needs More Than a Casual Approach
Folks dealing with chemicals pick up pretty quickly that each type brings its own baggage. N-N-Butylaniline, an organic compound used in dyes and chemical processes, stands out as one that definitely calls for respect in storage and disposal. The stuff doesn’t make headlines often, but health risks like skin and respiratory irritation can creep up, and environmental impacts run deeper than most realize. Labs and workplaces that get careless only set themselves up for trouble. OSHA and EPA guidelines point to big risks: it’s not just about fines, it’s about people, air, soil, and water.
How to Store N-N-Butylaniline Without Invite Trouble
Shoving bottles on a shelf in the back room doesn’t cut it with a chemical like this. Cool and dry beats warm and damp every time. Flammable liquid storage cabinets shut with proper seals offer a solid defense, and airflow from a fume hood or a back room vent makes a real difference. Glass or steel containers, tightly closed, cut the risk of slow leaks or fumes. Labels need to be plain — not just for inspectors, but also for folks who may grab the wrong bottle just because they were in a rush. After a summer storm, a wet floor in a shed turns into more than a nuisance if bottles aren’t on proper spill trays.
Colleagues have shared stories about warehouses catching a whiff of fishy or aniline-like odors months after a small spill went unnoticed. It always comes back to slipping on the basics: keeping incompatible chemicals apart (no mixing with strong oxidizers or acids), good ventilation, constant temperature, and checking caps and seals every so often. One lab went so far as to run checklists every Friday — it felt silly at first, but they stopped losing sleep over leak scares.
The Reality of Safe Disposal
Tossing N-N-Butylaniline in with the trash or flushing it down sinks would be reckless. Local wastewater plants and landfill managers don’t want it either. Licensed hazardous waste companies specialize in neutralizing or incinerating chemicals without putting workers or neighborhoods at risk. Certified companies document everything, from pickup to the final off-site process, which matters in audits and in case anyone gets sick. For once, bureaucracy makes sense: paper trails beat “I think somebody took care of that.”
Pouring extra effort into hazardous waste bins pays off, too. A drum with the right liner, labeled with both the chemical name and date, keeps things sorted and traceable. Some companies run training about what goes where — all that fuss upfront beats confusion after a spill. Small labs sometimes join a local waste share program, pooling pickups for chemicals like N-N-Butylaniline, which saves money and trims risk if you don’t use it often.
Better Choices Bring Real Results
Talking storage and disposal might sound like nit-picking, but nobody regrets careful steps after a week of breathing easy, without a single labmate coughing or complaining of headaches. Cutting corners rarely works out. A safety culture, reinforced by clear steps, steady checks, and using qualified waste handlers, keeps everyone out of the ER and city council meetings about pollution. If you ask around, the best stories always start with a cabinet kept cool and a drum labeled clear — not with a close call and an apology.
| Names | |
| Preferred IUPAC name | N-butylaniline |
| Other names |
N-Butylaniline N-Phenylbutanamine N-Butylbenzenamine Butylphenylamine N-Butyl-1-phenylamine |
| Pronunciation | /ɛn-ɛn-bjuːˈtɪlˌæn.ɪˌliːn/ |
| Identifiers | |
| CAS Number | 104-11-8 |
| Beilstein Reference | 1209241 |
| ChEBI | CHEBI:131673 |
| ChEMBL | CHEMBL32358 |
| ChemSpider | 16379 |
| DrugBank | DB13521 |
| ECHA InfoCard | 03e0c12a-15a9-43f9-9d03-c2262402cfd1 |
| EC Number | 202-981-2 |
| Gmelin Reference | 6926 |
| KEGG | C06397 |
| MeSH | D018369 |
| PubChem CID | 7504 |
| RTECS number | BY8575000 |
| UNII | 104C9I55X0 |
| UN number | UN2585 |
| CompTox Dashboard (EPA) | DTXSID7020635 |
| Properties | |
| Chemical formula | C10H15N |
| Molar mass | 177.29 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | ammonia-like |
| Density | 0.927 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble |
| log P | 3.6 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | pKa ≈ 4.6 |
| Basicity (pKb) | 3.20 |
| Magnetic susceptibility (χ) | -75.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.561 |
| Viscosity | 4.5 cP (20°C) |
| Dipole moment | 2.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 362.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -4811 kJ/mol |
| Pharmacology | |
| ATC code | N02CA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS07, GHS08 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H411 |
| Precautionary statements | P261, P280, P301+P312, P302+P352, P305+P351+P338, P312 |
| Flash point | 97°C |
| Autoignition temperature | 620°C |
| Explosive limits | Explosive limits: 0.7–6.0% |
| Lethal dose or concentration | LD50 Oral Rat 640 mg/kg |
| LD50 (median dose) | LD50 (median dose) of N-N-Butylaniline: 1750 mg/kg (oral, rat) |
| NIOSH | RN0410 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 200 mg/L |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Aniline N-Methylaniline N-Ethylaniline N-Propylaniline Diphenylamine N,N-Dibutylaniline |

