N-Methylperfluorooctanesulfonamide: A Story of Industry, Policy, and Responsibility
The Long Road of Development
N-Methylperfluorooctanesulfonamide, or N-MeFOSA for those who follow chemical trends, marks a shift in how industry has chased performance at any cost. This compound first started showing up in industrial applications in the late 1950s, driven by the search for water and oil repellency unmatched by natural chemistry. 3M and DuPont led the early charge, promising brighter colors and cleaner fabrics—seemingly innocent aims back in an era without talk of PFAS contamination. The additive soon spread through textiles, papers, and eventually firefighting foams, camping deep in the bloodstream of everyday products well before most folks realized what was happening. Looking back, the story of N-MeFOSA reminds me a lot about how technological optimism can outrun careful study; it’s easier to marvel at what works than to ask where it all ends up.
Understanding What We’re Dealing With
This chemical seems simple at first look: a methyl group stuck onto a hefty perfluorooctanesulfonamide tail, colorless in appearance, unyielding to almost everything it touches. Most folks never see it, but those clear, non-stick sprays and stainproof coatings owe their “miraculous” properties to this kind of chemistry. Scientists who measure these things have trouble breaking down the stability of these bonds; N-MeFOSA doesn’t burn easily, doesn’t dissolve in water, and hangs on to its molecular integrity through all sorts of punishing conditions—both a blessing for product designers and a curse for the environment.
From Synthesis to Spillage
Making N-MeFOSA starts with perfluorooctanesulfonyl fluoride as the backbone. A methylation step pulls the process toward its final form. These steps are straightforward for someone with industrial-capacity chemical reactors, but they also unavoidably produce waste streams, including side products nobody wants leaking into the municipal water table. Once made, this compound can mingle with other agents in the formulation of surface treatments, or undergo further chemical tweaks like ethoxylation or conversion into ionic surfactants. Its chemical stubbornness keeps it functional, but also sets the stage for environmental concerns that regulators, frankly, picked up on decades too late.
What Goes Out Must Come Back
Many trade names and synonyms crowd the regulatory paperwork for N-MeFOSA. For average consumers, though, all the product codes blend into a background hum. What matters more is that N-MeFOSA nearly always lands in places nobody anticipated: a creek running through a suburban neighborhood, a blood sample pulled in a distant town, dust in an elementary school. Decades of data across Europe, North America, and Asia have now linked PFAS compounds—including N-MeFOSA derivatives—with increased cancer risk, developmental delays, and immune suppression. In my view, waiting for market signals didn’t move industry fast enough. The story now stands as a hard lesson in the value of foresight and independent testing.
Labeling, Standards, and the Changing Ground
Technical specifications and labeling for N-MeFOSA have grown tighter. Most regulatory bodies with authority over chemicals insist now on hazard classifications, proper labeling, and documented exposure limits—at least for the most hazardous forms. There’s a split between how the U.S., Europe, and, say, Japan regulate PFAS, but a general agreement that unrestricted use crossed a red line years ago. The drive now isn’t for better coatings but for cleaner exit strategies: collection, containment, and destruction, where possible.
Shifting the Risk Calculus
With so much data pointing to toxicity—some studies find the stuff in polar bears, arctic ice, and all the way down food chains—no one can honestly say that the old “dilution is the solution to pollution” mindset works. The push to minimize worker exposure on industrial sites has met resistance from businesses touting legacy approaches, but regulations have teeth now. The world doesn’t run short of alternatives, despite what defenders might claim. Startups in green chemistry chase new surfactants every year, some offering plant-based substitutes that don’t persist for decades.
Weighing Applications Against the Cost
Application areas for N-MeFOSA never started as a public health challenge. Repellency, anti-fog treatments, and emulsion stability met needs no other chemical family could match. The difficult part comes with balance: Should society allow persistent compounds to stick around for the sake of a slightly cleaner carpet or a brighter raincoat? My own experience, watching close friends struggle with rare autoimmune disorders in communities ringing chemical plants, pushes my answer toward more honest accounting of the long-term consequences.
The Research Churn
Toxicity research has taken over where product promotion left off. Every year more peer-reviewed articles connect N-MeFOSA to hormone disruption or chronic disease. It’s a field now as crowded as any, but the headlines rarely trickle into consumer awareness unless a crisis breaks. Scientists map breakdown products, track their journey through wastewater, catalog their cumulative impact on living tissue. The science speaks clearly: persistence means these compounds accumulate, and organs that never evolved to handle PFAS face the bill. In my small corner of research consulting, I see nervousness among young chemists—they want careers that improve lives, not legacies that turn up in court documents or health studies.
Where Do We Go From Here?
Industry faces a crossroads with N-MeFOSA and its chemical cousins. Europe’s REACH program, U.S. state-level bans, and rising consumer awareness build pressure each year. Some companies now invest in safer alternatives and batch tracking that gives real accountability to chemical handlers. The future lies not just in finding substitutes, but also cleaning up old messes with new technology: advanced filtration, incineration at specialty centers, and pushing research further on biodegradable options that serve the same industrial purposes. Public oversight has to keep pace—otherwise the cycle repeats every generation. For those of us with skin in the game, whether through work or the simple act of drinking tap water, the demand remains stubbornly practical: safer chemistry and honest oversight, not more fine print.
What is N-Methylperfluorooctanesulfonamide used for?
What Is This Chemical Doing in Our Products?
N-Methylperfluorooctanesulfonamide, or NMeFOSA, pops up in places few people expect. Most folks run into this chemical without ever knowing. Industry folks use it to make water and stain-resistant coatings. A lot of early carpet and fabric treatments came from it. I remember visiting my aunt’s house and seeing water bead and roll right off her new sofa. Turns out that “magic” comes from fluorinated treatments—pretty wild, but also a reason for concern.
Why Do Manufacturers Lean on NMeFOSA?
This substance belongs to a family called per- and polyfluoroalkyl substances (PFAS). Chemists love PFAS for their ability to repel oil, dirt, and water. I’ve read lists of products that used these chemicals: food wrappers, firefighting foams, and even shoe sprays. That slick, never-dirty finish people wanted in the ‘90s and early 2000s often relied on NMeFOSA-based chemistry. Factories processed it into materials like perfluorooctanesulfonyl fluoride (PFOSF), which then made its way into fabric and paper coatings, surfactants, and even some insecticides.
Facts About Exposure and Risks
The tough part about NMeFOSA and its chemical relatives is how well they hang around. They do not easily break down, so they stick in the environment and inside our bodies. The CDC tracked PFAS levels in people from all walks of life. Results show these chemicals in almost every blood sample in the US. There’s mounting evidence linking things like PFOS—made from chemicals like NMeFOSA—to health issues. Research keeps pointing at changes in liver function, immune system effects, and higher cholesterol from long-term exposure.
Cleaning Up After Decades of Use
NMeFOSA didn’t draw public attention until scientists started sounding alarms about forever chemicals. Companies then started phasing out its use in North America and Europe. Still, older couches, carpets, and jackets could keep it circulating for years. I’ve seen neighbors replace their water filters after reading about PFAS in local wells. These chemicals head into water, soil, and sometimes the food chain.
What Can We Do?
Safe alternatives have started hitting the market, though it takes real effort. I look for plain, untreated fabrics and support brands that skip “nonstick” treatments. Lawmakers work to set stricter limits on all PFAS, not just NMeFOSA. Several US states already ban products with PFAS, urging manufacturers to find safer ingredients. On the health side, local water utilities run extra filtering steps and share test results. People need to check if their area faces PFAS issues and push for more clarity from companies selling treated products.
Moving Forward: Choices that Matter
Getting these substances out of everyday goods won’t happen overnight. Still, awareness has sparked changes. Seeing communities take action—sometimes just a few parents asking questions at a school board meeting—shows that everyday people can push companies and governments to act. I take that as a hopeful sign that the next generation can enjoy safer products and cleaner water.
Is N-Methylperfluorooctanesulfonamide hazardous to health?
What Is N-Methylperfluorooctanesulfonamide?
N-Methylperfluorooctanesulfonamide, known as NMeFOSA, turns up in some chemicals used for water, dirt, and oil resistance in things like paper, textiles, and cleaning sprays. If you’ve looked at old carpets or stain-resistant fabrics, there’s a chance this compound contributed to their tough coating. Over the years, some countries scaled back or banned NMeFOSA after learning more about the risks tied to its use.
How Does It Affect People?
It finds its way into air, dust, water, and the food chain. This means pretty much everyone gets exposed, whether through drinking water, dust at home, or eating fish from rivers that picked up pollution downstream. Once inside the body, it doesn’t break down easily. Research shows it sticks around in blood and tissues for years, not days or weeks. That’s not good news. The more something builds up, the greater the chance for trouble in the long run.
What Do Studies Reveal?
Animal studies point to damage in the liver, thyroid, and reproductive organs. In some reports, rodents exposed to high amounts of this compound experienced immune system problems and cancer risks rose. Scientists still debate how these risks play out in humans, but there’s little comfort in knowing you’re carrying something that lab animals do not handle well. The US Environmental Protection Agency and other health agencies treat NMeFOSA as a likely health hazard. Evidence in humans suggests possible links between long-term PFAS exposure—NMeFOSA falls under this family—and increased cholesterol, immune suppression, and even cancer.
Where Does That Leave Communities?
A lot of folks can’t choose what lands in their drinking water or carpets, especially in places near chemical plants or waste sites. This creates real worry for parents whose kids play on floors or eat local produce. The risk isn’t just personal, it’s collective. Communities with polluted water face higher costs for clean-up and can’t always trust what comes out of the tap.
Solutions and the Path Forward
Banning or restricting its use in products was a start, but the compound lingers in older goods, landfill sites, and some water supplies. Filtering technologies like activated carbon and high-pressure membranes help remove PFAS from water, but not every area can afford this gear. For people worried about daily exposure, using filtered water and avoiding older consumer goods known for PFAS treatment makes a difference.
Stronger regulations and regular public testing bring more accountability, forcing companies to look at safer alternatives. My own experience talking with neighbors near industrial sites showed just how much people value plain answers about what’s in their water. Some got involved with local clean-up groups, showing everyday folks can push companies and governments to step up.
Researchers continue to track long-term effects. Newer, less persistent chemicals enter the market, but we’ve learned to demand thorough safety checks up front, not after the fact. Open communication, tighter law enforcement, and empowering communities matter if we want to keep health risks in check.
What are the handling and storage recommendations for N-Methylperfluorooctanesulfonamide?
A Close Look at N-Methylperfluorooctanesulfonamide
N-Methylperfluorooctanesulfonamide has made a name for itself in flame retardants and industrial processing. The substance stands out for its unusual persistence in the environment. Once introduced, it clings to ecosystems, raising red flags in science journals and among environmentalists. When I first learned about it, the warnings about long-term exposure echoed strongly. I realized early that treating it as an average chemical brings unacceptable risks.
Safe Storage: The Basics Matter Most
I always start with containment. Store N-Methylperfluorooctanesulfonamide in a tightly-sealed container, away from direct sunlight or moisture. Shielding from heat sources cannot be overstated, since higher temperatures can trigger decomposition. Fire safety leans heavily on using materials that keep it far from reactive metals, acids, and oxidizers. Ventilated storerooms keep vapors from reaching dangerous concentrations.
During my time in a chemical plant, we marked every container with clear hazard symbols and full chemical names. It prevents accidents when an emergency forces quick action. A solid chemical log, instead of scribbles on notepads, helps trace quantities and track expiration dates. It’s not just bureaucracy; forgetting a barrel of this stuff can put workers and communities at risk.
Personal Safety and Spill Control
Early lessons taught me to never touch this material bare-handed. Go with impervious gloves, long sleeves, goggles, and face shields. Respiratory protection cuts down risk if fumes build up. Accidents may look small, but the cleanup goes beyond wiping up a spill. Use absorbent pads specifically rated for fluorinated substances. Bag the waste in clearly labeled, chemically-resistant liners before sending it for hazardous processing instead of general landfill.
For larger leaks, clear the area right away. Trained responders stop the spread using barriers and special neutralizers. Emergency kits stocked with those supplies keep response times short.
Environmental and Health Impacts
Several studies now link exposure to increased health risks. Workers exposed over long periods may face persistent headaches, nausea, and more severe chronic effects. Once this chemical seeps into soil or water, removal becomes expensive and difficult; it resists natural breakdown. My own neighborhood dealt with a minor chemical scare, and watching the cleanup teams work drove home just how slow and costly that decontamination can get.
Reducing Hazards Through Clear Rules and Education
Many facilities now train new hires using real spill scenarios, not just slides and checklists. Refresher courses improve muscle memory and response times. Access to updated safety data sheets lets everyone understand current science and practical risks. This openness builds trust among staff and the nearby community, which matters more than a polished safety award hanging on the wall.
Labels, logs, and regular inspections form the backbone of chemical safety. Although these steps call for time and discipline, neglecting them loads up future risks. Regulations continue tightening, nudging companies to rethink old routines, invest in better storage, and support cleaner alternatives where possible.
Looking Ahead
N-Methylperfluorooctanesulfonamide pushes all of us to take chemical safety seriously. Each barrel needs to come with a commitment. Gone are the days when a dusty storeroom or hasty cleanup could pass for safe. These habits must evolve, not just for compliance, but for our own well-being and the health of communities downstream.
What is the chemical structure of N-Methylperfluorooctanesulfonamide?
The Backbone of N-Methylperfluorooctanesulfonamide
N-Methylperfluorooctanesulfonamide, known among chemists as N-MeFOSA, features a backbone that sets it apart. Its structure includes a fully fluorinated eight-carbon chain. Chemists write this as C8F17. That chain attaches to a sulfonamide group, which is basically a sulfur atom double-bonded to two oxygen atoms and linked to a nitrogen atom. The methyl group links to the nitrogen, rounding out the ‘N-Methyl’ part of the name. So the full chemical structure can be represented as C8F17SO2NHCH3.
Perfluoroalkyl Chains: Why They Matter
Working in labs with these kinds of compounds, I’ve watched how this group of chemicals tends to stick around longer than almost anything else. The fluorine atoms hugging the carbon chain form an unbreakable bond. This structure resists heat, acids, and basic conditions. In daily terms, this means once N-MeFOSA gets into the environment, it’s not leaving any time soon. Scientists often call this group of chemicals ‘forever chemicals’ for a reason.
This seemingly innocent chemical backbone shows up in many industrial settings. Factories making water-repellent coatings, firefighting foams, and stain-proof finishes reach for compounds like N-MeFOSA. Its structure gives products an edge in terms of durability and resistance, but my experience tells me this edge has a sharp downside.
The Real-World Impact
Researchers studying water samples have detected N-MeFOSA and similar compounds in rivers, lakes, and even human blood. Data from the U.S. Centers for Disease Control and Prevention found PFAS chemicals (N-MeFOSA included in some studies) are now in the bloodstream of almost every American. This is not just an industrial oddity. It enters the food chain, sometimes through contaminated water, sometimes through packaging or dust.
The sulfonamide group attached to the main chain raises concerns about bioaccumulation, since it makes the molecule stay intact for years. Short of fire or concentrated chemical breakdown, it lingers. The chemical’s toughness in the face of natural decay means it keeps showing up in tests around the world.
Looking Toward Safer Solutions
Regulators in the United States and Europe have ramped up their monitoring and are now pressing manufacturers to limit the release of N-MeFOSA and related compounds. My own work in public health made it clear that relying on after-the-fact cleanup is expensive and not always effective. Better filters at industrial discharge points, more transparency from producers, and creative chemistry all stand out as lighter ways forward. Some manufacturers have started shifting away from long-chain perfluoroalkyls toward alternatives with shorter chains that, based on current evidence, move through the environment faster and don’t linger as long in the body.
Slow change frustrates people who depend on clean water and hope the regulators keep up with the science. The story of N-Methylperfluorooctanesulfonamide’s chemical structure highlights the trade-offs we face: the resilience that makes it desirable in manufacturing is the same resilience challenging public health. In chemistry, there is no true silver bullet—just a careful balance between utility and safety, best driven not just by the structure on paper, but by the real lives it touches.
How should N-Methylperfluorooctanesulfonamide be disposed of safely?
Understanding What’s at Stake
N-Methylperfluorooctanesulfonamide falls under the family of PFAS chemicals, which picked up the nickname "forever chemicals" for good reason. Once these substances slip into the environment, removal becomes nearly impossible. The danger isn't just about persistence. Growing scientific research links PFAS exposure to health problems, from hormonal shifts to increased cancer risks. Having handled chemicals in industrial settings, ignoring protocol isn’t something to shrug off—damage builds quietly, showing up years later.
Traditional Methods Just Won’t Cut It
Some folks still toss chemicals down the drain or ship them off with ordinary waste. This might seem easy, but “out of sight, out of mind” only leads to lakes, rivers, and soils laced with toxic residues. Even advanced sewage treatment plants struggle with PFAS; standard filtration won’t snag those stubborn carbon-fluorine bonds. Incineration can break them down, if done at extremely high temperatures with the right technology. Many facilities can’t guarantee complete destruction, and incomplete combustion creates hazardous byproducts. I remember seeing waste incinerators fumbling with stubborn industrial chemicals—the air didn’t feel right, and nearby communities felt the impact. Dumping or burning without controls solves nothing; it shifts the risk from one spot to another.
Secure Deep-Well Injection
Engineers sometimes recommend deep-well injection for hazardous chemicals. With proper design, chemicals go far below drinking water aquifers, locked away from people and the food chain. Yet, this isn’t foolproof. Wells require maintenance and monitoring, since a leaky seal can open a disaster decades down the line. Regulations in the U.S. classify certain forms of N-Methylperfluorooctanesulfonamide as hazardous waste, bringing strict controls dictated by the Resource Conservation and Recovery Act. I’ve seen responsible disposal through licensed contractors—there’s a mountain of paperwork, but mistakes track back to the source, keeping everyone accountable.
Thermal Destruction at Specialist Facilities
Some companies invest in high-temperature incinerators, which destroy fluorinated compounds at over 1,100°C. These plants feature scrubbers and monitors that catch toxic fumes before release. Japan, Germany, and the U.S. built dedicated PFAS facilities. Costs run high—the tech, training, and safety infrastructure do not come cheap. It makes sense for industry, not small labs or households. If you find N-Methylperfluorooctanesulfonamide in a storeroom, don’t improvise. Seek licensed disposal outfits. In my experience, quality contractors check every drum and log every ounce. They don’t cut corners, because the traceable record keeps everyone on the right side of the law.
Thinking Beyond the Waste
True safety starts long before disposal. Substitute safer chemicals, design products that don’t rely on persistent toxins, and pick suppliers who make transparency part of their business. Governments step in as well, enforcing reporting requirements, drafting stronger rules, and cracking down on shady practices. Scientists push for new destruction methods—things like supercritical water oxidation show promise—but technical solutions work best with policy muscle behind them. Safe disposal isn’t some side-show at the end of a product’s life. It’s an ongoing responsibility, shared by manufacturers, users, and regulators, every step of the way.
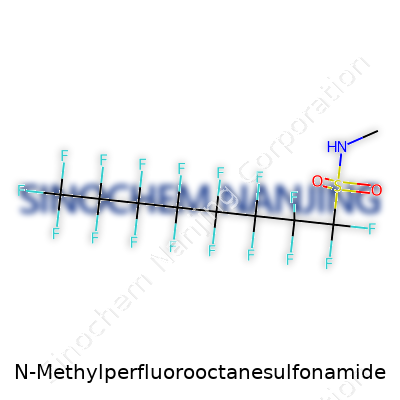
| Names | |
| Preferred IUPAC name | N-methyl-N-(perfluorooctyl)sulfonamide |
| Other names |
N-MeFOSA N-Methylperfluorooctanesulfonamide N-Methyl perfluorooctanesulfonamide Perfluorooctanesulfonamide, N-methyl- N-methyl-PFOSA |
| Pronunciation | /ɛn-ˈmɛθɪl-pɜːrˌfluːəˌrɒkˌteɪn-sʌlˈfɒnəˌmaɪd/ |
| Identifiers | |
| CAS Number | 315-34-6 |
| Beilstein Reference | 80290 |
| ChEBI | CHEBI:38840 |
| ChEMBL | CHEMBL60936 |
| ChemSpider | 22146 |
| DrugBank | DB02641 |
| ECHA InfoCard | 100.017.808 |
| EC Number | 221-615-5 |
| Gmelin Reference | 75590 |
| KEGG | C19354 |
| MeSH | D062728 |
| PubChem CID | 87749 |
| RTECS number | WN1400000 |
| UNII | Q60H5308SV |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C9H4F17NO2S |
| Molar mass | 499.17 g/mol |
| Appearance | Colorless liquid |
| Odor | Odorless |
| Density | 1.75 g/cm³ |
| Solubility in water | Insoluble |
| log P | 5.3 |
| Vapor pressure | 0.03 mmHg (25 °C) |
| Acidity (pKa) | 5.3 |
| Basicity (pKb) | 6.45 |
| Magnetic susceptibility (χ) | -6.7×10⁻⁶ |
| Refractive index (nD) | 1.310 |
| Viscosity | 15.8 cP (25°C) |
| Dipole moment | 3.1137 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 356.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1562 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P273, P280, P304+P340, P312, P405, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: |
| Flash point | 96.1 °C |
| Autoignition temperature | > 610°C |
| Lethal dose or concentration | LD50 oral rat 1120 mg/kg |
| LD50 (median dose) | LD50 (median dose): Mouse oral 1040 mg/kg |
| NIOSH | WA8300000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonamide N-Ethylperfluorooctanesulfonamide Perfluorooctanesulfonic acid Perfluorooctane |

