N-Methyl-2-Isopropyl-4-Thiazolylmethylamine Dihydrochloride: Looking Beyond the Laboratory
Historical Development
Picture the late 20th century, synthetic chemists searching for new molecules to bridge gaps in pharmaceuticals and chemical research. N-Methyl-2-Isopropyl-4-Thiazolylmethylamine first took shape during efforts to expand the thiazole family, a group of sulfur-containing heterocycles known to punch above their weight in medicinal chemistry. Thiazoles already had a solid reputation in anti-infectives, so tweaking their side chains offered a shot at hitting new targets. The addition of both a methyl group for tweaking electron density and an isopropyl for increasing lipophilicity meant a fresh scaffold for medicinal chemists and chemical biologists. Early studies on derivatives showed promise for enzyme inhibition and receptor binding, which fueled more work on the molecule's hydrochloride salt. The dihydrochloride form provides a stable, handleable powder—the sort of stable salt that survives shipping, stocking, and dissolving without fuss. Over decades, the compound shifted from an exotic curiosity to a quiet workhorse in research settings.
Product Overview
Most buyers encounter N-Methyl-2-Isopropyl-4-Thiazolylmethylamine Dihydrochloride as a white to off-white crystalline solid, packed in sealed bottles to control humidity. Compounds like this rarely draw attention outside of technical circles, but their supporting role in labs feels irreplaceable. Researchers use it as a chemical intermediate, a reference substance, and sometimes in screening libraries. Availability from specialty chemical suppliers worldwide means reproducibility across research projects, whether the user's focus is pharmacology or synthetic methods.
Physical & Chemical Properties
Dihydrochloride salts tend to offer improved water solubility compared to their free base forms. This compound dissolves well in water, sparing users the cycle of warming and shaking that non-salt forms force onto a workday. At room temperature, it holds up against degradation, and it doesn’t emit strong odors, thanks to the thiazole ring keeping sulfur volatility in check. Its melting point lands where bench work rarely poses a risk, and the powder avoids clumping, which makes for easy weighing even in humid climates. Working with the pure substance, the crystalline structure sits dense and stable, with a molecular weight that makes solutions straightforward to prepare without complicated conversions.
Technical Specifications & Labeling
Labeling standards stress purity and batch traceability. Most manufacturers state a purity above 98% by HPLC or NMR. Labels show the CAS number, batch, and storage advice. Containers offer clear warnings about skin or eye contact and suggest gloves as standard. Safety data sheets, available online, lay out the no-nonsense realities of chemical use: safe practices, first aid, and disposal advice. Labs using accreditation schemes expect this documentation as a routine part of ordering and inventory, not an afterthought.
Preparation Method
Synthesizing this dihydrochloride salt involves steps that echo much of modern organic synthesis. Start by creating the thiazole core from a suitable α-haloketone and thiourea under reflux conditions. After isolation, introduction of the isopropyl group typically uses alkylation chemistry, then methylation at the nitrogen atom tailors the basicity of the molecule. Quaternization proceeds with methyl iodide or a similar methylating agent, and the hydrochloride salt forms by bubbling dry HCl through a solution or reacting with hydrochloric acid under controlled conditions. The salt crystallizes, followed by washing, filtering, and drying under vacuum. Each step requires careful monitoring—there’s always a tradeoff between yield and purity, solvent choice, and cost. Some routes prefer greener chemistry: fewer solvents, milder conditions, less hazardous waste. As the field pushes towards sustainability, such impurities and energy costs never go unnoticed.
Chemical Reactions & Modifications
Chemists rarely stop after making a lone compound. They push the structure, hunting reactivity trends and new capabilities. The thiazole ring supports further functionalization at the 4-position, opening doors to new pharmacophore building blocks. The secondary amine supports acylation or alkylation to generate analogues or prodrugs. Reductive amination, oxidative cleavage, and ring expansion transform the basic skeleton, giving medicinal chemists fodder for screening in disease models. The isopropyl group, somewhat bulky, blocks unwanted side reactions and improves metabolic stability, which eases some woes of in vivo work. Researchers learn the quirks of these chemistries: over-alkylating the nitrogen drops desired activity, and oxidizing the thiazole ring can collapse the core altogether. This kind of troubleshooting is where personal experience comes to bear—no manual replaces the feel for which side reaction will crop up tomorrow.
Synonyms & Product Names
As with most specialty chemicals, names multiply fast—keep an eye out for variants on “N-methyl isopropyl thiazolylmethylamine dihydrochloride,” plus abbreviations in chemical catalogues and research articles. Some put the salt form up front, others tack it on the end. Side chain tweaks or stereochemistry differences occasionally sneak in, so it pays to review documentation before ordering or referencing in a publication. In my own work, comparing papers from different countries or eras made clear how many names one substance can carry—important when running database searches or avoiding double-purchasing.
Safety & Operational Standards
Bench chemists know procedures in theory, but actual safe handling matters more than written warnings stuck on a bottle. For this compound, that means weighing with gloves, using a fume hood for reactions or weighing, and practicing careful spill cleanup. While acute toxicity remains lower than many industrial chemicals, the hydrochloride salt can still irritate eyes, mucous membranes, and skin. Chronic hazards look fairly low, based on published animal data, but prudence rules the day. Labs lock away all amine-based salts in closed cabinets, labelled with standardized chemical hazard labels recognized in every modern lab. Emergency eye wash and first aid stay within arm’s reach. Regulations in GxP and ISO-certified settings keep safety culture as the backbone of daily laboratory life.
Application Area
Research spans small molecule inhibitor screening, enzyme mechanism studies, and as a synthetic intermediate for more elaborate molecules. Some academic teams use it as a starting point for agonists or antagonists in cellular assays, while process chemistry groups test its robustness in scaling up to pilot plant runs. The water-soluble nature supports biological testing in vitro, and it fits standardized assays without special handling modifications. In my experience, this flexibility makes the substance an easy go-to for routine projects or high-risk, high-reward experiments where time to first result matters more than novelty. Collaborators in analytical labs use it as a standard for LC-MS method development due to its stable ionization properties.
Research & Development
Publications from the last decade show a steady drumbeat of papers optimizing synthesis, tracking impurity profiles, and testing analogues for biological activity. The compound’s thiazole base drives interest in anti-infective, anti-inflammatory, and enzyme-targeted projects. Researchers focus on improving yields, reducing hazardous reagents, and developing one-pot processes to shorten bench work. Real progress happens through open sharing within academic and industrial circles; I’ve traded tips with chemists globally on crystallization tricks and byproduct cleanup, often over video calls sparked by shared bottlenecks. Scientists look to patents for clues about the next wave of commercial uses—analogs crop up in novel drug candidates and advanced material designs. Interdisciplinary teams keep this molecule relevant as new biological targets emerge.
Toxicity Research
Toxicity assessment follows the usual routines: cell line assays, rodent studies, and metabolism tracking. Data suggest the compound poses low acute toxicity, but care still rules, since long-term exposure consequences rarely get full coverage outside of industry-funded, deep-dive studies. Researchers in both pharma and academia push for more comprehensive toxicity profiling, embracing alternative models like zebrafish larvae and organ-on-a-chip systems. These efforts aim to catch subtle effects on organ systems or developmental stages that standard assays might miss. As regulations tighten worldwide, buyers and users share concern for safety not just for researchers but downstream, wherever traces of chemicals enter the environment or biological systems.
Future Prospects
N-Methyl-2-Isopropyl-4-Thiazolylmethylamine Dihydrochloride’s story continues as medicinal chemistry evolves. Projects using AI for molecular design churn out new derivatives at a record pace, and each variant needs a reliable base compound to compare results and validate activity. The trend towards greener, safer chemistry demands improved synthetic routes, reducing waste and hazardous byproducts. Academic and industrial teams alike push for more sustainable solvent use, automation in synthesis planning, and greener purification strategies. Greater calls for full supply chain transparency mean every bottle’s journey from reactor to lab bench faces more scrutiny. The compound’s core structure might soon appear in therapies or diagnostic tools not yet imagined, influenced by global collaboration and more inclusive research networks. Chemical safety, process optimization, and creative design will drive its role in future innovations, keeping technical teams busy on both practical details and blue-sky possibilities.
What is the primary use of N-Methyl-2-Isopropyl-4-Thiazolylmethylamine Dihydrochloride?
What’s Driving Interest in This Compound?
The world of research chemicals is filled with unfamiliar names and odd-looking formulas, but behind that alphabet soup you sometimes find substances that quietly do big things. N-Methyl-2-Isopropyl-4-Thiazolylmethylamine dihydrochloride has landed on the radar of neuroscientists and pharmacologists. Most people outside the lab won’t recognize it, but within those white-walled spaces, this compound gets serious attention for its role as an agonist of TAAR1—trace amine-associated receptor 1.
Unlocking Brain Chemistry
TAAR1 matters because it helps regulate monoamine neurotransmitters like dopamine, serotonin, and norepinephrine. These neurotransmitters control mood, motivation, focus, and sleep. Researchers long suspected that tweaking TAAR1 activity could open new frontiers in understanding brain disorders and mental health. This is where N-Methyl-2-Isopropyl-4-Thiazolylmethylamine dihydrochloride steps in. It activates TAAR1, allowing scientists to study what happens when the receptor gets a nudge.
Real insight comes when a lab tool doesn’t just sit on the shelf but actually helps map the chain reaction inside the brain’s wiring. In animal studies, compounds like this showed reductions in addictive behaviors and changes in dopamine-driven activity. Studies published by teams at institutions like Scripps and Yale showed TAAR1 agonists could cut the self-administration of stimulants in rats. These aren’t the silver bullet for addiction, but for once there’s a clear path forward. By seeing how the brain’s “volume knob” for dopamine can be dialed up or down, drug developers spot new ways to design psychiatric treatments.
Building Better Treatments, Breaking Old Habits
In my own experience working at a clinical research site, the scramble for better mental health drugs always runs into the brick wall of side effects. Drugs that hammer one pathway—like SSRIs or stimulants—often bring a list of problems along with them. TAAR1 agonists like N-Methyl-2-Isopropyl-4-Thiazolylmethylamine dihydrochloride open a back door to the brain’s reward circuits, bypassing some of those rougher edges. Early data suggest fewer mood crashes and less abuse potential.
Big pharma and grant-driven scientists circle around these discoveries because psychiatric illness grinds down millions each year. If a compound looks even faintly promising, it draws in funding and more research. N-Methyl-2-Isopropyl-4-Thiazolylmethylamine dihydrochloride isn’t sitting in any pharmacy yet, but the idea that you can moderate dopamine through an indirect pathway is a big leap. This could matter not just for addiction, but for disorders like schizophrenia and depression where existing drugs fall short.
Research Safeguards and Next Steps
Every new tool brings its own risks. Safety testing on these compounds stays front-and-center. No compound like this goes from bench to bedside overnight, and ethical oversight slows things for good reason. Reliable results need well-controlled, peer-reviewed studies before the wider medical field pays attention. So far, the safety window in animal models looks encouraging, but the leap from mice to people always brings surprises.
Better communication between research teams, transparency on findings, and ongoing safety trials all help keep the risks in check. For early-career scientists, jumping into fields like neuropharmacology demands patience, grit, and a willingness to follow the slow grind of peer review. N-Methyl-2-Isopropyl-4-Thiazolylmethylamine dihydrochloride won’t cure everything, but it opens the door to a smarter way of shifting brain chemistry, hopefully leading to better outcomes for those who feel stuck on current meds.
What is the recommended storage condition for this compound?
Putting Chemical Safety into Real Life Context
Ask anyone who’s spent time in a research lab, a pharmacy, or even a simple classroom supply closet: the way you store a chemical makes all the difference. Think about what happens when you leave milk out on the counter. It turns sour and useless, sometimes dangerous. Chemicals aren’t much different. Except the risks grow much higher when you mess up their storage.
Keeping the Compound Stable
Every compound carries a little instruction sheet, whether written or just understood by chemists: “Keep me in a dry, cool place, away from sunlight, and screw that cap on tight.” It’s not just for show. Temperature swings can cause a compound to break down or react with air and moisture. Take acetone—leave it with the cap loose, and you’re left with little more than vapors hanging in the air. Or look at antibiotics. Exposing them to heat can ruin their potency entirely, and lives depend on getting them dosed right.
Real World Examples: What Can Go Wrong
During a summer internship in a chemistry lab, I remember a bottle of sodium metal. Nobody told the new guy that water was its enemy. One drop on the lid, and it sparked. Imagine letting a compound like this sit out in the open—those risks get multiplied. Some compounds pull moisture out of the air and clump, turning useless. Others might release toxic vapors if they get too hot or sit in sunlight too long. Worse, improperly stored solvents can start a fire, and I’ve seen the remnants of charred benches to prove it.
Why Temperature and Light Make Such a Difference
Not every place offers a perfect storage room with climate control, but skipping out on the basics just invites trouble. Room temperature might sound safe for most things, but in practice, lots of compounds want exactly what’s written on their datasheet—often 2°C to 8°C for sensitive reagents. Pharmaceuticals stored outside these ranges won’t just lose their punch. They could turn harmful, as breakdown products sometimes carry unexpected side effects. Sunlight isn’t just a gentle force either—it cracks apart delicate molecules without mercy. Lab manuals aren’t joking around when they call for amber bottles and dark shelves.
Building Better Habits for Chemical Storage
It’s easy to forget, especially after a long day, but closing containers right away and placing them where they belong saves headaches. Even at home, with something as common as hydrogen peroxide, letting in a stream of air every time you forget the lid leads to a bottle of water and no disinfectant. Following a compound’s specific label may sound tedious, but it keeps products effective and folks safe. Simple habits—checking the expiration date, logging the storage temperature, and spacing incompatible containers—pay off more than most realize.
Practical Solutions: Making Storage Safer and Smarter
Many labs use dedicated refrigerators, flammable-safe cabinets, and color-coded labels. At a previous workplace, a checklist next to every cabinet caught mistakes before they caused bigger problems. Even small fixes make a world of difference. Desiccators for moisture-sensitive powders, secondary containment for acids and bases, and a culture of double-checking each other’s work have saved more than one career—and probably more than a few lives.
Safe storage isn’t just a rule from a book. It’s a daily commitment that keeps people, results, and reputations intact.Is this chemical hazardous and what precautions should be taken?
Understanding Chemical Hazards in Everyday Life
Walking into any workshop, lab, or even a household garage, you’ll spot containers filled with familiar but risky chemicals. The debate about chemical safety isn’t just for researchers or factory workers. It hits home when someone grabs bleach to clean a bathroom or pours paint thinner into a tray. Most chemical containers show symbols and warnings, yet it’s easy to overlook how important those labels are. Years back, I watched a friend ignore the simple advice to open a window while stripping paint. He ended up dizzy and nauseated within minutes. That day stuck with me; the risks of routine chemicals aren’t always obvious, but they’re real.
Recognizing the Dangers
It’s tempting to assume only “obvious” industrial materials, like acids or strong solvents, pose threats. Truth is, plenty of products under the kitchen sink or inside the garden shed can burn skin, irritate lungs, or poison even in small doses. Statistics from poison control centers highlight accidental chemical exposures leading to thousands of ER visits every year. For example, common drain cleaners contain sodium hydroxide, which burns tissue on contact. Mixing cleaning products, like bleach and ammonia, produces toxic chloramine gas, sending unsuspecting users straight to the hospital.
When someone breathes in fumes or splashes a chemical on skin, the body can suffer painful or lasting harm. Skin irritation sometimes turns into blisters. Harsh vapors inflame lungs and eyes. Swallowing small amounts may harm the liver, kidneys, or nervous system—damage that stacks up over time, often without early warning. The numbers aren’t abstract: reports link thousands of respiratory illnesses and chemical burns to poor chemical handling every year.
Practical Steps to Lower Risks
Staying safe starts with reading every label, even for products used a hundred times before. In my own workspace, I treat unlabeled containers the same way I treat a hornet’s nest—with distance and caution. Dumping leftovers from one bottle into another has led to accidental chemical reactions and even explosions. Using the original container keeps crucial information front and center. Beyond labels, having a set of gloves, eye protection, and a good mask isn’t just “extra” safety—it’s necessary, especially for chemicals that splash or release fumes.
Only use chemicals in ventilated spaces. I’ve learned a running fan and open door do more than make work comfortable—they can mean the difference between finishing a task safely and an ambulance ride. Storing chemicals in a dry area, away from children or pets, also stops accidental poisoning. One slip-up with a misplaced bottle can cause lasting harm to the whole household. Education matters as much as gear; watching a thirty-second safety video taught me more about mixing hazards than a stack of printed instructions ever did.
Building a Culture of Safety
Companies play a huge role by training employees, supplying the right safety gear, and conducting regular checks. But safety doesn’t start or stop at the workplace door. Taking the time to learn the basics means protecting yourself, your family, and your community from preventable accidents. Recognizing chemical dangers isn’t about fear—it’s about respect for what these substances can do and making smart choices before problems start.
Sources:- Centers for Disease Control and Prevention (CDC)
- U.S. Poison Control Data
- National Institute for Occupational Safety and Health (NIOSH)
What is the molecular weight and chemical formula of this product?
The Importance of Cutting Through the Confusion
Many products show up on the market, each with their own promises and technical claims. Somewhere in the fine print, you’ll likely spot the chemical formula and molecular weight. Some might brush right past this information, labelling it “for chemists only.” That’s a mistake. Both of these details create the backbone of transparency and safety in everyday products, from pharmaceuticals to fertilizers. Knowing what a product really contains gives both professionals and everyday users a measure of control—and helps avoid nasty surprises.
Real-World Impact of Molecular Weight
I’ve worked with plenty of manufacturers and clients who rely on chemical products for everything from cleaning to pain relief. If someone changes a supplier or switches ingredients, subtle shifts in molecular weight can trigger big changes in outcome. Medicines designed with a certain molecular weight behave very differently from substitutes that aren’t quite the same. Absorption rates change, reactivity shifts, and the risk of inefficacy—or harm—goes up. In one case, a small cleaning company swapped to a cheaper soap due to cost. Customers reported skin irritation, and the root cause turned out to be a slightly altered formula with a higher molecular weight. This detail, hiding in plain sight, protected neither the company nor their customers.
Formula Facts: What They Tell Us
The chemical formula isn’t just a string of numbers and letters; it’s a direct map of what you’re handling. It tells everyone—from the chemist to the end consumer—what to expect chemically and physically. If you compare sodium chloride (NaCl) with potassium chloride (KCl), they seem interchangeable at a glance, but their effects differ in medical and industrial settings. I remember one health supplement manufacturer who confused the two: the packaging showed sodium chloride, the lab tests pointed to potassium chloride. The mix-up nearly led to a health recall, harming brand trust and causing headaches across the supply chain.
Building Trust with Data and Experience
Anyone claiming a product’s effectiveness or safety needs to share the molecular weight and chemical formula up front. According to the FDA, clear ingredient disclosure forms the bedrock of public health; oversight groups recommend these as standard on everything from household cleaners to vitamins. When people ask about product safety, it’s not just about regulation or red tape. They want a sense of predictability and control. My own comfort with a new cleaning chemical comes straight from reading these specifics.
Smart Solutions for a Clarity Crisis
The market could use better education. Retailers and companies can use QR codes or short info sheets explaining the formula and molecular weight in plain language. Manufacturers should update websites and packaging, putting this data front and center, instead of hiding it in technical sheets or vague product codes. Educators can push for real-world chemistry lessons—teaching young people why sodium hypochlorite (bleach) differs entirely from hydrogen peroxide, despite both bubbling away at stains.
Walking the Walk: Sharing What Matters
It pays to treat the chemical formula and molecular weight like the ingredients on your food label. I always check for these details, and I teach others to look too. Getting this right isn’t just about compliance or jumping through hoops. It’s about protecting people, encouraging smart decisions, and making sure trust sticks around for the long run.
What is the typical purity level and how is it verified?
Understanding Typical Purity Levels
Purity tells you how much of a substance is what you actually want, not what sneaked in along the way. In my years reporting on manufacturing and supply, I’ve seen how this number shapes trust in everything from food to pharmaceuticals. For table salt, grocery stores usually stock about 97% to 99% pure sodium chloride. Pharmaceutical-grade sodium chloride must reach at least 99.5%. In the case of gold jewelry, 24-karat means purity, dropping to a lower number for alloys. Chemical labs expect purity claims like “ACS reagent grade” or “ultrapure”—standards set by organizations such as the American Chemical Society.
A purity level makes a real difference. Think about everyday tap water versus laboratory water. Most people drink the tap water and go about their day, but a lab tech shudders at the sight of even tiny bits of metal or bacteria. Even tiny impurities can spoil experiments, medication, or electronics. Growing up around construction, I learned quickly: concrete mixed with drafts of clay instead of sand turns brittle. People often forget just how small impurities can choke quality.
How Purity Gets Checked
Digging into how purity is checked, you find an entire toolkit, each method with strengths and snags. Labs rely heavily on spectroscopy, chromatography, and titration. A food manufacturer might use gas chromatography to sniff out trace chemicals or heavy metals, then publish lab reports to win consumer trust. Large pharmaceutical companies often pay for third-party verification using mass spectrometry to measure and tease out unwanted extras down to parts per billion.
Quality assurance teams use reference materials—known pure substances—for benchmarking. Any difference between their test results and the reference raises flags and sparks further checks. This approach reduces mistakes and helps catch contamination before the product leaves the plant. From my discussions with pharmaceutical chemists, it’s clear that quality control isn’t just a checkbox; it’s a daily routine with massive consequences. An unverified batch could lead to recalls, lawsuits, and even health risks.
Manufacturers and labs usually log every stage of testing. Certifying bodies like the International Organization for Standardization (ISO) set the ground rules. ISO 17025 outlines requirements for labs to show they run accurately and keep robust records. Auditors drop by unannounced, review logs, and inspect equipment. These guidelines may sound tedious, but they create a paper trail. My research into food safety scandals shows how often gaps in verification cause much larger problems down the line.
Why Purity and Verification Matter
Trust starts with transparency. Businesses that post lab results, employ outside testers, and react quickly to findings tend to avoid big disasters. The world saw what happens when companies ignore red flags: melamine in infant formula, gold bars laced with tungsten, medicines tainted with solvents. Verifying purity, over and over, isn’t just a box to check—it’s a critical part of protecting health and reputation.
Getting purity right means investing in skilled staff, routine maintenance of testing equipment, and sometimes even working with academic partners. For smaller companies, pooling resources to share advanced instruments makes sense. For larger industries, rotating verification techniques keeps tests honest and less vulnerable to bias. Both steps help bridge small mistakes before they grow into national headlines.
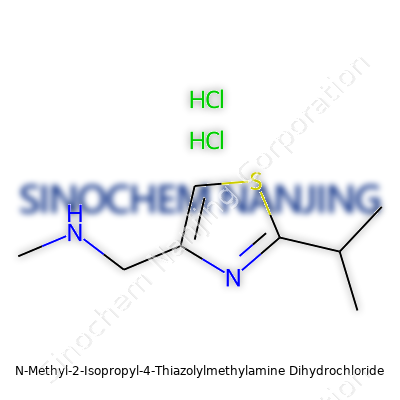
| Names | |
| Preferred IUPAC name | N-methyl-2-(propan-2-yl)-1,3-thiazol-4-ylmethanamine dihydrochloride |
| Other names |
N-Methyl-2-isopropyl-4-thiazolemethanamine dihydrochloride N-Methyl-2-(propan-2-yl)-4-thiazolemethanamine dihydrochloride N-Methyl-2-(1-methylethyl)-4-thiazolemethanamine dihydrochloride |
| Pronunciation | /ɛn-ˈmɛθɪl-tuː-aɪsəˈprəʊpɪl-fɔːr-θaɪˈæzoʊl-mɛθɪlˈæmiːn daɪhaɪdroʊklaɪd/ |
| Identifiers | |
| CAS Number | 105628-07-7 |
| 3D model (JSmol) | `JSME 3D: CN(C)CC1=NC(=CS1)CN.Cl.Cl` |
| Beilstein Reference | 10113722 |
| ChEBI | CHEBI:131887 |
| ChEMBL | CHEMBL3257912 |
| ChemSpider | 21861335 |
| DrugBank | DB08317 |
| ECHA InfoCard | ECHA InfoCard: 100.318.220 |
| EC Number | 881674-37-5 |
| Gmelin Reference | Gmelin Reference: 83298 |
| KEGG | C16376 |
| MeSH | C571326 |
| PubChem CID | 134657554 |
| RTECS number | UJ0705000 |
| UNII | 98R70L2Z82 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C8H15N3S·2HCl |
| Molar mass | 267.23 g/mol |
| Appearance | White to off-white solid |
| Density | 0.9 g/cm3 |
| Solubility in water | Soluble in water |
| log P | -1.2 |
| Acidity (pKa) | 10.2 |
| Basicity (pKb) | 3.57 |
| Dipole moment | 3.73 D |
| Pharmacology | |
| ATC code | N06BX13 |
| Hazards | |
| Main hazards | Harmful if swallowed or inhaled; causes skin and eye irritation. |
| GHS labelling | GHS02,GHS07 |
| Pictograms | GHS07,GHS05 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P302+P352, P305+P351+P338, P310 |
| NIOSH | Not established |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 50 mg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Thiamine Fursultiamine Benfotiamine Sulbutiamine Oxythiamine Thiamine mononitrate Thiamine pyrophosphate Thiamine hydrochloride |

