N-Isopropyl-N-Phenyl-Chloroacetamide: A Deep Dive
Historical Development
Chemistry has a habit of turning up valuable discoveries in unexpected places. That is how N-Isopropyl-N-Phenyl-Chloroacetamide entered the world’s chemical catalog. During the mid-20th century, the rapid growth in demand for acetylcholinesterase inhibitors and new synthetic herbicides pushed research labs to experiment with layered amides and substituted phenyl groups. Some eager minds, hunting for alternatives to tried-and-true acetanilides, landed on N-Isopropyl-N-Phenyl-Chloroacetamide. Over the decades, the subtle adjustment of its substituents allowed for exploration in pharmaceutical intermediates and specialty fine chemicals. By the late twentieth-century, it caught the attention of agricultural chemists who recognized its unique structural features for further derivatization. Chemists, often motivated by curiosity rather than initial application, gave us a molecule that has gradually found its way into more industrial processes and research shelves over time.
Product Overview
This compound stands out due to its modular design—a phenyl ring, an isopropyl group, and a reactive chloroacetamide moiety. That structure gives it a dual personality: a stable backbone and a reactive handle. You can get it as a crystalline solid, and the purity standards for laboratory use go beyond 98%. In my laboratory years, the arrival of a fresh shipment of N-Isopropyl-N-Phenyl-Chloroacetamide always meant unlocking new reaction possibilities and running fresh analytical tests. The consistency of well-produced batches determines reliability in chemical reactions, which matters when you’re fighting for reproducible results.
Physical & Chemical Properties
N-Isopropyl-N-Phenyl-Chloroacetamide appears as an off-white crystalline powder, most often with a faint aromatic odor that betrays its phenyl content. Melting points for high-purity samples usually settle between 95 and 98°C. Solubility stays moderate in polar organic solvents like ethanol or acetone, but struggles in water. Its density and refractive index reflect aromatic and amide character, but what truly marks its behavior is the reactive chlorine atom attached to the acetamide carbon. This group opens doors to substitution or elimination chemistry. The compound maintains relative stability at ambient temperatures but breaks down under prolonged strong acid or base contact, and reacts promptly with nucleophiles. These features lend appeal to synthetic organic chemists who want a reagent that’s both manageable and versatile.
Technical Specifications & Labeling
Manufacturers tend to provide technical data with clear reference to the CAS number and purity grade. Genuine analytical suppliers will offer products labeled with batch numbers, production date, storage recommendations (store below 25°C, keep dry), and a statement on shelf life, which hovers around two years in proper packaging. Hazard symbols related to irritancy and environmental risk get displayed, along with recommended handling instructions—always appreciated by bench chemists hoping to avoid trouble with regulatory compliance. Labels list chemical synonyms to assist with inventory control and prevent mix-ups. For new researchers, being able to match older literature references with current bottle labels has saved countless hours of confusion.
Preparation Method
The staple synthesis for N-Isopropyl-N-Phenyl-Chloroacetamide runs through an acylation step. Usually, phenylisopropylamine acts as the amino starting material, treated with chloroacetyl chloride in the presence of a non-nucleophilic base like triethylamine. Organic solvents including dichloromethane or toluene support the reaction by dissolving both starting materials and controlling heat dissipation. The exothermic reaction releases hydrochloric acid, so careful venting or addition of acid scavengers keeps things safe. Once the reaction settles, the product is purified through a series of washings and recrystallizations, leaving a high-purity crystalline material. On scale-up, chemists track byproducts and emphasize solvent recovery, since costs and environmental regulations force efficiency.
Chemical Reactions & Modifications
This molecule’s amide and chloro-functional groups lend themselves to a wide array of chemical manipulations. That chlorine serves as an anchor for nucleophilic substitution: swap it for oxygen, sulfur, nitrogen, or more elaborate organic fragments. I’ve seen it used to build up more complex agrochemical scaffolds with selective modifications of the aromatic ring. The isopropyl group bestows mild steric hindrance, making certain electrophilic substitutions more predictable than in straight acetanilides. Laboratories push this compound into routes towards amine derivatives, carboxylates, or even heterocyclic ring systems with a few thoughtful steps. Access to the phenyl ring allows for advanced functionalization through Friedel-Crafts and oxidation reactions. In the hands of medicinal chemists, the platform supports hundreds of routes for new lead candidates or analogs.
Synonyms & Product Names
The world’s chemical catalogs don’t always agree on naming, so researchers spot this compound under a range of monikers. You’ll find it labeled as N-(1-Methylethyl)-N-phenyl-2-chloroacetamide or Chloroacetanilide, isopropylated. Trade names turn up on some industrial supply lists, but academics stick to IUPAC or CAS-based identifiers. For chemical inventory control, cross-referencing these synonyms with regulatory lists safeguards against both double-ordering and accidental omission of hazardous substances. From my years in R&D inventory roles, mapping these names to storage bins has become almost as important as understanding their chemistry.
Safety & Operational Standards
No matter how familiar a lab worker feels, this compound calls for reliable safety protocols. Direct skin or eye contact generates severe irritation and potential long-term effects. That means gloves, eye protection, and working in a chemical fume hood at all times. Inhalation hazards exist because crystalline dust can irritate respiratory passages, so careful weighing and flask preparation matter. Disposal in accordance with regional hazardous waste regulations prevents downstream environmental load. I’ve participated in training sessions where one lapse led to an evacuation—reaction vessels overheating or acid-base effluent mixing with leftovers—reminding everyone to respect the material. Safety data sheets guide not just the lab worker but also support staff, storage handlers, and regulators in monitoring for leaks or spills.
Application Area
This compound sees most of its application as an intermediate in organic synthesis. Agrochemicals research teams look for ways to exploit the amide’s selectivity for crops versus weeds. Pharmaceutical scientists leverage the amide backbone to create new drug precursors, especially for molecules that require structural rigidity and well-placed electron density. Some dye and pigment manufacturers also draw on its aromatic structure for novel colorants. Over the years, its adaptability has allowed small- and large-scale shops to fit it into pilot campaigns for custom chemicals, whether aimed at seed protection or preparing for active pharmaceutical ingredient validation. The diversity in its core structure means many companies keep it stockpiled for last-minute synthetic rerouting.
Research & Development
R&D teams push the limits of N-Isopropyl-N-Phenyl-Chloroacetamide every year. The near-infinite options for structural modification have left this compound as a fixture in exploratory programs for next-generation pesticides and preclinical drugs. My collaborations with synthetic teams have turned up new heterocyclic ring systems, all beginning with this amide framework. Literature continues to grow, especially as researchers craft new screening libraries for biological and physicochemical properties. Teams build combinatorial sets starting from this molecule to maximize discovery in enzymes or cell lines. The compound rarely stays static; every year brings new techniques to fine-tune reactivity, analyze structure-activity relationships, and find greener ways to make, modify, or dispose of it.
Toxicity Research
Awareness of safety isn’t limited to the bench alone. Toxicology teams have found that exposure carries risks for acute irritation and organ sensitivity in higher-order mammals. Short-term contact can harm skin and respiratory mucosa, while longer exposure in animal trials raises flags for potential hepatic stress. Regulatory agencies keep tabs on threshold exposure limits, especially as derivatives migrate towards farm or consumer settings. From my perspective, early risk evaluation protects not just lab workers but end-users as well. Advances in analytical toxicology keep refining our understanding of metabolic pathways and downstream breakdown products. Responsible researchers stay up-to-date on safety data, incorporating it into both workplace practices and end-use recommendations.
Future Prospects
Looking at the near future, the adaptability and reactivity of N-Isopropyl-N-Phenyl-Chloroacetamide guarantee it a seat in innovation pipelines across industries. Synthetic chemists crave new building blocks for greener, safer molecules, and this compound’s features align with several high-priority goals. Regulatory challenges around hazardous substances may spur new effort to minimize toxicity and increase environmental compatibility. Research teams, including several I’ve worked with, are investigating biodegradable modifications or new functional groups that degrade safely. Big data chemoinformatics puts this molecule into virtual screening programs, linking up the next round of enzyme inhibitors, specialty agrochemicals, or advanced polymer additives. From my experience, the workflow rarely stands still—once a molecule like this lands on the broader radar, its story expands in dozens of directions at once.
What are the main uses of N-Isopropyl-N-Phenyl-Chloroacetamide?
Crop Protection and Growth
N-Isopropyl-N-Phenyl-Chloroacetamide turns up on many farms. It helps create herbicides that target weeds without hurting crops. Corn and rice fields, in particular, benefit from weed killers made with this compound. The reason for its effectiveness comes from how it disrupts weed growth at the seedling stage, giving crops a better shot at soaking up nutrients, sunlight, and available space. Farmers have relied on well-studied mixes containing this component for several decades. Scientists often point to its selectivity and low risk to non-target species. It directly improves food security by helping crops compete less with weeds and more with other world markets.
Role in Pharmaceutical Synthesis
Chemists like working with N-Isopropyl-N-Phenyl-Chloroacetamide for making certain medicines. Its chemical structure fits as a starting point for producing active pharmaceutical ingredients. In academic labs and industry, teams use it to kick off complex multi-step syntheses. I've met organic chemists who appreciate the way this compound behaves in reaction vessels, especially for producing molecules with precise side chains. Some painkillers and anti-inflammatory drugs have early-stage building blocks grown from this compound. The longstanding culture of chemistry journals talks about this reagent’s value in developing drugs that matter to people suffering from everyday pain or rare disease. Safety protocols need to be tight, but professionals highlight its stability and predictability during workups.
Intermediate for Agrochemicals
Agrochemical manufacturers see N-Isopropyl-N-Phenyl-Chloroacetamide as a useful intermediate. To keep pests and fungi at bay, companies build out analogs and derivatives with complex activity profiles. Research teams worldwide use its core chemistry to branch out into designing safer or more targeted crop sprays. By adjusting a side chain or adding to its structure, chemists can quickly test prototypes before scaling up for field tests. Experience in these labs shows that developing new generation pesticides or fungicides with improved environmental profiles often starts with such trusted intermediates. Regulatory bodies demand a lot of data—this compound’s records and history in the lab tend to check important boxes.
Environmental and Health Safeguards
Like any chemical meant for fields or factories, N-Isopropyl-N-Phenyl-Chloroacetamide needs responsible handling. Seasoned applicators and production staff take cues from safety data sheets: gloves, goggles, and strict storage rules. Residue analysis ensures water, soil, and crops stay safe. Some environmental groups still call for more independent testing and transparency about breakdown products, especially in aquatic settings. Farmers, chemists, and regulators continually improve best practices to protect both food and workers. Researchers see an opportunity here to design replacements or tweaked versions of the molecule, keeping plants robust but lower on the list of long-lasting residues.
Potential Solutions and Future Insight
To answer concerns about resistance, overuse, and runoff, the industry has turned toward integrated pest management and precision application of compounds like N-Isopropyl-N-Phenyl-Chloroacetamide. Tools like drone spraying, smart sensors, and molecular tweaking promise better results and fewer environmental impacts. Real progress, though, comes from listening to independent researchers, adapting local practices, and staying open to new science on both benefits and drawbacks. Looking at the world food challenge and the demand for safer pharmaceuticals, it’s clear lasting solutions will mix old tools like this one with new, smarter thinking.
What are the safety precautions when handling N-Isopropyl-N-Phenyl-Chloroacetamide?
Taking Chemical Hazards Seriously
Lab life teaches you pretty quickly that every chemical has its story. N-Isopropyl-N-Phenyl-Chloroacetamide, often used in research, comes with its own set of hazards. This compound doesn’t get as much limelight as some others, but its potential risks demand real respect. Any substance containing both a chloro and amide group can cause trouble if handled carelessly. Breathing problems, skin burns, eye irritation — those aren’t just warning labels. After seeing a lab mate suffer a chemical splash because of a tiny oversight, my view changed permanently: nothing replaces basic safety habits.
Personal Protection Comes First
Before popping open that bottle, personal protection needs to be squared away. Splash goggles aren’t optional. Just clearing foggy glasses after handling active ingredients made me understand the importance of a good seal against fumes — not any old eyewear will cut it. Standard lab coats help, but some synthetic substances eat right through cotton. Double up with nitrile gloves, not vinyl, and never trust gloves forever. Peer-reviewed studies have shown that nitrile stands up better to chemical penetrations, so swapping gloves every hour or after a spill pays off.
Mind the Air You Breathe
Volatile chemicals don’t care about invisible boundaries. Good ventilation blocks toxic vapors from building up. Opening a bottle outside of a fume hood invites risk, especially with a chemical that can irritate airways or cause headaches. I once worked in a space with a broken hood — the headaches that followed weren’t a coincidence. Proper airflow means peace of mind. Regular checks by safety officers can catch a poorly ducted hood before an irritant grabs you. Nearly every chemical safety guide from OSHA and NIOSH echoes this: don’t underestimate what you can’t see or smell.
Storage Isn’t Just About Space
Storing N-Isopropyl-N-Phenyl-Chloroacetamide with the rest of your chemicals could turn into a serious headache. Chlorinated compounds and acids sometimes get the urge to mix badly. Keeping them on a shelf above your workspace increases the odds of an accident. I found that storing such chemicals in a locked, cool, and segregated cabinet — and labeling containers clearly — let everyone sleep better at night. This habit matters even more at home labs or smaller clinics with fewer resources.
Spill Response Saves More Than Time
The time you spend prepping for accidents long before they happen pays off. Every experienced chemist I know keeps a spill kit nearby: absorbent materials, disposable suits, and neutralizing agents. After seeing a minor spill snowball because the team scrambled for materials, I learned to keep the basics within arm’s reach. Training protects not just yourself, but your teammates. Reviewing protocol isn’t just a chore from lab orientation — it means you can act fast in a real emergency.
Building a Real Safety Culture
Staying safe with chemicals like N-Isopropyl-N-Phenyl-Chloroacetamide has always been about more than just rules. Personal accountability and regular training shape a workplace where people spot danger signs early. Encouraging problem-solving by asking “How can this be safer?” helps. Investing in clear signage, routine updates to safety data sheets, and team-wide drills aren’t extras. Safety isn’t just policy, it’s culture. Raising the bar in this way — for yourself and others — stops treatable accidents from becoming disasters.
What is the chemical structure and molecular formula of N-Isopropyl-N-Phenyl-Chloroacetamide?
Breaking Down the Molecule
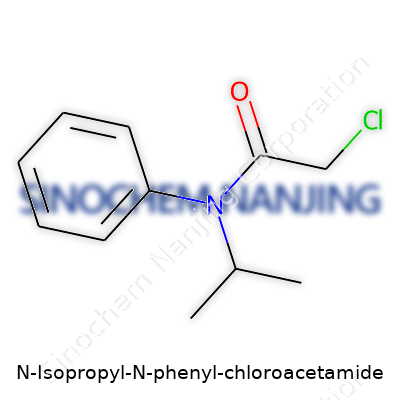
N-Isopropyl-N-Phenyl-Chloroacetamide brings together different groups in a single molecule. The backbone holds a chloroacetamide base; imagine a two-carbon chain with a chlorine atom on the first carbon and an amide group joining on the second. To that core, chemists attach two side branches—one phenyl (a ring made of six carbons in a classic hexagon, like the structure in benzene), and one isopropyl group (a three-carbon fork shaped like a small “Y”). N-methylacetamide, acetaminophen, and other well-known amide structures share some features, but this combination is far less common.
In plain chemical shorthand, its molecular formula is C11H14ClNO. The structure lines up as ClCH2CON(Ph)(iPr), where Ph stands for “phenyl” (C6H5) and iPr marks the isopropyl group (C3H7).
Use Cases and Real World Relevance
This molecule doesn’t pop up on store shelves, but its structure hints at why chemists care about it. That single chlorine atom, stuck on the acetamide’s side, acts as a chemical “handle,” allowing the molecule to extend into new compounds. Working in the lab, I’ve seen these “activated” acetamides function as building blocks for drug discovery or for new crop chemicals. Organic synthesis benefits from how such molecules can be changed; that one chlorine makes reactions move faster or in new directions.
When manufacturers need flexibility in chemical design, having a mixed amide like this lets them fine-tune a molecule’s behavior. Tweaking the isopropyl or phenyl side gives chemists a way to shift solubility, boost stability, or change what other molecules it interacts with. I’ve watched research teams shift a single side group and suddenly a chemical works as a pain reliever, or sometimes as a dye or a herbicide. The stakes get higher in drug research, where small changes in composition can make all the difference between useful medicine and something that stops working in the body.
Safety and Handling Concerns
Hands-on chemistry always demands respect for what a molecule can do. N-Isopropyl-N-Phenyl-Chloroacetamide contains that reactive chlorine; if it lands on skin or in the eyes, it can irritate or burn. There’s no reason to treat a chemical as just a pile of atoms—experience teaches that it matters to know what you’re working with and to plan how you handle spills or contact. Proper gloves, eyewear, and ventilation count for a lot. Storing it in a well-sealed bottle, far from sunlight or moisture, reduces breakdown and keeps reactions under control.
Toxicology data on this compound remains sparse in the public domain, so prudence stays front and center. Any lab with it on the bench uses proper labeling and keeps waste separately. Regulators worldwide require rigorous testing, even at the early stages, to ensure nothing slips through that could harm people, animals, or the environment. Anyone introducing new acetamide derivatives into research or commerce faces growing scrutiny, and rightly so.
Looking Forward: Practical Paths
With new molecules like N-Isopropyl-N-Phenyl-Chloroacetamide, honesty about unknowns matters as much as technical skill. Labs testing such compounds pool data, share insights, and push for safer, cleaner synthesis routes. Green chemistry, which stresses responsible choices in reagents and waste, increasingly impacts how chemists approach molecules with reactive groups like chloroacetamide.
As someone who has spent long hours troubleshooting reactions, I respect scientists and regulatory teams asking more questions before a chemical goes widespread. The path forward pairs innovation with accountability, putting health and sustainability at the center of every new research project, especially for compounds with untapped potential like this one.
How should N-Isopropyl-N-Phenyl-Chloroacetamide be stored?
Safety Starts With Knowing What's on the Label
N-Isopropyl-N-Phenyl-Chloroacetamide isn’t just a handful to say. Behind the long name, the chemical brings risks you don’t want sneaking up on workers in a lab or warehouse. I’ve seen plenty of places overlook basics just because a bottle looks sealed or harmless. Yet one cracked cap, and things can get expensive fast—not just in cash, but in health.
Temperature and Light Turn Chemicals Foul
I once worked in a lab where sunbeams reached right across a chemical bench. Trouble came soon after, with residues and labels curling. Organics, especially those holding a chlorine atom, get twitchy with heat and light. Aim for a spot that keeps things between 2°C and 8°C—basic fridge territory but not the kitchen at home. Direct sunlight or heat from radiators can kick off slow reactions, turning safe stock into a clean-up problem. Chemicals don’t get tired or worn out, but they get meaner over time from these exposures.
Airtight, Not Just Closed
I’ve come across bottles that looked fine, but odors leaked out as soon as you handled them. Ordinary screw tops sometimes let solvent vapors creep out or oxygen slip in—especially after repeated use. Go with tightly sealed containers. If the packaging looks cheap or shows cracked plastic, switch it out right away. Secondary containment, like sealed bins or trays, gives extra insurance against accidental drops or leaks. Once, a single bottle leak had us scrubbing shelves and discarding stock around it for weeks. Lesson learned: even a minor breach matters.
Get the Neighbors Right—Segregate Chemicals
Chemicals play nice until they don’t. This one holds both isopropyl and chloroacetamide groups, which tell you it’ll be happiest away from oxidizers, acids, and alkalis. We kept organics grouped apart and clear of anything reactive or flammable. At one job, someone stored acids on a shelf above solvents, which ended with both streams trickling down the same drain during a spill. Don’t repeat those mistakes. Use shelves built to handle leaks and sorted by hazard class—not by what fits where.
Clear Labels and Up-to-Date Inventory
Labels rub off. Sometimes print fades or the adhesive goes. Relying on memory for what’s inside is a way to gamble with everyone’s safety. Every container deserves a fresh label with the name, concentration, and date received. Stock rotation might feel tedious, but it beats trusting a bottle that’s been sitting untouched since last fiscal year. I stick to regular inventories, updating lists and checking for old or damaged stock. It keeps labs efficient and prevents expired compounds from sneaking back into experiments or work flows.
What Works for Disposal Also Works for Storage
If something sits in a “hazardous waste” drum after use, that’s a big clue about how it should live on the shelf. Sturdy containers, solid labeling, and physical separation help in both cases. It’s about not dropping the ball—because the rules around chemical storage always lean on common sense. I worked with folks who saw every bottle as a potential hazard, not an odds-and-ends from yesterday. That mindset kept every shift running without trips to urgent care.
Don't Treat Chemical Storage as an Afterthought
I’ve seen fiery warnings on news sites and official memos. Most problems could’ve been prevented with a cool, dark space, an unbroken seal, simple labels, and a few minutes spent planning out shelving. Keep it simple, keep it vigilant, and practice what gets overlooked in a rush: put thought and respect into every bottle, every time.
What are the potential health hazards of N-Isopropyl-N-Phenyl-Chloroacetamide exposure?
Understanding the Chemical
N-Isopropyl-N-Phenyl-Chloroacetamide belongs to a group of organic compounds that sometimes show up in specific manufacturing settings. Most folks outside the chemical industry rarely cross paths with this material, but many who work in labs, factories, or chemical storage facilities see the risk. If something can pose health problems for workers, it’s not worth turning a blind eye.
How Exposure Happens
Exposure usually happens by inhaling dust or fumes, spilling on the skin, or accidentally ingesting tiny amounts on contaminated hands or surfaces. Some processes which cut corners or ignore safety make these kinds of accidents more likely. People handling this chemical without gloves, masks, or good airflow end up at higher risk. Even a clumsy moment—leaving a bottle open or skipping the quick cleanup—can turn into a real problem.
Short-Term Hazards
Irritation stands out as the most common immediate reaction—eyes, skin, and lungs feel the impact. Red, itchy patches after a splash hint at the compound’s toxicity. Breathing fumes from even a small spill can trigger coughing or throat burning. Some workers talk about feeling dizzy or nauseous after long shifts near vapors, arguing it drags down their energy and leaves them foggy for hours afterward. These symptoms indicate that the compound attacks sensitive membranes and disrupts normal respiratory function.
Long-Term Effects
Longer exposure stories tend to paint a more troubling picture. Prolonged contact may cause skin to peel, develop sores, or feel raw to the touch. People with repeated exposure risk chronic irritation or asthmatic problems. Research points toward more complex dangers if exposure keeps happening—some lab tests on similar amide compounds have shown liver and kidney stress and immune changes in rodents, meaning organs steadily lose resilience. Scientists raise alarms because chemicals like N-Isopropyl-N-Phenyl-Chloroacetamide sometimes carry mutagenic or carcinogenic risks if used without respect for safety measures. Nobody wants to end up with a nasty diagnosis because a work process skipped a safeguard.
Who Faces the Greatest Risk?
Maintenance crews, lab technicians, and line workers often deal with the greatest hazards. They handle containers, clean spills, or work around ventilation systems. In my own years in a chemical warehouse, it wasn’t always the chemists at risk, but the guys moving crates and maintenance teams fixing broken pumps. Younger workers or trainees, sometimes unaware of strict protocols, have experienced rashes or coughing fits before even learning exactly what made them sick. Each year, workplace case reports back up the fact that hands-on staff face these risks far more than office or admin personnel.
Safety Steps and Solutions
Good practice starts with training. Hands-on demonstrations—how to suit up, clean spills, ventilate workspaces—save people from trouble. Nobody should trust broken gloves or leave open containers unattended. Emergency showers, easy-to-read signage, and regular air checks go a long way in stopping chronic exposure. Regular health checkups catch problems before they turn serious.
Lab-tested substitutes or process changes targeting lower risk alternatives deserve more attention. Smart companies push for less hazardous chemicals that fill the same role. Tighter industry regulation, combined with worker empowerment to speak up about unsafe situations, creates lasting cultural change. After all, keeping teams safe always outweighs cutting costs or skipping steps for speed.
| Names | |
| Preferred IUPAC name | N-(propan-2-yl)-N-phenyl-2-chloroacetamide |
| Other names |
N-Isopropyl-N-phenylchloroacetamide Chloroacetic acid N-isopropyl-N-phenylanilide Chloroacetic acid N-isopropyl-N-phenylamide Chlorpropham CIPC |
| Pronunciation | /ɛn-aɪ-soʊˈproʊpɪl-ɛn-ˈfiːnɪl-klɔːroʊəˈsiːtəˌmaɪd/ |
| Identifiers | |
| CAS Number | [31507-99-8] |
| 3D model (JSmol) | `CC(C)N(C(=O)CCl)c1ccccc1` |
| Beilstein Reference | 1207934 |
| ChEBI | CHEBI:91113 |
| ChEMBL | CHEMBL603716 |
| ChemSpider | 21883603 |
| DrugBank | DB08796 |
| ECHA InfoCard | 13-1-00-61748 |
| EC Number | 202-246-9 |
| Gmelin Reference | 38872 |
| KEGG | C18761 |
| MeSH | D017967 |
| PubChem CID | 36674 |
| RTECS number | KB3325000 |
| UNII | 65L74P6LLS |
| UN number | 2811 |
| Properties | |
| Chemical formula | C11H14ClNO |
| Molar mass | 197.68 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.15 g/cm³ |
| Solubility in water | Insoluble |
| log P | 2.3 |
| Vapor pressure | 0.00123 mmHg at 25°C |
| Acidity (pKa) | 14.9 |
| Basicity (pKb) | pKb = 12.85 |
| Magnetic susceptibility (χ) | -63.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.573 |
| Viscosity | 2.74 mPa·s (25 °C) |
| Dipole moment | 3.76 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 369.5 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N03GA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07, GHS05 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P304+P340, P405, P501 |
| Flash point | 135 °C |
| Autoignition temperature | 360 °C |
| Lethal dose or concentration | RAT oral LD₅₀: 640 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 640 mg/kg |
| NIOSH | BZ2975000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Chloroacetamide N-Phenyl-Chloroacetamide N-Isopropyl-Chloroacetamide N-Isopropyl-N-Phenyl-Acetamide N,N-Diisopropyl-Chloroacetamide |

