N-Hexyl Acetate: A Deep-Dive Commentary
Historical Development
N-Hexyl acetate didn’t just show up in the world of chemicals one morning. Creating and studying esters, including n-hexyl acetate, grew out of classic organic chemistry research in the 19th and 20th centuries. Chemists wanted to understand natural flavors and fragrances, and the search for reliable synthetic routes to these familiar compounds kept turning up esters. N-hexyl acetate, with its pleasant, fruity aroma, entered the radar through the exploration of minor components that give foods and beverages subtle notes—think apples, pears, even rum. Its commercial development started as the flavor and fragrance industry matured. Now, you’ll find its trace almost everywhere, from botanicals to engineered scents, with its story echoing the history of how chemistry and daily life tangle up together, guided by the needs of industry and the curiosity of researchers.
Product Overview
N-Hexyl acetate stands as a clear, neutral-colored ester. It’s valued not just for its sensory impact, but for its workhorse function in both food science and manufacturing. Producers use it to boost the taste profile of sweets and fruit-flavored snacks, and to impart cleanliness to personal care products. On the industrial end, it works great as a solvent in coatings and printing inks, able to dissolve certain resins plastics leave behind. Its application list covers both ends of the spectrum: food additives, industrial solvents, and perfume fixatives alike. Chemists pick up a flask expecting both versatility and stability from this compound.
Physical & Chemical Properties
N-Hexyl acetate appears almost like water, but with a far different scent—light, fruity, and pleasing. It sits with a boiling point just above 171°C, and a density at room temperature around 0.87 g/cm³. Solubility in water runs low, making it easier for blending with hydrocarbons or alcohols in flavor or solvent applications. Its vapor pressure sits moderate, which ties back to its role in fragrance release. As an ester, it contains both a carbonyl (C=O) and an ether (C-O) bond, leaving it stable at room temperature, but still reactive enough for further chemical work. Its flash point keeps it off the list of heavily flammable solvents, yet care remains necessary due to volatility.
Technical Specifications & Labeling
In manufacturing and packaging, n-hexyl acetate demands tight tolerances. Most commercial lots specify minimum purity at or above 98%. Labeling falls under strict guidelines for chemical identity, hazard warnings, and sometimes flavor or fragrance grade. Transport containers highlight its classification as a combustible liquid, and manufacturers often add batch tracking and expiration details. End-users expect a technical data sheet with figures for refractive index, assay, acidity, and water content, connecting lab data to the real-world outcomes for each batch. For food and flavor use, regulatory approval details get printed next to the chemical specification.
Preparation Method
Synthesizing n-hexyl acetate follows the familiar route of Fischer esterification. Chemists combine n-hexanol and acetic acid, using an acid catalyst, often sulfuric acid, to drive the process. The reaction mixture heats under reflux, sometimes with water removal to push the reaction forward. As the ester forms, separation and purification refine it further, typically using distillation to peel off impurities and voilà—high-grade n-hexyl acetate. Industrial sites scale the process up, always watching for waste minimization and consistent product quality across batches. This method stands as a time-tested way to make many esters, but it’s especially straightforward for n-hexyl acetate thanks to its manageable boiling points and reactant availability.
Chemical Reactions & Modifications
Once on hand, n-hexyl acetate stays relatively stable under typical storage, but can break down if exposed to strong bases or acids, turning back to n-hexanol and acetic acid. Hydrolysis is the main breakdown route, mirroring the reverse of its synthesis. Other labs see interest in modifying its carbon skeleton to create a wider set of esters or related alcohols, either for fragrance design or for tweaking solvent capabilities. Its acetate group gives some versatility, as related compounds like butyl or octyl acetates come from similar chemistry, just with different carbon chain lengths. This flexibility anchors n-hexyl acetate as both a standalone ingredient and a stepping stone to further chemical engineering.
Synonyms & Product Names
Chemists and buyers know n-hexyl acetate by several names. In the scientific world, it counts synonyms like acetic acid, hexyl ester, 1-hexanol acetate, or hexanol acetate. Commercial suppliers may add product codes or brand names, like “HexAcet” or “Hexestol-A.” On a regulatory front, globally recognized identifiers like its CAS number or EC number keep things clear for import/export paperwork and compliance audits. This tangle of synonyms underlines the necessity to check both chemical structure and global naming systems before making purchases or running cross-reference checks.
Safety & Operational Standards
Working with n-hexyl acetate always brings responsibility. It can irritate the skin or eyes, and its vapor, if concentrated, will upset the respiratory tract. Gloves, safety glasses, and good ventilation are basics in any lab. On a bigger scale, chemical storage standards call for cool, dry environments and separation from reactive materials like acids and bases. Transport handlers have their own set of operating rules, treating it as a flammable liquid and marking shipping documents for local fire brigades to check. Waste handling needs care too, since disposal as regular waste leads to both pollution and regulatory trouble—chemical incineration or professional recycling stand as recommended routes.
Application Area
The power of n-hexyl acetate comes alive in both sensory and industrial settings. Perfumers reach for it to introduce top notes that last just long enough to intrigue. Soft drinks and candies use it for gentle, apple-pear-like overtones that don’t overwhelm the main flavors. Print shops and paint manufacturers need it as a safe, strong solvent, capable of smoothing out resins and dyes without heavy odors or toxicity. Even the automotive sector saw growing use in eco-friendly solvents and cleaning agents. Everyone from chefs to chemists, from perfumers to process engineers, finds a reason to keep a jug of it on the shelf.
Research & Development
Lab teams keep poking at n-hexyl acetate’s properties, always searching for improved synthesis, greener chemistry, and new uses. Biochemistry labs want to design biosynthesis pathways using engineered microbes, bypassing fossil feedstocks and harsh acids. Food scientists focus on recreating natural fruit aromas more efficiently. Cleaning-product engineers test new formulations with n-hexyl acetate as a central solvent, seeking lower toxicity and better biodegradability. With demand shifting toward sustainable materials, R&D budgets increasingly turn toward routes that cut waste or offer biodegradable esters. Academic studies and white papers push this envelope, showing the importance of asking how everyday chemicals can work harder for both industry and the environment.
Toxicity Research
Toxicity remains a key sticking point. Lab rats exposed by inhalation or skin contact showed low acute toxicity, but high concentrations can still bring respiratory distress and irritation. Regulators pay special attention to chronic exposure—workers or manufacturers need tight occupational limits. In food use, experts watch for metabolites produced during digestion, monitoring liver and kidney function in animal studies. Most research points to relatively low bioaccumulation potential, but every year brings new data on safe exposure levels, competition with related solvents, and new pathways for environmental breakdown. Animal and cell culture studies track not just acute but long-term, sub-toxic effects to ensure products meet evolving safety expectations.
Future Prospects
N-Hexyl acetate holds a place in the ongoing shift toward safer, greener chemicals. Synthetic biologists want to mimic its production with bioengineered yeast, reducing both carbon footprint and hazardous waste streams. Regulatory changes are pushing bigger companies to rethink solvent use, shifting away from legacy options toward those showing lower toxicity and faster breakdown in nature. Fragrance and flavor houses follow consumer demand for “natural” labels while maintaining consistent, reliable composition, often leaning on fermentation-derived n-hexyl acetate. The journey forward will blend green chemistry, regulatory rigor, and constant quality control, as buyers—from small startups to global suppliers—push for safer, smarter, and more sustainable options in both consumer and industrial markets.
What is N-Hexyl Acetate used for?
Where You’ll Find N-Hexyl Acetate Working Its Magic
Open a bottle of perfume and you might catch a whiff of something fruity and fresh that reminds you of pears, apples, and even a touch of banana. That signature aroma often owes a lot to N-Hexyl Acetate. Perfumers like me look for molecules that fill out a scent, not just with punch, but with roundness and depth. N-Hexyl Acetate steps into this role almost invisibly—it never hogs center stage but binds the chorus of notes together. In the fragrance industry, its soft, floral, apple-pear odor shows up in everything from fancy perfumes to everyday deodorant and household air fresheners.
Beyond the world of scents, it’s a crowd-pleaser in flavor creation. The food industry adds it to candies, drinks, baked goodies, and even ice cream—anywhere a clean, sweet, juicy fruit flavor feels right. It doesn’t take much to transport a product from bland to craveable. N-Hexyl Acetate isn’t the star of any natural food, but in tiny doses, it gives that pleasant ripe-pear flavor boosted by a crisp apple tone.
Industry Relies On N-Hexyl Acetate
Factories and formulating labs like N-Hexyl Acetate because it plays well with oils, waxes, and solvents. It acts as a carrier for flavors and fragrances, helping them blend and stick where they belong, whether in soap, shampoo, or floor polish. Chemical companies take advantage of its good solubility profile. It breaks down and lifts grease in industrial cleaners. I spent some years in a contract manufacturing plant, and N-Hexyl Acetate always turned up when engineers needed a solvent that performed but didn’t reek of harsh chemicals.
In paints and coatings, this solvent helps smooth out surfaces and reduces bubbling in gloss finishes. It’s found in some varnishes, where it helps ingredients spread evenly and dry properly. Factory workers appreciate products that don’t give off aggressive fumes, and N-Hexyl Acetate earns points for its mild scent compared to most industrial chemicals.
What About Safety?
Any product touching food or skin invites questions about safety. Regulatory bodies like the U.S. Food and Drug Administration review food additives for risk. N-Hexyl Acetate appears on the “generally recognized as safe” (GRAS) list for food when used within strict limits—too much, and you only taste chemical. In a fragrance lab, proper ventilation keeps exposure low. While it's naturally found in small amounts in some fruits, pure N-Hexyl Acetate can irritate eyes or skin with heavy exposure. It also burns if mishandled, so factories keep it away from flames.
Better Choices for the Future
Every time we use chemicals, we need to ask: Is there a gentler, safer way to get the same effect? Though N-Hexyl Acetate is considered safe at low levels, people studying green chemistry keep searching for alternatives with even lighter footprints on health and the environment. Recyclable solvents and natural isolates could play a bigger role in the years ahead. For now, education around use and proper storage matters most. That means workers learn the ins and outs of chemical handling, and companies follow strict rules about emissions and waste. As customers, reading labels helps us know which ingredients we invite into our kitchens, bathrooms, and everyday lives.
A Supporting Player With Big Impact
My years formulating both fragrances and cleaning products taught me to appreciate these unsung molecules. N-Hexyl Acetate isn’t famous, but its soft fruit aroma and easy blending properties give it an essential job in many things we use and enjoy. A sharp nose can spot its influence, but for most of us, it works quietly in the background, making food taste fresher and daily chores a little more pleasant.
What is the chemical formula of N-Hexyl Acetate?
The Basics: Chemical Identity
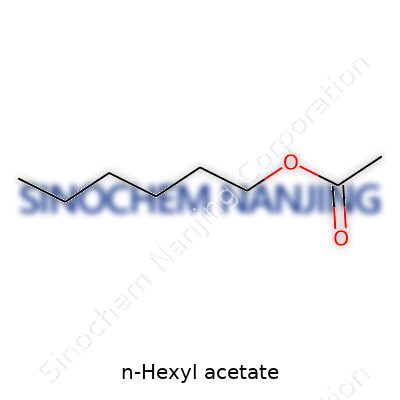
N-Hexyl acetate carries the chemical formula C8H16O2. In plain terms, it’s an ester formed from acetic acid and n-hexanol. The structure includes a six-carbon straight chain attached to the familiar acetate group. The arrangement plays a big part in this molecule’s sweet, fruity scent. Pick up a bottle of pear-flavored soda and that lifted note often owes itself to compounds like this one. You’ll also find n-hexyl acetate in perfumes, food flavorings, and certain household cleaners.
Why This Molecule Matters in Everyday Life
Fragrance industries rely on simple molecules that can evoke complex aromas. N-hexyl acetate fits this bill. The intense, almost juicy smell closely resembles natural fruits—especially pears. Pirouetting in the air, its vapor lingers softly, so perfumers and flavorists pick this ester to create or enhance desirable bouquets. In foods and drinks, small amounts add an extra layer of flavor without introducing sharp undertones.
Looking back on time in a bakery, watching workers flavor fruit fillings, I saw bottles of n-hexyl acetate sitting alongside other flavoring agents. The chemist explained that the right balance could fix a batch of bland pears, giving them an unmistakable lift that mimics freshly picked produce. That's a real-world example of food science quietly shaping our sensory experiences.
N-Hexyl Acetate in Industry
Beyond tastes and smells, n-hexyl acetate steps up as a reliable solvent. Paints, coatings, and even some adhesives benefit from its moderate evaporation rate and compatibility with both oil and water-based systems. This makes it a go-to choice during formulation work in industrial settings where flexibility and performance matter. Its relatively lower toxicity compared to many industrial solvents means less worry during manufacturing and application, keeping exposure risks in check.
Safety Concerns and Handling
No one should think that a pleasant-smelling ester is free of safety concerns. N-hexyl acetate can still irritate skin and eyes, and excessive inhalation isn’t healthy. Responsible workplaces rely on fume hoods, gloves, and proper ventilation. Training helps workers understand how to store and use such chemicals properly, cutting down on risky exposure. Regulatory bodies set workplace limits on exposure and demand proper labeling to keep everyone informed. Reading about an incident where a worker splashed solvent on their skin brought home that even familiar materials deserve respect.
Sustainability and Alternatives
As demand rises for green chemistry, manufacturers seek ways to produce n-hexyl acetate from renewable feedstocks. Modern processes sometimes use biotech routes, tweaking microorganisms to generate n-hexanol, the precursor, directly from plant sugars. This sidesteps fossil fuels, decreasing the carbon footprint of the final product. Some food scientists and fragrance experts also explore how to replicate the compound’s aroma using naturally derived alternatives, blending essential oils and extracts.
Continuous Progress in the Chemical World
Familiar molecules serve as building blocks across many fields, making daily products better. Understanding the formula behind something as unassuming as n-hexyl acetate leads to bigger questions about safety, sourcing, and the constant push toward cleaner production. Every bottle in a lab or on a factory floor reconnects people to choices about sustainability and health, making chemical formulas like C8H16O2 more than just numbers and letters.
Is N-Hexyl Acetate safe to use?
What N-Hexyl Acetate Does in Everyday Life
N-Hexyl acetate doesn’t pop up in conversation much, but anyone who’s ever opened a bottle of pear-flavored soda or cleaned with fruity-smelling products might have crossed paths with it. This chemical walks into plenty of homes through flavors, fragrances, and industrial cleaners. Companies lean on it because of a pleasant, sweet smell—something closer to pears than chemicals. That easygoing aroma draws people in, but most folks never stop to ask whether it’s actually safe to be around, inhale, or eat in small amounts.
What Research Really Says About Its Safety
Years of experience working around labs and asking toxicologists tough questions makes me look for three things: a chemical’s toxicity, how much you actually get exposed to, and what the evidence says about long-term health. Let’s get down to brass tacks. The U.S. Food and Drug Administration allows N-hexyl acetate as a flavoring agent in food, but they keep the doses tiny. The European Food Safety Authority says it doesn’t cause concern at current levels in food products, as long as the amounts stay extremely low.
Breathing in the vapors is a different story. Workers handling larger quantities in factories wear gloves and masks, not just for show, but because exposure in big doses can irritate lungs and eyes. High concentrations in the air come with headaches or dizziness. That’s common sense for most organic solvents—my own nose has felt the burn after sniffing a spilled bottle during an old college chemistry lab. Skin contact over time can dry things out or cause mild irritation. I’m careful not to brush off these signs. The quantities in home products or foods drag well below the threshold that raises concern. Still, no chemical belongs in the hands of young children unsupervised, and anyone with asthma or pre-existing sensitivities might want to steer clear of strong scents in poorly ventilated spaces.
Why Information Transparency Matters
Plenty of people get nervous about “chemical” names in products, and with good reason: safety isn’t always a given just because something smells nice. Companies and regulators bear a responsibility to show the work, not just wave it away. I track global databases like ECHA (European Chemicals Agency) and read up on reports: At low doses in food and fragrance, N-hexyl acetate doesn’t appear carcinogenic or mutagenic, based on available studies. No routine links show up between regular low-level use and chronic health outcomes in the general population.
Still, it’s not the kind of chemical you want leaking into water systems or drifting in the air around industrial plants. Careful labeling and handling instructions matter, especially since not everyone reads the fine print or takes time to search out studies. Ingredients lists hide behind vague terms like “flavoring” or “fragrance,” leaving consumers with questions and products without specifics. Full disclosure doesn’t fix every problem, but it would help shoppers make better choices—particularly in an age where allergies and environmental sensitivities affect more lives than most realize.
Shape Up Policy and Personal Choices
Experience teaches the value of reading those tiny hazard icons and learning the terms behind product names. Regulations help, but people also need basic education on what these ingredients mean for health. If you work with chemicals, push for safer workplace practices. At home, good ventilation and care with concentrated products go far. Whenever possible, choose transparency from brands—ask questions, demand details, because the right to know what’s in food, drinks, and cleaning sprays isn’t a luxury, it’s part of looking after family and community. That’s true for N-hexyl acetate—and for every other ingredient name that sparks a little curiosity or concern.
What are the storage and handling requirements for N-Hexyl Acetate?
Recognizing the Risks and Realities
N-Hexyl acetate often shows up in laboratories, factories, and within flavors and fragrance businesses. With its fruity scent, the liquid finds a home in many workspaces—but storing and handling it can bring headaches to those in charge of safety. People sometimes treat it like any other solvent, but this liquid comes with its own set of rules because of its flash point and reactivity.
Safe Storage: Staying Ahead of Trouble
Flammable liquids spark worry, and N-hexyl acetate falls into that category. Law or not, nobody enjoys the panic of an unexpected fire or chemical spill. The warehouse manager in me remembers how careless storage decisions can turn a regular Friday into a cleanup operation and a pile of paperwork. Keeping this solvent cool and away from heat stops trouble before it starts. Direct sunlight acts as the enemy here, driving containers toward unsafe temperatures and raising pressure inside drums.
Metal cabinets rated for flammable storage help, as do rooms set up with proper ventilation. Storing drums off the ground matters because concrete sweats, and moisture corrodes bulk containers over time. Pallet racking and some elevation keep drips and leaks more visible—nobody wants to discover a silent leak by the smell alone.
Chemical Handling: A Matter of Routine and Respect
Transferring N-hexyl acetate from drums or smaller cans boils down to attention and basic protection. Eye shields and gloves often prove their worth. After seeing one joiner splash a little on his sleeve, the quick dash to the eyewash station reminded me how even seasoned workers make mistakes. It irritates eyes and skin, and fumes catch up if the workplace skimps on airflow.
Leaning too close over an open container brings on headaches faster than most expect. Good local exhaust hoods stop fumes from building up around faces and workbenches. Pouring or pumping this solvent works best when everyone follows a clear routine: static discharge straps, slow dispensing, and the absence of open flames nearby. Sparks from tools or static can surprise the overconfident, leading to fires that don’t care about reputation or milestones met.
Labeling and Documentation: The Paperwork that Matters
Labels often look unimportant, but losing track of what’s inside a can brings chaos. Custodians and shift workers deserve to know the risks of what they handle, not just through training but on every container. Clear hazard warnings and up-to-date Safety Data Sheets close the gap between rules on paper and reality on the floor.
Mix-ups between similar-looking liquids almost always start with lazy labeling. My years in manufacturing have shown that strong habits beat empty reminders. Checking inventory dates and shelf lives avoids waste, saves money, and prevents unwelcome surprises when it’s time to dispose of older, unstable stock.
Reducing Environmental Harm
Spills and leaks from N-hexyl acetate don’t just risk human health. Discharges find their way to drains if floors slope the wrong way or drains go unprotected. Catch basins and absorbent kits help, but they work only if everyone knows where to find them and how to use them. Secondary containment, like bunded pallets, often stops trouble from spreading across a warehouse.
Routine inspections for rust, swelling, or leaks keep stocks safe. Disposing of residues and used absorbents in line with local regulations matters for community well-being and keeps companies clear of fines.
Final Thoughts
Treating N-hexyl acetate with care keeps workers safe and neighbors happy. The solution to safe handling starts not with fear, but with honest preparation, simple habits, and respect for every step of the process.
What is the shelf life of N-Hexyl Acetate?
Real Stories from the Storage Room
N-Hexyl acetate pops up everywhere—paints, food flavors, perfumes. My first time handling it was in a cramped college lab, the label smudged, the air tinged with that sweet, pear-like aroma. Back then, nobody on my team could say how long that chemical would hold up. Later on, after years working in chemical distribution, I learned the hard truth: shelf life isn’t just about a number. It’s also about trust, process, and, sometimes, luck.
What Actually Happens as Time Passes
Stored right, n-hexyl acetate lasts two years, sometimes longer. Oxygen, heat, and sunlight stand ready to turn it sour—literally, since acids start creeping in as it breaks down. The real risk is hydrolysis. Even a trace of water starts splitting the ester, so the liquid loses its clean aroma and reliable behavior in manufacturing. Quality sours before anyone spots the change on a spreadsheet.
Data supports this decay. Chemists measure acid value to track breakdown, and a rising number means trouble ahead. With an original purity over 98%, manufacturers promise the stuff stays stable sealed tight at room temperature and out of bright light for at least 24 months. One batch might hang in there for 30, but that requires not just a warehouse’s word, but documented storage records and responsible supply chain partners.
Why Shelf Life Calls for Respect
Anyone working with food and fragrance knows the headache that comes from using a spoiled ingredient. Even trace amounts of degradation byproducts can ruin a mix, cause off-flavors, or wreck lab results. Many companies build trust with customers based on their guarantee of quality. Fudging shelf life erodes that trust, even if no law gets broken. I once watched a client lose a contract because an old batch delivered perfume oil that soured months before it hit stores. One slip, and the reputation everyone sweated to build melts away.
Smart Moves for Long-Term Storage
Air and water always chip away at these compounds. A tight seal, dry environment, and consistent temperature near 20°C limit the damage. Stainless steel or glass wins over plastic; even good HDPE leeches trace chemicals if left too long. Each big supplier relies on periodic retesting—pulling samples, measuring the acid value, and confirming nothing’s slipped since the lot left the plant.
Logistics matters too. “First-in, first-out” isn’t just warehouse jargon; it’s necessary. Labels must list both LOT and expiry dates, not just for compliance, but because people—real humans—move jobs, take vacations, and misplace files. Tracking every step, from loading dock to bottling line, keeps surprises at bay.
What Can Go Wrong—and How to Fix It
The best fix is prevention. That’s not just my take; leading regulatory and industry bodies urge fresh air monitoring, real-time tracking, and routine staff training in chemical handling. Mistakes will still sneak in. Quick detection helps—smell, color, and titration are the frontline tools. Most quality slip-ups get caught before they hit the customer, but recycling wasted material is expensive. Erring on the conservative side isn’t just good practice; it saves serious money and avoids legal headaches later.
Looking Forward
Science keeps updating, and so should storage and tracking methods. Regular audits, creative use of automation, and a culture of speaking up when something smells off—that keeps the shelf life promise real. With climate and supply chains more unpredictable than ever, no company can afford to get lazy. That’s not overkill; it’s the baseline for staying in business and protecting real people along the way.
| Names | |
| Preferred IUPAC name | hexyl acetate |
| Other names |
Acetic acid hexyl ester Hexyl ethanoate 1-Hexanol acetate Hexyl acetate |
| Pronunciation | /ɛn-ˈhɛks.ɪl ˈæs.ɪ.teɪt/ |
| Identifiers | |
| CAS Number | 112-06-1 |
| 3D model (JSmol) | CCCCCCOC(=O)C |
| Beilstein Reference | 1208736 |
| ChEBI | CHEBI:51232 |
| ChEMBL | CHEMBL31866 |
| ChemSpider | 12929 |
| DrugBank | DB14097 |
| ECHA InfoCard | 100.011.452 |
| EC Number | EC 203-683-2 |
| Gmelin Reference | 822309 |
| KEGG | C09848 |
| MeSH | D000408 |
| PubChem CID | 8094 |
| RTECS number | AF3675000 |
| UNII | 8O0327C1Z2 |
| UN number | UN2264 |
| CompTox Dashboard (EPA) | DTXSID8021248 |
| Properties | |
| Chemical formula | C8H16O2 |
| Molar mass | 130.22 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.867 g/cm3 |
| Solubility in water | insoluble |
| log P | 2.79 |
| Vapor pressure | 0.53 mmHg (25°C) |
| Acidity (pKa) | pKa ≈ 25 |
| Magnetic susceptibility (χ) | -7.41×10⁻⁶ |
| Refractive index (nD) | 1.409 - 1.411 |
| Viscosity | 1.1 mPa·s (at 25 °C) |
| Dipole moment | 4.92 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 269.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -428.15 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3997 kJ/mol |
| Pharmacology | |
| ATC code | V03AB36 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | Precautionary statements for N-Hexyl Acetate: "P210, P233, P240, P241, P242, P243, P261, P264, P271, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 68 °C |
| Autoignition temperature | 225 °C |
| Explosive limits | Explosive limits: 0.7–7.5% |
| Lethal dose or concentration | LD50 oral rat 5,000 mg/kg |
| LD50 (median dose) | 5300 mg/kg (rat, oral) |
| NIOSH | NIOSH: MN9200000 |
| PEL (Permissible) | PEL: 50 ppm (240 mg/m³) |
| REL (Recommended) | 300 mg/m³ |
| IDLH (Immediate danger) | 200 ppm |
| Related compounds | |
| Related compounds |
Ethyl acetate Butyl acetate Isoamyl acetate Pentyl acetate Octyl acetate Methyl acetate Hexyl alcohol Acetic acid |

