N-Ethylperfluorooctanesulfonamide: Commentary on Its Role, Development, and Impact
Historical Roots and Industry Evolution
N-Ethylperfluorooctanesulfonamide, often referenced by researchers as N-EtFOSA or known among industry insiders as a byproduct of older surfactant fabrication lines, has a complicated history marked by impressive technological ambition and, later on, hard lessons. In decades past, when stain-resistant coatings and fire-fighting foams formed the cutting edge of protective chemistry, this compound piqued serious interest. Standing at the intersection of fluorine marvels and real industrial needs, it took off during a time when the world connected “performance chemicals” to progress. Technical literature from the late 20th century tells a story not just about synthesis but about how chemists viewed the very boundaries of water repellency and stain resistance. Supply chains funneled tons of this compound into factories manufacturing textiles, leather goods, and paper coatings, each business trying to improve consumer goods resilience with invisible magic. Nobody at the time, including regulatory boards, truly anticipated the long shelf life those decisions would have in the natural world.
Practical Overview and Daily Product Connections
Anyone with a few years’ experience working on the production floor or researching surface protectants has run up against this chemical’s fingerprints. N-EtFOSA gave raincoats, carpets, and food wrappers a sort of permanent shield against moisture and grease. Walk down any grocery store aisle in the 1990s or 2000s, and you saw the real results without thinking much about the chemistry behind it. Manufacturers relied not only on its ability to form flexible, thin layers but on its endurance through repeated wash cycles, temperature swings, and exposure to oils. As I found during a stint consulting for packaging suppliers, food compliance meant more than just barrier strength—it meant confidence that pizza boxes or fast-food wrappers would never break down in a consumer’s hands. That reliability, achieved with very little actual product per box, made it even more attractive to producers looking to trim costs and meet aggressive client expectations. People rarely discussed what happened after these wrappers made their way into the landfill or water table, though.
Physical and Chemical Realities from the Lab Bench
The chemistry of N-Ethylperfluorooctanesulfonamide fascinated our lab teams not just for its mouthful of a name but for how it behaved in testing. At room temperature, this compound sits in a white to off-white powder or waxy solid form, with a faint chemical odor—a sensory detail that anyone who’s ever opened a fresh drum in a pilot plant can describe. It dissolves in most organic solvents but shrugs off water, and that feature alone drove its explosive adoption in waterproofing jobs. Its molecular structure, bristling with fluorine atoms linked to a sulfonamide group, grants it near invulnerability when it comes to heat, acids, or UV light. Even under harsh processing conditions used in plastic molding or paper manufacturing, it held its ground. Colleagues trying to tweak the pH of solutions or run accelerated aging tests bumped up against just how persistent its perfluorinated chain really was—it didn’t just outlast the competition, it outlasted most other chemicals on the bench.
Technical Data, Labeling, and Industry Requirements from the Field
On the warehouse rack, barrels labeled with proper hazard codes and regulatory warnings delivered more than regulatory compliance—they warned plant workers that this wasn’t an ordinary feedstock. Product datagrams usually note a melting point above 50°C, an impressive boiling range north of 200°C, and minimal volatility at room conditions. Working at a mid-sized specialty chemicals plant, I remember the level of training that preceded any transfer or weighing job. Material Safety Data Sheets (MSDS) spelled out both the promise and the risk: thermal stability offered long product shelf life, but transport regulations treated this material as potentially hazardous to aquatic life. Operators learned quickly that slip-ups didn’t just invite chemical burns or leaks—they posed long-term cleanup headaches, both for health and for environmental reasons. Clear technical details, accurate safety labeling, and batch traceability separated responsible handlers from the shortcut takers.
Making N-Ethylperfluorooctanesulfonamide: Preparation and Cost Factors
Commercial production of N-Ethylperfluorooctanesulfonamide draws from some of the most intense synthetic chemistry practiced outside of pharmaceuticals. The go-to route starts with perfluorooctane sulfonyl fluoride, which gets reacted with ethylamine under tightly controlled conditions. This synthesis demands high temperatures, careful venting of byproducts, and vigilance to prevent worker exposures. Costs rack up fast—not just from specialized reactors resistant to fluorine corrosion, but from waste treatment requirements and handling strict emissions caps enforced since the 2000s. During my time as a junior process chemist, scouting for alternative methods or greener catalysts wasn’t just a nod to sustainability—it was a search for economic survival as regulatory fees and raw materials soared. Every operator along the line had a stake in minimizing spills and off-spec batches, which turned into costly reprocessing jobs and sometimes ugly discussions with waste contractors.
Synonyms, Product Names, and Navigating Regulatory Databases
Any researcher investigating the global market, or even a procurement manager looking for the right grade, eventually compiles a list of alternate identifiers for N-EtFOSA. Some databases and safety registries keep track of aliases such as NEtFOSA, perfluorooctanesulfonamidoethane, and even regional trade names. Each carries important differences, from small tweaks in purity to company-specific stabilizers. Navigating such a tangle of names became second nature after years of searching regulatory databases, particularly when cross-checking against European REACH listings or US EPA chemical inventories. Not only did this help avoid duplicate orders or regulatory violations, but it also spotlighted how product harmonization lags behind actual supply chain reality. Some smaller suppliers overseas continue to circulate less-documented versions, sparking compliance worries for importers.
Operational, Storage, and Worker Safety on Site
From an operational standpoint, workplace safety isn’t about posters—it’s about habits and memory, reinforced by close calls. At larger production sites, strict access controls separate handling and weighing rooms from general warehouse space. Respiratory protection, nitrile gloves, and full-face shields became part of my daily uniform, and fire suppression systems in these buildings always incorporated both chemical foam and conventional sprinklers, reflecting the dual threats of organic solvents and stubborn perfluorinated powders like N-EtFOSA. Even simple tasks, such as decanting barrels for day use, required double-checks of air flow and prompt cleanup of even small spills. This discipline grew more intense as safety studies flagged possible links between occupational exposures and troubling symptoms—no crew wanted to face investigation by regulators or health authorities after a preventable incident. Senior operators enforced logbook entries and shift checklists, not as red tape, but as insurance that nobody would leave a shift with unexplained rashes, headaches, or in the worst case, respiratory irritation.
From Raincoats to Circuit Boards: Where It Makes the Difference
The reach of this chemical stretches across industries often overlooked in environmental debates. N-Ethylperfluorooctanesulfonamide found homes not just in consumer textiles and paper coatings but across electronics fabrication, oil recovery, and even lubricants for machinery exposed to harsh industrial conditions. Early fire-fighting foams used by airport emergency crews depended on its unmatched capacity to suppress flammable vapors—something that safer replacements still struggle to rival in some scenarios. In my own work with industrial hygiene teams, I saw how electronics manufacturers increased product yields as this compound improved photoresist etching baths, shaving losses and allowing finer circuit patterns without messy breakdowns or residue. Its value, measured in increased uptime, reduced scrap, and consistent results, made it an industry favorite until mounting health and environmental concerns forced a shift toward substitutes, sometimes with a hit to product quality or cost structure.
R&D on a Tightrope: Searching for Better Answers
No conference on advanced materials or sustainable chemistry goes without a debate on what comes next. From university research groups to corporate R&D wings, the hunt for replacements runs in parallel with efforts to better understand lingering contamination. Researchers, myself included, often run against the same stubborn problems: how to match the water-and-oil resistance offered by perfluorinated sulfonamides without saddling the world with the same headaches. Recent studies, published on both sides of the Atlantic, look at biodegradable substitutes, dynamic covalent chemistries, and radically different approaches like nano-coatings. These aren’t just theoretical events—funding keeps going into field pilots, government-backed innovation labs, and start-ups banking on consumer pressure for cleaner alternatives. Yet every test that comes close to matching performance seems to stumble on cost, durability, or scale, showing just how high the bar sits.
Research on Toxicity: No Simple Answers
Toxicological reviews on N-Ethylperfluorooctanesulfonamide bring out hard truths with each new wave of journal articles. Animal studies proved what many suspected: this class of compounds does not break down easily. Early bioassays showed persistence in liver and kidney tissue in lab animals, raising flags for possible human impacts. Follow-up work tied higher blood levels in manufacturing workers to specific health symptoms, and wider environmental sampling started to pick these molecules out of remote lakes and arctic wildlife, far from any chemical plant. Managers reading through the latest regulatory advisories soon realized the risk portfolio went beyond factory gates and cleanup budgets—the legacy of widespread use meant groundwater and even food chains could carry low but steady loads for years. Many sites went through costly site remediation, with soil incursions prompting long-term monitoring and, in some cases, whole-slab removal or state-mandated filtration. Lately, regulatory handbooks treat this family of chemicals with a seriousness similar to PCBs or dioxins, with allowable residue thresholds shrinking as new detection methods pull up more sensitive numbers.
Looking Forward: Hard Choices and Real Accountability
Communities around old production facilities, both in North America and Europe, keep track of regulatory changes to N-Ethylperfluorooctanesulfonamide with a sharp interest born out of hard experience. In my conversations with city water managers and local public health staff, the emphasis comes back to accountability—not just controlling current emissions but funding cleanup of what’s already out there. The future for this compound, like many other so-called “forever chemicals,” seems to be one of managed phase-out, relentless monitoring, and enforced substitution by newer, hopefully safer chemistries. The right path won’t just show up from more lab data or tighter laws—it depends on real collaboration between regulators, producers, researchers, and communities already living with the results of past decisions. That means more than just shifting to new molecules; it calls for open audit trails, increased worker safety, and honest reporting on health impacts. As public scrutiny continues, the world won’t ignore either the marvel or the fallout of this compound’s journey from lab benchtop to landfill and, sometimes, drinking water taps.
What is N-Ethylperfluorooctanesulfonamide used for?
What’s the deal with this chemical?
N-Ethylperfluorooctanesulfonamide (sometimes called N-EtFOSE) isn’t just some obscure compound from a chemistry textbook. It pops up in places that are downright ordinary, and that's what makes it worth some honest attention. This chemical grew in use throughout the ‘80s and ‘90s, landing in everything from stain repellents for carpets to firefighting foams. Companies loved it for the same reason parents love easy-clean surfaces — nothing sticks, water beads up, and stains don’t stand a chance. If you’ve ever spilled grape juice on a “stain-proof” rug, you were likely relying on chemistry like this.
The Everyday Impact
N-Ethylperfluorooctanesulfonamide doesn’t fade away after it’s used. It lingers. That means years after that sofa gets tossed, this chemical can still show up in landfill runoff, water, or even in wildlife far from suburbia. The molecular structure — loaded with tough carbon-fluorine bonds — resists breaking down. Even after decades, scientists keep finding traces in lakes and even in some drinking water supplies.
I spent a few years working in water quality testing, and we’d get samples that shouldn’t have any connection to PFAS chemicals (N-EtFOSE belongs to this group), yet there they were, in tiny but stubborn amounts. That’s the kind of persistence that gets regulators nervous because we’re all potential drinkers and bathers, not just chemical engineers.
Where it Shows Up
Before anyone knew better, products like Scotchgard and other coatings used this compound. That meant sofas, jackets, popcorn bags, even some cosmetics packed a little bit of N-Ethylperfluorooctanesulfonamide into people’s homes. Firefighters depended on foams laced with it to smother burning fuel. Industry found it handy for making things resistant to water and grease, especially during an era that prioritized convenience over long-term risk.
Health and Environment Concerns
Research started catching up with reality. Studies flagged that N-Ethylperfluorooctanesulfonamide can accumulate in human tissue. Lab tests on animals raised alarms about liver and kidney effects. Further data suggested that the compound crosses over into breast milk and can reach infants, which is pretty unsettling. The EPA keeps tracking new data, and countries including the US and those in Europe responded by setting stricter rules or banning new uses.
Wildlife studies show some sobering trends. Birds and fish living near industrial zones tend to build up the compound in their bodies. Some researchers have pointed toward possible reproductive problems in animals linked to this exposure. For a country that values clean water and safe food, ignoring these connections makes little sense.
Where Do We Go From Here?
Industry started swapping out N-EtFOSE for newer chemicals thought to break down faster. That is only part of the answer. I’ve learned from my time in environmental labs that solutions need a double dose of science and common sense. We should keep tight oversight on chemicals used in stuff that washes down the drain or ends up in the garbage. Consumer labels can go further to tell people what’s in each product, so folks know what they’re bringing home. Old household goods with stain-proofing shouldn’t land in regular dumps. Hazardous waste collection or take-back programs offer better paths.
Nothing replaces vigilance. Scientists, government agencies, and the public all have stakes here. Chemicals like N-Ethylperfluorooctanesulfonamide serve as reminders — what gets designed for convenience can stick around, and that persistence ends up on everyone’s tab.
Is N-Ethylperfluorooctanesulfonamide hazardous to health or the environment?
The Real-World Impact of N-Ethylperfluorooctanesulfonamide
N-Ethylperfluorooctanesulfonamide—often shortened to NEtFOSA—crops up most often in the story of older stain-resistant coatings and firefighting foams. People don’t see this chemical in everyday life, but that doesn’t mean its footprint’s small. If you’ve heard of PFAS, sometimes called “forever chemicals,” this compound falls within that group. Its unique properties made it valuable, but they also shaped its risk profile, both to health and the places we live.
Stubborn by Design
I learned firsthand about environmental stubbornness after moving near a river with a legacy of chemical dumping. Like other PFAS, NEtFOSA doesn’t easily break down. The chemical hangs around for decades—maybe longer—in soil and groundwater. Not only does it stick around, but it travels. Small amounts on a factory site or a landfill drift through water, sometimes surfacing far downstream. Drinking water systems, agricultural land, and even household dust pick up a share along the way.
Health Risks—What the Research Tells Us
Through studies by the EPA and researchers in Europe, we’ve learned that exposure to NEtFOSA and chemicals like it brings a mix of concerns. Lab tests have shown liver and thyroid problems in animals, though data in people aren’t as complete. Still, a pattern stands out: PFAS show up in blood samples almost everywhere scientists look. Early science links high levels to changes in cholesterol, immune system disruption, and—in some populations—problems with infant birth weight and cancer risks.
Environmental Effects Aren’t Confined to Water
After more than a decade of reports from local fishers and water quality teams, it’s clear wildlife can’t dodge these chemicals either. NEtFOSA accumulates in fish and birds, climbing higher in the food web as bigger animals eat smaller ones. Scientists found it in polar bears and eagles. These animals never saw a factory, but they carry the chemical inside them, showing just how far and wide the reach extends.
Why This All Matters to Communities
Most people worry about what goes into their water or air, but NEtFOSA’s story shows why it’s about more than one product or pollution site. It’s about trust in what’s supposed to protect us. Once such a chemical enters a community, removing it becomes nearly impossible with today’s water treatment. The sense of security fades, especially when parents learn about forever chemicals building up in local water or playgrounds.
Better Choices for the Future
A growing number of scientists and advocates have pushed for tighter controls on PFAS and related compounds. Several countries have phased out or greatly restricted NEtFOSA use. Groups like the Interstate Technology & Regulatory Council and the Environmental Working Group urge more transparency, faster cleanup, and innovation in safer alternatives. My own experience in community action tells me local voices play a big role. Testing water, pushing for bans, and sharing honest information forces lawmakers and companies to act.
Building on Lessons Learned
Chemicals like NEtFOSA teach tough lessons. Real progress comes from listening to people affected, taking science seriously, and not settling for quick fixes. Safer products and stronger rules protect not only our health, but also our confidence in the places we call home.
What are the typical applications of N-Ethylperfluorooctanesulfonamide?
Navigating Chemical Utility in Everyday Life
People rarely hear about N-Ethylperfluorooctanesulfonamide (often shortened to NEtFOSA), but its footprint appears in places most folks wouldn’t expect. Having some years in environmental science, I’ve seen how specialized chemicals shape products we rely on daily. NEtFOSA forms the backbone of several performance-enhancing additives. Even when you don’t notice it, it quietly transforms the way coatings, foams, textiles, and firefighting products behave.
The Backbone of Stain and Water Resistance
Over the last few decades, furniture and carpets have boasted “stain resistant” claims. Dig into those features and you’ll find compounds like NEtFOSA. This molecule stands out for its unique ability to repel both oil and water. Carpets, upholstery, and even some outerwear leverage this chemical so spills don’t soak in right away, offering people—especially parents with young children—a fighting chance to clean up before a permanent mark sets in.
From personal experience, nothing beats knowing your new white couch can survive a spill or two. In the textile industry, NEtFOSA-based treatments offer a practical upgrade to everything from rain jackets to seat cushions. Companies use it to keep their products cleaner and less prone to stains, which translates to fewer replacements and less waste. Many outdoor enthusiasts trust this kind of chemical help when miles from home, counting on their gear to repel rain, mud, and oil.
Boosting Firefighting Efforts and Industrial Safety
Not all the magic happens in the living room. Firefighting foams have relied on chemicals derived from NEtFOSA for their knack at spreading evenly over flammable liquids. This matters any time a fuel spill ignites on a tarmac or industrial site—foam cuts off oxygen more efficiently, containing fire and giving emergency crews a fighting edge. Such performance isn’t just a technical win; it saves lives and reduces property loss, an impact you can’t overstate.
In electronics and plating factories, NEtFOSA again makes its mark. Manufacturers add it to chrome plating baths and other processes, since it helps suppress mist creation and reduces worker exposure to hazardous materials. Manufacturing jobs come packed with enough risks; if a chemical can cut down breathing in something dangerous, companies have a responsibility to step up. My visits to older plants showed that every layer of added safety matters, and chemical controls like these belong in discussions about worker protection.
Environmental Challenges and Safer Paths Forward
There’s a hard truth with many perfluorinated chemicals like NEtFOSA: they last. They stay in the environment long after their useful life, which leads to buildup in water and living tissue. This persistence brings real concerns—studies link similar compounds to health issues. Calls for stronger regulation have grown as scientists gather more data. Some companies now look for alternatives or safer methods to get the same effect without the risks.
Switching to newer, less bioaccumulative substances, tightening industry standards, and expanding water-treatment systems can help limit harm. Consumers can pay attention to product labels, and pressure brands to step up transparency around chemical use. Everyone along the chain—from chemical labs to home users—plays a part in balancing performance with responsibility.
How should N-Ethylperfluorooctanesulfonamide be stored and handled?
Recognizing the Stakes
I’ve watched labs, warehouses, and factories act casual with specialty chemicals, only to pay for small mistakes. N-Ethylperfluorooctanesulfonamide—commonly known as NEtFOSA—does not forgive neglect. It appears as a powder or a solid with a reputation for chemical toughness thanks to its fluorine-rich structure. Industries have counted on it for water-repellent treatments and surfactants since the late 20th century, but its persistence in the environment and the risks it can pose inside storage rooms invite respect.
Respecting Chemical Stability
This material doesn’t break down easily, but that doesn’t mean anything goes. I’ve always told new technicians: lock volatile fluorochemicals and strong acids behind reinforced doors. Keep NEtFOSA completely dry and sealed tight in high-density polyethylene, glass, or fluoropolymer containers. It seems like overkill until you consider what happens when water, high heat, or reactive chemicals cross paths with NEtFOSA. Tanks, drums, and even bench bottles should stay out of direct sunlight; move them as far from steam pipes and hot process lines as possible. Chemical stability lasts longest out of the light, at cool to moderate room temperature.
Thinking Through Containment
Spillage changes everything in a hurry. Perfluorinated compounds don’t simply wash away, and they follow anyone who forgets simple things like secondary containment trays. I always put NEtFOSA on a dedicated shelf—never on the floor, and never above head height. Fumbling with a container or dropping a bag seems rare until you see a sour smell drifting across a whole floor; then, everyone pays attention. Waterproof gloves, safety glasses, and a fitted lab coat stay non-negotiable with this stuff. In bigger facilities, air extraction systems and chemical fume hoods keep accidental dust or vapor from spreading into shared spaces.
Training Makes the Difference
Experience tells me safety data sheets only go so far. I’ve seen experienced workers freeze up the first time they handle a powder like NEtFOSA. It’s worth taking a few extra minutes in the morning to review transfer steps and double-check respirator fit. Label every container, and mark the chemical’s hazards using GHS pictograms. People forget, and labeling never does. Run drills for spill cleanup—never just talk about the theoretical steps. Dedicated disposal bins for contaminated gloves and wipes cut down chaos during a real incident.
Controlling Exposure Risk
Long-term health questions surround many old PFAS chemicals, including NEtFOSA. Dozens of scientific reports point out links to organ toxicity and environmental persistence. I’ve learned that ventilation wins every time. Never cut corners by pouring or weighing this chemical near food, drinks, or break rooms. Keep eating areas in another building if possible. Wash hands thoroughly before leaving the storage zone. With sheets of hard data and real-world cleanups under my belt, I always push management to plan for the material’s lifetime—from delivery to waste drum—because small exposures can add up in surprising ways.
Pushing for Better Habits
Regulators track these compounds more closely every year. That’s not just bureaucracy at work—it reflects lessons learned the hard way by workers and communities. Safety never really stays static. Audit your storage routines. Rotate backup gloves and safety supplies. Push for worker feedback on new ways to make containment and cleanup easier. NEtFOSA calls for a thoughtful approach from everyone on the site. Ignoring those lessons never ends well, not for people, not for property, and definitely not for the surrounding environment.
Is N-Ethylperfluorooctanesulfonamide regulated or restricted in any countries?
Why Regulators Watch N-Ethylperfluorooctanesulfonamide Closely
Most people have never heard of N-Ethylperfluorooctanesulfonamide, often called NEtFOSA. This chemical belongs to the PFAS family—often tagged now as “forever chemicals.” These substances don’t break down easily and linger in water, soil, and our bodies. I stumbled across PFAS years ago, working in an environmental lab, and realized just how tough they are to remove from wastewater. NEtFOSA, specifically, pops up in firefighting foam, carpet treatments, and stain resistance products. That convenience comes at a price: it hangs around long after its job is done.
Regulations Around the World
The European Union treats NEtFOSA with caution. Under REACH, which tracks and limits hazardous chemicals, NEtFOSA and a handful of PFAS relatives face strong controls. Use in consumer products gets clamped down. The EU’s Stockholm Convention listing for these chemicals means no more manufacture or sale for most applications. Even imported goods have to show compliance, a reality that shakes up supply chains. As someone who’s worked on regulatory compliance, staying ahead of these rules means the difference between doing business freely and facing product recalls.
Looking at North America, Canada put NEtFOSA and its breakdown products like NEtFOSAA and NEtFOSE under its Prohibition of Certain Toxic Substances Regulations. It’s not allowed in goods imported to Canada or made within the country. The United States has taken steps too. The EPA keeps a tight leash on manufacturers and importers through significant new use rules (SNURs). If a company tries to bring NEtFOSA or similar chemicals into the market for new uses, they have to notify the EPA first and often clear tough safety hurdles.
Why Some Countries Move Faster Than Others
Regulation speed depends a lot on local concerns. European countries faced strong public pushback after PFAS contamination stories hit the news. The drinking water crisis in some small towns made politicians act fast. In the U.S., state-level bans often move ahead of federal action. Michigan and New York, for example, target PFAS in firefighting foams and consumer goods. For many nations with fewer resources, PFAS issues fly under the radar. Lack of testing capacity and public awareness means fewer restrictions, at least for now. I’ve seen factories in Southeast Asia still use PFAS-based supplies because they cost less and local rules don’t block them yet.
The Health and Environmental Stakes
People worry for good reason. NEtFOSA and its cousins resist breaking down in the environment. Those chemicals enter drinking water and build up in wildlife and people. Studies link PFAS exposure to problems like immune system suppression, thyroid disruption, and certain cancers. I’ve met folks living near contaminated sites who worry every time they pour a glass of water. Limiting these chemicals goes beyond technical debates; it’s about real risks for people and ecosystems.
Better Choices and Possible Solutions
Companies aiming for safer products already look for PFAS alternatives. Restaurants switch to plant-based fiber containers, and fabric producers test new coatings. Governments fund research on better disposal methods and reliable detection techniques. But cost and performance challenges persist, especially in industries reliant on the unique properties of PFAS. Strong monitoring, clear labeling, and global cooperation help close loopholes, making it harder for banned chemicals to get shuffled across borders.
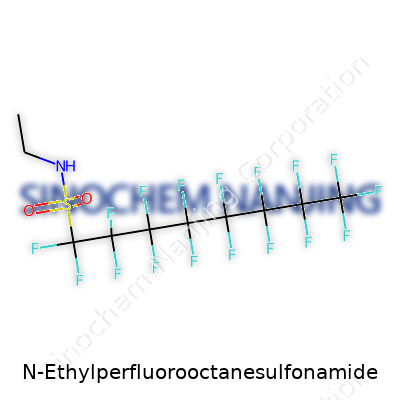
| Names | |
| Preferred IUPAC name | N-ethyl-1,1,2,2,3,3,4,4,5,5,6,6,7,7,8,8,8-heptadecafluorooctane-1-sulfonamide |
| Other names |
NEtFOSA N-ethyl perfluorooctane sulfonamide N-ethylperfluorooctanesulphonamide Perfluorooctanesulfonamide, N-ethyl derivative N-ethyl-PFOSA |
| Pronunciation | /ˌɛnˌɛθɪlˌpɜrˌflʊəroʊˌɑkˌteɪnsʌlˈfɒnəˌmaɪd/ |
| Identifiers | |
| CAS Number | 2991-50-6 |
| Beilstein Reference | 85937 |
| ChEBI | CHEBI:132709 |
| ChEMBL | CHEMBL36379 |
| ChemSpider | 84629 |
| DrugBank | DB01575 |
| ECHA InfoCard | 100.102.457 |
| EC Number | 206-203-2 |
| Gmelin Reference | 88108 |
| KEGG | C19609 |
| MeSH | D020087 |
| PubChem CID | 21740 |
| RTECS number | GO1100000 |
| UNII | Z8N2I3XWGC |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | urn:epa.comptox/dashboard/DTXSID9020835 |
| Properties | |
| Chemical formula | C10H6F17NO2S |
| Molar mass | C10H6F17NO2S, 499.20 g/mol |
| Appearance | White to off-white powder |
| Odor | Odorless |
| Density | 1.7 g/mL |
| Solubility in water | Insoluble in water |
| log P | 2.8 |
| Vapor pressure | 1 mm Hg (at 20 °C) |
| Acidity (pKa) | 5.3 |
| Basicity (pKb) | 5.3 |
| Magnetic susceptibility (χ) | -67.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.355 |
| Viscosity | 13.6 mPa·s (25 °C) |
| Dipole moment | 3.5631 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 519.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1530.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1355.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C08GA01 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P273, P280, P304+P340, P312, P337+P313 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Flash point | 134°C |
| Lethal dose or concentration | LD50 oral rat 1200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 241 mg/kg |
| NIOSH | KN4900000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for N-Ethylperfluorooctanesulfonamide: Not established |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonamide Perfluorooctanesulfonyl fluoride Perfluorooctanesulfonic acid N-Methylperfluorooctanesulfonamide |

