N-Benzyl-N-Ethylaniline: A Closer Look at a Chemist’s Old Familiar
Historical Development
Long before niche specialty chemicals earned their place in major pharmaceutical or industrial toolkits, N-Benzyl-N-Ethylaniline made its quiet debut in labs exploring aniline derivatives for new applications. Chemists in the early 20th century started tinkering with these structures, looking for cleaner, more workable pathways for synthesis and modification across fields ranging from dyestuffs to medicine. N-Benzyl-N-Ethylaniline stepped out quietly through the work of researchers mapping out the vast landscape of substituted anilines. Over time, its name turned up in research journals, especially in papers discussing intermediary products for more complex organic syntheses. Early researchers weren’t looking for media attention. They just wanted to see if tweaking aniline with benzyl and ethyl groups opened up routes you couldn’t get from the parent compound. In the end, their efforts paid off. You can trace the spread of this compound’s influence across patents filed in Europe and the United States, usually buried in lists of reaction intermediates or targeted modifications, building blocks in larger chemical adventures.
Product Overview
N-Benzyl-N-Ethylaniline has a structure brimming with possibilities: morphing from a straightforward lab reagent into a foothold for advanced organic chemistry. Built around an aniline backbone with added ethyl and benzyl groups attached to the nitrogen, the molecule pops up in a surprising number of chemical conversations. Labs and workshops reach for it when they want something a little more versatile than plain aniline, benefiting from changes in its reactivity and physical traits. In day-to-day terms, this compound helps chemists solve practical problems by making certain syntheses a bit more accommodating, especially in dye, pharmaceutical, and advanced material projects.
Physical and Chemical Properties
Step into any lab and N-Benzyl-N-Ethylaniline stands out, mostly as a clear or yellowish liquid, a bit denser and oilier than water. With a melting point well below room temperature, it keeps its fluid nature over a wide range—handy for syntheses. Its relatively high boiling point and low solubility in water steer its use toward nonpolar systems, and it mixes easily with a range of organic solvents, including ether and chloroform. Thanks to its aromatic rings, the compound often gives off a faint, recognizable amine smell that signals its presence even before the bottle’s fully open. The chemical stability here lets it tolerate mild acids and bases; still, it doesn’t take abuse kindly from strong oxidizers.
Technical Specifications and Labeling
In a laboratory context, N-Benzyl-N-Ethylaniline typically arrives labeled with its CAS number and an unambiguous chemical structure printed right on the bottle for easy reference. Purity specs usually sit above 98 percent for research and more demanding industrial uses. The compound’s lot number and production date often get scrawled onto paper or digital batch records, essential for maintaining traceability during audits or research replications. Technical grades rarely contain more than trace anilines or related contaminants, since those could interfere with downstream reactions, especially for pharmaceutical labs where impurity profiles matter as much as yield.
Preparation Method
N-Benzyl-N-Ethylaniline rarely appears on its own in nature, so synthetic routes drive its supply. Most chemists lean toward alkylation reactions for its preparation, a method that typically involves starting with N-ethylaniline and carefully adding a benzyl halide under basic conditions. The setup resembles so many foundational reactions taught in organic chemistry courses, though scale and cleanup steps get more demanding as batches grow bigger. The real art sits with the choice of solvent and temperature, a balancing act between getting the reaction to completion and avoiding the kinds of side products nobody wants to purify away at the end. After the reaction, extra care goes into distillation or crystallization to ensure product purity.
Chemical Reactions and Modifications
This molecule opens doors for further chemistry, acting as both a substrate and intermediate. Its structure, with two substituents on the nitrogen, changes traditional reactivity, making it a bit more resistant to some reactions while offering new attachment points for others. Electrophilic aromatic substitution on the aniline ring, for example, needs the right catalysts and milder conditions; the nitrogen’s bulkier substitution slows down reactions like nitration or sulfonation. On the flip side, the benzyl and ethyl groups can serve as launching pads for more ambitious modifications—think oxidative cleavage, halogenation, or even dealkylation, depending on what the downstream process requires. All these possibilities explain why you’ll spot reference to this compound in patents and journals relating to the creation of tailored chemical libraries.
Synonyms and Product Names
Though most chemists stick to calling it N-Benzyl-N-Ethylaniline in the lab, you may see synonyms like N-Ethyl-N-Phenylmethylaniline or even casual shorthand—“Benzylethylaniline”—in notebook margins. These alternate names show up in supply catalogs and chemical registries, especially when searching cross-border or historical documentation. Each iteration hints at the compound’s structural motif, making it easier for anyone scanning large compound libraries to spot functional similarities across different naming conventions. This naming versatility helps scientists avoid confusion as terms shift over time and between suppliers.
Safety and Operational Standards
Any chemical with an aniline base brings a history of hazards, both in the workplace and outside. N-Benzyl-N-Ethylaniline doesn’t disappoint. The aromatic rings and amine backbone hint at potential concerns: absorption through the skin, vapor inhalation, and risk of longer-term exposure effects. While not as notorious as some aromatic amines, the compound gets handled with the same caution—personal protective equipment, chemical fume hoods, and strict protocols on storage. Organizations like OSHA and international hazard labeling systems demand clear warning statements. Waste disposal follows local and international regulations to prevent environmental contamination. To build a culture of safety, labs lean into practical measures: clear labelling, regular safety audits, and staff training to reinforce best practices.
Application Area
N-Benzyl-N-Ethylaniline finds its utility in diverse fields, mainly as a building block rather than a final product. Historically, chemists in the dyestuff industry relied on it to create intermediates for colorants with tailored properties, especially when the shift to synthetic dyes put pressure on manufacturers to innovate quickly. Pharmaceutical research groups see value in its reactivity, using it to try new scaffolds, especially when hunting for bioactive molecules with modified toxicity or solubility. Materials scientists exploring conductive polymers or new adhesives also bring this compound to the table, seeking subtle tweaks in backbone flexibility or electron transport characteristics. Across every field, its core appeal rests in being more than a simple amine, allowing chemists to test creative reaction strategies on a stable platform.
Research and Development
In my own work, running exploratory reactions for pharmaceuticals and functional materials, N-Benzyl-N-Ethylaniline’s reputation always arrives before the bottle. Academic and corporate labs worldwide tap its potential as they test new reaction methodologies—organocatalysis, photo-induced transformations, transition metal cross-coupling. Several published works cite it as a model substrate for optimizing yields, especially in processes where steric hindrance from the nitrogen substituents acts as a proving ground for new catalyst activity. As researchers look to expand green chemistry, interest grows in finding renewable feedstocks for its synthesis, moving away from petroleum-derived inputs. The compound’s adaptability continually raises new questions, fueling the next round of investigation.
Toxicity Research
Much of what we know about N-Benzyl-N-Ethylaniline’s toxicology comes from broader studies into aniline derivatives, a group with a mixed reputation for causing everything from acute irritation to longer-term organ effects. Exposure studies have highlighted the compound’s capacity for mild acute toxicity in animal models, especially upon inhalation or skin contact, though the compound doesn’t seem to bioaccumulate in ways that worry most toxicologists. Regulatory agencies often set occupational exposure limits based on broader aniline family data, nudging chemists toward robust engineering controls and careful monitoring. Ongoing studies keep a close eye on metabolites and breakdown products, hunting for any surprises in the way the human body handles this molecule.
Future Prospects
Looking ahead, N-Benzyl-N-Ethylaniline’s story is far from over. Researchers in the pharmaceutical and specialty materials fields show no signs of slowing down their use of this molecule as a foundation for new syntheses. With green chemistry pushing everyone toward more sustainable processes, there’s growing interest in better, safer synthesis routes using renewable reagents and minimizing hazardous waste. Advances in catalysis could soon make its preparation more efficient, even at large industrial scale. At the same time, the drive to understand long-term health and ecological risks keeps attention on finding less toxic alternatives or improved containment. As chemists, we benefit from the lessons learned in working with complex molecules like N-Benzyl-N-Ethylaniline—balancing utility, risk, and innovation, one experiment at a time.
What is the chemical structure of N-Benzyl-N-Ethylaniline?
Mapping Out the Structure
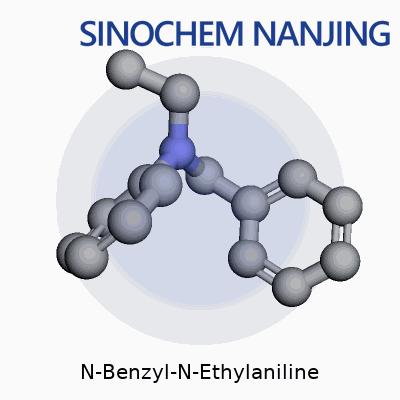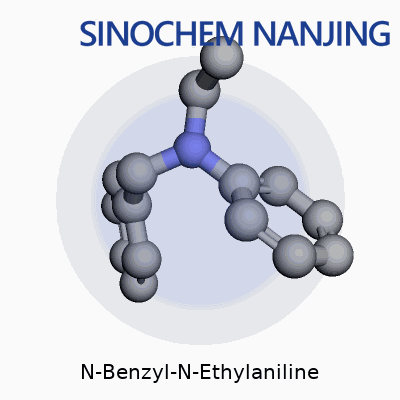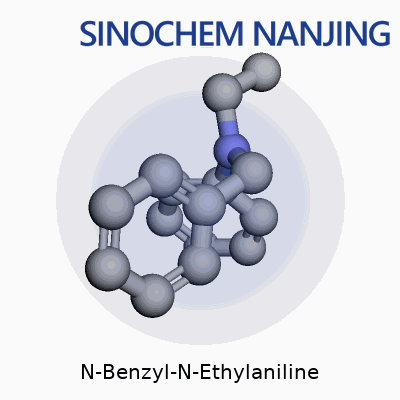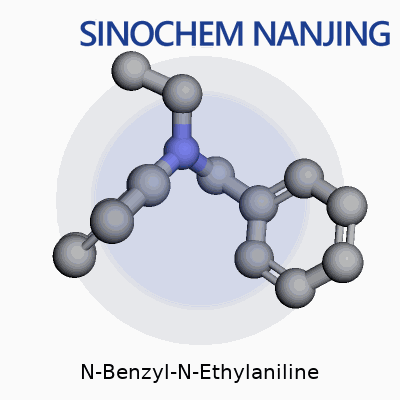
Chemistry tends to feel like an abstract puzzle. N-Benzyl-N-Ethylaniline sounds intimidating, but it draws from familiar building blocks. This molecule matches its name. There’s an aniline core—think of a benzene ring holding on to an amine group (NH2). N-Benzyl-N-Ethylaniline swaps those two hydrogen atoms on the nitrogen for a benzyl group (a phenyl ring attached through a CH2) and an ethyl group (two carbons, C2H5). Staring at its skeletal formula, you spot three clear zones: the main benzene ring (the aniline nucleus), the ethyl tail, and a second benzene ring arriving with the benzyl group through a short linker.
Why Structure Shapes Use
It’s easy to underestimate what that structure means in practice. Working with chemicals like N-Benzyl-N-Ethylaniline, I’ve found that every tweak to a molecule’s skeleton changes more than just what shows up on paper. Modifying the nitrogen’s attachments—an ethyl group giving some flexibility, a benzyl group adding bulk and aromatic character—transforms not only solubility but also how this molecule interacts with its neighbors, whether in a lab bench reaction or in industry.
The arrangement keeps it relatively non-polar, which often leads to practical challenges for people handling it in solution. That increased hydrophobicity, plus the two aromatic rings, can drive stacking interactions in chemical synthesis. Anyone who’s ever tried to mix up dyes or work with organic intermediates finds out just how much these bits of structure control performance.
Connections to Real-World Application
Organic chemists often look at N-substituted anilines when creating fine chemicals, new pharmaceuticals, or specialty materials. The basic atom arrangement in N-Benzyl-N-Ethylaniline shows up all over complicated synthesis strategies, especially those focused on building blocks for more elaborate molecules.
Take dyes, for example. Compounds like this pop up as intermediates in the creation of vivid colors, sometimes affecting not just shade but how stable a dye remains under light or washing. I’ve seen cases where tweaking a substituent—trading an ethyl for something bulkier, or flipping a benzyl group into a different aromatic ring—completely changes the colorfastness of the resulting ink or textile dye. N-Benzyl-N-Ethylaniline fills a gap for chemists wanting just the right balance between reactivity and stability.
Looking at Safety and Sustainability
Anyone who’s handled aromatic amines knows about the ongoing concerns over long-term safety, both for researchers and for the environment. Like most substituted anilines, this chemical asks for strict precautions: gloves, fume hood, smart waste management. It can be tempting to skip steps or cut corners, but the reality is that these compounds sometimes have carcinogenic risks.
I’ve learned that simple actions—choosing greener solvents, keeping good airflow, using newer methods to restrict emissions—make a difference. Whenever possible, research teams lean toward microscale synthesis or safer analogues. Regulations keep growing, especially around waste disposal, and I see more labs shifting toward careful recordkeeping and green-chemistry approaches. It’s a small but real step in making work with molecules like N-Benzyl-N-Ethylaniline safer.
Building Deeper Understanding
Peering into this molecule’s structure reveals more than a string of atoms and bonds. For chemists, students, safety officers, and industry workers, N-Benzyl-N-Ethylaniline represents the blend of precision, practical challenges, and ethical choices that modern chemistry brings. Approaching this not just as a formula to memorize, but as a real-world tool to use wisely, gives us more control—and more responsibility—over its impact.
What are the common uses or applications of N-Benzyl-N-Ethylaniline?
Pigment Pastes and Dyes: The Color We Take For Granted
Think about all the bold colors on your clothes, the vibrant shades in printing inks, or the paint you roll onto walls to brighten up a room. N-Benzyl-N-Ethylaniline helps shape much of that world of color, thanks to its role as an intermediate for pigment and dye manufacturing. Chemical factories lean on this compound when they want to build up “triarylmethane” dyes, which pop up in everything from fabric coloring to printer inks. I remember watching a friend work in a textile mill, describing how precise adjustments in the chemistry determined the richness of blue or green in garments. Subtle changes in molecular structure, guided by additives like N-Benzyl-N-Ethylaniline, decide how well a color withstands sun or how long it sticks through dozens of washes.
Building Block in Specialty Chemicals
Plenty of industries look for molecules that anchor bigger chemical structures—N-Benzyl-N-Ethylaniline fits that role. Chemical companies mix it into synthesis reactions to create surfactants, stabilizers, or even pharmaceuticals. If you’ve ever visited a facility where specialty coatings get made, you’ll see this sort of compound making the rounds, not only for color but for specific molecular tweaks. Research articles in organic synthesis journals show how chemists experiment with molecules like this one, producing variations that offer different sticking power, gloss, or resistance in coatings or electronics.
Functional Additive in Lubricants and Oils
Cars on the road, machines churning in factories, agricultural tools pulling through fields—they all need reliable lubrication. Producers include N-Benzyl-N-Ethylaniline in select lubricants and transformer oils. Its chemical backbone helps these fluids last longer, keeping engines or gearboxes from getting gummed up by breakdown products. During summers, I’ve watched old diesel tractors start up before dawn, freshly serviced with specially formulated oils; the difference in performance always pointed to small tweaks in their additive blends. Studies from the American Petroleum Institute demonstrate that nitrogen-containing molecules, such as those related to N-Benzyl-N-Ethylaniline, play a direct part in minimizing wear and protecting metal surfaces inside machinery.
Challenges and Safer Practices
While this chemical brings a lot of benefits, it calls for careful handling. Its toxicity remains under scrutiny, meaning production workers and lab staff must use strict protective gear and ventilation. Years ago, a friend working in quality control spoke about the regular training sessions she attended to make sure there was zero slip-up in safety measures while handling materials like this one. Regulations from organizations like OSHA and REACH in Europe set firm boundaries for exposure and waste management.
Looking ahead, there’s growing talk in the industry about greener synthesis routes. Safer alternatives or improved recycling practices for waste streams might not grab headlines, but they support both people in the workplace and the wider ecosystem. Research labs continue searching for new intermediates and changing reaction conditions, aiming to keep production efficient and risks lower. My own visits to modern chemical plants leave a clear impression: safety and sustainability are now a core part of the business, not just an afterthought.
What are the safety and handling precautions for N-Benzyl-N-Ethylaniline?
What’s at Stake With N-Benzyl-N-Ethylaniline?
N-Benzyl-N-Ethylaniline shows up in a handful of chemical processes, mostly within research, chemical synthesis, or as a reaction intermediate. People in chemical labs work with it for what it can help create, yet overlook the less glamorous part: safety concerns. I’ve spent my fair share of hours in cramped labs and ventilated cages, so I know that each compound carries its own risks, and this one’s no different. Getting comfortable around unfamiliar organic chemicals lands people in trouble faster than they’d like to admit.
Health Hazards Are Real
This compound brings classic hazards. Short-term exposure can leave you with headaches or dizziness if you breathe its vapors. Skin or eye contact can lead to redness or irritation. Spend enough time around leaky vials, poke at spills with a naked hand, and you risk bigger problems: long-term effects can reach from nerve system issues to unknown chronic health concerns because some aromatic amines in its class have flagged cancer risk in animal studies.
I once watched a colleague wipe down a contaminated bench, thinking she’d been careful, and end up in the nurse’s office—skin contact from a single spill. Any time handling organic amines, I never forget gloves. Anyone using this chemical must respect the risks, not just for their own sake, but the safety of others in that shared space.
No Substitute for Good Ventilation
All the gloves in the world won’t matter if vapors fill the workspace. Organic solvents and aromatic amines don’t mix well with lung tissue. I always worked with N-Benzyl-N-Ethylaniline inside a fume hood—never out in the open. Even a minor spill or flash evaporation could send fumes into your breathing zone. Trying to work in a poorly ventilated space risks cumulative exposure over time. It’s not enough to “crack a window” or rely on tabletop fans. If your lab’s air system isn’t moving several full air exchanges per hour, or if a chemical fume hood doesn’t alarm for airflow loss, you’re asking for trouble.
Personal Protection Matters
Proper lab coats, chemical-resistant gloves, and safety goggles are the real-life armor in these situations. I favor nitrile gloves for extra resistance, and I avoid latex because it breaks down too easily against organics. Aprons and closed-toe shoes further keep liquids from soaking into fabric or skin. It’s a boring routine, but it stops trouble before it starts.
Storage and Waste: Contain and Track
N-Benzyl-N-Ethylaniline doesn’t like warm, sunny rooms. It belongs in a tightly sealed container, out of reach of light and heat. I label every jar in fat, legible print—never trust a half-torn sticker from a past user. Spent material or spill cleanups never wind up in general trash. Local regulations treat waste containing aromatic amines as hazardous material. I keep contaminated rags and gloves sealed in chemical waste containers for the safety team to collect. Any shortcut with this process endangers janitors, landfill workers, or groundwater down the line.
Training Sets the Standard
Working with unfamiliar or toxic chemicals isn’t a solo journey. Real-world mentorship, step-by-step training, and written SOPs prevent mistakes. In my lab, everyone discusses the hazards before opening any new bottle, and we all have a say when protocols seem risky. Respect for the risks, plus honesty about your limits, sets a standard that makes safety automatic. That’s how you keep accidents at bay and chemicals in the bottle—never underestimated, always respected.
What is the molecular weight and formula of N-Benzyl-N-Ethylaniline?
Digging Into the Formula and Molecular Weight
Talking chemistry basics, N-Benzyl-N-Ethylaniline draws attention with a formula of C15H17N. That means 15 carbon atoms, 17 hydrogens, and one nitrogen make up this organic compound. For anyone curious about its molecular weight, you land at about 211.3 g/mol. This number doesn’t just come from a table—it stands as the sum of the atomic masses: carbon weighs in at 12.01, hydrogen at 1.008, and nitrogen at 14.01. Blend these in the right ratio, and you get the consistent figure that researchers and labs depend on.
Why Specifics Matter in Practical Research
Pinning down the right formula matters more than most people realize. N-Benzyl-N-Ethylaniline shows up in dye chemistry, pharmaceutical research, and sometimes as an intermediate during fine chemical synthesis. Getting the molecular weight right lets formulations stay on point, so reactions don’t go sideways. I remember running thin-layer chromatographies in grad school and realizing mistakes in molecular weights throw off yields. Even a slip-up of a decimal can waste time, reagents, and energy.
The right formula streamlines safety checks, too. Safety data sheets and regulatory filings need these numbers squared away, especially in academic and industrial labs where mistakes have expensive or dangerous consequences. Over the years, chemists have avoided serious mishaps just by triple-checking the math on weights and formulas.
Connections to Broader Concerns
Not everyone uses N-Benzyl-N-Ethylaniline daily, but millions rely on products or breakthroughs touched by organic compounds like this one. Modern drugs, high-performance pigments, and test reagents in diagnostics often come from processes that depend on the exactness of such chemicals. In the real world, reliability matters more than ever. Companies get pressure from regulators to provide traceable proof that their supply chains understand each molecule’s pedigree.
Transparency in chemical sourcing links back to formula and weight accuracy. Authorities expect consistency and documentation tracing every bottle and shipment, and for good reason. A mislabel, mixed up with the wrong statistics, can put a halt to production lines, research projects, or even trigger a recall of finished products.
Solutions and Best Practices
Better training helps. Most problems crop up where staff get rushed, rely on memory, or lack up-to-date references. Digital databases and easy-to-use lab software take human error out of routine calculations. Spending a few minutes to double-check paperwork—especially when ordering, receiving, and labeling chemicals—saves hours of fixing mistakes down the road. I once watched a shipping clerk stop a shipment for review because a label had one number off; the lab manager called it the cheapest insurance ever.
More open communication between purchasing agents, bench chemists, and managers can also catch mistakes early. Sharing firsthand stories of mix-ups makes procedures real instead of just rules in a manual. The industry also benefits when everyone agrees on one trusted set of references—no shortcutting with half-remembered trivia. Relying on recognized scientific databases and keeping Certificate of Analysis documents close helps prevent headaches later.
Wrapping Up the Numbers
Details, like those behind the formula C15H17N and the number 211.3 for N-Benzyl-N-Ethylaniline, drive good science and smooth business. Precision at this level carves the path for large-scale innovations, keeps jobs safe, and helps companies pull ahead without trouble from regulators. The difference between progress and a pile of setbacks often comes down to knowing, and trusting, the numbers before the work even starts.
How should N-Benzyl-N-Ethylaniline be stored and transported?
Personal Perspective and Experience
Working in labs where chemicals like N-Benzyl-N-Ethylaniline show up on the shelves has taught me not to underestimate the risks, even if a chemical doesn’t sound as notorious as others in the news. Years ago, I saw what happens when storage gets sloppy—a single spill led to long hours of cleanup, investigations, and a lot of stress. The lesson: thinking ahead saves trouble, money, and sometimes, health.
Understanding the Requirements
N-Benzyl-N-Ethylaniline stands as an organic compound and many chemical companies list it as a substance that’s irritating and possibly flammable. Earlier safety data sheets I checked didn’t mince words: keep this out of your eyes, and don’t let it mix with ignition sources. That reminds me of how important the right storage is, not just for safety, but to prevent loss from accidents.
Practical Advice for Storage
From hands-on experience, containers should feature clearly labeled hazard warnings and tight seals. N-Benzyl-N-Ethylaniline performs best in a cool, dry space, away from sources of heat or sparks. Direct sunlight sometimes speeds up chemical changes, and nobody wants mysterious byproducts when precision matters. Locks or access controls help keep only trained techs in charge of material like this.
Don’t forget the basics here—never stack heavy objects on top of a chemical drum or store incompatible substances nearby. Fires in labs often start not from the main workbench, but from a forgotten shelf with a flammable chemical sitting too close to oxidizers. Investing in proper storage cabinets, especially flame-resistant ones, pays back every time an accident doesn’t happen.
Focus on Transport: Keeping It Contained
Transit brings its own risks. I’ve seen too many reports of containers breaking during a rough ride in the back of a truck. Only use strong, tested packaging that can handle jolts and sudden stops. Last thing anyone wants is an accident in a crowded place because a drum leaked or burst. If a spill happens during shipping, quick cleanup protocols and first-responder kits must be there at all times. Proper documentation, addressed by every responsible shipper, adds another level of assurance—you know what’s inside, and so does the next person handling it.
Most shipping providers recommend certified drums, clear hazard labeling on all sides, and protective cushioning inside transport vehicles. These aren’t just red tape. I speak from seeing how small mistakes turn into major emergencies—loose caps and casual labeling mean wasted product and sometimes calls to fire departments.
Health, Environment, and Ethical Responsibility
Working safely with N-Benzyl-N-Ethylaniline protects more than lab workers. Mishandled chemicals often end up affecting nearby communities, factories, or rivers. Companies can cut costs by skipping safety, but the long-term reputation and human impact mean everybody ends up paying more. Proper chemical management stands as a basic sign of respect for communities and ecosystems, not just compliance.
Looking Forward: Some Solutions
Training goes further than any storage cabinet ever will. Anyone who handles chemicals needs up-to-date safety lessons. Many workplaces hold drills for spills or fires, but in smaller outfits, these sometimes get skipped. This can’t keep happening.
Using tech such as electronic inventory tracking and automated alarms sets a new standard. These systems flag temperature changes or unauthorized access—small changes that catch small mistakes before they become big problems. Leaders who take chemical management seriously invest in these solutions not for show, but because they know the real cost of doing nothing.
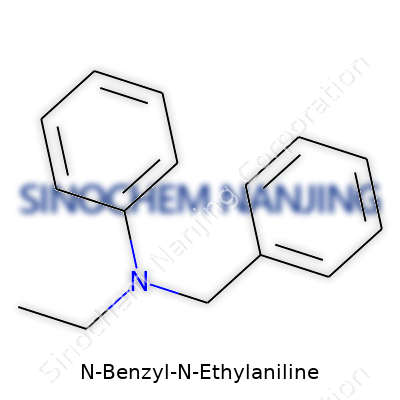
| Names | |
| Preferred IUPAC name | N-benzyl-N-ethylbenzenamine |
| Other names |
N-Benzyl-N-ethyldiphenylamine N-Ethyl-N-phenylbenzenemethanamine N-Ethyl-N-benzylaniline Benzenemethanamine, N-ethyl-N-phenyl- |
| Pronunciation | /ɛnˈbɛnzɪl ɛnˈɛθɪlˌænɪliːn/ |
| Identifiers | |
| CAS Number | 92-54-6 |
| Beilstein Reference | Beilstein Reference: 107875 |
| ChEBI | CHEBI:132747 |
| ChEMBL | CHEMBL156383 |
| ChemSpider | 22833 |
| DrugBank | DB08313 |
| ECHA InfoCard | 100.053.163 |
| EC Number | 202-858-4 |
| Gmelin Reference | 79257 |
| KEGG | C19642 |
| MeSH | D052020 |
| PubChem CID | 162424 |
| RTECS number | BU5425000 |
| UNII | C7B1XAT6TE |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID60137207 |
| Properties | |
| Chemical formula | C15H17N |
| Molar mass | 241.34 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | aromatic |
| Density | 1.015 g/mL at 25 °C |
| Solubility in water | insoluble |
| log P | 4.2 |
| Vapor pressure | 0.01 mmHg (20°C) |
| Acidity (pKa) | 4.8 |
| Basicity (pKb) | 5.98 |
| Magnetic susceptibility (χ) | -74.8×10^-6 cm³/mol |
| Refractive index (nD) | 1.5790 |
| Viscosity | 2.42 cP (20°C) |
| Dipole moment | 3.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -5647.6 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P330, P337+P313 |
| Flash point | > 135 °C |
| Autoignition temperature | 481°C |
| Lethal dose or concentration | Lethal dose or concentration (LD50, oral, rat): >2000 mg/kg |
| LD50 (median dose) | Rat oral LD50: 1880 mg/kg |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg |
| Related compounds | |
| Related compounds |
N-Ethylaniline N-Benzylaniline Aniline Benzylamine Ethylaniline |

