N-Ethyl-P-Toluidine: Tracing Its Roots to Today’s Chemical Industry
Historical Development
Looking back, N-Ethyl-P-Toluidine reflects the traditions of modern chemistry. Chemists in the late 19th and early 20th centuries scoured coal tar and petroleum for new aromatic amines, eager to satisfy the rapid growth in industries like dyes and pharmaceuticals. Over time, both laboratory and industrial minds found N-Ethyl-P-Toluidine a handy intermediate. The structure—a methyl group on a benzene ring paired with an amino group and an ethyl side-chain—opened doors for synthetic work. Historical records credit German chemical industries with much of the early research, as competition for brighter aniline dyes and innovative intermediates pushed experimentation. As the field grew, so did protocols for synthesis and purification, giving way to today’s batch and continuous processing.
Product Overview
Today, N-Ethyl-P-Toluidine shows up in high-purity forms for pharmaceuticals, pigments, and various specialty chemicals. The pure compound arrives as a colorless to pale yellow liquid, with a strong amine-like odor familiar to anyone working around aromatic amines. Its production supports a chain of industries, especially those seeking intermediates with consistent reactivity. Whether for laboratory-scale research or plant-scale manufacture, the demand keeps steady, reflecting both tradition and a need for versatile amine intermediates.
Physical & Chemical Properties
Most chemists recognize N-Ethyl-P-Toluidine by its chemical formula, C9H13N, and its molecular weight of about 135.21 g/mol. It stays liquid under ordinary conditions, with a melting point below -10°C and a boiling point hovering near 218°C. Vapors can irritate eyes and airways, so good ventilation becomes essential. Water solubility stays modest, but it dissolves with ease in organic solvents such as ethanol, ether, and benzene, making it suitable for reactions demanding such media. Chemical stability sits high under normal storage, yet exposure to strong oxidizers or acids usually prompts rapid reactions.
Technical Specifications & Labeling
Suppliers attach clear technical details to every drum or bottle: purity typically reaches at least 99%, as measured by gas chromatography. Color, acidity, and the presence of common impurities, like aniline or residual solvents, draw close scrutiny. Flash point falls near 93°C, giving clear direction for fire safety. Labels carry hazard pictograms warning of toxic effects and skin or eye hazard, signaling the need for gloves and goggles. Transportation companies classify it under UN2810, Toxic Liquid, ensuring consistent treatment worldwide.
Preparation Method
Industrial synthesis relies mostly on the alkylation of p-toluidine with ethyl halides or ethyl sulfate under basic conditions. Classic lab techniques use sodium hydroxide and ethyl bromide, with careful temperature control to maximize selectivity. Process tweaks—for instance, switching to phase-transfer catalysis or continuous flow reactors—aim to boost yield and limit side reactions. Controlling stoichiometry ensures excess ethylating agent doesn’t transform the product further or introduce impurities. Waste streams undergo amine recovery or incineration in line with regulatory standards to cut environmental harm.
Chemical Reactions & Modifications
N-Ethyl-P-Toluidine displays reactivity typical of substituted anilines. Nitration brings on new groups for dye work. Oxidation, whether by permanganate or chromate reagents, shifts functional groups and color properties, which researchers tap for analytical studies. In industry, acylation or sulfonation changes solubility or boosts affinity for fibers. It also acts as a nucleophile in electrophilic substitution, opening it up for further customization by creative chemists. Anyone engineering dyes or drug precursors values these versatile reactions.
Synonyms & Product Names
The catalogues echo a range of synonyms: 4-Methylaniline, N-Ethyl-4-toluidine, N-Ethyl-p-toluidine, and Ethyl-p-toluidine stand out. Sometimes legacy names like N-Ethyl-4-methylaniline pop up, especially in older patents or research papers. Commercial suppliers build their inventory around these synonyms, and regulatory documents rely on these aliases for accurate import-export handling.
Safety & Operational Standards
Toxicity demands cautious handling. Direct skin or eye contact can cause severe burns and allergic reactions; inhalation of vapors brings on headaches, nausea, or confusion. Chronic exposure, particularly in poorly ventilated plants, raises risks of hemoglobin damage. The industry demands chemical fume hoods, full-face shields, nitrile gloves, and splash aprons whenever large volumes get handled. Spill kits and eye-wash stations stay on hand. Emergency procedures and training in first-aid responses ensure both lab technicians and factory staff understand what to do if an accident happens. National and international workplace standards, such as those from OSHA and REACH, lay out the expectation for exposure monitoring and engineering controls.
Application Area
The biggest markets for N-Ethyl-P-Toluidine cluster in dyes, pigments, and specialty chemicals. Its amine backbone lets it participate in azo coupling and other colorant processes. Agrichemical firms pull from its reactivity to prepare herbicide and fungicide intermediates. In the pharmaceutical sector, it sometimes acts as a stepping-stone in preparing APIs—especially antihistamines or analgesics—where tailored rings and substituents can transform activity. Polymer chemists use it as a stabilizer or a hardener for epoxy resins. Electrochemistry researchers often examine its redox properties for sensor applications. Every year, specialty labs report new derivatives for analytical chemistry or specialty photoresists, revealing its ongoing value.
Research & Development
Recent research takes several directions. Green chemistry advocates look for routes that replace hazardous solvents, cut energy demands, or incorporate renewable feedstocks, paying attention to lifecycle impacts. Novel catalytic systems—phase-transfer catalysts, ionic liquids—grab attention for lower emissions and higher selectivity. In pharmaceuticals, researchers chase derivatives with greater bioactivity and better metabolic profiles while minimizing toxicity. Analytical chemists, on the other hand, seek new detection methods for both trace impurities and environmental residues. Patents keep rolling in for new uses in organic electronics and sensing technology, driven by tinkering with its electronic properties.
Toxicity Research
Animal studies and cell assays do not paint a benign picture. N-Ethyl-P-Toluidine has shown methemoglobinemia effects, altering oxygen carriage in blood after exposure. Mutagenicity assays sometimes record potential genetic damage, though conclusive links to cancer in humans stay uncertain. Regulations force monitoring of exposure not just among workers but also in wastewater streams, as aromatic amines have a reputation for persistence and possible environmental effects. Toxicology teams push for safer handling packaging and strive to invent less hazardous analogs that still provide necessary performance for industry.
Future Prospects
Moving forward, the compound’s future rides on tightening environmental rules, changing customer demand, and innovation in synthesis. Alternative manufacturing methods—using enzymes or sustainable catalysts—could replace legacy chemistry with cleaner, safer options. Regulatory clamps in major markets force companies to develop better containment and recovery systems. Research continues to tweak its molecular structure for new applications in green solvents, specialty resins, and organic sensors. As demand for advanced materials and safer production methods advances, those who invest in responsible process design and green chemistry know-how will shape what comes next for N-Ethyl-P-Toluidine and its chemical cousins.
What is N-Ethyl-P-Toluidine used for?
How Industries Rely on N-Ethyl-P-Toluidine
Walk into any auto body shop and the smell of paint hits fast. Most people don’t think twice about what it takes to make automotive finishes smooth and tough. N-Ethyl-P-Toluidine makes this happen. Companies making paints, especially those for cars, use this chemical as a curing agent to help resins set hard and even. Tough coatings don’t just make a car look shiny; they keep rust out, which matters when you want to keep that big repair bill away. Epoxy and polyurethane coatings also rely on it, which means garage floors and industrial machines get protection that lasts longer than a typical seasonal upgrade.
Printing factories rely on fast-drying inks. N-Ethyl-P-Toluidine acts as an accelerator in inks. Magazines, labels, and packaging often cycle through presses at speeds so fast, ink has to set before the next roll hits—nobody wants smudged labels or unreadable barcodes. This compound keeps print shops running at high efficiency, especially for flexographic and screen printing. Some dyes used in textiles and cosmetics also link back to this chemical as a building block. Even though most consumers never see it in the final product, it quietly helps deliver consistent color and coverage that people appreciate in clothes and packaging.
Health and Safety Concerns
Exposure risks get more attention these days. Chemical factories that handle or produce N-Ethyl-P-Toluidine must follow strict rules—for good reason. Studies point out that this compound can cause headaches, dizziness, or worse symptoms if not handled carefully. Chronic exposure could have long-term effects that aren’t always obvious upfront. Production staff need personal protective equipment, proper ventilation, and routine monitoring. Mistakes or shortcuts hurt real people, and nobody wants to see preventable accidents upend lives or families.
Products containing this compound should never get into children’s hands. If someone works with it at home for any reason—rare, but sometimes it happens—they should treat it with respect, keep containers closed tight, and work in open air. Following instructions lowers risk, and that advice comes from years dealing with chemical safety at different worksites. Labels and safety sheets don’t just fill out paperwork; they matter in practice.
Questions About Environmental Impact
Wastewater coming from factories can carry N-Ethyl-P-Toluidine if corners get cut somewhere in the process. Soil and river contamination can harm fish and plants, with those effects spiraling out to people who rely on the land. Eco-friendly alternatives should get a real look, even if switching takes time and money. Better capture technologies and stricter waste treatment help, but companies have to stick to their promises. I once sat in a city council meeting where parents demanded more information on chemical plant runoff, and the public pressure brought actual improvements—community voices really matter in these debates.
Better Choices and Honest Responsibility
Switching to greener chemistry doesn’t come easy, especially for large manufacturing or printing operations used to older methods. But real progress means trialing replacements, investing in new research, and pushing suppliers. Industry groups, governments, and environmental advocates need to keep the conversation going. For buyers, paying attention to what’s inside products—and asking questions—makes a difference, too. Clean manufacturing supports everyone, from workers managing chemicals to families living nearby. That responsibility starts with companies, but watchdogs, local leaders, and everyday people push for better standards every year.
What is the chemical formula and structure of N-Ethyl-P-Toluidine?
Chemical Makeup of N-Ethyl-P-Toluidine
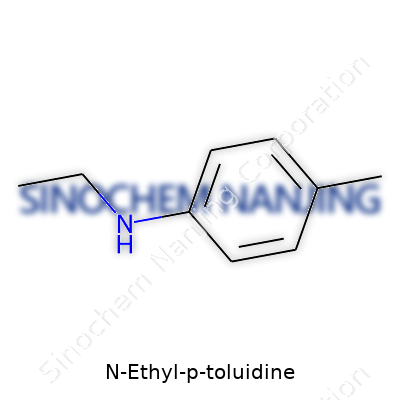
N-Ethyl-P-Toluidine’s chemical formula, C9H13N, might look like mere characters to most, but beneath those nine carbon atoms, thirteen hydrogen atoms, and a single nitrogen atom lies a specific arrangement that tells a much bigger story. The structure features an aromatic benzene ring. Attached to that ring sits a methyl group (–CH3) at the para (or 4th) position relative to an amino group. The nitrogen in the amino group connects to an ethyl chain (–C2H5). To chemists, the molecule’s name comes from the layout: “toluidine” refers to the methyl-substituted aniline, and “N-ethyl” marks the ethyl group linked directly to the nitrogen atom.
Take a look at the structure:
- A central benzene ring
- A methyl group at the para position
- An ethyl group bonded to the nitrogen of the amino group opposite the methyl
The Significance of This Compound
N-Ethyl-P-Toluidine pops up in lots of places, especially in specialty chemistry labs and industries that turn out dyes, pigments, and intermediates for pharmaceuticals. Back in my college lab, handling toluidine derivatives required focus and respect for safety. A whiff of its distinctive, rather pungent odor stuck with me during late-night experiments.
People often overlook how critical these molecules become in broader manufacturing. The world’s thirst for brightly colored textiles, certain inks for printers, or even specific polymers all ties back to foundational chemicals like N-Ethyl-P-Toluidine. Its particular structure—especially the ethyl group at the nitrogen—lets it serve as a building block. Each small tweak in the molecule shapes what the end product does, how stable it becomes, and even how it interacts with other chemicals down the line.
Health, Safety, and Environmental Considerations
Compounds like N-Ethyl-P-Toluidine bring up important safety discussions. The molecule has shown potential harm, with concerns about toxicity if inhaled or absorbed through the skin. Over time, some research has flagged aromatic amines (the chemical family N-Ethyl-P-Toluidine belongs to) for links to certain health conditions. In busy industrial settings, workers have to rely on strong ventilation, gloves, and airtight handling procedures to avoid unnecessary risk. The substance doesn’t just disappear; improper disposal risks contaminating soil or water. Experience has taught me that the protocols in place aren’t just legal hoops. They’re there because a single careless move can lead to real harm.
Looking Toward Responsible Use
The chemical industry faces a crossroad. Demand keeps rising for molecules that deliver on color, performance, and function. Yet, regulations around worker safety and environmental protection grow stricter. Some smart researchers have started designing alternative pathways or swapping in greener chemicals where possible. At the same time, companies invest in recovery and recycling methods for aromatic amines. The ask isn’t just about following rules—it’s about recognizing the real-world stakes. Truth is, every bottle and barrel of N-Ethyl-P-Toluidine connects to somebody’s safety and the health of the environment beyond the lab doors.
Is N-Ethyl-P-Toluidine hazardous or toxic?
Getting to the Root of the Problem
N-Ethyl-P-Toluidine does not make headlines like plastics or oil spills, but it sits in plenty of workplaces across industries. If you have ever glanced at a safety data sheet in a chemical plant or a laboratory, you might have seen this name. Its use shows up in dyes, certain printing inks, and some processing chemicals. There is also a sliver of it lurking in polymer chemistry. Why do I care? After years spent around industrial sites and research labs, I have learned one thing about unfamiliar chemicals—healthy suspicion protects you just as much as goggles and gloves.
Understanding the Hazards in Plain Language
So, is N-Ethyl-P-Toluidine toxic or hazardous? The answer, based on decades of toxicological reports and government safety data, is yes. If someone breathes it in, it irritates the nose and throat. Exposure can lead to headaches, dizziness, and in some cases, worse—blue skin and lips, which point toward methemoglobinemia. That’s a mouthful, but what it really means is the blood cannot carry oxygen as well, choking off healthy function. In my time observing field workers, I have seen these symptoms with chemicals in the same class, and it’s unsettling.
Touching liquid N-Ethyl-P-Toluidine poses danger to skin and eyes, causing redness and pain. Once it gets to your bloodstream through skin or inhalation, it can head straight to your organs. Studies in animals show that repeated exposure may trigger damage to the liver, kidneys, and perhaps even lead to cancers. The National Institute for Occupational Safety and Health flags it as a possible carcinogen. To me, the word “possible” here acts as a warning siren—maybe the research is ongoing, but the smart move is to treat it as a threat.
Real-World Experience Speaks Loudest
On factory floors, safety steps can slip. Folk cut corners for speed, or gear gets uncomfortable, especially in summer. But comfort is no match for chronic illness. I have worked alongside seasoned workers who lost sensation in hands or struggled with breathing, all linked to poor chemical handling. Small exposures repeated over months or years leave lasting marks. That’s hard evidence, harder than anything in a textbook. People tend to trust what they can see today and forget what can creep up later. And companies, hungry for profit, sometimes skip the extras that secure workers’ long-term health. It’s only after legal battles or hospital bills stack up that anyone pays attention to the warning signs.
Paths Forward—What Actually Works
Banning a chemical sounds dramatic, but more often the best move is strict control. Every worksite using N-Ethyl-P-Toluidine should make personal protective equipment non-negotiable—gloves, goggles, respirators. Well-ventilated areas cut risk, but scheduled air sampling makes sure that invisible threats get picked up. Training helps, but it needs to be routine, not a once-and-done exercise at hiring. Signs and simple instructions keep safety top of mind. Labels should call out the risk, clear and honest, not buried under legal jargon.
For smaller labs or businesses, substitution often proves smarter. Many non-aromatic alternatives on the market offer the same industrial results without the persistent health cloud. For me, advocacy means pushing industry and regulators to speed up independent testing and release all findings, not just the ones that clear liability. Doctors and safety officers need up-to-date facts to connect cryptic symptoms in workers back to their real cause.
Why Talking About Toxicity Matters
If we ignore the issues with substances like N-Ethyl-P-Toluidine, we pay the price in rising cancer rates and preventable accidents. Beyond statistics, the real cost falls on families and communities who shoulder the burden of care. Good science and tight controls do not exist just on paper—they play out in the daily health and safety of real people. Your health stands as reason enough to scrutinize what comes into your space. No workplace shortcut or product tweak justifies putting lives on the line.
What are the storage and handling recommendations for N-Ethyl-P-Toluidine?
Understanding the Risks
N-Ethyl-P-Toluidine shows up in chemical labs and industrial sites where it plays a role in dyes, pharmaceuticals, and polymers. Even if you trust your equipment and routine, cutting corners with storage or handling has real consequences. Accidental exposure can bring headaches, dizziness, or even more serious health issues. Direct skin contact sometimes leads to irritation or worse. Since vapors can be flammable and may irritate the respiratory system, mishandling spills or leaks nearly always creates danger that spreads fast across a workspace.
Storing N-Ethyl-P-Toluidine Right
Chemical safety often starts with picking the right place to keep things. N-Ethyl-P-Toluidine needs a storage area that stays dry, cool, and out of direct sunlight. Locked cabinets work well, especially those meant for flammable liquids. Store this substance away from any oxidizers, acids, or food supplies. Leaks and cross-contamination run common in cramped storage, so always separate incompatible substances.
Just about every chemical warehouse I have visited uses tightly sealed containers, often glass or heavy-duty plastic. Metal cans cause problems, thanks to corrosion over time, especially in humid climates. If a container does look damaged, arrange for proper disposal instead of keeping it around for convenience’s sake. Avoid using reused containers from other chemicals to stop unexpected reactions.
Handling Precautions that Count
Direct handling of N-Ethyl-P-Toluidine asks for gloves, goggles, and solid lab coats. A well-ventilated room with approved chemical fume hoods cuts down the risk when vapors escape, even for quick tasks. Breathing in vapors brings risk, so many labs add an extra layer of respiratory protection, especially if handling more than a few milliliters at a time.
Label every container clearly with the substance name and hazard warnings. This sounds obvious, but rushed or temporary tags lead to mix-ups, especially across shift changes. In my experience, most accidents stem from overlooked labeling rather than rare equipment failures. It’s not just about following the rules on paper—those signs mean something when every second counts in an emergency.
Fact-Based Solutions to Common Issues
Spills can escalate without fast action. Absorbent pads designed for chemicals clean up minor accidents before they spread. Dispose of all waste (solid, liquid, and absorbent material) through a licensed hazardous waste facility, not regular trash or drains. If you ever catch that sharp, amine-like odor, check all containers and ventilation systems. Minor leaks from poor seals or left-open lids build up over weeks and raise risk across the whole site.
Training staff on emergency protocols stays just as important as safety gear. Regular drills and refreshers beat any single sign or poster. Many teams benefit from having clear emergency showers and eyewash stations a few steps from any chemical handling bench.
Why Personal Experience Matters
I’ve seen more than a few blown fuses and rushed mistakes after a long shift. Stressed teams skip glove checks or ignore that fading warning label. Even highly experienced technicians get blindsided by shortcuts built over time. Treat N-Ethyl-P-Toluidine with respect, in line with its safety data sheet, and accidents lose any chance to get out of control. Sound routines make safe chemistry look simple, but that simplicity gets built on smart choices every day.
What are the typical purity specifications for N-Ethyl-P-Toluidine?
Looking Beyond the Chemical Name
The mention of N-Ethyl-P-Toluidine triggers a lot of technical talk for people in labs and on production floors. I've seen chemists and plant operators glance over purity specs, only to later struggle with a reaction that stalled or an ink batch gone off-color. Some folks see purity as a dull number on a label. Out in the real world, though, these numbers separate productive days from money lost on unexpected troubleshooting.
What Purity Looks Like in Daily Practice
Most reputable suppliers offer N-Ethyl-P-Toluidine at purities between 98% and 99.5%. This range isn’t just a formality. In dye manufacturing, even small traces of related amines or other organics can create hues that customers reject outright. A biotech team using the compound for synthesis can’t afford purity slippage either; any residual dimethylaniline or toluidine by-products end up lowering the yield and sometimes cause failed quality checks. I've watched batches get dumped in the name of those last two percent.
Impurity Impact: Keeping Tabs on the Small Stuff
Suppliers often test for specific impurities along with the overall purity percentage. Water content usually gets capped below 0.5%, and heavy metals stay well under 10 ppm. Free acids create trouble in chemical reactions, so their limits typically go below 0.1%. People sometimes overlook color and appearance, but yellow tints or unexpected crystal formation can point to problems before the instrument reports even come back.
During several audits, I worked with quality teams who flagged N-Ethyl-P-Toluidine from lesser-known vendors because of off-spec “moisture content.” Water’s easy to forget in organics, but it can act as a silent reaction-stopper or an unwanted sidekick in storage tanks. Problems like these do not show up in a textbook. Real-world checks, like a Karl Fischer titration or a quick evaporation test, turn out to be invaluable.
Certification and Analytical Realities
Every batch should come with a certificate of analysis. Labs rely on gas chromatography and mass spectrometry for tracking the main compound and any by-products. Spectral purity gets cross-checked since trace contaminants often hide where the eye can’t see. Over the years, I’ve asked suppliers tough questions about their testing procedures. Not all certificates are equally trustworthy, so relationships with suppliers who invest in decent equipment and skilled analysts help keep surprises to a minimum.
Getting Purity Right: Practical Solutions
Individuals working with N-Ethyl-P-Toluidine can take some simple steps to keep problems away. Storing the compound in sealed, non-reactive containers at room temperature slows down degradation. If you run a smaller lab, it makes sense to split shipments to use only fresh material—humidity and heat create impurity headaches fast.
For buyers, it pays to look at more than just the top figure on the spec sheet. Asking for details about water, free acid, and heavy metal testing can avoid wasted hours later. When teams push for transparency in how suppliers measure and confirm purity, they help the whole chain of production run smoother.
Getting a handle on these purity details means fewer production interruptions, more reliable products, and a safer work space. People who care about their finished result always watch the small numbers, long before a problem ever turns up.
| Names | |
| Preferred IUPAC name | N-ethyl-4-methylaniline |
| Other names |
4-Ethylaminotoluene N-Ethyl-4-methylaniline N-Ethyl-p-toluidine N-Ethyl-1-methyl-4-aminobenzene N-Ethyl-4-methylbenzenamine |
| Pronunciation | /ɛnˈɛθɪl piː təˈluːɪdiːn/ |
| Identifiers | |
| CAS Number | 612-00-0 |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:38570 |
| ChEMBL | CHEMBL20113 |
| ChemSpider | 10972 |
| DrugBank | DB14018 |
| ECHA InfoCard | 100.011.231 |
| EC Number | 202-805-4 |
| Gmelin Reference | 8323 |
| KEGG | C14318 |
| MeSH | D014046 |
| PubChem CID | 7507 |
| RTECS number | XN8575000 |
| UNII | 6SV640R62K |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID6020289 |
| Properties | |
| Chemical formula | C9H13N |
| Molar mass | 135.21 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | amine-like |
| Density | 0.956 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.15 mmHg (20°C) |
| Acidity (pKa) | 5.1 |
| Basicity (pKb) | 7.68 |
| Magnetic susceptibility (χ) | -63.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.560 |
| Viscosity | 15.32 cP (25°C) |
| Dipole moment | 2.03 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 296.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3561.8 kJ/mol |
| Pharmacology | |
| ATC code | N05CM18 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes skin irritation. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | ["GHS07", "GHS08"] |
| Signal word | Danger |
| Hazard statements | H302 + H312 + H332: Harmful if swallowed, in contact with skin or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P280, P301+P310, P302+P352, P304+P340, P308+P313, P405, P501 |
| Flash point | 87 °C |
| Autoignition temperature | 405°C |
| Explosive limits | Explosive limits: 1.2–7% |
| Lethal dose or concentration | LD50 oral rat 870 mg/kg |
| LD50 (median dose) | 1,650 mg/kg (rat, oral) |
| NIOSH | RN 91-67-8 |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended): 2 mg/m³ |
| IDLH (Immediate danger) | IDLH: 500 mg/m3 |
| Related compounds | |
| Related compounds |
N-Methylaniline N,N-Diethylaniline N-Ethylaniline N-Phenylethanamine P-Toluidine 4-Ethylaniline |

