N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide: A Closer Look at its Journey, Impact, and Future
Historical Development
Decades back, the story of N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide started in industrial labs looking for fluorinated materials with unique surface-energy characteristics. Manufacturers hunted for ways to repel water, oil, and dirt in textiles, firefighting foams, paper coatings, and cleaners. Out of these experiments, perfluorooctanesulfonamides flourished, offering properties that beat old waxes and silicones. As demand for non-stick, stain-resistant products grew, so did research into manipulating sulfonamides like this one, driven largely by breakthroughs in polymer chemistry. The compound saw prime use in the 1970s and 1980s thanks to its stability and performance under tough conditions. Regulatory pressure on persistent organic pollutants forced the field to look harder at toxicity and environmental persistence.
Product Overview
N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide belongs to a class of fluorosurfactants well known by those in manufacturing for its pronounced ability to lower surface tension. In practice, that means it helps keep coatings even and boosts the finish of finished goods. My work in textile labs means I’ve watched workers rely on fluoroalkyls for stubborn stain-fighting performance or for forms of streak-free surface treatment that can outlast harsh scrubbing or weather. Each time a new end-use appeared, users pushed for better formulations built around the core structure of this sulfonamide, always chasing a mix of performance with manageable costs.
Physical & Chemical Properties
With a perfluorinated carbon tail matched to a polar head, this molecule acts like a chemical Swiss Army knife—unusually stable, highly resistant to both acids and bases, and more persistent in the environment than most hydrocarbons. In plain terms, it doesn’t break down easily, making it especially valued for demanding industrial processes. It's typically a waxy solid or viscous liquid at room temperature, undergoing only slight color changes under most storage conditions, and it barely dissolves in water, which means it sticks tightly to the things it treats. These properties greatly increase its value, but they also mean that it hangs around in ecosystems for years, igniting big debates about long-term toxic effects.
Technical Specifications & Labeling
Suppliers in this niche provide N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide to strict quality standards, usually outlining the minimum fluorine content and permissible levels of trace organics. I’ve reviewed countless safety data sheets listing flash points, temperature ranges for storage, and requirements for moisture control to avoid hydrolysis. Most packages arrive in sealed drums or containers marked with UN hazard symbols for irritant or environmental hazard, warning anyone in downstream handling roles to use gloves and eye protection. In my time working with these chemicals, I’ve seen guidelines evolve as new research revealed the need for better inventory tracking and clear, easy-to-read identification labels for those outside the lab.
Preparation Method
The most common process relies on reacting perfluorooctanesulfonyl fluoride with ethylamine and 2-aminoethanol under carefully controlled temperature and pH conditions, usually with batch reactors built to handle corrosive intermediates. Operators monitor pH and exothermicity continuously, as things can get out of hand with too much residual fluoride. Post-reaction workup involves vacuum distillation or solvent extraction to purify the crude product. In labs I’ve toured, teams invest in custom glassware or stainless-steel vessels to prevent etching and breakdown, limiting contamination and safeguarding purity for use in downstream specialty applications.
Chemical Reactions & Modifications
Chemists use this sulfonamide as a building block for further modifications. The hydroxyethyl group opens doors to polymer attachment, crosslinking, or even transformation into new surfactants with tighter tailoring for detergents, fire-fighting foams, and repellency treatments. Companies often graft this molecule onto backbone polymers to yield products with lasting non-stick or anti-soiling qualities. I once watched a team demo new syntheses based on this compound, adding reactive sites for UV-cured coatings and pushing formulations to withstand harsh industrial cleaning. Results often depended heavily on fine-tuning pH, reaction temperature, and the presence of trace metals, which can act as unwanted catalysts or contaminants.
Synonyms & Product Names
In technical literature and on safety reports, this substance often appears under names like N-Ethyl-N-(2-hydroxyethyl)perfluorooctane sulfonamide, FOSE, EFOSE, or more cryptic codes from legacy manufacturers. Some call it by trade names designed to conceal its identity in finished blends. In my experience, confusion in naming conventions has sparked costly errors, especially when translated across regulatory regimes or between companies working from decades-old stockpiles. Clear documentation of CAS numbers and synonyms has helped prevent these mix-ups, especially for emergency response teams.
Safety & Operational Standards
This is one of those substances you only want in the hands of trained staff. Touching or breathing dust or aerosol can irritate skin, eyes, and lungs, sometimes for hours. Standard operating procedures demand chemical-resistant gloves, goggles, and local exhaust ventilation. Air monitoring becomes key in larger facilities that process kilograms at a time, while those in research or pilot-scale runs often train new staff with spiked air samples and practice spills to reinforce muscle memory. I’ve seen major improvements in how safety drills are handled, prompted by evolving data on chronic toxicity and persistence. Fire departments responding to lab incidents now carry specialized cleanup kits because old foam extinguishers sometimes react with these compounds, creating highly persistent residues.
Application Area
Few materials span as many uses as this one. From paper coatings that resist wine and oil, to high-performance fire-fighting foams on airport runways, to industrial cleaners for electronics fabrication, this chemical quietly forms the backbone of surface-protection strategies. In factories processing textiles and carpets, workers rely on its ability to hold up against repeat washing and rough use. In firefighting, crews swear by the foam’s ability to form stable barriers, trapping volatile fuels and slowing vapor release during emergencies. Growing restrictions on legacy fluorochemicals drive more targeted applications, moving away from broad spray use toward specialty coatings or closed-system industrial reagents, though global demand stays steady due to persistent technical advantages.
Research & Development
Pushes for greener alternatives fill specialty journals every year. Researchers, often driven by regulatory deadlines or disclosure requirements, experiment with biological breakdown pathways, less persistent analogs, and process tweaks to limit run-off or bioaccumulation. I remember sitting in meetings where teams debated data on trace soil and water contamination, arguing over how to measure environmental half-life or model the transfer up food chains. Driven by community feedback and hazard reports, R&D budgets grow for safer substitutes, smarter recovery processes, and materials capable of maintaining benefits while avoiding deep environmental costs. Partnerships between academic labs and manufacturers make up a big part of these efforts; field data handed off to modellers lets industry plan transitions with fewer surprises.
Toxicity Research
Toxicologists keep a close watch on perfluorooctanesulfonamide derivatives like this one. Lab animal studies reported liver and thyroid effects, developmental delays, and bioaccumulation in food webs. Analytical advances let researchers pick out trace levels in polar bears, drinking water, and even humans living near waste sites. This body of evidence led to regulatory limits in many countries. I’ve watched risk assessors balance public pressure with lagging toxicology studies, racing to set limits while industry called for more robust data. Comprehensive reviews from groups like the EPA or ECHA slow down only due to the massive complexity of fluorinated chemistries. Calls for blood-level monitoring, health surveys near factories, and long-term monitoring programs reflect concern that low-level, chronic exposure still raises unknowns for community health.
Future Prospects
Facing mounting pressure to balance industrial utility with health and environmental stewardship, producers and users of N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide rethink every stage of its lifecycle. International treaties and national rules on persistent organic pollutants shape the future, pushing supply chains toward recycled content, closed-loop manufacturing, and stricter recordkeeping for waste and end-of-life handling. My colleagues in product development invest more in alternative chemistries with similar benefits but proven degradability or lower bioaccumulation. Communities near plants push for transparency, on-site containment, and support for exposed residents, steering industry innovation toward safer, more responsible models. For those of us in the technical community, the lessons learned here echo in every conversation about new synthetic chemistry—urgency for responsibility, a hunger for real-world data, and a steady focus on protecting workers, consumers, and environments alike.
What is N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide used for?
What This Compound Does
N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide, also known as N-EtFOSE, falls in the class of fluorinated surfactants. On paper, it seems like just another mouthful from a chemistry textbook. In day-to-day life, it has shaped a lot of everyday products—often in ways most people never see. Its main role links back to its abilities to lower surface tension, resist stains, and help liquids spread evenly. These traits make it an easy pick for manufacturers in industries ranging from textiles to firefighting.
Real-World Applications
Think about the time spilled coffee slid right off your pants or the way oil beads up on treated carpet. Companies have used fluorinated compounds like N-EtFOSE behind the scenes for years in stain-resistant coatings. Anyone who remembers Scotchgard on sofas or water-repellent jackets already knows what these chemicals can do.
Firefighting foams, especially older formulations called aqueous film-forming foams (AFFF), leaned on N-EtFOSE for their punch. The foam creates a film that chokes out flames stronger and faster than traditional water. In electronics, it helped in manufacturing semiconductors by stopping static and acting as a wetting agent. Even some floor waxes and cleaners once relied on it to boost performance.
Health and Environmental Concerns
Stories about perfluorinated chemicals have filled the news in recent years. Scientists started drawing connections between these compounds and health problems: liver issues, hormonal disruption, and even certain cancers have been tied to long-term exposure. N-EtFOSE breaks down in the body and the environment into perfluorooctanesulfonic acid (PFOS), a substance that lingers for years in soil or water. This stubborn persistence earned PFOS and related compounds the nickname “forever chemicals.”
In my work with community groups near industrial sites, I have seen firsthand the anxiety these chemicals cause. Families worry about contaminated drinking water, fishing stocks vanish, and local wildlife numbers drop when perfluorinated chemicals wash downstream. Headlines about these chemicals showing up in polar bears’ blood and Arctic ice keep raising the temperature on this issue.
Regulatory Shifts and Safer Options
By the early 2000s, research had triggered tough new regulations. Production of N-EtFOSE, especially in North America and Europe, began to shrink. Big names like 3M phased out many uses. New standards forced companies to rethink the recipes they depended on for decades. Today, consumer demand for greener, safer products keeps growing.
Switching out chemicals like N-EtFOSE means real challenges for industry—often, safer substitutes come with trade-offs in performance or price. Plenty of new coatings still struggle to mimic the water and oil resistance without the environmental baggage. Some companies now use shorter-chain fluorochemicals or even turn to non-fluorinated options like silicon-based coatings and advanced waxes.
Moving away from long-lasting pollutants calls for regulatory grit, transparent research, and honest communication with the public. Safe drinking water and clean air matter much more than making life a bit easier with stain-proof carpets. The story of N-EtFOSE reminds us: the best solutions leave behind fewer problems for the next generation.
Is N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide hazardous to health?
A Look at the Chemical’s Use and Exposure
People who spend any time researching industrial chemicals often stumble across complicated names like N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide, sometimes called N-EtFOSE, which shows up in older fire-fighting foams and stain-resistant coatings. Companies liked it because of its resistance to heat, oil, and water. Folks who grew up in places with heavy manufacturing probably remember advertisements for magical products that would keep your carpets spotless and your raincoats dry.
Why Do We Care About N-EtFOSE?
N-EtFOSE belongs to a family of chemicals called PFAS, or “forever chemicals,” which means it hangs around in the environment without breaking down. Water supplies in unexpected places—from small towns in Maine to suburbs in Michigan—have tested positive for PFAS. Exposure happens mostly through water, though workplace inhalation in factories once posed a risk. Studies show these compounds build up in human blood over time.
Scientific Evidence on Health Impact
Researchers in the scientific community have linked high levels of PFAS, including N-EtFOSE, to health problems. Some results point toward an increased risk for cancer, immune system disruptions, liver damage, and problems during pregnancy. The National Academies and World Health Organization flagged PFAS for their persistence and evidence of toxic effects, especially after long-term exposure. In 2001, 3M, one of the main manufacturers, decided to pull these chemicals from shelves after growing pressure and concerning results in animal studies.
I’ve watched people in my own circle struggle with confusing language on chemical exposure. It’s tough to translate dense research into everyday worries. Still, the health consequences aren’t theoretical anymore; several lawsuits and local protests have grown out of real, documented health complaints.
Regulation and Clean-Up Efforts
Government agencies in the US and Europe now set stricter drinking water limits for PFAS. The EPA has started listing chemicals like N-EtFOSE as hazardous substances, which means new obligations for clean-up and transparency. These rules matter: people living near contaminated sites push for strict enforcement to keep families safe.
Dealing with contamination costs money and resources. Filtration systems get installed in affected towns, but the catch is that these systems only catch some PFAS, not all of them. Scientists keep working on better ways to remove or destroy the chemicals. Some companies moved away from N-EtFOSE to less persistent alternatives, but newer replacements don’t always solve the underlying problem.
Pursuing Safer Alternatives
Some manufacturers now market products as “PFAS-free,” a response to growing consumer demand for safer household goods. People want to know that the water out of their tap and the items in their kitchen won’t lead to trouble down the line. Raising awareness, supporting stronger disclosure, and pressing for investment in safer chemistry all play a role here.
Whether you work in a lab, run a small business, or look after a family, people share common worries over quiet contaminants that slip into daily life. The story of N-EtFOSE brings a lesson from yesterday, but also sets the direction for future choices: keep pushing for science-backed limits, better clean-up, and smarter alternatives.
What are the storage and handling requirements for N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide?
Why Safe Storage Matters
Storing chemicals like N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide (often called NEtFOSE) isn't some dry checklist. I’ve spent enough time in labs to know that treating chemical storage casually invites headaches or worse. NEtFOSE lands on regulatory watchlists because it belongs to the PFAS family. These substances earned the “forever chemicals” label thanks to their stubborn resistance to breakdown. NEtFOSE builds up in both the environment and animals. Unsafe storage can mean leaks, and leaks can find their way into water or the ground more easily than folks think.
Real Storage Requirements
For NEtFOSE, storage begins with proper containers. Skip the steel drums unless you’re sure about their resistance to corrosion. Polyethylene or fluorinated containers hold up better in most cases. Keep the lid or cap tightly closed. In a crowded storage room, somebody always knocks something over. A loosely closed container invites spills, and this compound isn’t one anybody wants on the floor or in the sink.
Temperature control sounds tedious, but temperature swings speed up chemical reactions, which can turn a harmless storage cabinet into a source of trouble. Store NEtFOSE away from direct sunlight and heat. I once watched a coworker forget this rule; a summer sunbeam hit a row of chemical jugs, and cleanup was ugly. 15-25°C (59-77°F) works well in most labs and warehouses.
Humidity counts too. Moisture collects on caps and labels, smears markings, and blurs hazard warnings. Water sometimes reacts with the chemical itself. Keep NEtFOSE dry at all times. Use desiccant packs if humidity gets high.
Handling and Personal Protection
Chemical gloves aren’t all equal. Regular latex rips too easily, and NEtFOSE slips through tiny pinholes. Nitrile or neoprene gloves last longer and offer better resistance. Full goggles matter. Even careful workers rub their eyes now and then, especially at the end of a shift.
Work in a well-ventilated area. My old lab had a windowless storeroom, and the first time we opened a long-abandoned container, fumes filled the air. Always open NEtFOSE containers under a chemical hood or with good air flow. Accumulated vapors sneak up on people and can lead to headaches, dizziness, or worse. Don’t trust your nose; odor isn’t a reliable warning.
Labeling and Emergency Readiness
One truth stands out from every safety training I’ve sat through: unlabeled chemicals cause panic. Properly label the container with both full chemical name and the accepted short name, date received, and hazard symbols. This isn’t about box-ticking—responders, coworkers, and even you, weeks later, need to know what’s inside.
Spill kits with absorbent material should sit close at hand. Regular sweeping, checking for leaks, and updating records doesn’t just please auditors. It cuts down the chance of cleanup nightmares or lost inventory. Local regulations may require special storage conditions for PFAS, so get familiar with them or risk costly surprises.
Toward Safer Practices
Long experience shows shortcuts rarely stay hidden. Relying on proper containers, clear labels, and controlled climates ensures NEtFOSE stays put and doesn’t become an environmental or health hazard. Facilities with high chemical throughput often automate tracking and maintain strict access, but even small labs benefit from regular audits and staff reminders.
Some places now work to phase out PFAS compounds like NEtFOSE. Until safer substitutes become routine, secure storage and careful handling provide the main line of defense. Even basic upgrades—better ventilation, up-to-date labels, and staff training—raise the safety bar without breaking the bank. Safe habits protect people and the planet from mistakes that only become obvious after real harm.
How should N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide be disposed of safely?
The Realities of Chemical Legacy
People working in science, manufacturing, or environmental cleanup might recognize long chemical names like N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide. For the rest of us, it points straight to perfluorinated compounds — the "forever chemicals" that don’t just break down. Once in the environment, these substances can persist for decades, even centuries. I’ve followed the trajectory of these compounds from distant factories to drinking water across the world, and it’s never a cheerful story.
This specific chemical, used in everything from surface treatments to firefighting foam, reflects why modern waste management faces huge challenges. Nobody can afford shortcuts — for public safety and for the trust we place in companies and governments.
Health Risks Aren’t Just Hype
It’s easy to brush aside chemical risk as “not my problem.” But with this substance, research links its relatives to immune suppression, thyroid issues, and change in cholesterol. Veterans and workers exposed to related compounds sometimes talk about mysterious illnesses that only make sense years later. The human cost from environmental carelessness showed itself near industrial sites: unexpected birth defects, cancer clusters among fishers, and soil that no farmer can sell.
Dumping or flushing this chemical down the drain means adding a little more to a global problem. Standard sewage plants don’t break down these compounds. Instead, they end up in rivers, then into drinking water, and even bottled water. You risk a domino effect that affects everyone—wildlife, livestock, families.
How the World Responds
Across North America, Europe, and parts of Asia, regulations tightened after citizens demanded protection. Now, many regions block perfluorinated chemicals from landfill or incinerators without tight controls. Some states, like New York and Michigan, fund collection drives and public education, especially near fire stations and airfields known for heavy contamination.
What works best so far? High-temperature incineration at facilities equipped with advanced scrubbers captures most of the harmful byproducts. Landfills designed for hazardous waste, with double liners and leachate controls, give another option for materials that resist burning. A few labs now test plasma-based destruction that promises fewer emissions, although costs still run high. Handling this chemical safely often means shipping it far from its point of use, which adds complexity and risk. Yet, each step toward proper containment reduces what leaches into soil or water.
Solutions Begin With Taking Responsibility
I’d argue personal and company-level responsibility make the biggest difference. Facility managers set policies guiding employees to separate, label, and document each container. Workers demand clear procedures and are the first to notice unsafe storage. Regular audits — ideally, by outsiders — keep everyone honest.
There’s a public right to know if this material sits unused in closets or ends up at local dumps. Laws like the Emergency Planning and Community Right-to-Know Act serve as a watchdog, but communities need support for real enforcement. Reporting concerns shouldn’t bring retaliation.
Practical Steps Forward
If someone handles this chemical, consulting local hazardous waste authorities is the first step. Never mix it with general trash or pour it down the sink. Use sealed, labeled containers and keep records tracking the chain of custody. Push for more research into destruction technologies. Push companies to replace “forever chemicals” with safer alternatives wherever possible. Cleanups will cost less if we stop adding to the pile today.
In the end, no single community can fight these risks in isolation. Global cooperation shapes every solution. When people care, they press regulators, business leaders, and scientists to put health first — for us and for generations coming after.
What is the chemical structure and molecular weight of N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide?
Breaking Down the Structure
Chemistry sometimes feels out of reach, but it only takes a closer look to see why certain compounds keep popping up in headlines. N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide, for instance, stands out for its unique structure and the wake it leaves across industries and science.
The name helps decode what it contains. On one end, you’ve got the “perfluorooctane” part; that’s an eight-carbon chain where all hydrogens are swapped with fluorines. These carbon-fluorine bonds run deep, making the molecule stubborn against breakdown. Attached to this chain sits a sulfonamide group—SO2NH—and on the nitrogen, you get two side arms: an ethyl group (CH2CH3), and a hydroxyethyl group (CH2CH2OH).
Visualizing Atoms and Connections
Lay out the atoms, and you end up with the chemical formula C12H8F17NO3S. This compound boasts 12 carbons, 8 hydrogens, and a wall of 17 fluorine atoms, an oxygen triplet, one nitrogen, and a single sulfur. Look at its molecular weight: you find the sum tips the scale at about 499.25 g/mol. That heavy hit comes from the fluorines and the beefy carbon backbone.
I’ve seen plenty of formulas, but this one always jumps out for those numbers alone. In the lab, it stands out for its resistance to just about everything: acids, bases, and even time. Try to cut one of those carbon-fluorine bonds, and the world’s best chemists often throw up their hands.
Why It Matters Beyond the Lab
Pull this molecule out of a chemistry book, and it becomes clear why it still shows up in gear meant to resist stains or water. I remember using old carpet sprays at home that never seemed to wear out. Later I learned that this kind of perfluorinated compound gave those results: it slides liquid away, never absorbs oils, and just won’t quit.
People tie this molecule and its family to a bigger story about “forever chemicals,” those tricky substances that hide in drinking water, soil, even blood. Their stability goes from benefit to hurdle in no time flat. One study from the CDC found these chemicals in the blood of nearly every American they tested. Perfluorooctanesulfonamides like this one are fingered in countless papers for these reasons.
Taking Responsibility and Exploring Solutions
As more evidence piles up, my respect grows for chemists and policymakers working to bring these compounds under control. Tools like advanced filtration, including activated carbon and reverse osmosis, show real promise in stripping these molecules from water sources. Research groups keep pushing for safer substitutes in fields like firefighting foams, textiles, and electronics.
It’s easy to see why understanding this chemical’s structure and weight matters. Every new fact helps sharpen the choices we get to make—whether it’s consumers voting with their wallets, companies exploring greener choices, or governments setting boundaries that put public health first. The right mix of vigilance, transparency, and creativity can push us toward a healthier, cleaner future, all starting with one well-understood molecule.
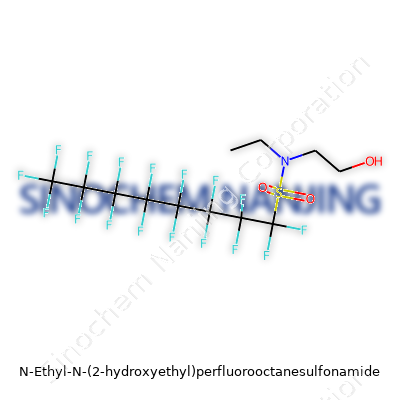
| Names | |
| Preferred IUPAC name | N-ethyl-N-(2-hydroxyethyl)perfluorooctane-1-sulfonamide |
| Other names |
N-Ethyl perfluorooctanesulfonamidoethanol N-Ethyl perfluorooctanesulfonamidoethanol (N-EtFOSE) N-Ethyl-N-(2-hydroxyethyl) perfluorooctanesulfonamide 2-(N-Ethylperfluorooctanesulfonamido)ethanol EtFOSE |
| Pronunciation | /ɛn-ˈɛθɪl-ɛn-ˈtuː-haɪˌdrɒk.siˌɛθɪl-pɜrˌfluːrəˌɒkˈteɪnˌsʌlˈfɒnəˌmaɪd/ |
| Identifiers | |
| CAS Number | 2991-50-6 |
| 3D model (JSmol) | `3D Model (JSmol) String:` `C(CN(CCO)S(=O)(=O)C8F17)` This is the SMILES string, which can be loaded into JSmol or other molecular visualization tools to render the 3D structure. |
| Beilstein Reference | 1361194 |
| ChEBI | CHEBI:131452 |
| ChEMBL | CHEMBL572602 |
| ChemSpider | 492028 |
| DrugBank | DB02586 |
| ECHA InfoCard | 03e9916e-b5ef-41e5-beb5-3b532b7c371e |
| EC Number | 251099-16-7 |
| Gmelin Reference | 97755 |
| KEGG | C19228 |
| MeSH | D010983 |
| PubChem CID | 30836 |
| RTECS number | YJ8570000 |
| UNII | Y06ONGS6EZ |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C10H20F17NO3S |
| Molar mass | C10H20F17NO3S, 499.32 g/mol |
| Appearance | Colorless to pale yellow transparent liquid |
| Odor | Odorless |
| Density | 1.52 g/mL at 25 °C |
| Solubility in water | insoluble |
| log P | 3.73 |
| Vapor pressure | <1 mm Hg (20 °C) |
| Acidity (pKa) | 5.3 |
| Basicity (pKb) | 5.34 |
| Magnetic susceptibility (χ) | -59.5×10^-6 cm³/mol |
| Refractive index (nD) | 1.355 |
| Viscosity | 12-18 cP (25°C) |
| Dipole moment | 3.49 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 527.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1511.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -8022 kJ/mol |
| Pharmacology | |
| ATC code | D08AJ18 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Harmful to aquatic life with long lasting effects. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335 |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P337+P313 |
| Flash point | > 110°C (230°F) |
| Lethal dose or concentration | LD50 Rat Oral > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 5000 mg/kg |
| PEL (Permissible) | PEL (Permissible) for N-Ethyl-N-(2-Hydroxyethyl)Perfluorooctanesulfonamide is not specifically established by OSHA. |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonamide N-Ethylperfluorooctanesulfonamide N-Methyl-N-(2-hydroxyethyl)perfluorooctanesulfonamide Perfluorooctanesulfonyl fluoride Perfluorooctanoic acid |

