N-Ethyl-1-Naphthylamine: Commentary on an Underappreciated Aromatic Compound
Historical Development
N-Ethyl-1-naphthylamine came into chemical circles during the late 19th and early 20th centuries, a period marked by rapid growth in synthetic organic chemistry. Researchers chasing new dyes and intermediates stumbled upon a host of arylamines. Naphthylamines, in particular, showed promise for their deep color, reactivity, and versatility in industrial applications, and N-alkyl substituted forms quickly drew interest. Chemists tracked the use of the naphthalene ring as a platform for building more complex molecules. While not as loud as its cousins like aniline, N-ethyl-1-naphthylamine gained ground quietly, appearing as a specialized intermediate in dye and pigment manufacturing and organic synthesis. Its history traces closely to advances in coal tar chemistry and the steady expansion of synthetic dyes. As labs grew more focused on health, safety, and mechanistic study in the 20th century, this compound kept its place, sometimes overshadowed, but never irrelevant.
Product Overview
N-Ethyl-1-naphthylamine appears as a yellowish to brownish crystalline powder. Looking at a sample, most chemists would first notice its faint aromatic odor. Companies dealing in fine chemicals label it as an intermediate for research, especially where clients seek to build up larger, more complex aromatic frameworks. The cost of production often comes up, since demand remains steady but not overwhelming. Distribution follows the demands of specialty fields—mostly small batch, often pre-ordered with specific purity levels, and supported by data sheets spelling out every lot’s characteristics.
Physical & Chemical Properties
The molecule carries a fused-ring naphthalene backbone with a single ethyl substituent on the nitrogen atom. This arrangement leads to a melting point typically in the 65-75°C range, though impure samples can show broad melting behavior. Its solubility profile favors organic solvents—ethanol, ether, chloroform—over water, which fits with most aromatic amines. The compound’s stability holds up at room temperature as long as it stays out of direct sunlight or strong oxidants. In the lab, the faint color can deepen upon exposure to air, signaling slow oxidation, which remains a point of interest in handling and storage decisions.
Technical Specifications & Labeling
Commercial suppliers publish technical sheets detailing purity, moisture content, melting point, and residual solvents. Analysts use chromatography and spectroscopy—typically HPLC and NMR—for identity confirmation. Labels list hazard codes under GHS, especially indicating dermal and respiratory risks, due to the aromatic amine structure. Packaging typically features amber glass or metal tins to keep light away. In my experience, every chemist values the extra care in labeling, especially with aromatic amines that can cause confusion if containers get mixed up.
Preparation Method
Synthetic routes most often involve the direct alkylation of 1-naphthylamine with ethyl halides under basic conditions. Some labs use phase-transfer catalysts to increase yields and favor the N-alkylation over ring substitution. The process usually starts by dissolving 1-naphthylamine in an organic solvent, introducing an ethylating agent—commonly ethyl bromide—and adding a base like potassium carbonate. The reaction agitates for several hours, sometimes with gentle heating. Careful extraction and purification steps, usually crystallization or vacuum distillation, follow to remove unreacted materials. Waste management always enters the discussion here, given the toxicity concerns around both the starting and byproduct amines, a lesson I learned early from senior colleagues who handled these reactions with a steady hand and steady nerves.
Chemical Reactions & Modifications
The lone electron pair on the nitrogen atom lends the molecule considerable reactivity. N-Ethyl-1-naphthylamine couples readily with diazonium salts, forming vivid azo dyes that once painted much of the world’s textiles. The aromatic system also welcomes electrophilic substitution. Sulfonation, acylation, and halogenation open up further derivatization, enabling scientists to tailor-make custom intermediates for pigments, pharmaceuticals, or analytical reagents. In my own work, I once saw a project transform a kilogram batch through oxidation and subsequent coupling, watching the color change as fresh molecules emerged at every step.
Synonyms & Product Names
Catalogs and papers bounce between several names. N-Ethyl-1-naphthylamine can also appear as 1-(Ethylamino)naphthalene, Ethylnaphthylamine, or SEPA. In dye chemistry, some legacy naming lingers, with old labels like C.I. 37110. Accurate identification avoids costly mistakes, whether searching in a database or placing an order for a time-sensitive batch. Attention to these names pays off, since even a misplaced digit leads to entirely different compounds.
Safety & Operational Standards
Aromatic amines deserve respect for their health risks, including sensitization and possible carcinogenicity with chronic exposure. N-Ethyl-1-naphthylamine requires proper personal protective equipment—gloves, eye protection, and well-ventilated workspace. Guidelines from agencies like OSHA and the EU’s REACH regulation guide handling. Spills draw attention to the need for immediate containment, as these compounds can be absorbed through skin or present inhalation hazards when dust forms. Experienced operators rely on fume hoods and avoid working alone during scale-up runs. Disposal follows strict hazardous waste protocols, echoing long-standing advice from occupational safety officers.
Application Area
The market for N-Ethyl-1-naphthylamine leans heavily toward industrial and research settings. Dye manufacturers value it for its performance in coupling reactions, delivering reliable shades in both textiles and pigments for inks or plastics. Analytical chemistry textbooks reference it in nitrite determination, where its color-forming ability creates sensitive tests for trace analysis. Specialty pharmaceutical synthesis sometimes taps its reactivity to build larger molecular frameworks. In my own graduate research days, I encountered the compound as a building block for polycyclic aromatic projects that aimed at high-performance materials.
Research & Development
Modern R&D continues to explore both the core structure and modifications of N-Ethyl-1-naphthylamine. Efforts target safer synthesis, greener solvents, and improved specificity in color reactions for analytical tests. Chemical engineers weigh in on continuous flow methods to increase safety and yield, hoping to sidestep batch-to-batch variability. Computational chemists examine the molecule’s electron density and aromaticity, chasing deeper understanding for both synthesis and regulatory compliance. Collaboration between academic labs and industry keeps pushing boundaries, sometimes chasing exotic dyes and other times working to understand environmental impacts and safer disposal techniques.
Toxicity Research
Toxicologists devote careful study to aromatic amines, including N-Ethyl-1-naphthylamine, because of their links to methemoglobinemia and possible carcinogenicity with chronic exposure. Animal studies and cell culture work both point to structural alerts common to arylamines; they sometimes undergo metabolism in the body that turns harmless molecules into potent electrophiles capable of damaging DNA. While this compound hasn’t featured in infamous public health disasters, regulatory agencies urge a cautious approach, recommending exposure limits and calling for regular monitoring in workplaces handling larger volumes. Personal stories in the lab underline the importance of risk assessments and alertness to even minor spills.
Future Prospects
The horizon for N-Ethyl-1-naphthylamine stretches along two lines: synthetic opportunity and regulatory challenge. The push for non-toxic dyes and pigments challenges the industry to innovate safer alternatives or to modify existing molecules to lower their hazard profiles. Analytical chemistry keeps the compound in toolkits for its selectivity, while materials science wonders how modified naphthylamines might contribute to organic electronics or photonics applications. Regulatory scrutiny will only grow, driving improvement in production safety, waste management, and environmental monitoring. Investment in green chemistry holds promise, as does deepening mechanistic understanding enabled by modern computation and instrumentation. Hands-on experience, both past and present, underscores the need for thoughtful stewardship and creative problem-solving as the field adapts to changing demand and rising safety standards.
What is N-Ethyl-1-Naphthylamine used for?
Chemistry That Shows Up in Daily Life
N-Ethyl-1-naphthylamine might sound like something only a chemist worries about, but this compound finds its place in some interesting corners of industry and science. The main point with chemicals like this isn’t complexity – it’s what people manage to do with them. Most folks won’t run into a jar of N-Ethyl-1-naphthylamine at the grocery store, but it often plays a role in what researchers, manufacturers, and even some environmental experts do behind the scenes.
A Closer Look at Laboratory Uses
In colorimetric analysis, this material matters. It reacts in specific ways that help chemists figure out the amount of nitrite in water samples. I remember seeing a team use color tests on river water to spot pollution. N-Ethyl-1-naphthylamine paired with sulfanilic acid gives a pink color if nitrite exists in the sample. The color shows up, the result gets measured, and a real answer – not just a guess – tells officials if there’s a problem with contamination.
Dyes and Pigments: Deep Colors with Small Quantities
The dye world relies on raw materials like this quite a lot. Making certain azo dyes wouldn’t be possible without such amines as building blocks. Textile production and ink manufacturing care about reliability and cost, and these naphthylamine compounds come into play because they handle both. Years ago, I worked with a designer who wanted a specific shade of orange for a project. The pigment source traced back to reactions involving N-alkyl naphthylamines just like this one. It reminded me that, even if most users never see the raw chemicals, they notice the colors they create.
Challenges Surrounding Safety and Regulation
Chemicals always attract attention over potential hazards. N-Ethyl-1-naphthylamine isn’t one you’d let spill across the kitchen table. Skin contact, inhalation, or improper storage can go badly, so industry regulations exist for a reason. Labs train staff on safe handling, and the material gets stored with labeling that leaves no guessing. Reports from the European Chemicals Agency and other groups warn about the risks. Companies follow rules about quantities, registration, and disposal because a shortcut could hurt workers or the environment.
Can We Do Better? Exploring Alternatives
The search never really ends for safer or greener chemicals. Some research looks for substitutes that do the same job in nitrite analysis or dye preparation, but with less risk. I've talked with chemists who see this as gradual work. New testing agents might arrive, but it often takes years to balance effectiveness, safety, and cost. At the same time, stricter rules on labeling and disposal keep encouraging improvements, even for established compounds.
Why Paying Attention Pays Off
There’s sometimes a gap between lab science and the impact on daily living. N-Ethyl-1-naphthylamine proves that what happens in the background shapes what people see, touch, and use every day – whether clean water, bright fabrics, or tested ink. When industry and science stay honest about risks and keep looking for smarter solutions, everyone benefits. That mix of vigilance and innovation shows why understanding even obscure chemicals fits into a bigger story about progress and public trust.
What is the molecular formula of N-Ethyl-1-Naphthylamine?
Why Chemical Formulas Matter in Our Daily World
Anybody who has spent time mixing things together—be it in a kitchen, garage, or science lab—knows how quickly things can go wrong when the ingredients aren’t just right. The same goes for chemistry. The molecular formula of a compound is not a badge or a label, but a crucial bit of information that tells us what is in a molecule and how those pieces connect. For N-Ethyl-1-Naphthylamine, the molecular formula is C12H13N.
Pulling Apart the Name: What’s Really Involved?
N-Ethyl-1-Naphthylamine comes off as a complicated name at first glance, but it tells a story. The “naphthyl” part points straight to naphthalene, those old-school mothballs that many remember from their grandmother’s closet. A naphthalene ring—two benzene rings fused together—gives us the backbone here. Add “amine,” and you’re adding a nitrogen atom tagged onto the ring at its number 1 position.
The “N-ethyl” means there’s an ethyl group—basically two carbons and five hydrogens—attached directly to the nitrogen atom instead of to the ring itself. So, folks in a lab or in industry can look at “C12H13N” and know right away what atoms they’ll be working with. This isn’t just trivia; when chemical reactions happen, those numbers decide the ratios and the safety precautions.
Why Do Scientists and Engineers Keep Coming Back to This Compound?
N-Ethyl-1-Naphthylamine shows up often in dye production, in biological research, and in industrial analysis. Its usefulness comes from the naphthyl structure, which easily absorbs light, and the amine group, which connects easily with acids and other chemical groups.
Lab techs and scientists keep careful records because the world runs better when there are fewer surprises about what goes into reactors or test tubes. Precise knowledge of that C12H13N formula helps chemists avoid expensive mistakes or dangerous waste. In my own experience, mixing reagents or scaling up reactions without sharp attention to these details almost always leads to a problem—anything from weird smells to explosions. Getting those numbers wrong costs more than pride; sometimes, it’s real money or safety on the line.
What Gets in the Way of Clear Chemical Communication?
One ongoing issue in chemical industries and research is mislabeling or data entry mistakes that can lead to incorrect handling of substances like N-Ethyl-1-Naphthylamine. Human errors slip into the process, especially in fast-paced environments. As a fix, double-checking molecular formulas before running a reaction or inputting data into analysis tools helps engineers and lab staff dodge costly mix-ups. Training newcomers to respect the difference between an ethyl and a methyl group, or between a naphthyl and a phenyl, makes a world of difference.
Digital tools now help keep formulas and structures straight, but the best safety net involves developing a habit of checking the basics—starting with formulas like C12H13N. Relying on clear labels, frequent audits, and honest communication nips confusion in the bud and lets labs and production lines run smoothly. Instead of glossing over these details, the focus on molecular formulas keeps both research and industry anchored in accuracy.
Is N-Ethyl-1-Naphthylamine hazardous to health?
What We Know About N-Ethyl-1-Naphthylamine
N-Ethyl-1-naphthylamine shows up in industrial and laboratory settings. Workers may see it used in dye manufacturing, certain lab tests, and chemical analysis. At first glance, the compound looks like any other chemical, blending into a long list of substances with complex names. That often leads to people not thinking about risks until they become unavoidable. Ignoring the risks doesn’t make them disappear, especially with compounds that pack surprises for the human body.
Documented Hazards and Routes of Exposure
This chemical doesn’t feature heavily in mainstream headlines, but medical research flags it for health watchers. Breathing in or touching N-ethyl-1-naphthylamine raises alarms because it can travel into the body through the skin or lungs. Workers without protective measures face more than short-term irritation. The compound finds its way into the bloodstream, where it may harm organs over time. I’ve seen coworkers in busy labs forget gloves or delay mask use, thinking short exposure won’t matter. The reality? Even quick contact builds up in unexpected ways.
Scientific papers from toxicologists highlight links between aromatic amines (the family where this compound belongs) and a higher risk of certain cancers. The International Agency for Research on Cancer treats related substances with suspicion. Chronic exposure pushes up cancer risk, especially bladder cancer. Repeated or long exposure at low doses often slips under the radar, but that’s where the long-term damage hides. Skin, lungs, and the liver may feel the brunt, showing symptoms—rashes, difficulty breathing, or headaches—long before a chronic problem surfaces.
Regulatory Gaps and Ongoing Research
Many countries lack clear, enforceable limits for N-ethyl-1-naphthylamine exposure. The U.S. Occupational Safety and Health Administration does not list a specific permissible exposure limit for this chemical. Absence of regulation doesn’t equal safety. Scientific organizations like the American Conference of Governmental Industrial Hygienists call for caution and recommend lung and skin protection for anyone handling aromatic amines. From my time working with hazardous substances, no news about limits often means regulators haven’t caught up with scientific findings yet. That leaves people relying on their own judgment or their employer’s decisions, making the workplace a risky place for mistakes.
Preventing Hazard: Lessons from the Lab and Factory Floor
Personal experience working in chemical environments teaches one thing: safety measures work only if people believe the risk is real. Lab directors and plant managers who set up regular training, keep protective equipment accessible, and enforce mandatory gear use always see better outcomes. Proper gloves, fitted respirators, and ventilation systems act as the first and last defense against exposure. Good labeling and storage practices help avoid accidental contact. Employees who push for safety audits and who keep lines of communication open with supervisors find more protection against chronic hazards. Bystanders, including janitors or delivery workers, also deserve clear warnings and training since chemicals don’t discriminate with their risks.
Finding a Healthier Path Forward
Chemical companies and industrial leaders can support independent testing, demand up-to-date safety data, and apply the highest standard for worker safety, even if regulations don’t yet require it. Research into safer alternatives should take priority, reducing dependency on chemicals with a dangerous history. Sharing lessons from health case studies, alongside enforcing controls, can help move the culture in the right direction. For those on the ground, speaking up about unsafe practices lifts the standard for everyone and avoids repeating tragedies seen with similar industrial chemicals. Trust in safety starts with good information and real protection, not just crossed fingers.
What are the storage conditions for N-Ethyl-1-Naphthylamine?
Why Safe Storage Matters
Working around chemicals brings back memories of my early days in the lab. N-Ethyl-1-Naphthylamine sat on one of our crowded shelves, right next to a host of aromatic amines. One spilled bottle taught me that treating chemical storage lightly courts real danger. This compound, much like its close relatives, can break down with light, heat, or air exposure, putting people and experiments at risk.
Keep It Cool and Dry
This chemical doesn’t play well with moist air. Humid spaces can lead to chemical degradation, costing both safety and data integrity. Storing it below 25°C keeps most unwanted reactions at bay. Our fridge, which rarely climbed above 20°C, kept supplies stable through the summer months. Even a warm storeroom can nudge sensitive chemicals toward unwanted breakdown, and old stock rarely lasted long in such conditions.
Tight Containers Mean Fewer Headaches
At work, I learned not to leave things halfway sealed. N-Ethyl-1-Naphthylamine needs an airtight container—polyethylene bottles work, but amber glass bottles give even more security against light and oxygen. Volatile organic compounds can evaporate or pick up moisture, leading to tainted samples. A forgotten lid once wrecked a week’s worth of research. Clamps and screw tops, routinely checked, take only a minute to secure and save hours of cleanup and risk.
Away From Sunlight and Sparks
Direct sunlight and lab lamps heat chemicals quickly. Shading the shelf made a huge difference in our old storeroom; nothing broke down or yellowed as it did before. N-Ethyl-1-Naphthylamine can react if exposed to light and strong oxidizing agents. I never kept it near bleach or nitrates because a simple mistake could set off a chain reaction. Fire safety officers always reminded us: don’t stack strong reactants anywhere close.
Label Everything. Then Check Again.
Faded labels and stray markers cause confusion in even the most organized storage spaces. Clear labeling with large print and hazard information keeps everyone on the same page. Chemical names, dates received, and hazard symbols should be visible at a glance. A routine sweep for leaking bottles and illegible labels caught problems early, long before they turned into emergencies.
What Happens If You Ignore the Rules?
I’ve seen the aftermath of poor storage—crystallized clumps, odd fumes, and sometimes headaches just from walking by. Decomposition products sometimes prove more toxic than the parent chemical, and reordering rare supplies isn’t always easy. Old samples can sabotage test results or create costly delays. One spill can mean a day spent in protective gear, ventilating and cleaning instead of actually working.
Solutions for Safer Labs
Investing in controlled storage saves money and hassle. Climate-controlled cabinets, detailed inventories, and routine training sessions saw our incident rate drop. Routine safety meetings and checklists ensured nobody forgot old stocks in the back corners. Everyone in the room knew mishandling one bottle could ruin a week’s work or worse, send a colleague to the emergency room. Better habits kept us safe, kept our science honest, and left everyone less anxious after hours.
What is the purity specification of N-Ethyl-1-Naphthylamine?
Getting Down to Practical Purity
N-Ethyl-1-Naphthylamine finds itself used in plenty of chemical labs and specialty manufacturing setups. Ask any analyst or QC manager about this compound, and you’ll often hear frustration about conflicting numbers on specs. Tabbing through different suppliers’ data sheets, you’ll see purities quoted at 98%, sometimes up to 99% — but every tiny variance means something big for the person doing the actual experiment or synthesis.
Why Purity Matters in Daily Operations
From my time working in a pigment lab, quality of this material could make or break a whole batch. N-Ethyl-1-Naphthylamine plays a major role in diazo coupling reactions, and running it under less-than-ideal purity led to odd colors or even full-on failed reactions. Imagine a supplier promising 98% pure stock, but that last two percent sneaks in unknown amines or junk. You spend money, time, and lose trust along the way. Most buyers push for over 98% on the Certificate of Analysis because nobody wants to guess what trace impurities will show up on the chromatogram.
Understanding Typical Impurities
The main impurities often crop up as related naphthylamines, leftover reagents from the manufacturing route, or decomposition products that slip in during handling. Running a sample by HPLC, I’d generally notice tiny blips that—while small—couldn’t be ignored for sensitive catalyst work. I recall one batch where a half-percent of starting naphthylamine completely blocked a downstream reaction. It didn’t look like a big deal on paper, but on the bench, all that paper meant nothing.
Industry-Driven Purity Standards
Producers usually promise a figure at or above 98%—not as some arbitrary goal, but because that purity cuts down troubleshooting and keeps reactions more predictable. Pharmaceutical outfits often demand even tighter specs, like 99% minimum, backed by GC-MS and NMR data. Environmental labs, on the other hand, watch for trace amines since these can mimic pollutants in analytical runs. Regulators lean on this, too: a shipment labeled at one purity really has to match up on testing, or audits and import problems start cropping up.
Tools That Separate Good from Great
Checking N-Ethyl-1-Naphthylamine quality, I’ve always trusted a solid set of high-resolution HPLC or GC results. TLC won’t cut it for high-stakes work. Nobody likes interpreting peaks with ghost impurities lurking. These days, top suppliers document every detected impurity down to 0.1% or even finer. Certificates typically show both minimum assay (the main compound) and maximum for total impurities.
Next Steps: Holding Suppliers Accountable
Many labs burn through excess time clearing up bad material when suppliers slack on documentation. It pays to ask for recent, traceable batch data and maybe even run side-by-side tests before scaling up. For university researchers, a bottle labeled 97% might look like a steal, but any big project deserves double-checking before risking publication credibility. Short-term savings by cutting corners on purity rarely beats the cost of rework, lost time, or inaccurate results.
Moving Toward Transparency
As N-Ethyl-1-Naphthylamine’s uses keep expanding, the demand for transparent reporting follows suit. Labs no longer accept vague promises. They want to see real numbers and real batch data. This openness increases trust across the board, making both production and research easier for everyone involved.
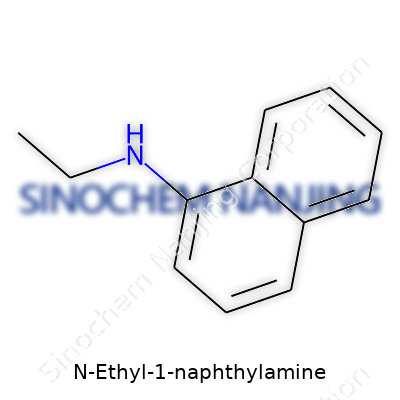
| Names | |
| Preferred IUPAC name | N-ethyl-1-naphthalenamine |
| Other names |
N-Ethyl-1-naphthalenamine 1-Naphthyl ethylamine N-Ethyl-1-naphthylamin N-Ethyl-α-naphthylamine |
| Pronunciation | /ɛn-ˈɛθɪl-wʌn-ˈnæfθɪl-əˈmiːn/ |
| Identifiers | |
| CAS Number | 612-12-4 |
| Beilstein Reference | **1208737** |
| ChEBI | CHEBI:34317 |
| ChEMBL | CHEMBL157826 |
| ChemSpider | 83456 |
| DrugBank | DB04257 |
| ECHA InfoCard | 100.008.488 |
| EC Number | 202-235-6 |
| Gmelin Reference | 79277 |
| KEGG | C06217 |
| MeSH | D009663 |
| PubChem CID | 11851 |
| RTECS number | KK7525000 |
| UNII | Z42V8U9B1Z |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID1022923 |
| Properties | |
| Chemical formula | C12H13N |
| Molar mass | 207.29 g/mol |
| Appearance | Light yellow to brown crystal |
| Odor | amine-like |
| Density | 1.023 g/mL at 25 °C |
| Solubility in water | insoluble |
| log P | 3.1 |
| Vapor pressure | 0.0133 hPa (25 °C) |
| Acidity (pKa) | 5.3 |
| Basicity (pKb) | 5.97 |
| Magnetic susceptibility (χ) | -74.1·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.637 |
| Viscosity | 110.00 cP (20°C) |
| Dipole moment | 3.03 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 318.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 85.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3703 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause an allergic skin reaction, suspected of causing cancer |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P304+P340, P330, P405, P501 |
| Flash point | Flash point: 157°C |
| Autoignition temperature | 455°C |
| Lethal dose or concentration | LD50 oral rat 1680 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat: 1230 mg/kg |
| NIOSH | RN0956 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.5 mg/m3 |
| Related compounds | |
| Related compounds |
N-Methyl-1-naphthylamine N,N-Diethyl-1-naphthylamine 1-Naphthylamine 2-Naphthylamine N-Ethyl-2-naphthylamine |

