N-Cyclohexylcyclohexanamine Nitrite: More Than Just a Compound
Tracing the Roots
Chemistry, much like any other science, advances through everyday observation and persistent human curiosity. N-Cyclohexylcyclohexanamine nitrite, though not a household name, stands as a result of decades of exploration into organic amines and nitrites. People sometimes overlook how substances like this grew from basic laboratory reactions to occupy valuable space in materials science and specialty synthesis. This compound, born from simpler cyclohexyl amine derivatives, reflects just how deep synthetic organic chemistry reaches into complexities. Interest in such compounds often surged after World War II, when synthetic pathways rapidly evolved thanks to new industrial catalysts and improved reactors. Today, those old flasks and handwritten lab notebooks have given way to digital modeling and automated syntheses, yet the core motivations haven’t changed—curiosity, efficiency, and the promise of new applications.
Understanding the Compound
N-Cyclohexylcyclohexanamine nitrite features a fused cyclohexyl group attached to an amine nitrogen, embraced by a nitrite moiety. Even if you’ve never worked with it, you can imagine the complexity once you see the molecular diagram: the molecule keeps a certain structural rigidity thanks to its two rings. In my own experience, compounds like this typically arrive as waxy solids or slightly yellow oils, often described by chemists not for their beauty, but for their stubbornness during purification. In practical work, subtle shifts in temperature or solvent selection can change yield and quality dramatically. For those handling lab-scale volumes, synesthetic smells and slow-developing color changes offer busy hints of a reaction’s end—elements that stay with you after a long day at the bench.
Diving Into Properties
On the bench, N-Cyclohexylcyclohexanamine nitrite doesn’t behave like a simple salt or oil. Its melting point usually shows sensitivity to trace contaminants, a trait that often frustrates even steady hands in purification. The nitrite group reacts keenly to strong acids and reducing agents, prompting swift releases of nitrogen oxides, which nobody wants in a small, closed workspace. This property alone underscores why proper ventilation and personal protection demand respect, not shortcutting. In solution, the compound prefers nonpolar or slightly polar solvents. Sometimes, manipulating these solubilities lets you tease out greater purity or separate stubborn by-products. Watching it under UV sometimes hints at low-level impurities, though most researchers depend on NMR and chromatography for reliable results.
Technical Requirements and Labels
Every chemical compound deserves careful labeling, both for safety and efficient workflow. In any research environment I’ve visited, there’s always a detailed record, not only for compliance but for tracking questionable batches or strange results. Labels on N-Cyclohexylcyclohexanamine nitrite don’t let you forget the presence of a nitrite. Most bottles flash warning symbols and storage temperature ranges. Whether stored in amber glass or thick plastic, the compound needs a stable, dry place, away from oxidizers or acids. Mislabeling, even by a decimal point, can spark confusion for weeks in a high-turnover lab. I once witnessed a near-miss when a mislabeled amine led to reactive waste production—cleaning that situation forced everyone to rethink their recording methods.
Crafting the Compound
Synthesis of N-Cyclohexylcyclohexanamine nitrite doesn’t follow a back-of-the-envelope recipe. Most methods employ the diazotization of the corresponding amine using sodium nitrite and an acid, often done at low temperature to avoid uncontrolled decomposition or gas evolution. This step requires patience and a steady approach—rushing never helps, especially as the reaction sometimes creeps instead of explodes. Watching for the endpoint, typically through color change or gas evolution, gives seasoned researchers an edge over newcomers. Work-up often includes extraction with nonpolar solvents, followed by vacuum distillation or column chromatography. Losses mount unless everything stays dry. Thorough washing and careful separation reduce nasty surprises when you next open the container.
Tuning and Tweaking: Chemical Reactions
Chemists don’t just stop at basic synthesis. N-Cyclohexylcyclohexanamine nitrite slips into further reactions, acting as a versatile building block or intermediate. Reductive cleavage yields various other cyclohexylamines, while heating can promote changes to oximes or more exotic nitrogen-containing rings. Some groups use the nitrite’s cis/trans isomerism to probe reaction pathways or influence physical handling. Others seek to block reactive sites for more targeted downstream coupling, especially in pharmaceutical discovery projects. My old lab group once spent weeks chasing orthogonally protected versions—very high-yielding only on paper, yet never quite matching theoretical promise in practice. That unpredictability keeps things interesting and frustrates those who crave only black-and-white outcomes.
A Matter of Many Names
Chemical synonym lists sometimes read like genealogy charts. N-Cyclohexylcyclohexanamine nitrite pops up in registries under names that echo its structure or hint at former uses—like dicyclohexylamine nitrite, or simply cyclohexylamine nitrite. Catalog searches often return trade names or manufacturer tweaks, leading to more confusion than clarity. I’ve spent late hours sorting shades of difference in supplier catalogs, only to find that most references point backward to the same core chemical. That muddle can waste time and resources for inexperienced researchers digging for true novelty.
Rules, Risks, and Responsibility
Safety standards have climbed since the days when chemists sniffed unknown substances without a second thought. Handling nitrites, especially ones tethered to hydrophobic rings, brings its own teaching moments. Nitrite esters and related amines can trigger serious health effects. Exposure brings risks of methemoglobinemia, headaches, and even respiratory issues, problems that demand respect for gloves, glasses, and fume hoods. Regulatory checklists push teams to ask tough questions about disposal and accident recovery. I remember a training exercise that made everyone map out routes for containing chemical spills—nobody enjoyed the process, but everyone knew it could save a life. Training, retraining, and regular drills help keep day-to-day work grounded in precaution over convenience.
Where the Work Happens
Application remains the final test for any specialty chemical. In industrial circles, amine nitrites like this often find themselves as intermediates, stepping stones on the way to more potent drugs, agricultural additives, or stabilizers in coatings. Sometimes, the niche nature of the material means only a handful of labs worldwide ever handle more than a few grams at a time. On a personal note, it’s rare to see such compounds go straight into consumer products; risk profiles and production costs tend to hold them back. Research teams—in both business and academia—often reach for these compounds during method development, prodding for new reactivity or safer derivatives of existing functional groups. Each project uncovers new quirks, sometimes leading to publications and patents, more often ending in silent, gritty lessons learned at great expense.
Digging Deeper in Research
N-Cyclohexylcyclohexanamine nitrite grabs attention in toxicology studies, as researchers look to answer outstanding questions about chronic exposure and organ-specific targets. Toxicity research hinges on reproducibility, and studies draw from both cell cultures and animal models. A few routes point toward environmental persistence, raising old worries about accidental spills or improper waste handling. Regulatory agencies set increasingly strict limits on discharge and exposure, reflecting the faint traces sometimes detected downstream of production facilities. Researchers face a tough balancing act—pushing toward breakthrough applications while honoring community oversight and staying transparent about hazards.
Looking Down the Line
Future prospects rarely stand still in chemical development. Automation and better computer modeling promise safer, more efficient production, hopefully moving away from high-risk reagents or energy-intensive conditions. Some groups push for greener syntheses, swapping hazardous nitrites for milder oxidants whenever possible, or employing continuous-flow systems that can tamp down volatile intermediates. Others fixate on regulatory forecasts, factoring in pressure from global health groups or environmental watchdogs. In all this, the value of strong ethical standards and open data sharing keeps things grounded. Imagining the future for N-Cyclohexylcyclohexanamine nitrite means wrestling with risk, reward, and the creative mischief that fuels chemistry at every level. Progress depends on smart, committed people who never lose sight of the real-world impact—people who keep their sleeves rolled up and their tools sharp, always questioning, always looking one step further.
What are the main uses of N-Cyclohexylcyclohexanamine Nitrite?
The Role in Chemical Synthesis
Chemists and manufacturers working on specialty products look for reagents like N-Cyclohexylcyclohexanamine Nitrite because it helps switch up and build new molecular structures quickly. It acts as a building block, especially for fine chemicals, agrochemicals, and certain dyes. It helps attach nitrite groups, create nitroso compounds, and sometimes shuffle atoms in a molecule to make new versions of a product. The long name aside, these shifts in molecular make-up matter for anyone improving performance or safety in products used every day—whether that's coloring in textiles or active ingredients in protective coatings.
Intermediate for Pharmaceuticals
My work in chemical safety makes it clear: every complex medicine starts with a batch of intermediates, and N-Cyclohexylcyclohexanamine Nitrite shows up in some of those recipes. Pharmaceutical researchers count on it to deliver precise modifications during early steps. It acts as a springboard, allowing scientists to connect the right chemical groups that become new health therapies. If you’ve been in a lab, you know that skipping corners never works; every step in a medicine’s creation has huge weight on what you get at the end. Quality, purity, and traceability shape the final outcome—people care about their health more than catchy marketing.
Polymer and Plastic Additives
Polymer makers face tough questions from customers who need durability, color, flexibility, and resistance to breakdown in the sun or harsh conditions. Here, specialty nitrites like N-Cyclohexylcyclohexanamine Nitrite become useful. Manufacturers blend small amounts of such compounds to tune the physical properties of plastics and rubbers: things like stability under heat, better colorfastness, and less yellowing over time. In factory settings, even a gram per ton can make a difference over how a window frame or an outdoor seat holds up year after year. I’ve seen testing labs run panels through crazy weather cycles—nobody wants a lawn chair that cracks after one season.
Research and Development Applications
In research circles, this compound earns attention because it lets scientists try out new reaction pathways and product ideas. Small-scale syntheses in the lab allow researchers to test potential pesticides, performance coatings, or additives—all without betting the farm on large production batches. From what I’ve seen in universities and industrial innovation centers, access to these chemicals brings faster progress. Such tools turn the “what ifs” in someone’s notebook into real prototypes that can get tested and measured. Most new products today grow out of this trial-and-error loop; speed here means gaining an edge.
Responsible Handling and Safety
Every step in using N-Cyclohexylcyclohexanamine Nitrite demands respect for health and safety standards. Long exposure or careless handling causes skin and eye irritation, and inhaling vapors can bring real harm. Facilities set up storage and ventilation systems, train staff in safe procedures, and keep emergency equipment nearby. Proper documentation and transparent supply chains matter just as much as technical know-how. Producers who take shortcuts endanger not just workers, but also the communities around them. Following chemical safety rules isn’t a burden—it’s the only ethical way to do business.
Is N-Cyclohexylcyclohexanamine Nitrite safe to handle and use?
The Basics that Matter
Every time I’ve worked with lesser-known chemicals, I’ve seen how easy it is to miss the risks when there’s little chatter or no familiar hazard symbol. N-Cyclohexylcyclohexanamine nitrite, not as widely discussed as some big-name substances, slips under the radar. Folks often ask whether it’s safe to handle or use, and the short answer goes back to what’s true about most nitrite compounds – respect the risks, or pay the price.
Nitrites react with organic matter and break down in the presence of acids or heat. They can produce toxic gases like nitrogen oxides, especially in poorly ventilated spaces. N-Cyclohexylcyclohexanamine nitrite shares these harmful traits. You might not see stories about it in mainstream news, but chemical reports back up health and safety concerns: skin and eye irritation, respiratory issues, and the ever-present threat of accidental ingestion or inhalation.
Learning from Industry Practices
I remember one job where procedural shortcuts nearly led to a major scare. The chemical wasn’t this exact nitrite, but the lesson stays the same. Safety data sheets aren’t just paperwork – they spell out vital precautions. Proper gloves and eye protection shouldn't ever stay in storage. Reliable chemical fume hoods and tight-fitting respirators shield workers from accidental exposure. Loose habits and time crunches might tempt people to compromise, but short-term gains only boost long-term risks.
EPA and OSHA haven’t named N-Cyclohexylcyclohexanamine nitrite on any banned list, but guidance draws from close relatives. Most nitrite warnings stress wearing personal protective equipment, storing away from acids, and never mixing around heat or sunlight. Factory settings stress regular training – people don’t memorize every chemical’s quirks and hazards, but they should build a reflex: check labels, check air flow, never rush disposal.
Risks for Curious Newcomers and Hobbyists
DIY chemistry attracts all sorts of curious minds. Online forums stir up interest in exotic-sounding names. Access to raw chemicals has never been easier, but so has the chance for amateur slip-ups. Data from the CDC highlights rising calls to poison control centers, often linked to people mixing or handling seemingly harmless substances at home.
Internet recipes often skip safe disposal steps or miss the risks tied to improper storage. Sometimes, new experimenters simply don’t know what vapor is forming or how a spill can turn serious. I’ve seen close calls – not because people ignored advice, but because good advice seemed invisible until something went wrong.
Raising the Bar for Safety
Community knowledge offers the best defense. Chemical manufacturers and suppliers shoulder responsibility. They need to send clear, plain-language hazard info instead of dense legal jargon. Local governments can keep up with inspections and spot checks, nudging labs and warehouses toward safe practices, not just compliance.
Schools and universities play a huge role. Practical lab safety has to come before memorizing equations. That means real-life walkthroughs, not just posters, so tomorrow’s scientists don’t get comfortable with “close enough” standards.
Better Choices Go a Long Way
Chemicals like N-Cyclohexylcyclohexanamine nitrite demand the same respect as more infamous lab substances. Safety rules can’t protect anyone unless people actually use them. That means not just reading the labels, but making them easy for the next person to spot and follow. Honest conversations between suppliers, workers, and curious non-professionals do more good than another stack of fine print warnings nobody reads.
Respecting a chemical’s risks never killed anyone. Ignoring them still does.
What are the storage and handling recommendations for this compound?
The Basics: Keeping Chemicals Safe and Stable
Storing chemicals might sound simple, but a few common sense steps can make all the difference between a safe workspace and a costly, even dangerous mistake. I’ve seen too many labs take shortcuts—leaving bottles on benches, skipping labels, or tucking solvents in random cabinets. The result: ruined runs or, worse, hazardous spills. Chemistry, after all, doesn’t always forgive carelessness.
Every compound comes with its own quirks. Organic solvents often ask for a cool spot with steady temperatures, away from ignition sources. Acids and bases prefer their own shelves with lots of ventilation, and some peroxides will spoil quietly if left near sunlight. From firsthand experience, tossing incompatible substances together in one cabinet breeds headaches. Segregation by hazard class is a rule worth following. The few minutes spent on organized storage saves hours cleaning up after a disaster.
Temperature, Light, and Humidity
Humidity and heat can silently degrade many compounds. This isn’t just about pharmaceuticals or exotic reagents; plain bleach or hydrogen peroxide breaks down faster in warm, damp air. I’ve watched flasks cake up and lose potency after only a week on a humid shelf. Cold rooms, dedicated fridges, and dry cabinets should not be optional. Each year, the cost of replacing spoiled material reminds me that trust in the basics—low, dry, dark—pays off.
Don’t ignore manufacturer’s instructions. These usually aren’t there for liability alone. For my own work, silica gel packets and amber glass bottles became crucial. Silica grabs extra water vapor; amber bottles shield light-sensitive stock. Over the years, I’ve learned that a little preventive care with packaging matters more than chasing expiration dates.
Container Choice and Labelling
Not all containers are created equal. Strong acids eat through thin plastic, and certain organics seep out of soft polyethylene. I remember discovering a puddle under a shelf where the wrong bottle met the wrong liquid. Glass with tight caps, lined with PTFE, remains a gold standard for a reason. Reused bottles need careful washing—trace residue leads to surprises.
Labelling deserves respect. No one likes hunting for faded or missing tags during an emergency. Full chemical names, concentration, hazard warnings, and the date of receipt or opening—these details guide safe handling. Once, during my early days in the lab, a simple error in labelling sodium azide nearly resulted in mixing with copper. Anyone who’s studied chemical reactions knows what happens next; it rarely ends well. Proper labelling keeps confusion at bay.
Safe Handling Starts Long Before Use
Routine makes safety second nature. Wearing goggles, gloves, and lab coats, along with using proper fume hoods, can save you from costly health scares. Once, a friend ended up with burns after a flask leaked; he’s a veteran chemist but skipped gloves “just this once.” Mistakes happen quickly—safety gear always wins.
SOPs (standard operating procedures) should be written in plain language. Training new team members in these protocols means less guessing and fewer accidents. In my experience, clear signage—hazard diamond stickers, color-coded tape, and posted instructions—helps everyone remember best practices during a busy day.
Solutions for Safer Workspaces
Investing in storage planning and regular audits cuts risk dramatically. Periodic checks of stock for leaks, expiry, and cross-contamination prevent accidents and reduce waste. Digital inventory systems, which send alerts for low stock or aging chemicals, proved useful in my last research lab. Good habits sustain safe environments long after initial enthusiasm fades. Storage, labeling, and handling should not feel like chores; they’re the pillars supporting productive, responsible work.
Are there any known side effects or hazards associated with N-Cyclohexylcyclohexanamine Nitrite?
Looking Past the Chemistry: People and Safety
N-Cyclohexylcyclohexanamine Nitrite rarely shows up in everyday conversations, but for people working with chemicals, safety isn’t up for debate. I remember a time handling less complicated chemicals in a university lab. The rules drilled into us felt strict—until I saw someone pay the price for breaking them. Fainting, headaches, and wild fluctuations in blood pressure can happen fast with nitrites, especially in poorly ventilated rooms. The body doesn’t shrug these off. Chemical exposure always leaves a mark, especially the ones we don’t see coming.
Side Effects: What Science and Experience Show
Nitrites, no matter the name in front, mess with how blood carries oxygen. Someone exposed to N-Cyclohexylcyclohexanamine Nitrite—maybe through inhalation or skin contact—is rolling the dice with their health. Methemoglobinemia, a long medical word, basically means blood cells stop holding onto oxygen like they should. People turn blue, get dizzy or even lose consciousness. Hospital visits for this aren’t rare in places where safety slips. I’ve seen colleagues lose days at work because of headaches, nausea, and other symptoms that doctors trace right back to nitrite exposure. That isn’t worth any shortcut.
The science backs this up every year. According to the Centers for Disease Control and Prevention, nitrite compounds act fast in the bloodstream. A small dose might cause mild effects—redness, rapid heartbeat, maybe nausea. Larger exposure sends people to the ER. There’s no glamour in finding this out the hard way. In animal studies, major organ damage and even death followed high enough doses. The unpredictability adds another layer. Some folks show signs at low levels, others tolerate a bit more—nobody escapes unharmed indefinitely.
Risks Beyond the Obvious
Beyond immediate symptoms, chronic effects matter too. Week after week exposure—think poorly maintained jobsites—can mean long-term nervous system problems. Studies link organ damage, trouble concentrating, and mood swings to nitrites. Over time, the risks sneak up. The American Industrial Hygiene Association mentions skin gets irritated, eyes start stinging, and lungs cough when exposed for weeks on end. I’ve heard stories of people developing sensitivity to all sorts of chemicals later on in life after years around nitrites.
Solutions Start with Respect—for Science and People
No amount of bravado makes up for real safety. Strong ventilation, strict use of personal protective gear, and up-to-date workplace training make a difference. My own routine changed after too many close calls: double-gloving, changing clothes right after a shift, opening windows even when the boss grumbled about energy bills. I check chemical labels twice every time. Companies owe it to their teams to keep safety data sheets within reach, with clear instructions anyone can follow. It helps to reward teams who spot risks before they become accidents.
I’ve learned to treat every new chemical with caution until experts prove otherwise. Health agencies suggest regular health checks and monitoring for anyone who works around nitrites. Trust in science. Tell coworkers what you see, even if it seems minor—catching a headache early costs nothing compared to a hospital stay. Over the years I’ve come to believe: chemicals don’t forgive mistakes. They only wait to show what happens if we stop paying attention.
Where can I purchase N-Cyclohexylcyclohexanamine Nitrite and what is its price?
Understanding the Compound
N-Cyclohexylcyclohexanamine Nitrite stands out as a chemical used mostly in research settings, sometimes in specialized industrial applications. Its molecular structure makes it a relatively niche product, not something you typically find on the shelves of local supply stores or large online retailers. Most people interested in this compound work in scientific labs, universities, or companies dealing with advanced chemical synthesis.
Market Availability
Trying to buy a rare chemical comes with its own set of hurdles. Most buyers turn to established chemical suppliers. Big names in laboratory supplies such as Sigma-Aldrich, TCI, or Alfa Aesar offer a range of specialty chemicals, but not every compound ends up in their catalogs. Even for professionals with accounts, you might not see a listing for N-Cyclohexylcyclohexanamine Nitrite online. If you do, it usually appears with very little public information. Companies ask for proof of research credentials or verified business purposes. In my experience, purchasing obscure chemicals usually requires paperwork—sometimes import permits, safety data sheets, and end-use declarations.
Price Expectations
Prices for fine and specialty chemicals change all the time. Cost depends on how difficult the synthesis is, overall demand, purity, and packaging. When dealing with compounds similar to N-Cyclohexylcyclohexanamine Nitrite, lab-scale orders—anything from a few grams to a couple hundred grams—tend to cost more per gram than larger, industrial-sized shipments. For hard-to-find chemicals, the price can reach several hundred dollars per gram, but you won’t find this clearly posted in most cases. You typically need to request a formal quote. In some past situations, even getting a price means back-and-forth with a sales rep to confirm you aren’t seeking the chemical for unapproved or illegal uses.
Legal Rules and Safety Measures
Ordering and handling chemicals like N-Cyclohexylcyclohexanamine Nitrite puts responsibility squarely on the buyer. Safety rules control who can own or store such substances, especially if they have toxicity or a connection to other regulated chemicals. Countries enforce their own sets of rules. In some places, you can’t import certain chemicals without paperwork from a government agency. In the US, the DEA, EPA, and state laws layer on extra restrictions. Not every chemical on paper seems dangerous, but regulations often run broad, and getting caught skipping them carries serious penalties. In Europe and the UK, REACH and other local rules get involved. Always work with a supplier with a solid reputation. Reliable companies won’t risk their license by overselling or shipping to an unverified buyer.
Best Approach for Potential Buyers
If you have a legitimate need, start with an inquiry to a reputable chemical supplier. Bring clear documentation showing academic, industrial, or scientific use. Describe your project and provide supporting licenses. Ask for a formal quote, lead time, purity options, and shipping conditions. Some suppliers only ship to institutional addresses, so a home address almost always gets rejected. If one company does not carry the compound, a custom synthesis house might help, but expect a much higher price and long wait times. Using trusted networks or industry contacts can speed up the process, but eventually, paperwork always comes due.
The Demand for Transparency
Chemicals like N-Cyclohexylcyclohexanamine Nitrite call for strong ethical handling. As a buyer, your due diligence protects your organization and the public. Strict record-keeping, risk assessment, and knowledge of downstream effects set apart responsible buyers from reckless ones. If more organizations embraced this approach, broader risks in the global chemical trade would get smaller.
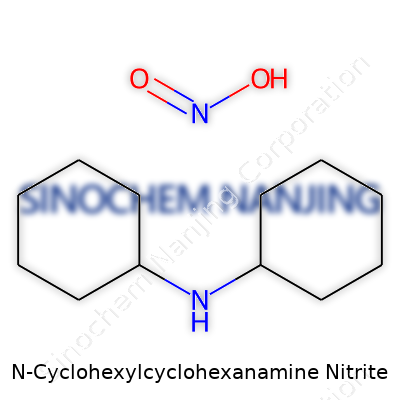
| Names | |
| Preferred IUPAC name | N-cyclohexylcyclohexanamine nitrite |
| Other names |
Nitrous acid N-cyclohexylcyclohexylamine ester N-Cyclohexylcyclohexylamine nitrite |
| Pronunciation | /ɛn-saɪ.kloʊˈhɛk.sɪl.saɪ.kloʊˈhɛk.səˌnæm.iːn ˈnaɪ.traɪt/ |
| Identifiers | |
| CAS Number | [3886-70-2] |
| Beilstein Reference | 1633883 |
| ChEBI | CHEBI:131452 |
| ChEMBL | CHEMBL2106618 |
| ChemSpider | 22408573 |
| DrugBank | DB11439 |
| ECHA InfoCard | 03ed39e6-6920-4ba9-8d18-0e0a9d324082 |
| EC Number | 218-760-9 |
| Gmelin Reference | 66403 |
| KEGG | C18994 |
| MeSH | D008410 |
| PubChem CID | 160215 |
| RTECS number | GV5950000 |
| UNII | KED7J0D84H |
| UN number | UN3339 |
| CompTox Dashboard (EPA) | DTXSID0044652 |
| Properties | |
| Chemical formula | C12H22N2O2 |
| Molar mass | 184.29 g/mol |
| Appearance | Light yellow liquid |
| Odor | Amine-like odor |
| Density | 0.97 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 4.3 |
| Vapor pressure | 0.00922 mmHg at 25°C |
| Acidity (pKa) | 10.7 |
| Magnetic susceptibility (χ) | -8.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.507 |
| Dipole moment | 3.2052 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 307.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | N02BX |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H312, H314 |
| Precautionary statements | P261, P273, P280, P302+P352, P305+P351+P338, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 97.0 °C |
| Lethal dose or concentration | LD50 (oral, rat): 156 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral LD50 640 mg/kg |
| NIOSH | SW-7592400 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.01 ppm |
| IDLH (Immediate danger) | Not established |

