N-butylimidazole: Insight into a Versatile Chemical
Historical Development
Chemists first explored the realm of imidazoles in the late nineteenth century, keen on discovering compounds with new properties for industrial and pharmaceutical uses. Among these, N-butylimidazole attracted attention for its unique structure—a five-membered ring with nitrogen atoms that set it apart from other bases. Over the decades, researchers understood that attaching a butyl group to the nitrogen not only modified the physical profile but also unlocked chemical pathways and industrial uses that simple imidazole could not provide. Laboratories and factories gradually ramped up its production, chasing new applications in synthesis, catalysis, and specialty materials, showing how incremental chemical tweaks can ripple across technology.
Product Overview
N-butylimidazole appears as a colorless to pale yellow liquid, offers a faintly amine-like odor, and packs a punch in several applications. Its structure lets it act as both a base and a ligand, which means it helps catalyze key reactions in the lab or industry. In solvent extraction, it stands out for its ability to dissolve both polar and non-polar compounds—handy when working with complicated mixtures. The pharmaceutical and coatings industries often rely on it to help stitch together more complex molecules, or as a building block on the way towards harsher synthetic targets. For most of us, the benefits of products like N-butylimidazole are felt in things like more efficient drugs, advanced coatings, and cleaner extraction technologies, even if most won't ever see a bottle of it on a shelf.
Physical & Chemical Properties
N-butylimidazole, with a molecular formula of C7H12N2, weighs in at 124.18 g/mol and typically boils just over 237°C. Its density hovers near 0.96 g/cm³ at room temperature. Not much water disrupts it—this chemical dissolves modestly in water but much better in many organic solvents, a handy trait when trying to transport substances from one chemical world to another. Its pKa leans toward basicity, enough to help in acid-scavenging within sensitive reaction mixtures. The butyl group tacked onto the imidazole ring boosts its lipophilicity, which changes how it behaves compared to its parent molecule—think solubility, volatility, and reactivity. For laboratories, knowing such specifics means safer storage and better reaction planning.
Technical Specifications & Labeling
N-butylimidazole sourced from reputable suppliers arrives labeled with clear concentration, purity grade (which usually reaches above 98%), water content, and lot number for traceability. The container specifies hazards, safety instructions, and preferred storage conditions—usually tight, dry, and cool, far from sunlight. Workers and lab techs rely on this data for proper handling: material safety data sheets back up the labeling, detailing risks, first aid measures, and reactivity. Having handled similar chemicals, I check the labeling closely, as inconsistent purity or moisture can wreck hours of careful reaction work, or worse, create safety risks.
Preparation Method
To make N-butylimidazole, chemists usually react imidazole with n-butyl halide (like n-butyl bromide or chloride) in a polar solvent. A mild base—sodium hydride or potassium carbonate—helps pull the hydrogen off imidazole, setting up the nucleophilic substitution attack. After stirring, sometimes for hours under reflux, workers extract the product, typically washing, drying, and purifying it by distillation. The preparation seems simple on paper, but controlling reaction conditions remains crucial. Unwanted side products creep in unless temperatures and stoichiometry are fine-tuned. Scale-up for industry demands even more attention, so dozens of iterations occur before a process reaches the necessary quality and yield.
Chemical Reactions & Modifications
N-butylimidazole serves as a partner or starting point for dozens of transformations. The lone pair on the imidazole nitrogen lets it interact with electrophiles, accepting protons or forming coordination complexes with transition metals. This behavior underpins its use in catalysis and organic synthesis—both in making drugs and specialty materials. Modifying the butyl chain further yields analogues with different lipophilicity or reactivity, allowing chemists to tailor function. Some labs test other alkyl groups, shifting reaction rates or properties to suit certain conditions, which means N-butylimidazole often acts as a stepping-stone toward even more specialized compounds.
Synonyms & Product Names
N-butylimidazole appears under several names depending on context. You may spot labels like 1-butylimidazole, N-butyl-1H-imidazole, or simply butylimidazole on commercial containers. Some chemical catalogs use registry numbers or abbreviations, such as NBI. Inconsistent naming can cause confusion in inventory or procurement, but cross-referencing with CAS numbers and reputable supplier lists usually clears up ambiguity. In my own work, I double-check every new bottle, especially where different regions use overlapping names for related imidazole derivatives.
Safety & Operational Standards
Handling N-butylimidazole demands respect. Eye, skin, or respiratory contact can irritate, so gloves, goggles, and lab coats are standard. Spills get treated as chemical hazards—ventilation and containment come first, with all waste gathered in labeled, dedicated containers. Emergency eyewash and spill kits stay close at hand. In production environments, automated dispensing and contained transfer systems reduce worker risk. Regular training ensures everyone knows procedures for accidental exposure, and standardized documentation supports quick action. Over time, experienced technicians recognize subtle signs—a small leak or unfamiliar smell—and intervene before little problems escalate.
Application Area
Industries turn to N-butylimidazole for its ability to smooth over tricky chemical conversions. Catalyst systems in organic synthesis gain efficiency thanks to its base properties; sometimes it helps solubilize metals or activate specific reagents. It shows up in resin production, contributing to well-cured coatings or adhesives. Extraction processes for pharmaceuticals or rare metals also make use of its solvent capacity. Research operations, particularly in medicinal chemistry, value it as a building block for drug scaffolds, with downstream modifications adding new functionality. Day-to-day, few workers outside of manufacturing or specialized labs interact with it directly, but its presence improves yields, reduces unnecessary waste, and opens new routes to advanced products.
Research & Development
Development teams study new uses for N-butylimidazole, often by running reactions with it as a base or ligand in tandem with emerging catalysts. Green chemistry initiatives push researchers to maximize atom economy and minimize hazardous waste, and this compound fits into schemes seeking milder reaction conditions or better selectivity. Academic groups test its derivatives in biochemistry, probing how structural changes affect pharmacology, toxicity, or bioavailability. This chemical also serves as a scaffold for ionic liquids, which attract attention for their role in environmentally friendlier manufacturing. Funding agencies now support efforts that harness its unique properties for efficiency or sustainability.
Toxicity Research
Toxicological studies reveal N-butylimidazole can irritate mucous membranes and skin, and inhalation at high concentrations brings risk of central nervous system effects. Animal testing—conducted according to ethical and legal requirements—shows dose-dependent toxicity above certain thresholds, which guides occupational exposure limits in the workplace. Long-term or large-scale exposure data remain sparser, so researchers continue to study its breakdown products in the environment and their persistence. Regular monitoring and conservative handling protocols help keep risks in check. As with many chemicals, upfront transparency on toxicity supports both regulatory compliance and public trust in its continued use.
Future Prospects
Interest in N-butylimidazole follows trends in green chemistry, where milder, more selective chemicals gain favor in industry. Emerging catalytic systems—especially for pharmaceutical or materials applications—depend on efficient, reusable ligands and bases. As researchers chase more sustainable solvent and extraction processes, demand for this compound likely grows. Continued R&D could yield analogues or formulations with reduced toxicity or improved performance, broadening its appeal. With stricter environmental standards and economic pressures, industries constantly revisit process chemicals, so those with established performance and flexible application, like N-butylimidazole, remain in play. If newer regulations or scientific discoveries reshape its risk/benefit profile, responsive adaptation in manufacturing and safety standards will determine its ongoing role in technology and industry.
What is N-butylimidazole used for?
What It Really Does
N-butylimidazole comes up in labs and factories for good reason. This chemical works quietly behind the scenes, showing up in processes that touch daily life, even though most people have never heard of it. You find N-butylimidazole in the manufacture of specialty chemicals, especially where tweaking molecules means a better product or smoother reaction. Its role as a base and a catalyst proves valuable in several organic syntheses. That means it helps chemical reactions happen more efficiently, and sometimes even makes them possible in the first place.
Why Chemists Rely on It
I spent my early career watching researchers try to shave seconds off reaction times and cut down on by-products. N-butylimidazole often came out during conversations about cleaner syntheses. In pharmaceuticals, for example, getting the right reaction without creating a mess of unwanted side products means better safety for workers and less environmental waste. Its structure offers stability while helping steer outcomes. This saves money and time, especially when companies look to scale up their work.
Pushing Green Chemistry Forward
The push for greener chemistry keeps picking up speed. Many industrial processes used to rely on harsher bases and solvents, but switch to compounds like N-butylimidazole when feasible. Less corrosive and often more selective, this chemical has helped replace tougher substances that wore down equipment or created bigger disposal problems. Since I’ve seen older plants wrestle with the costs of leaking pipes or hazardous waste, the move toward friendlier reagents stands out as a real step forward. More selective reactions mean less cleaning, fewer accidents, and lower bills over time.
Practical Applications in Electronics
Modern electronics ask for precise chemicals at nearly every stage. N-butylimidazole finds use in polymer research, especially in the development of conductive materials or specialized coatings. Its ability to work as both a ligand and a catalyst means researchers can shape new materials to better match demands like heat resistance, improved conductivity, or longer shelf life. It doesn’t grab headlines, but small changes in chemical formulations can push entire industries forward. The smartphones, wiring, or screens we use every day owe a debt to such behind-the-scenes refinements.
Risks and the Road Ahead
N-butylimidazole’s advantages don’t absolve users from responsibility. I still recall my first safety training: new compounds create new hazards. Handle it carelessly, and you’re looking at irritation or worse. Good training and solid ventilation keep accidents low, but oversight matters. United States and European regulators keep a watchful eye on chemicals with wide industrial applications. The trend—especially after a few high-profile spills—is to document everything, from sourcing to disposal. That raises costs for compliance, but safer workplaces and cleaner water make those investments worthwhile.
What We Can Do
The conversation about specialty chemicals like N-butylimidazole moves past their formulas. Knowledge-sharing among researchers helps catch risks before they grow. Clear labeling and clear communication cut down on mistakes, while investing in research keeps safer substitutions on the horizon. From my side of the lab bench, I’ve seen how small improvements—better gloves, clearer signage—can prevent major headaches. Industry players should keep talking with regulators and the public, making sure new advances land safely and benefit everyone involved.
What are the safety precautions when handling N-butylimidazole?
Recognizing What You’re Dealing With
N-butylimidazole isn’t a household chemical—working with it introduces real risk. This liquid often shows up in labs dealing with organic synthesis, and it carries hazards that you might not spot at first glance. Direct contact can irritate the skin or eyes. Inhalation causes headaches, dizziness, and deeper breathing problems. When I started out in chemical research, a seasoned technician showed me scars from one splash—lesson learned fast.
Personal Protective Equipment Matters
The basics sound simple, but a missed glove or forgotten pair of goggles can ruin your month. Chemical-resistant gloves go first, and nitrile ones usually stand up to N-butylimidazole. Lab coat, long pants, and closed shoes shield the rest of your body. Chemical splash goggles beat safety glasses for anything likely to spritz or splash. I always double-check my sleeves and collar, since gaps attract trouble. If lab work demands higher risk, throw on a face shield.
Workouts for Fume Hoods
Fume hoods turn into your best ally when vapor risk rises. Even careful handling creates fumes you don’t want in your lungs. Too many people crack a reagent cap on the bench “just for a second” and regret it. Decent ventilation doesn’t cost much, but it keeps your lungs clear and the headache away. At group meetings, people trade stories about how fume hood alarms saved their skin by alerting them when airflow dropped.
Spill Planning for Peace of Mind
Everyone thinks a spill kit is overkill until someone tips a beaker. If N-butylimidazole lands on your skin, the response from your team has to be fast—lots of water, quick removal of contaminated clothes, and reporting the incident so the safety officer calls the right shots. The right absorbents (not paper towels) can lock up most spills. Test your group with mock drills, just like fire drills in school. People stumble over the steps the first time. With practice, speed and calm set in.
Labeling and Storage: Good Habits Pay Off
Clear, accurate labeling cuts guesswork. I’ve seen too many shelves loaded with mystery bottles. If the label peels off, replace it on the spot. Avoid stacking containers or lining them up too close together. Here, less clutter equals less risk. Store N-butylimidazole far from heat, sparks, or sunlight, since it’s not just the hazard itself—uncontrolled temperature can build up pressure or cause leaks. Separate it from acids, oxidizers, and foods.
Disposal Done Right
Tossing leftover N-butylimidazole down the drain isn’t just wrong—it’s usually illegal. Collect all waste in properly marked, sealable containers. Disposal companies, not garbage bins, finish the job. Ask an environmental safety officer if there’s doubt about waste rules. Skip the shortcuts to avoid heavy fines or worse, toxic incidents for building maintenance crews.
Training Protects Everyone
Most chemical lab accidents hit new or distracted workers hardest. Refresher training, real incident reviews, and walk-throughs with new hires turn safety rules from words on a poster into lived habits. Even old hands benefit from stories and lessons. Sharing facts—like N-butylimidazole’s toxicity and flammability ratings—proves the risks aren’t just theoretical. No one wants to end up as the warning example for next year’s class.
What is the chemical structure of N-butylimidazole?
A Close Look at N-Butylimidazole’s Blueprint
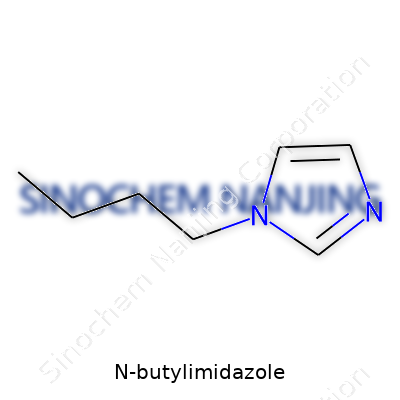
Walk through any well-stocked chemistry lab, and you’ll find shelves with glass bottles, odd labels, and a quiet sense of untapped energy. Among these is N-butylimidazole. At face value, the name hints at complexity, but the structure tells a straightforward story. N-butylimidazole forms when a butyl group, a four-carbon chain (CH3–CH2–CH2–CH2–), attaches at the nitrogen atom of an imidazole ring. That imidazole ring, a five-membered system featuring two nitrogen atoms at positions 1 and 3, keeps things versatile by hosting many substitutions.
The Structure on Paper, in Labs, and Out in the World
Drawing it out, N-butylimidazole features a core five-membered ring. Nitrogen at position 1 connects to a straight four-carbon butyl tail. The ring carries three carbons and another nitrogen at the third position, with ring hydrogens bringing up the count. The skeletal formula looks like this: C7H12N2. But on paper, and especially in the way it behaves, each atom’s place matters.
I spent a summer working with heterocyclic compounds. In those weeks, there was no substitute for getting hands-on with molecular models. Snapping the butyl group onto the right nitrogen changes not just the molecule’s shape, but its feel—how it dissolves, what it smells like, and what reactions it can start. Imidazole rings, small and unassuming, stay at the heart of pharmaceutical and industrial chemistry. Swap in an alkyl group, and suddenly a molecule’s solubility, reactivity, and target activity can switch. This isn’t just theoretical. The difference between an imidazole and an N-alkylimidazole (like N-butylimidazole) influences everything from corrosion inhibitors to catalysts in organic synthesis.
Why Structure Affects Performance—and Our Choices
Each atom that gets added or moved shifts how researchers and industries use these molecules. Adding the four-carbon butyl chain to the imidazole ring changes its polarity, which is key for anyone trying to dissolve this compound in solvents that plain imidazole might not tolerate. Imidazoles serve as tiny engines for many chemical reactions, with their unsubstituted forms often used as building blocks for pharmaceuticals. As chemistry has pushed into greener territory, N-alkylimidazoles like N-butylimidazole have emerged as neat problem-solvers, sometimes showing low toxicity and decent biodegradability compared to bulkier, more aromatic cousins.
Chemists value compounds with predictable behavior. With N-butylimidazole, that comes from its structure. It hardly ever surprises in how it reacts with acids, bases, or even in catalysis. That makes it a steady choice where consistency is prized, such as in making ionic liquids or as ligands in modern metal-organic frameworks.
Addressing Challenges and Planning Ahead
Sourcing pure N-butylimidazole sometimes brings up safety and environmental concerns. Residual solvents or byproducts from synthesis can make a difference in lab outcomes and downstream applications. So, labs often look to better purification techniques—simple distillation at small scales, or more advanced chromatography in larger operations. Once the chemistry settles into reliable procedures, the compound’s benefits multiply across agriculture, energy storage, and medicine.
The demand for safe, greener, and affordable functional molecules in synthesis keeps growing. Research keeps pushing for options where N-alkylimidazoles play central roles. Students in advanced lab classes pick N-butylimidazole to test how simple substitutions can lead to real-world improvements. Science teachers, too, sometimes use its straightforward structure to teach why every group and atom counts.
How should N-butylimidazole be stored?
Why Storage Matters
Anyone who’s worked with specialty chemicals like N-butylimidazole knows the risks aren’t just theory—they’re real. I remember watching a lab tech scramble to contain a spill simply because a lid wasn’t tight enough. This compound isn’t outrageously dangerous compared to strong acids or explosive solvents, but it sure reacts poorly when left in the wrong spot or exposed to humid air. N-butylimidazole’s specific structure allows it to absorb moisture from the environment, which ends up causing decomposition or even unwanted byproducts in sensitive processes.
What Science Says about Safe Storage
Peer-reviewed journals and chemical safety data all say one thing: store this compound in a cool, dry place—always. Research shows that N-butylimidazole reacts with strong oxidizers and can degrade over time if it sits in damp conditions or hits high temperature swings. Chemical manufacturers usually recommend keeping it in tightly sealed containers, far away from sunlight and incompatible reagents.
I once saw someone leave an open flask out over the weekend; the result was yellowish gunk and an annoyed supervisor. That’s just more proof that exposure leads to degraded purity fast. Analytical studies confirm this: even a few days exposed to air impacts the effectiveness for next use. Quality control teams from leading suppliers follow cGMP guidelines, keeping samples at stable temperatures, often below 25°C, inside shatterproof amber bottles to fight off both UV and excess heat.
What Good Storage Looks Like
The best spot for N-butylimidazole is a locked, chemical-grade storage cabinet. Think fire suppression features, ventilation, and segregated shelves. Don’t let acids or oxidizers live nearby—accidental mixing gets messy. In a shared lab, everyone respects color-coded bins and clear labels. Keeping only the amount you need on hand cuts risk, too. Nobody wants to babysit a half-empty drum over a holiday break.
Moisture control plays a huge role. Silica gel desiccants help dry out any humidity that sneaks into a bottle. I keep small pouches in every cabinet, swapping them out as soon as they look pink or soggy. If your facility runs warm in summer, aim for a temperature-stable room or cold storage; even slight heat can speed up unwanted reactions or vapor loss.
Damage Control and Spill Safety
No matter how careful you are, accidents happen. Good storage means easy access to spill kits and eye wash stations. All staff handling N-butylimidazole train and drill for leaks. Having clear standard operating procedures keeps panic low, and incident reports help catch storage slip-ups quickly. These aren’t burdens—they’re lessons learned from labs that skimped on preparation.
Building a Culture of Safe Handling
Safety advice sticks when experienced chemists share real stories, not just lectures. I’ve found that new lab techs pay closer attention when they hear about actual “close calls” rather than abstract safety slogans. Employers hold the biggest responsibility. Supplying proper PPE, regular hazard awareness check-ins, and easy-to-follow signage in the storage area keeps everyone engaged and aware.
Smart storage of N-butylimidazole doesn’t just protect the people in the room—it keeps down contamination, waste, and budget headaches. It’s about treating the chemical as valuable: not just for the experiment, but for everyone’s peace of mind. Responsible storage builds trust in the process and respect for the science behind it.
What is the purity and available packaging of N-butylimidazole?
Understanding Purity in Chemical Procurement
Purity isn’t just a checkbox on a lab report; it decides almost everything about how a chemical will perform in a real-world application. With N-butylimidazole, levels commonly reach 98% or higher, and for many clients, that’s considered the working gold standard. You find options labeled at 98%, 99%, and sometimes lower for less critical processes. Even a one-percent difference can matter, especially in pharmaceutical research, specialty solvents, or custom catalysts. If contaminants sneak in, you’re looking at skewed data, unpredictable reactions, and wasted raw materials. The stakes only rise when the end products interact with living systems or tight process controls.
Purity Impacts More Than Just Research Outcomes
Experience teaches you not to take purity specs at face value. Reliable suppliers back up their claims with documentation—Certificate of Analysis (COA), chromatograms, or independent third-party results. Each document tells its own story about batch consistency, residual solvents, moisture, and byproduct traces. Cutting corners to save on price can backfire; once, an improperly vetted drum cost a team weeks of downtime chasing an elusive contaminant that ruined critical tests. No one enjoys that headache.
The broader chemical supply chain pays close attention. Regulations keep tightening for anything even remotely linked to pharmaceuticals or food processing. Even industrial users—electronics, polymers, coatings—favor higher-purity batches for predictable performance. Globally, market data shows the trend: manufacturers are investing in advanced purification methods and tracking sharper purity grades year after year.
Packaging Shapes Handling and Storage
Packaging is more than a box or a drum; it decides shelf life, safety, and lab workflow. For N-butylimidazole, standard formats range from 500 milliliter bottles to bulk drums (20 kg, 25 kg, sometimes up to 200 liters for high-volume users). Amber glass bottles get the nod for research-grade orders: they block UV light, which can help slow degradation over long storage periods. Tightly sealed polyethylene or fluorinated containers show up for bulk delivery—nobody wants a slow leak or cross-contamination from incompatible plastics.
I’ve seen labs switch suppliers because poorly designed caps led to lost product, or returned drums because of unclear labeling. Small things make a big difference: child-proof lids, tamper-evident seals, clear expiry dates, and hazard symbols. It’s about risk management, not just convenience. Every transfer or decanting step opens a door to spills, airborne vapors, or operator exposure. A good packaging choice can cut down those risks and keep regulatory inspectors happy.
Working Toward Better Solutions
The market has an appetite for smarter, safer, and greener options. Recyclable drums and reusable totes are catching on as the next step in sustainable chemical handling. Some distributors have started offering just-in-time refill services to slash waste and keep shelf stocks fresh. Producers open to feedback often hear from customers tired of over-sized packaging or wasteful single-use containers. Improvements come from paying attention to these real pain points rather than just chasing margins.
Transparency goes a long way. Suppliers who openly share production certificates, lot histories, and guarantee tamper-proof packaging usually earn customer trust quickly. That’s not just marketing—it’s a lived reality for quality-conscious labs and production sites. Better practices save money, cut pain, and keep everybody safer, from dock workers to end users.
| Names | |
| Preferred IUPAC name | 1-butyl-1H-imidazole |
| Other names |
1-Butylimidazole N-Butyl-1H-imidazole 1H-Imidazole, 1-butyl- |
| Pronunciation | /ɛn-ˌbjuːtɪlˌɪmɪˈdɑːzəʊl/ |
| Identifiers | |
| CAS Number | 14341-33-2 |
| Beilstein Reference | 120924 |
| ChEBI | CHEBI:83547 |
| ChEMBL | CHEMBL186492 |
| ChemSpider | 78616 |
| DrugBank | DB08798 |
| ECHA InfoCard | 13b5bc78-1ee0-480f-b03a-37011b391e13 |
| EC Number | 911-544-4 |
| Gmelin Reference | 70773 |
| KEGG | C19316 |
| MeSH | D018331 |
| PubChem CID | 86641 |
| RTECS number | UJ4375000 |
| UNII | 2672B1QNHQ |
| UN number | UN2810 |
| Properties | |
| Chemical formula | C7H12N2 |
| Molar mass | 138.21 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 0.968 g/mL at 25 °C |
| Solubility in water | soluble |
| log P | 0.89 |
| Vapor pressure | 0.0598 mmHg (25 °C) |
| Acidity (pKa) | 7.0 |
| Basicity (pKb) | 7.07 |
| Magnetic susceptibility (χ) | -63.5×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.489 |
| Viscosity | 1.155 mPa·s (20 °C) |
| Dipole moment | 2.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -18.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3458.6 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H314 |
| Precautionary statements | P280, P261, P305+P351+P338, P337+P313 |
| Flash point | 76 °C |
| Autoignition temperature | 410°C |
| Explosive limits | Explosive limits: 1.4–9.5% (V) |
| Lethal dose or concentration | LD50 (oral, rat): 657 mg/kg |
| LD50 (median dose) | LD50 (median dose) of N-butylimidazole: 398 mg/kg (rat, oral) |
| NIOSH | NA1240000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 100 mg/L |
| Related compounds | |
| Related compounds |
N-methylimidazole N-ethylimidazole N-propylimidazole N-hexylimidazole 2-methylimidazole Imidazole |

