N-Butylbenzene: Shaping Industry with Chemistry and Caution
Historical Development
N-Butylbenzene entered the scene during the early expansion of the petrochemical industry in the twentieth century. Chemists searching for unique hydrocarbon structures quickly set their sights on alkylbenzenes, molecules built for versatility. The discovery of more efficient alkylation techniques gave the sector a boost, as researchers learned that benzene and butyl halides could react in the presence of Lewis acids like AlCl3 to deliver a straightforward path to n-butylbenzene. As petroleum refining became more advanced and global industry pushed for newer functional chemicals, the production of this compound became a benchmark in many organic chemistry labs. Discussions in historical journals, including those from the American Chemical Society during the postwar chemical boom, point out how this compound threaded itself into the synthesis stories of pharmaceuticals, surfactants, and innovative plasticizers.
Product Overview
The hydrocarbon n-butylbenzene displays a simple structure—a benzene ring with a linear butyl group attached. This compound’s core value lies in its purity and reactivity, qualities sought by both researchers and industry managers. In bottles or drums, it shows up as a colorless, oily liquid, known for its dispensability and efficient solvency. The chemical comes labeled under a variety of product codes, catering to analytical labs, pilot plants, and manufacturers working with specialty solvents. Companies trading in laboratory reagents or custom organics offer n-butylbenzene in several grades, with purity ranging from technical to analytical standards. Most users in scientific and industrial settings expect clear labeling, providing molecular formula (C10H14), CAS number, hazard signal words, and batch-specific data.
Physical & Chemical Properties
N-Butylbenzene comes with a set of properties that set it apart in organic laboratories. It brings a boiling point around 183°C and a melting point near -94°C, letting it stay liquid far past normal European and American winter lows. Its flash point stands close to 53°C, which means routine handling calls for fire safety mindfulness. This oily solvent doesn’t mix with water, but it blends well with most organic liquids, making it a popular choice in extraction chemistry. In terms of molecular weight, it clocks in at 134.22 g/mol. Its density, a touch under 0.87 g/cm3, keeps it lighter than water. The aromatic core and hydrocarbon tail drive its performance as both a solvent and a synthetic precursor.
Technical Specifications & Labeling
Professional packaging of n-butylbenzene always focuses on safety and clarity. Each container comes marked with hazard pictograms that speak volumes to experienced hands—flammability, environmental risk, and potential health concerns stand out. Documents accompanying shipments include expiration dates and recommended storage temperature, emphasizing the need to keep the chemical in a cool, ventilated location, away from ignition sources. Analytical-grade versions list impurities measured in parts per million, along with batch traceability for audits and regulatory checks. Large scale procurement often involves specifications for peroxide content, halide residue, and water content, in alignment with downstream process compatibility. Some buyers request lot samples for independent gas chromatography testing before green-lighting a full purchase.
Preparation Method
The classic route to n-butylbenzene begins with straightforward Friedel–Crafts alkylation. Chemists introduce benzene to n-butyl chloride or bromide, sometimes using n-butyl alcohol and an acid catalyst as a substitute route. The aluminum chloride catalyst facilitates electrophilic attack, favoring the formation of the desired linear isomer instead of its branched cousins. Reaction control demands careful temperature monitoring—side reactions cost money and time due to unwanted polyalkylation or isomerization. Once the reaction completes, standard workup includes water quenching, neutralization of residual acid, multiple washings, and distillation under reduced pressure to capture a high-purity product. Advanced process reactors in continuous production lines allow fine-tuning of residence time and recovery of solvents for circular economic benefit.
Chemical Reactions & Modifications
N-Butylbenzene takes part in a range of transformations, giving it a strategic edge in synthetic organic chemistry. The aromatic ring welcomes substitutions—nitration and sulfonation serve as classic examples, yielding intermediates for dyes, pharmaceutical APIs, and specialty resins. Oxidative conditions attack the butyl group, sometimes leading to benzoic acids or corresponding alcohol/aldehyde derivatives, useful for further polymerization or as building blocks for surfactants. The compound’s hydrocarbon tail, being unbranched, resists severe rearrangement, limiting the formation of byproducts and easing downstream purification. In combinatorial synthesis projects, n-butylbenzene serves as both a substrate and a model compound, letting researchers test new catalysts and reaction conditions for sustainable chemistry initiatives.
Synonyms & Product Names
The chemical literature and supply chain directories list n-butylbenzene under several names, adding another layer to tracking and regulation. You’ll find it as 1-phenylbutane, butylbenzene, and sometimes phenylbutane depending on the convention. International suppliers may carry codes referring to it as NBZ or use catalog numbers for streamlined ordering. Customs paperwork, regulatory compliance filings, and safety data sheets all demand precise naming to avoid mistakes and delays. These varying product names often reflect legacy branding from earlier importers, or subtle shifts in how regional standards list organics. Knowing these synonyms prevents confusion during compliance review and ensures globally consistent shipments.
Safety & Operational Standards
Hazards linked with n-butylbenzene steer users toward strict best practices. In the lab, it’s not enough to rely on fume hoods—everyone sticks to flame-proof storage and routine leak checks because the flash point isn’t forgiving, especially in confined spaces. NIOSH and OSHA guidelines underline the need for eye and skin protection, since the liquid can defat tissue or provoke irritation after repeated exposure. In manufacturing spaces, local exhaust ventilation and proper labeling cut the odds of fire, and routine safety drills include cleanup of hydrocarbon spills as a core skill. Chemical handlers look for label updates as regulations shift under GHS and REACH frameworks, double-checking MSDS sheets for permissible exposure limits before green-lighting new contracts. Over the years, hazard awareness has grown thanks to community forums, industry training programs, and stepped-up regulatory inspections. Direct experience with volatile organics has pushed most facilities to update their fire suppression systems and quick-evacuation protocols.
Application Area
The reach of n-butylbenzene runs deep in the petrochemical world. Industrial plants often rely on it as a solvent for resins, dyes, and specialty coatings, pursuing consistent quality in everything from automotive paints to synthetic rubber formulations. It acts as an intermediate in the production of more complex surfactants and lubricating oils, where engineers optimize formulations for performance at both high and low temperatures. Laboratory teams use it as an internal standard for calibrating analytical instruments, banks of HPLC and GC detectors humming with reference samples day after day. Its low polarity and chemical stability give it an edge in nonaqueous extraction systems, making it a frequent choice during sample prep for soil, air, and water testing. R&D chemists continue to experiment, scouting new catalyst systems and greener process routes that build off the robust core of this classic alkylbenzene.
Research & Development
Research circles continue to test n-butylbenzene for new functions. Pharmaceutical companies investigate its value as a starting block in the search for active molecules that treat infectious disease or inflammatory conditions. Polymer chemists tune its incorporation into polystyrene blends and trial modifications that may enhance durability or add flame retardancy. Academic projects, funded in part by government innovation grants, explore the environmental impact of n-butylbenzene released from fuel and plastic manufacturing sites—data feeds into updated exposure models and risk assessments. Bench scientists look for catalytic systems that can crack the benzene ring more selectively, hoping to minimize waste or improve the pathway to higher-value specialty aromatics. In energy labs, n-butylbenzene is trialed in model fuel systems thanks to its resemblance to some actual gasoline tail molecules, contributing valuable combustion property data. The need to balance industrial value with health and environmental safety remains front and center for every innovation.
Toxicity Research
Interpreting the toxicological profile of n-butylbenzene calls for sober reflection. Old studies from the 1970s laid the foundations, flagging skin and respiratory irritation risks at higher concentrations and pointing toward possible nervous system effects in poorly ventilated shops. Environmental testing shows this hydrocarbon does not rapidly break down in soil and water, raising long-term exposure concerns in spill-prone regions. Toxicology labs tasked with running in-vitro and in-vivo assays note that metabolic byproducts can accumulate in aquatic organisms; regulatory attention has grown as these findings enter public health debates. Overexposure to vapors, accepted decades ago as part of the daily grind, now brings increased scrutiny under OSHA and EU REACH rules, with calls for enhanced PPE and monitoring. Long-term inhalation and oral exposure studies continue, aiming to calibrate reference values that can protect workers and residents living downwind from large industrial centers. Environmental activism has driven funding into more exhaustive ecosystem studies, as communities demand tighter reporting and more effective remedial action for chemical leaks.
Future Prospects
Looking forward, the pathway for n-butylbenzene feels closely linked with broader movements toward cleaner chemistry and responsible industry. Plant managers know that synthesis steps anchored in traditional aromatics will continue to matter, but they’re under pressure to cut emissions and shrink the lifecycle footprint. The shift toward closed-loop reactors, solvent recycling, and continuous monitoring meets both economic and environmental expectations. Research teams are constantly pressured to design new catalysts that unlock greater selectivity or rely on renewable feedstocks, so the entire supply chain can demonstrate progress toward sustainability targets. Updated toxicity data, combined with more accurate environmental models, equips policymakers to set tighter limits on workplace and community exposure. Investments in greener chemical processes signal a future where components like n-butylbenzene remain integral, but industry meets sharper standards of care for people and planet alike.
What is N-Butylbenzene used for?
What Is N-Butylbenzene?
N-Butylbenzene may sound like a mouthful, but if you’ve ever worked in a lab, browsed through an organic chemistry set, or followed the flow of petrochemical products into the world around us, you’ve probably crossed paths with it. The stuff appears clear and colorless—totally unassuming—but has a distinct, oily odor. It’s made by sticking a butyl group onto a benzene ring, which puts it in the category of aromatic hydrocarbons.
The Routes It Takes in Industry
N-Butylbenzene plays a big part in the world of chemicals used to make other products. For people in the chemical manufacturing field, it serves as an important intermediate. That means it’s not usually the end product; it gets mixed and altered so other chemicals can be created. You’ll see it show up as a starting point for synthesizing plasticizers, which help make plastic bend and twist without snapping. Plastics show up everywhere, from garden hoses to electrical wiring, and plasticizers often depend on sturdy intermediates like N-Butylbenzene to keep finished goods safe and flexible.
This compound also works as a solvent. In simple terms, solvents help dissolve things or create mixtures that are more useful than the original parts. Paints, coatings, dyes, resins, and even some laboratory processes rely on solvents. Because N-Butylbenzene avoids reacting with most other materials under typical conditions, it’s trusted to mix things that otherwise wouldn’t come together so easily. My time mixing ink formulas in an old print shop would have stalled without proper solvents, and N-Butylbenzene helped bind color with oil for a smooth result.
Fuel Research and Testing
Specialists in oil refining and energy development rely on N-Butylbenzene to understand complex petroleum mixtures. Petrochemical engineers sometimes use it as a calibration standard—think of this like keeping a yardstick handy whenever you run a test. In fuel research, N-Butylbenzene acts like a stand-in for other compounds, letting researchers predict how real fuels might behave without burning rare or dangerous chemicals. That saves time, resources, and keeps things safer in the testing lab.
Concerns in Handling and the Push for Safety
Handling N-Butylbenzene requires caution. Spills and accidents can lead to health problems, especially if fumes drift around unventilated rooms. Reports of headaches and dizziness among workers highlight the real-world risks. The Environmental Protection Agency (EPA) and the Occupational Safety and Health Administration (OSHA) both keep a close eye on how workplaces store and transport this chemical, setting exposure limits and encouraging the use of gloves and masks on site.
From experience, most safety lapses happen not because rules are missing, but because people get comfortable and skip steps. Industry could do better by making safety routines more practical—checklists, air quality monitoring, and regular training matter more than one-and-done briefings. Investing in spill-proof containers and extraction hoods for fumes improves both worker health and quality control.
Alternatives and the Road Ahead
The push to replace older, riskier chemicals keeps gaining steam. Green chemistry programs look for solvents and intermediates that won’t hang around in the environment or build up in animal tissue. Some labs now test bio-based solvents, which break down faster and avoid the health impacts of traditional compounds. Shifting toward greener options depends on steady investment: both in research to prove new chemicals work as well, and in the infrastructure to use them safely at scale. For now, N-Butylbenzene holds its place as an important chemical in manufacturing, but the search for safer substitutes will keep growing.
What are the safety precautions when handling N-Butylbenzene?
Clear Risks Call for Real Action
N-Butylbenzene can smell sweet, but don’t let that fool anyone who works with it. This organic solvent shows up in labs and industrial settings where chemists and techs use it to make other compounds or test out new reactions. I’ve seen people get casual around it just because it doesn’t bite like acids or fume like ammonium, but that’s a shortcut to trouble. Long days around these solvents can put workers’ health on the line. Eye and skin contact make a fast path for irritation—and breathing too much vapor in a space without fresh air brings on headaches, dizziness, or worse.
Don’t Skip Good Ventilation
It’s tempting to cut corners when pressure’s on, especially during a heavy production run or a late-night experiment. Yet even a quick job with N-Butylbenzene in a poorly aired room can do damage. I remember my early years on the bench, learning fast about headaches from vapors that hung around longer than I liked. There’s no replacement for working under a real fume hood or at minimum, cracked windows and moving air. Labs and workshops need these setups, not just as a precaution, but as the first line of defense.
Personal Protective Equipment Isn’t Optional
You can’t trust bare hands with solvents like this. Nitrile gloves hold up better than latex if there’s a spill or a splash. I always check the glove box for splits or pinholes before starting in. Protective glasses or goggles go on even if just measuring out a beaker, because nobody expects a splash until it happens. If splashes get on clothes, lab coats resist more than cotton does. After handling, there’s no shortcut—wash hands thoroughly. These small moves matter every single time.
Spill Response—Speed and Know-How
Leaks and drips seem minor right up until someone tracks solvent onto a desk or, worse, the floor. Absorbent pads or sand work fast, but only if they’re ready and waiting, not buried under junk on a cluttered shelf. While mopping up, there’s no racing—move slow, avoid stirring up fumes, and bag waste into the right container. N-Butylbenzene-contaminated trash shouldn’t join the regular garbage. Hazardous waste bins stop bigger headaches for everyone around.
Training Makes the Difference
From my years in the field, nothing beats solid training. New techs can freeze up or guess wrong under pressure, so detailed walk-throughs matter—both for fresh hires and seasoned folks. Companies can set aside a day just for review, no shortcuts, no brush-offs. People remember hands-on practice with equipment and mock spills way better than reading a manual. Accessible Material Safety Data Sheets (MSDS) bridge the gap during emergencies or confusion, and every staff area should keep current copies.
Thinking Beyond the Job
Handling N-Butylbenzene safely reaches beyond personal health. Neighbors—both in the next lab and in the surrounding community—count on responsible management. Regular inspections, honest reporting of near-misses, and updating safety practices cut down on risks piling up. People bring home the lessons learned at work, passing habits on to family. That kind of ripple effect—driven by good training and respect for the chemicals in use—builds long-term safety one shift at a time.
Solutions Rely on Consistent Habits
Routine safety checks, proper labeling, and a culture where anyone can speak up about unsafe gear lift everyone up. When teams make a point of sharing near-misses or fixes, no one needs to hide mistakes. That openness makes it much easier to work safely in the long run. Good habits, patient training, and plain respect for risk make N-Butylbenzene manageable. There’s no magic fix, just careful steps repeated until they’re second nature.
What is the chemical formula and structure of N-Butylbenzene?
The Backbone of N-Butylbenzene
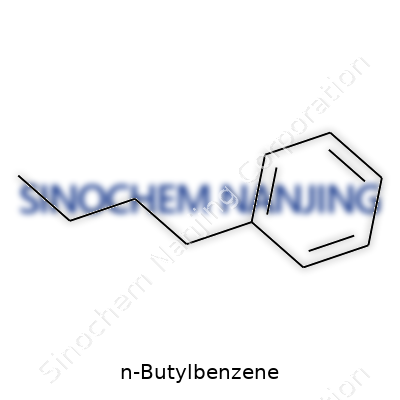
N-Butylbenzene carries the chemical formula C10H14. Each molecule holds a benzene ring fused to a straight-chain butyl group. If you place a benzene ring next to a simple four-carbon chain, you’ve got the basic idea. Drawing the skeletal formula, you would see the hexagonal ring marked for benzene, with a four-carbon tail attached to it. The IUPAC system calls it 1-phenylbutane. A chemist could sketch it in seconds, but it pays to know what these lines and letters represent in real materials.
Real-World Importance
Lab work doesn’t always feel practical until it moves from a textbook to an actual production line. N-Butylbenzene shows up often around petroleum distillation and solvent manufacture. In my time working in a university chemistry lab, someone always seemed to have a bottle on a shelf. Behind that label sat a liquid, transparent and oily, just below water in density yet noticeably more aromatic. Too many chemicals go unnoticed until their health risks emerge. N-Butylbenzene deserves close storage and handling—its fumes rise easily, and they can irritate skin or upper airways after exposure.
Chemical Behavior in Everyday Contexts
Take that four-carbon tail—it doesn’t just extend the structure; it ramps the molecule’s nonpolarity. Putting it next to plain benzene, N-Butylbenzene resists water and dissolves in organic solvents like hexane or ether. This quality matters in product formulation. People have used it to help separate mixtures or introduce specific properties to resins and adhesives. You won’t find N-Butylbenzene at the top of pop culture news, but more industrial labs use it than many realize.
Potential Hazards and Health Risks
Handling organic solvents always brings up safety. I recall an instance during an undergraduate organic chemistry lab: a spilled vial of N-Butylbenzene ended with a strong odor, some coughing, and a trip outside for fresh air. It doesn’t take long for vapor to become noticeable—volatile organics such as N-Butylbenzene can produce dizziness, headaches, and sometimes allergic skin reactions with repeated exposure. Proper gloves, goggles, and fume hoods form the most effective safeguards.
Environmental Considerations
Spills of N-Butylbenzene can leach into groundwater and linger due to its slow breakdown. Unlike ethanol or vinegar, which degrade in days, this chemical hangs around far longer. Waste disposal experts and those working near waterways keep a close eye on storage tanks, avoiding accidental releases. There’s no magic fix, but collecting wastes carefully and switching to less persistent alternatives for routine cleaning or extraction helps limit long-term impact.
Looking Ahead: Solutions in Chemical Safety
Small changes in handling mean a world of difference. Swapping out bulk storage for tightly sealed, single-use containers helped my lab cut accidental releases by half. Updating training for newcomers also worked—weekly refreshers kept everyone on alert, especially during late-night experiments. Where possible, researchers and manufacturers switch to greener solvents, but plenty of work remains.
Structure at a Glance
N-Butylbenzene doesn’t exist in isolation. It plays its part in dozens of reactions and product lines, driven by a straightforward formula—C10H14—with a ring and a tail. Understanding its structure, uses, and hazards stands as an essential step for anyone working with organics or overseeing environmental safety in a modern lab or plant.
How should N-Butylbenzene be stored?
Why Proper Storage Matters
If you've spent any time around labs or industrial sites, you know how much headaches a single slipup with chemicals brings. N-Butylbenzene seems pretty tame in the grand scheme of organic solvents—it isn’t unstable, and doesn’t explode just by looking at it. Still, some risks lurk quietly in the background. Flammable liquids always demand respect, no matter how common they feel. A careless bottle left under a sunny window or forgotten in a hot storeroom almost dares nature to remind us why the safety data sheets exist in the first place.
The Real Hazards, No Sugarcoating
I once visited a warehouse that kept drums of solvents, and overheard an old timer say, “Anything that burns on its own belongs behind a locked door.” He wasn’t kidding. N-Butylbenzene catches fire easily. Vapors from a leaky cap can turn an electrical spark from routine maintenance into a nightmare. The National Fire Protection Association (NFPA) classifies materials like this as flammable, so there’s nothing theoretical about the risk.
Beyond fire, keep in mind that airborne vapors irritate eyes and lungs. Prolonged storage with no ventilation turns a storage room into a health hazard, even if nobody lights a match. People don’t always talk enough about cumulative low-level exposure, but the toll shows up in headaches, dizziness, and worse. Cutting corners with storage means gambling with employees’ health and careers.
What Works: Storage Solutions Backed by Experience
For something with N-Butylbenzene’s profile, give it the same treatment reserved for trusted solvents. Choose metal safety cans or high-density polyethylene containers with a secure seal. Store inside a fire-rated chemical cabinet—room temperature, away from heat sources, out of direct sunlight. Every facility that values continuity and safety in their operations keeps chemical cabinets labeled and locked, usually with spill trays just in case luck runs out. Don’t let containers sit near oxidizers or acids; these pairings spell trouble if anything leaks or breaks.
Every label should be facing out, clear and legible—no peeling stickers or missing names. I’ve seen too many labs with mystery bottles, leading to last-minute panic during an emergency. A sharpie or printed label now saves confusion and stress later. Place containers on shelves that won’t collapse if bumped, far from exits or walkways. The point here isn’t about fussiness. It’s about having everyone walk out in one piece if something goes sideways.
Ventilation and Checks—Not Optional
Fresh air matters. A storage area with working exhaust fans keeps vapor levels low and the workplace compliant with OSHA’s occupational exposure limits. Don’t stash N-Butylbenzene anywhere that air gets stale or stuffy. Regular walk-throughs help you spot bad habits, leaks, or containers that look swollen or damaged. Nobody likes taking inventory, but missing these red flags costs more in the long run. Keep spill response kits nearby, and train everyone, not just supervisors, to use them fast and well. If something does go wrong, seconds matter. Quick, confident people keep accidents small.
Personal Responsibility—Everyone Plays a Role
The burden for safety doesn’t fall on managers alone. I’ve seen chemical safety programs succeed when everyone pays attention, not just the people with fancy titles. If you notice a container out of place or a cabinet that isn’t locked, speak up. Share what you know—knowledge spreads quickly in a tight crew. Safe storage only works if every person understands why these steps matter and stands ready to act before minor issues grow teeth.
Is N-Butylbenzene hazardous to health or the environment?
What is N-Butylbenzene?
N-Butylbenzene hides in labs, chemical plants, and pops up as an intermediate during the synthesis of plastics, resins, and a batch of specialized chemicals. Old-school chemistry tutorials mention it, but outside the lab, barely anyone recognizes the name. This stuff looks innocent, a clear oily liquid, but stories from colleagues who’ve handled it say otherwise.
Personal Glimpses From the Lab
Old memories from grad school come up. Handling chemicals like N-Butylbenzene never really felt risky—a bit like using old paint thinners. Gloves go on, goggles per protocol, but sometimes the room fills with a sweet aroma, and then you remember, “Ah, yeah, that’s the solvent.” Once in a while, a headache lingers, and you wonder what’s floating through the air vents. Bureau of Labor stats show that regular solvent handlers report nausea, migraines, and sore throats at higher rates than most office folks.
Health Risks — More Than Just Irritation
Diving into the data, N-Butylbenzene causes skin and eye irritation. It doesn’t take a spill on the clothes to pick up that something’s off—just a bit splashing on the skin feels like a burn after a few minutes. Inhalation triggers sore throats and headaches, and a day in the lab sometimes ends with late-night coughing. Safety Data Sheets list potential nervous system effects, and the chemical structure shares too much resemblance to other aromatic solvents linked years later to worse outcomes–liver stress, kidney damage, and trouble concentrating.
Environmental Concerns That Won’t Go Away
One of the biggest headaches comes from disposal. N-Butylbenzene doesn’t break down fast. Dumping leftovers down the drain never crossed anyone’s mind where I worked, but not every facility keeps up. This chemical seeps through soil, disrupting aquatic life; scientific reviews mark it toxic to fish even at low concentrations. A 2022 environmental study tracked persistent solvent pollution near sites that handled aromatic hydrocarbons and found elevated benzene derivatives in groundwater for years, raising questions about drinking water safety and lingering risks for rural communities.
Why It Matters—And How Change Happens
Ignoring bulk chemicals until the community or regulators flag a problem has bitten companies before—benzene got the axe only after decades of denial. Folks often shrug off “minor irritants” like N-Butylbenzene, thinking it’s handled with gloves, masks, or fume hoods, but rules do slip. In poorly ventilated workplaces, vapor can build up, and symptoms pile on. EPA records show fewer than half of violations for improper solvent disposal lead to quick fixes or fines.
Routine air monitoring helps, but labs running on a tight budget return to bare minimum safety steps. Proper storage, periodic health monitoring for handlers, and labeling go much farther than paperwork. Companies moving to greener solvents—where breakdown is fast and less toxic residue builds up—show fewer worker complaints. Some have swapped out N-Butylbenzene for emerging bio-based solvents, which cut accident records in half over the past decade, according to a 2021 industrial safety audit.
Facing Up to the Risks
N-Butylbenzene isn’t a name on most people’s radar, but stories from people working with it point to hidden risks both for health and the environment. Slow action or looking the other way leaves communities with pollution nobody agreed to. Industry push to rethink chemical hazards and smarter choices for substitutes make all the difference—not just for compliance, but for protecting those who handle the chemicals and those who live nearby.
| Names | |
| Preferred IUPAC name | butylbenzene |
| Other names |
1-Phenylbutane Benzene, butyl- Butylbenzene n-Butyl benzene |
| Pronunciation | /ˌɛnˌbjuːtɪlˈbɛnziːn/ |
| Identifiers | |
| CAS Number | 104-51-8 |
| Beilstein Reference | 1209244 |
| ChEBI | CHEBI:38841 |
| ChEMBL | CHEMBL14001 |
| ChemSpider | 12022 |
| DrugBank | DB01604 |
| ECHA InfoCard | 100.123.018 |
| EC Number | 202-065-6 |
| Gmelin Reference | 82217 |
| KEGG | C01799 |
| MeSH | D000687 |
| PubChem CID | 7737 |
| RTECS number | CU4375000 |
| UNII | Y3S068B08W |
| UN number | UN2669 |
| Properties | |
| Chemical formula | C10H14 |
| Molar mass | 134.22 g/mol |
| Appearance | Colorless liquid |
| Odor | Aromatic |
| Density | 0.862 g/mL at 25 °C (lit.) |
| Solubility in water | insoluble |
| log P | 3.7 |
| Vapor pressure | 0.38 mmHg (25°C) |
| Acidity (pKa) | 43 |
| Basicity (pKb) | 15.96 |
| Magnetic susceptibility (χ) | -73.0e-6 cm³/mol |
| Refractive index (nD) | 1.491 |
| Viscosity | 2.55 mPa·s (20 °C) |
| Dipole moment | 1.20 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 353.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -10.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6612.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H304, H315, H319, H335, H411 |
| Precautionary statements | P210, P261, P280, P301+P310, P303+P361+P353, P305+P351+P338, P331, P370+P378 |
| Flash point | 112 °C (234 °F; 385 K) |
| Autoignition temperature | 550 °C |
| Explosive limits | '1.1–6.4%' |
| Lethal dose or concentration | Lethal dose or concentration for N-Butylbenzene: "LD50 (oral, rat): 4,300 mg/kg |
| LD50 (median dose) | LD50 (median dose) of N-Butylbenzene: **3,005 mg/kg (oral, rat)** |
| NIOSH | NIOSH: DN2625000 |
| PEL (Permissible) | 50 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Toluene Ethylbenzene Propylbenzene Sec-butylbenzene Tert-butylbenzene Cumene Diphenylmethane n-Butylcyclohexane |

