N-Butyl Isocyanate: A Close Look at Its Story, Science, and Future
Historical Development
N-Butyl Isocyanate didn’t just appear out of thin air as a go-to intermediate for the chemical industry. Back in the postwar era, researchers looked for compounds that would improve performance in resins, agrochemicals, and even pharmaceuticals. Along that journey, isocyanates became the backbone for various innovative chemistries. N-Butyl Isocyanate, specifically, found its place because of its balance between reactivity and manageable volatility. Chemists saw early on that the butyl group offered a sweet spot for synthesis — long enough to add flexibility, short enough to avoid complications in reaction control. By the 1960s, notable research publications, patents, and early industrial scaling made it clear that this molecule had more to offer than just being another member of the isocyanate family. Its adoption kept growing as manufacturers became more confident in handling its hazards and realized the efficiency gains in diverse applications.
Product Overview
N-Butyl Isocyanate ends up used in many sectors because its functionality fits needs in coatings, adhesives, and active pharmaceutical ingredients. Chemists and process engineers value it for its straightforward single functional group that lets them target reactions and substitutions without scrambling to protect secondary positions. If you've ever wondered why certain agrochemical products deliver consistent performance or how binder systems in paints maintain flexibility, there's a decent chance this compound played a part somewhere in the manufacturing chain.
Physical & Chemical Properties
This molecule comes off as colorless and nearly odorless at room temperature, which sometimes makes folks underestimate its potency. Its boiling point hovers just under 158°C, and its density falls around 0.89 g/cm³. It dissolves in most organic solvents like acetone, toluene, and diethyl ether, which opens doors for both lab technicians and industrial-scale chemists. Reactivity jumps out even under mild conditions, especially with water, alcohols, or amines, which is both a blessing for fast reactions and a risk if exposed to the ambient environment. Some chemists have likened handling it to carrying around an eager student, always ready to react but quick to mess up if you lose focus.
Technical Specifications & Labeling
Products hit the market after batch testing for consistency of purity, typically greater than 98%, with water content kept below 0.05%. Impurities, like residual butylamine or unwanted side-products, demand strict quality thresholds to prevent unwanted side reactions down the line. Labels usually show the CAS number 111-36-4, and transport containers require UN hazard codes for toxic flammable liquids. Proper categorization isn't just regulatory bureaucracy — it keeps workers aware and businesses compliant, helping avoid headaches with transport, storage, and workplace incidents.
Preparation Method
Factories rely mostly on a phosgenation route, which might haunt anyone familiar with phosgene’s reputation. The process starts with n-butylamine, reacting with phosgene under controlled temperature and pressure, followed by strict workup procedures to eliminate corrosive by-products. Some firms have experimented with phosgene-free routes, using carbonyl diimidazole and urea derivatives, but these paths struggle to reach the same scale or cost-effectiveness. Process safety always sits front and center. Every step, from batch charging to post-reaction purification, demands constant attention to pressure, temperature, and venting systems. Operators working with these setups get plenty of hands-on training, not only to hit yield targets but to protect themselves and the community.
Chemical Reactions & Modifications
People in the lab rarely work with N-Butyl Isocyanate by itself for long. It reacts with alcohols to make carbamates, which then head on to become pesticides or specialty coatings. It tackles amines for the quick formation of ureas, giving rise to materials ranging from foam to active drugs. Its isocyanate group also acts as a tag for ‘click chemistry’ style reactions, where researchers build new molecules efficiently. Modifications sometimes include adding stabilizers to minimize polymerization, especially if storage conditions might fluctuate. Some even attach butyl isocyanate units to larger scaffolds, all to exploit the reactivity without exposing themselves to free isocyanate. The molecule’s versatility keeps research interesting — with some chemists winning patents for new linkages every year.
Synonyms & Product Names
Over the years, chemists have jotted down a variety of names: 1-Isocyanatobutane, Butyl isocyanate, n-Butylcarbylamine oxide, and even n-Butyl isocyanide (which actually confuses folks since isocyanates and isocyanides behave quite differently). Commercial products often stick with the clean “N-Butyl Isocyanate” label, printed clearly on both small bottles and large drums. For anyone ordering or trying to cross-reference suppliers, the CAS number helps sidestep any terminology hiccups.
Safety & Operational Standards
Working with this chemical calls for more than gloves and goggles. Respirators, splash shields, and effective local exhaust ventilation become daily gear for plant workers and lab techs alike. The compound irritates eyes, skin, and especially airways — even at concentrations below odor detection limits. Emergency response plans usually include high-flow showers, neutralizing powders, and detailed spill procedures. Occupational exposure limits draw from health agency data, with frequent air monitoring and medical surveillance for workers. Anyone storing it learns you can’t leave it near moisture, heat, or incompatible chemicals like acids. Training programs stick with clear language about risks, running drills for leaks or fires, and making sure every step — from barrel opening to waste disposal — follows a written, rehearsed plan.
Application Area
Companies use N-Butyl Isocyanate as a key building block for herbicides, insecticides, and fungicides, especially as part of carbamate pesticide synthesis. Its role in specialty coatings gives toughness to flooring, automotive finishes, and industrial machinery. Pharma researchers test versions of urea drugs built off this molecule as enzyme inhibitors or anti-cancer agents. The adhesive and binder industry also turns to it for making polyurethane systems, where flexibility and resilience matter. Sometimes it’s phased in for smaller-scale research projects, like synthesizing custom molecules to probe protein-ligand interactions or to build novel polymer backbones.
Research & Development
Research labs push N-Butyl Isocyanate into new synthetic pathways to invent better drugs, greener pesticides, and smarter coating systems. The hunt for safer, phosgene-free production methods remains a big target — not just because of public pressure, but because companies see the benefits in safety and sustainability. Some teams focus on tuning reactivity, adding molecular “handles” so chemists can tune product specificity or slow down unwanted side reactions. Universities invest in data modeling and AI-driven reaction optimization to shave months off development timelines. A few ambitious startups chase ‘closed-loop’ manufacturing, recycling the compound or turning waste into new products, aiming for a future where chemical sustainability goes hand in hand with profitability.
Toxicity Research
Toxicologists have set up dozens of experiments to understand how this molecule affects living systems. Inhalation and skin contact lead to clear irritation in rabbit, rat, and human studies. Chronic exposure seems tied to respiratory issues and in some cases sensitization, making warehouse air-monitoring systems more of a necessity than a luxury. Agencies classify it as acutely toxic, flagging it as a risk for anyone not wearing full personal protective equipment. Studies test metabolites and breakdown products, hoping to understand long-term risks and improve cleanup procedures in case of spills. The research clearly shows the gap between lab hazards and those faced by field workers — highlighting the need for workplace controls and community outreach wherever the chemical gets stored and handled.
Future Prospects
The road ahead promises plenty of challenges and opportunities. The biggest push right now comes from environmental forces — tightening regulations on toxic intermediates and phosgene-derived processes pressure manufacturers to innovate. Plant operators and research directors invest time and money searching for greener synthesis routes or safer alternative isocyanate sources. Meanwhile, new uses keep emerging in pharmaceuticals, materials science, and even in organic electronics where isocyanates help build conductive polymers or adhesives that stand up to harsh environments. Collaborative efforts between government, academia, and industry may help shape safer handling standards and encourage the adoption of both cleaner production and end-of-life management. Watching these directions unfold firsthand, it’s clear that N-Butyl Isocyanate’s role in science and manufacturing will keep evolving, balancing the chemical’s reactivity and value with practical concern for health, safety, and the planet.
What is N-Butyl Isocyanate used for?
Behind the Scenes in the Chemical Industry
I’ve spent enough time in and around industrial labs to know most folks have never heard of N-Butyl Isocyanate. This colorless liquid, carrying a sharp odor, stays behind the curtain in conversations about manufacturing. Yet there’s real reason for regular people—engineers, plant workers, even local communities—to pay attention. N-Butyl Isocyanate finds its way into products that touch daily life.
Where You’ll See Its Impact
This compound starts with simple roots. Chemical plants use N-Butyl Isocyanate to build larger molecules called ureas and carbamates. Paint companies rely on it for specialty coatings—think finishes on cars, electronics, and industrial equipment. The reason? It bonds strongly with many types of materials, giving coatings a tough, protective edge. In adhesives, product developers chase that same principle: secure bonds that stand up under stress.
Agriculture also leans on this isocyanate. Certain crop protection agents trace back to reactions using N-Butyl Isocyanate. Without specialty chemicals like this one, fields face tougher pests with fewer tools. On the pharmaceutical side, companies often tinker with N-Butyl Isocyanate in early drug synthesis. Researchers stitch together new molecules hoping to find treatments for old diseases, and this chemical sometimes acts as the starting point.
Health and Safety: Looking Beyond the Factory Fence
I can’t talk about N-Butyl Isocyanate without thinking about safety. My time visiting plants convinced me that risks travel beyond lab coats and goggles. Breathing in its vapors can irritate eyes and lungs. Spills need immediate attention to keep workers safe. Emergency rooms don’t want regular visits from people exposed to chemicals like this one, so strong training and proper equipment in any facility using N-Butyl Isocyanate matter at every stage.
Communities near chemical plants often carry worries about exposure. News stories about leaks or accidents—rare, but never impossible—stick in people’s minds. Companies have a responsibility to set up control systems, track emissions, and have emergency plans that work not just on paper but in real life. Regulators like OSHA and the EPA keep close tabs, but a real culture of safety comes from people who understand the chemical up close.
Finding a Smarter Way Forward
Any industry relying on specialized chemicals faces a crossroads: use what works, push for innovation, or both. N-Butyl Isocyanate delivers results in coatings, adhesives, and medicines that can’t be ignored. Still, it makes sense to ask tough questions. Are there safer alternatives that get the job done? How quickly can safer handling technologies roll out? In my experience, open conversation between industry, researchers, and communities shines a light on solutions that last. Swapping information about best practices, sharing accident reports openly, and keeping workers in the loop keeps everyone a step ahead of trouble.
Sometimes the most important work happens out of sight. N-Butyl Isocyanate rarely grabs headlines, but its effects ripple into everyday products and public health. Recognizing its power—and its risks—calls for care, honesty, and steady improvement on all sides.
What are the safety precautions when handling N-Butyl Isocyanate?
Understanding the Risks
N-Butyl Isocyanate carries some hefty risks. This chemical doesn’t just give off sharp fumes; it can provoke coughing, burn your nose, set off asthma, and irritate skin on contact. Splashing it in your eyes can seriously damage your vision. Folks working with it need to understand just how reactive and toxic it is. A little slip can cost you more than a headache — longer exposure has made some people sick for days.
Personal Protective Gear Counts
I remember handling isocyanate compounds in an old university lab. Gloves and goggles felt like armor, but they made the job a lot safer. Basic nitrile gloves aren’t enough for N-Butyl Isocyanate; you want thick butyl rubber gloves, chemical splash goggles, and a face shield. Wear a sturdy lab coat and, for bigger jobs, add a chemical-resistant apron. Clothes that fully cover arms and legs offer another line of defense.
Respiratory gear matters as well. An organic vapor cartridge respirator blocks harmful vapors. In tiny, unventilated spaces, air-purifying respirators won’t cut it; switch to supplied-air systems. Masks might sound like overkill, but they keep lungs clear after a hundred small exposures.
Handling and Storage: Zero Margin for Error
Poor habits often spell trouble. Tighten caps, double-check containers for leaks, and never store this chemical near food or open flames. N-Butyl Isocyanate reacts sharply with water and alcohols and can set off fires when it meets oxidizers. Store in a dry, cool cabinet with a chemical label that stands out. I’ve seen accidents sparked just by missing labels — don’t let it slide.
Always use N-Butyl Isocyanate in rooms where fresh air gets pulled in and fumes get pushed out. Fume hoods work wonders, but opening a window doesn’t. If a spill happens, quickly grab a spill kit with absorbent materials and neutralizing agents. Leaving the job half-done means someone else could pay a price.
Training Everyone on Site
Handling dangerous chemicals isn’t just about following protocols. Knowing exactly how to wash out eyes, clean up small spills, or evacuate the building makes a big difference. Regular training — with short drills or quick team reminders — builds habits. Most chemical accidents I’ve seen happened to smart people who thought they could skip steps or take shortcuts.
Key safety signs outside storage and work areas provide reminders, especially in places where multiple chemicals get used every day. Emergency showers, eyewash stations, and first aid kits should never be out of reach. If a coworker gets splashed, seconds matter. Anyone can freeze up, so talk out what to do long before any emergency.
Building a Safety Culture
The science behind N-Butyl Isocyanate is clear: it can do real harm. Just one wrong move can lead to lasting health effects, lost workdays, or even legal tangles. Prioritizing strict safety habits keeps everyone off the injury list and reassures your team they won’t come home with chemical burns or breathing problems. Using strong facts, practical gear, and a no-shortcut mindset keeps the lab, factory, or workshop running clean. Taking care of each other — that goes a long way toward a safer workplace.
What is the chemical formula of N-Butyl Isocyanate?
Getting to Know N-Butyl Isocyanate
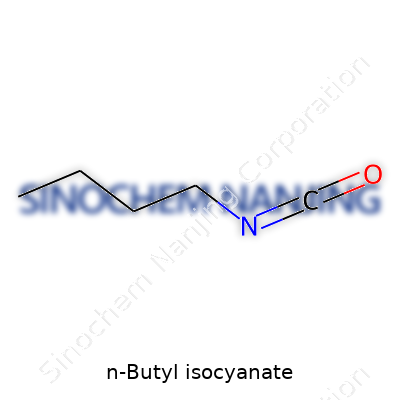
N-Butyl isocyanate, known for its use in organic synthesis, features a simple formula: C5H9NO. The structure carries a four-carbon chain, a single nitrogen, and one oxygen atom, arranged so that the isocyanate group attaches to the end of the butyl chain. More than a mere collection of letters and numbers, this formula reveals plenty about how the molecule acts and reacts. In the world of surface coatings, adhesives, and pharmaceuticals, this small but powerful compound often turns up during the process of making more complex chemicals.
Why Formulas Like C5H9NO Aren’t Just for Chemists
During my studies in college, a classmate spilled a tiny bottle of N-butyl isocyanate in a lab—instantly, everyone noticed the intense odor. Quick reference to its formula in our notes reminded us that even a small molecule can have a big presence. The isocyanate group in its structure makes handling even basic lab work a lesson in safety. The formula gives professionals crucial information about how to respond if something goes wrong, including the risks connected with inhalation or exposure. Reliable knowledge about the chemical’s structure and its reactive groups helps protect workers in factories or students in labs, keeping both people and the environment out of harm's way.
Industry and Health Impacts
Chemicals like N-butyl isocyanate might never grab headlines, yet they play a quiet role in everything from polyurethane foams to pest control products. The formula, C5H9NO, points to the reason why regulations surround its use. The presence of the –NCO group signals a volatile and potentially harmful substance. Factory workers and manufacturers trust labels and data sheets to figure out the best way to store and use it safely. Since compounds containing isocyanate groups can irritate the respiratory tract or cause sensitization, clear knowledge anchored in its formula aids in creating well-controlled environments with protective measures in place. In my own work, reading about hospitalizations from isocyanate overexposure drove home the message that chemical awareness starts with an honest look at structure and formula.
Seeking Safer Alternatives and Sustainable Solutions
Years of industry pressure have led many companies to invest in less hazardous substitutes, but some processes still require precise reactivity offered by the isocyanate group in N-butyl isocyanate. Awareness about the formula guides this search—chemists tailor new molecules to match or exceed the performance of older ingredients, minus the risks. Regulatory agencies set exposure limits and call for improved ventilation in spaces where this compound appears, based on trusted scientific evidence of toxicity and environmental persistence. These steps create value far beyond compliance, offering sustainable pathways for industries while uplifting both worker health and community safety.
Responsible Handling and Future Prospects
Chemists, manufacturers, and policymakers all rely on strong, factual understanding of substances like N-butyl isocyanate. The formula C5H9NO carries more than the weight of its atoms; it signals a need for vigilance and partnership across science, industry, and public health. Prioritizing knowledge, education, and real-world safety practices will continue to drive progress—ensuring this chemical, like many others, supports innovation without putting well-being on the line.
How should N-Butyl Isocyanate be stored?
An Often Overlooked Chemical, But Not Without Its Hazards
N-Butyl Isocyanate doesn't always get much attention outside specialized circles, but anyone who's spent time in a chemical storeroom knows its presence means donning gloves and double-checking labels. Years ago, I shared a cramped campus lab where one cracked seal or missed warning made for scary afternoons. N-Butyl Isocyanate, with its strong odor and volatility, never did much to calm nerves.
Keeping the Workspace Safe
Let's skip the textbook talk. This chemical will irritate skin, eyes, and lungs if mishandled. One small spill can become a bigger problem, setting off alarms or exposing entire rooms. And believe me, no one wants to play "what’s that smell?" in a workspace that should feel safe.
Storage starts with location. Don’t tuck N-Butyl Isocyanate next to acids, moisture, or any kind of oxidizer just to save shelf space. It reacts far too eagerly, and mixing it with water can generate toxic fumes before anyone even knows there's a leak. Concrete floors, metal shelving, and solid walls help, but only if the air moves freely and fire suppression fits the hazards. Vents, alarms, and easy escape routes turn a risky corner shelf into something much less nerve-wracking.
Choosing the Right Containers Really Matters
This isn’t the place to reuse some old glass jar. Sealed steel drums or tight plastic containers work best, but only if the lids fit tight and the label can stand up to drips. I’ve seen folks reach for unlabeled cans after lunch and spend an afternoon scrubbing—it isn’t worth the shortcut. Even a clean room loses its value if the smallest bit of vapor gets out.
Temperature Makes a Big Difference
Let the temperature go up just a few notches, and the vapor builds up fast. In my early days, a stuffy store-room in mid-July sent us running to check all the seals. Keeping things cool—below 30°C—means fewer surprises and much less chance of pressure swelling inside a drum. Dry spots with stable air run quieter, and the worry about sudden leaks fades a bit.
Planning for Emergencies
Even careful storage isn't everything. Accidents can happen, and plans save time when things go wrong. Chemical absorbent pads, eyewash stations, and a working ventilation hood become friends in the rare, stressful moments. Don’t forget to run a spur-of-the-moment drill once in a while, just so no one forgets how to act when the alarms sound.
Documentation and Routine Checks
Logs and regular inspections beat memory every time, especially after weeks away from the bench. Maintenance schedules, clear signage, and shared records keep everyone in sync—no wondering who last checked the seal or wiped down the benchtop. Good practices flow from shared experience, and older workers guiding newer faces make the whole room more confident.
Moving Toward Better Practices
Safer storage improves daily routines. One mistake won’t erase months of careful planning, but staying alert builds a culture where everyone watches out for the unexpected. Sharing tips between shifts or across teams has kept my own workspaces quieter and healthier, and that’s reason enough to keep those rules at the front of the lab book.
What are the potential health risks of exposure to N-Butyl Isocyanate?
Why N-Butyl Isocyanate Gets Noticed in Health Talks
N-Butyl Isocyanate often shows up in labs and factories where chemists turn out pharmaceuticals, pesticides, and plastics. Anyone stepping foot in those spaces probably recognizes the sharp, eye-watering smell of isocyanates. I spent some time working in a research facility that handled isocyanates, and seeing co-workers suit up in heavy gloves and respirators always drove home the potential danger. This chemical doesn’t ask much: just a splash on skin or a breath in your nose, and you’ll know it found you.
Immediate Effects That Hit Hard
Symptoms show up quickly. Most people report burning eyes, lots of coughing, or skin that feels on fire. Folks without proper protection can experience difficulty breathing, similar to a bad asthma attack. That isn’t an exaggeration—I watched a colleague wheeze his way to the emergency room after a small spill. The CDC ranks isocyanates among hazardous chemicals with good reason; short-term exposure injures eyes, lungs, and skin all at once.
Runny noses, sneezing, tight chests crop up even in small spills—these aren’t just annoyances that fade away, because some effects stick longer than you'd hope. Prolonged contact runs a real risk of skin becoming sensitive or allergic over time, which can haunt a person years after leaving the job.
Long-Term Damage Goes Deeper Than You Think
I learned from colleagues who had worked decades around chemicals: repeated exposure can trigger lifelong asthma or chronic bronchitis. The Mayo Clinic and OSHA list N-Butyl Isocyanate as a known cause of occupational asthma. Once sensitivities develop, even brief contact can launch severe attacks. Some people lose the ability to work in their industry after developing an allergy or chronic lung problem.
The chemical also brings up cancer concerns. Animal studies from regulatory bodies link certain isocyanates to increased cancer risk, especially with poor personal protection practices. Although more research is needed for this specific compound, the industry assumes that chronic isocyanate inhalation brings more risks than what records currently prove.
Responsible Ways to Cut Down Risks
Nobody has ever regretted doubling up on safety gear with this chemical. Working with N-Butyl Isocyanate means reaching for a fit-tested respirator, chemical-resistant gloves, and face shields on every shift. The American Industrial Hygiene Association stresses the role of local exhaust ventilation. At my old job, I saw firsthand how a well-maintained fume hood with regular filter changes kept the work atmosphere clearer and staff healthier.
Workplaces benefit when staff gets trained not just on handling, but on regular spill drills and first aid responses. Some employers go cheap on safety resources, but the cost of medical care or missed work days adds up fast. Well-written policies, careful storage, and airtight containers, plus spill response kits—these measures pay off day to day. No shortcuts work where powerful chemicals are involved.
Protecting Everyone—Not Just Lab Coats
Most people picture danger only in a lab, but there’s a bigger web. N-Butyl Isocyanate moves through supply chains, transport hubs, and waste sites. I’ve spoken with truck drivers who haul these chemicals and learned how easy it is for accidents to affect whole communities if something leaks on the road. That’s why regulations covering emergency notification and containment already exist, but they depend on people following the rules every day.
If you’re working anywhere near isocyanates, don’t assume someone else will catch mistakes. Safety comes from covering up, staying alert, and making sure everyone on the team puts protection before convenience. With chemicals this powerful, every small choice matters.
| Names | |
| Preferred IUPAC name | N-butyl isocyanate |
| Other names |
1-Isocyanatobutane n-Butyl isocyanate Butyl isocyanate Butylcarbyl isocyanate |
| Pronunciation | /ˈɛnˌbjuːtɪlˌaɪ.səʊ.kaɪˈæ.neɪt/ |
| Identifiers | |
| CAS Number | 111-36-4 |
| Beilstein Reference | 3539173 |
| ChEBI | CHEBI:132977 |
| ChEMBL | CHEMBL159269 |
| ChemSpider | 61954 |
| DrugBank | DB04209 |
| ECHA InfoCard | 03e6c9e9-5db2-4de0-baa7-c48d40cb9569 |
| EC Number | 209-817-5 |
| Gmelin Reference | 7548 |
| KEGG | C18969 |
| MeSH | D017905 |
| PubChem CID | 82136 |
| RTECS number | NX8575000 |
| UNII | W74D3C658G |
| UN number | UN2289 |
| CompTox Dashboard (EPA) | DTXSID7020953 |
| Properties | |
| Chemical formula | C5H9NO |
| Molar mass | 129.18 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | strong, pungent |
| Density | 0.868 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 1.97 |
| Vapor pressure | 4 mmHg (20°C) |
| Acidity (pKa) | 15.2 |
| Basicity (pKb) | 13.20 |
| Magnetic susceptibility (χ) | -7.03 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.398 |
| Viscosity | 0.657 mPa·s (at 20 °C) |
| Dipole moment | 4.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 379.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -86.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4786.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS06 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314, H317, H330, H334, H335 |
| Precautionary statements | P210, P261, P280, P302+P352, P304+P340, P305+P351+P338, P310, P403+P233 |
| NFPA 704 (fire diamond) | 3-3-2-W |
| Flash point | 50 °C |
| Autoignition temperature | 205 °C (401 °F; 478 K) |
| Explosive limits | Explosive limits: 1.4% - 9.8% |
| Lethal dose or concentration | LD50 oral rat 930 mg/kg |
| LD50 (median dose) | LD50 (median dose) Oral rat: 230 mg/kg |
| NIOSH | NIOSH: SA 9485000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for N-Butyl Isocyanate: "0.05 ppm (0.19 mg/m3) as TWA |
| REL (Recommended) | 0.5 ppm |
| IDLH (Immediate danger) | IDLH: 6 ppm |
| Related compounds | |
| Related compounds |
Isocyanic acid Methyl isocyanate Ethyl isocyanate Propyl isocyanate Phenyl isocyanate |

