N-Boc-4-Oxo-L-Proline Tert-Butyl Ester: Shaping Advances in Modern Chemistry
Historical Development
Years of progress in amino acid chemistry brought forward compounds like N-Boc-4-oxo-L-proline tert-butyl ester. Early investigations into proline derivatives opened the door for intricate modifications of simple structures, letting researchers design building blocks for complex molecules. The introduction of the Boc (tert-butoxycarbonyl) protecting group set a new standard in peptide synthesis. Chemists saw that protecting key reactive sites on proline made handling and downstream chemistry far easier. Development picked up speed during the late twentieth century, as laboratories started focusing on orthogonally protected amino acids for advanced pharmaceutical and biomedical projects. Over time, protocols improved, purity levels increased, and the compound’s role in research expanded rapidly.
Product Overview
N-Boc-4-oxo-L-proline tert-butyl ester stands out as a heavily modified proline derivative, blending a Boc-protected nitrogen with an esterified carboxy group and a ketone sitting at the four-position of the ring. Such multi-functional modulation makes the molecule a prime tool for organic chemists. Its stability and reactivity brought it into favor for crafting complex architectures and producing peptidomimetics and other bioactive materials. Sourcing high-purity material matters since impurities can disrupt the strict demands of medicinal chemistry. Laboratories avoid generic versions and source directly from experienced suppliers to ensure every reaction goes as planned.
Physical & Chemical Properties
On the bench, this compound usually appears as a white to off-white crystalline solid, resistant to modest temperature shifts. It has a molecular formula of C14H21NO6 and a molecular weight near 299.3 g/mol. The Boc and tert-butyl ester units boost the molecule’s weight and impact its solubility profile. N-Boc-4-oxo-L-proline tert-butyl ester dissolves well in organic solvents like dichloromethane, ether, tetrahydrofuran, and acetonitrile but remains only sparingly soluble in water. It holds a melting point typically between 92°C and 98°C, and chemists rely on infrared and NMR spectroscopy for identity verification, though routine TLC and HPLC methods work well for tracking purity through synthesis. The Boc and ester groups protect against hydrolysis under neutral and slightly acidic conditions but break down under strong acid or base.
Technical Specifications & Labeling
Reliable suppliers provide clear, thorough labeling for N-Boc-4-oxo-L-proline tert-butyl ester. This includes the correct IUPAC name, batch number, expiration and packing date, as well as purity level (frequently above 98%). Labels call out proper storage—dry and at low temperature—since the Boc group hydrolyzes over time, especially if moisture sneaks in. Packaging usually uses amber glass or polyethylene to avoid photodegradation and chemical interactions. Safety data sheets break down hazards like eye irritation or respiratory risk, stressing mask and glove use. Regulations may classify it as a non-hazardous research chemical, but adherence to good laboratory practice stays non-negotiable.
Preparation Method
Synthesis involves several steps, each tailored for efficiency and safety. Chemists start with L-proline, oxidizing the ring at the four-position to introduce the ketone. Protecting the amino group with Boc2O comes next, followed by esterifying the carboxy group with tert-butanol. Each phase uses controlled pH monitoring, temperature control, and time management to limit byproducts and improve yield. Organic solvents used in these steps—including dichloromethane, DMF, and acetonitrile—demand careful handling. After workup, recrystallization or preparative chromatography produces a pure, ready-to-use final product.
Chemical Reactions & Modifications
On the synthetic front, this ester acts as a workhorse. The Boc group comes off easily with trifluoroacetic acid or HCl in dioxane, regenerating the free amine where needed. Hydrolysis under basic conditions removes the tert-butyl ester, liberating the carboxylate for downstream reactions. The four-position ketone functions as a platform for further transformations—think reductive amination, Wittig reactions, or addition of nucleophiles for ring expansion. These modifications help generate large, intricate molecules, giving drug developers tools to tune pharmacokinetic and pharmacodynamic properties.
Synonyms & Product Names
You won’t always find this compound under one title. Researchers might list it as “tert-butyl 1-(tert-butoxycarbonyl)-4-oxo-L-proline”, “Boc-4-oxo-proline tert-butyl ester”, or less commonly, “N-Boc-4-keto-L-proline tert-butyl ester”. Catalog numbers and CAS registration streamline procurement, reducing the risk of costly mistakes, especially in multinational teams or labs sourcing from different continents.
Safety & Operational Standards
Working with this molecule doesn’t mean skipping on safety. Even in skilled hands, exposure risks exist, from inhalation of dust to skin contact from accidental spills. Inhaling fine powders could irritate the throat or cause coughing, while skin contact leads to mild redness or dryness if unprotected. Eye contact remains a concern, so goggles consistently make sense. Labs handling the compound operate with fume hoods, nitrile gloves, and closed containment protocols. Waste solvents from synthesis fall under hazardous chemical procedures. Spills mean immediate cleanup using wet wipes—not dry sweeping—to prevent aerosolization. Regulatory bodies may not flag this material as acutely toxic, but that never translates to carelessness on the bench.
Application Area
Application hinges mainly on advanced organic synthesis. Peptide chemists use this ester as a protected intermediate, letting them run complex stepwise reactions without scrambling amino acids’ natural order. Academia employs it to build non-natural peptides for probing protein interactions. Pharma companies depend on it during the early stages of drug discovery and production, searching for improved stability and target specificity in their therapeutic leads. Medicinal chemistry groups also reach for this compound when studying enzymes—or designing inhibitors that block specific prolyl residues in disease pathways. My own time in academic synthesis taught me to appreciate how much frustration a well-crafted building block can save. Sometimes, just swapping to a similar protected proline variant removes days of purification headaches.
Research & Development
Continuous R&D efforts drive innovation using N-Boc-4-oxo-L-proline tert-butyl ester. Research groups regularly tailor the scaffold to develop new functional materials or produce drug candidates with optimized biological properties. Cross-disciplinary partnerships push boundaries by integrating computational chemistry, high-throughput screening, and automation. Recent studies use the ketone site for selective labeling, making it easier to track molecular movements in live cells. Collaborations with material scientists open the door to polymers and nanostructures carrying this proline core for targeted delivery systems. As more researchers recognize its flexibility, demand keeps increasing, challenging suppliers to deliver higher purity and larger quantities at lower environmental cost.
Toxicity Research
The research community pays close attention to any molecule’s potential biological risks. Early studies show minimal acute toxicity in standard laboratory models, likely due to the bulky protecting groups limiting bioavailability. Any derivative that enters animal studies or preclinical protocols undergoes strict scrutiny—testing for off-target effects, metabolism, and possible accumulation. Chronic exposure data remain sparse; more research is ongoing to address questions about metabolic products and their pharmacology. Handling practices reflect these unknowns: researchers work in well-ventilated spaces, avoid eating or drinking nearby, and dispose of waste through approved chemical disposal channels.
Future Prospects
N-Boc-4-oxo-L-proline tert-butyl ester’s future looks bright given the surge in peptide therapeutics, custom materials, and enzyme-targeted drug discovery. The toolkit for modifying natural amino acids keeps expanding. This compound serves as a platform—one with the practical staying power that chemists value during scale-up and the flexibility that discovery scientists crave for one-off projects. Automation and machine learning may soon streamline its synthesis, making once-rare derivatives “off-the-shelf” rather than custom, accelerating the journey from bench to bedside. At the same time, green chemistry initiatives push for cleaner methods and solvent recovery. Research landscapes don’t stand still, and neither do the tools at the heart of molecular innovation.
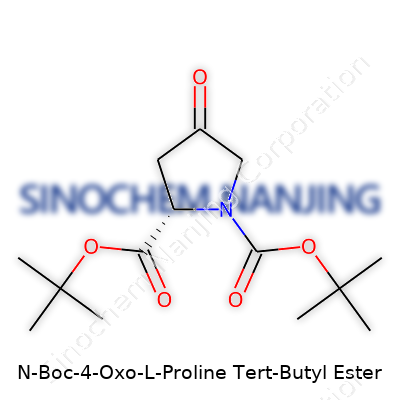
What is the chemical structure of N-Boc-4-Oxo-L-Proline Tert-Butyl Ester?
What Draws Scientists to This Molecule?
N-Boc-4-Oxo-L-Proline Tert-Butyl Ester carries value not just for its chemical quirks, but because it’s a stepping stone in pharmaceutical development. Organic chemists look for such compounds since the functional groups offer real flexibility. The name alone unpacks a story: “N-Boc” refers to a tert-butoxycarbonyl protecting group on the nitrogen; “4-oxo” points to a ketone sitting on the fourth position of the proline ring; “tert-butyl ester” marks the carboxylic acid protected as an ester. Every part of this name shapes how the molecule will react and interact with others down the line.
The Backbone: Understanding Its Chemical Anatomy
Dissecting the structure, start with the base: proline. This amino acid’s five-membered pyrrolidine ring forms the skeleton. Once the “oxo” group lands at the fourth carbon, this ring develops a sharp edge—adding reactivity thanks to that carbonyl’s electron-withdrawing power. The nitrogen on the ring doesn’t get left bare; it wears the Boc group, a common face among amino acid derivatives. That Boc piece shields the nitrogen from unwanted reactions, giving chemists more control as they build complex molecules. For those who remember their undergraduate days, this protection step saves headaches in peptide synthesis.
On the carboxylic side, instead of a bare acid, we find a tert-butyl ester. This feature offers the same protection logic—avoiding unwanted side reactions and keeping the acid tamed until the right moment arises. The molecule, taken as a whole, acts as a protected, reactive intermediate. For those who work on coupling reactions or custom peptide chains, its structure means you get the ring rigidity needed for specificity, without the chaos that comes from unprotected amines and acids.
Why All This Protecting?
Ask anyone who’s tried multi-step organic synthesis, and they’ll tell you protection is everything. The Boc group gets popular thanks to its easy removal—one shot of mild acid and it’s gone, leaving the amine free and clean for action. Tert-butyl esters play the same game, cleaved smoothly by acids like trifluoroacetic acid, which makes purification easier for the next stage. Without this structure, side reactions would litter every synthesis.
Importance in Pharma and Research
Pharmaceuticals demand precision. N-Boc-4-Oxo-L-Proline Tert-Butyl Ester lets researchers tweak drug candidates with predictable outcomes. Expect to see derivatives of this compound pop up in enzyme inhibitor libraries or as fine-tuned scaffolds for targeting bioactive pockets. The stability and handling offered by Boc and tert-butyl groups save both time and materials, reducing stress during scale-up and batch production. Tiny changes in molecular features can swing a compound from inactive to effective, so every protecting group makes a real world difference.
Addressing Potential Issues
Compounds like this open doors, but they also spotlight chemistry’s sustainability question. The reagents used for protecting and deprotecting aren’t always easy on the environment. Chemists look for greener alternatives, recycling routes, or milder conditions to clear Boc and tert-butyl groups without harsh waste. The future will likely see more work toward enzymes or catalysts that handle these tasks, minimizing byproduct formation.
Life in the lab hinges on reliable, well-characterized intermediates. Students, researchers, and industrial chemists stake progress on the trustworthiness of chemical structures. With every functional group and protecting group, choices should always lean toward safety, efficiency, and transparency—core principles that underpin both scientific advance and public trust.
What are the common applications of N-Boc-4-Oxo-L-Proline Tert-Butyl Ester in research or synthesis?
A Tool on Every Synthetic Chemist’s Bench
Science throws a lot of jargon around, but talk with anyone doing peptide synthesis or tinkering with small molecules, and chances are they mention intermediates like N-Boc-4-Oxo-L-Proline tert-butyl ester. This molecule finds a home in labs working on next‑gen drugs and new materials. It’s not flashy, but it keeps projects moving. Having spent hours setting up reactions where every step had to go right, I know the relief of reaching for a stable, predictable intermediate.
Makes Peptide Synthesis Manageable
Peptide chemistry can feel like threading a needle in the dark. Protecting groups matter. The Boc group, sitting on the nitrogen, gives chemists a sigh of relief because it holds strong through most steps and then lets go cleanly when it’s time. N-Boc-4-Oxo-L-Proline tert-butyl ester simplifies tough sequences. It lets teams build proline rings or modify peptide backbones without watching their building blocks fall apart or stick together by mistake.
Solid-phase peptide synthesis—often the main playground for biomedical advances—depends on building blocks that stay put until the right moment. The tert-butyl ester keeps the carboxylic acid from reacting early. Only once assembly ends do harsh acids remove it, giving access to the final active compound. Working with this kind of protected amino acid has helped groups step up new analogs for cancer therapies and enzyme design.
Opening Doors for Analog Development
Pharmaceutical research crowds the literature with analogs, all searching for that perfect tweak—better fit, improved action, reduced side effects. N-Boc-4-Oxo-L-Proline tert-butyl ester has shown up in projects testing new inhibitors for proteases and designing next-gen antibiotics. Flexible enough for a range of modifications, it becomes a launchpoint for cyclic or constrained analogs.
Tough targets, like some viral enzymes or stubborn cancer cell pathways, demand creative chemistry. Laboratories use this ester in routes toward macrocycles and peptidomimetics that mimic protein surfaces or loop regions. Making rigid, stable rings is part art, part grit. I remember whole weeks dedicated to coaxing one bond to form—starting from a reliable protected proline always helped.
Enabling Safer, Greener Synthesis
Cleaner reactions matter now more than ever. Teams seek reagents that cut down on unwanted byproducts or toxic intermediates. N-Boc-4-Oxo-L-Proline tert-butyl ester fits modern demands by combining stability with ease of removal in the final cleavage step. Large-scale peptide development leans on the safety profile this approach offers. Less risk to researchers, less waste to treat downstream.
Academic labs, contract manufacturers, and biotech startups have all leaned into strategies that reduce steps, shrink solvent use, and create purer products. No shortcut replaces skill, but plenty of headaches disappear using robust intermediates like this one. Talking over lunch with colleagues, I’ve heard plenty sigh—the right protecting group makes or breaks the week.
Pushing Boundaries in Chemical Biology
Beyond peptides, folks in chemical biology reach for N-Boc-4-Oxo-L-Proline tert-butyl ester when they need a foothold for probe design or labeling studies. Its reactivity profile and reliability allow fine-tuning, making it possible to attach tracers, fluorophores, or targeting motifs. Introducing precise changes in a protein or small molecule probe relies on these building blocks.
As research chases more targeted therapies and engineered proteins, solid, versatile compounds like this keep things grounded. It’s rarely the star, but chemists trust it with their hardest challenges. Years spent building molecules from scratch have shown me—progress in the lab depends on strategic choices. N-Boc-4-Oxo-L-Proline tert-butyl ester has quietly shaped the pathways leading to today’s breakthroughs.
What is the recommended storage condition for N-Boc-4-Oxo-L-Proline Tert-Butyl Ester?
In academic labs and commercial settings alike, reliable storage practices for fine chemicals draw a clear line between a successful project and frustrating setbacks. N-Boc-4-Oxo-L-Proline Tert-Butyl Ester sits among those compounds that can quietly spoil an entire synthesis if left in a warm, humid, or unprotected environment. Experience in the chemical research trenches has taught me the importance of nailing down the basics: temperature, humidity, light, and container choice. Cutting corners here doesn't just risk losing a compound—a missed step means lost time, uncertainty in results, and even money down the drain.
Temperature: Cool Heads Prevail
Chemists know the subtle anxiety that comes with pulling a sensitive ester off a shelf, only to find degradation. N-Boc-4-Oxo-L-Proline Tert-Butyl Ester holds best in a fridge, usually set between 2°C and 8°C. Room temperature storage brings little reassurance, especially in warmer climates where the relentless march of time and heat can erode product quality. Storing chemicals in well-calibrated refrigerators reduces risk, preserves reactivity, and upholds batch-to-batch consistency, a lifeline for anyone scaling up reactions or defending reproducibility in publications.
Humidity: The Hidden Enemy
Those just starting out often underestimate moisture's quiet threat. Like other Boc-protected amino acid derivatives, this ester attracts water if you let humidity creep up. A small clump can mask more insidious problems—chemical hydrolysis doesn’t always wave a red flag. Every failed TLC or NMR spectrum traces back to the simple act of not screwing a cap tight. Keep storage areas dry and the jar sealed. Invest in desiccants. Silica gel packs don’t break the bank, but they do keep the ester in top shape for when you need it, whether weeks or months later.
Light Sensitivity: Shield for Stability
Some forget to guard against light. Overexposed chemicals sometimes fade, as do dreams of reliable synthesis. Amber glass bottles make a big difference. If you can’t find one close at hand, wrap the bottle with aluminum foil. Ultraviolet rays have their place in the lab, just not on your chemical bench. A dark, closed cabinet, away from stray light, keeps everything stable for the long haul.
Air: Oxygen and its Unwanted Hand in Degradation
Oxygen exposure does its share of silent damage to esters and other sensitive organics. Many researchers I know flush such containers with argon or nitrogen gas, especially if they expect storage to last beyond a couple of days. Less headspace cuts down on oxidation, and for more precious batches, it’s a step worth adding. Every milligram counts when a rare intermediate took hours to synthesize, or when budgets limit fresh starts.
Labeling, Documentation, and Practical Solutions
Staying organized seems basic, but labels fade, stickers peel, and vials shift shelves. Each jar should carry its arrival date and original supplier info. Encourage colleagues and students to log their own samples. Regular audits of storage conditions catch problems before they end careers. All this stems from a culture that values quality and respect for the next person using the reagent.
Why Good Storage Matters
Quality research stands on trustworthy data. Poorly stored intermediates undermine everything from purity to yields, muddying mechanistic studies and sending teams back to square one. Companies depend on robust quality assurance, and universities need credible published results. Good storage practices help ensure people downstream can build on what’s gone before, instead of starting at a disadvantage. For anyone handling N-Boc-4-Oxo-L-Proline Tert-Butyl Ester, attention to these ordinary but crucial details adds up to real scientific progress.
What is the purity and CAS number of N-Boc-4-Oxo-L-Proline Tert-Butyl Ester?
The Identity Behind a Name: CAS Number Matters
N-Boc-4-Oxo-L-Proline Tert-Butyl Ester isn't just a mouthful—it's a specialty compound with real-world use in labs and industry. People ask about the CAS number for a reason. In this case, the CAS registry assigns 138844-62-1 to this specific molecule. The number itself serves as a fingerprint, allowing chemists and suppliers around the world to speak the same language. I remember many times a miswritten CAS number meant the wrong box on the loading dock. That delays research and costs money. Reliable sourcing depends on these digits matching up every step from bench to bottle.
Purity: Not Just a Buzzword for Scientists
Purity comes into play right after the CAS number. Suppliers of N-Boc-4-Oxo-L-Proline Tert-Butyl Ester typically advertise purities of 97% and up. For chemists, those couple of percentage points matter. Impurities can alter a reaction or skew data, especially in peptide synthesis or drug research. In my years working with sensitive synthesis, a batch dropping to 95% purity could stall a whole week’s work. The higher the percentage, the fewer unknowns enter the experimental chain. Confirming these numbers often demands more than a glance at paperwork. Analytical checks like NMR, HPLC, and mass spectrometry give that peace of mind that clean materials deliver consistent results. Mistakes cost more in time and dollars than the few pennies saved on a lower-grade order.
How Purity and Traceability Shape Outcomes
People outside the lab sometimes overlook the significance of formal documentation. Reputable suppliers include certificates of analysis and batch records. Cheap imports or ambiguous labels leave researchers guessing. I’ve heard stories of promising experiments ruined when a contaminated reagent slipped in from a no-name supplier. By trusting verified sources and double-checking CAS numbers, users cut down the risk of rework and waste. That practice supports safer workplaces, too.
Looking Ahead: Building Better Supply Lines
No single chemical solves every problem in research or manufacturing, but consistency in identity and purity goes a long way. N-Boc-4-Oxo-L-Proline Tert-Butyl Ester often operates as a protected intermediate and as a building block for peptides and other functional molecules. Every missed impurity means less trouble with purification and fewer hiccups in scale-up. This isn’t just lab talk—the further downstream you find a mistake, the more effort and material go out the window. Transparent traceability from supplier to scientist closes a major gap. I’ve seen organizations that require supplier audits and purity certificates end up with smoother workflows and less waste. In the end, keeping the CAS number straight and purity high protects both budgets and reputations.
Are there any specific safety or handling precautions for N-Boc-4-Oxo-L-Proline Tert-Butyl Ester?
Why This Chemical Needs Respect in the Lab
Lab work has a way of reminding you that even the most routine-looking compound deserves attention. N-Boc-4-Oxo-L-Proline Tert-Butyl Ester often pops up as an intermediate for making peptides and other bioactive molecules. Exposure isn’t worth brushing off. I’ve seen colleagues underestimating solvents or similar esters—usually because the bottle didn’t look menacing—and regretting it. Here, a little caution easily outweighs the inconvenience.
What GHS and Reliable Data Say
Reliable manufacturers and suppliers list this compound with the GHS exclamation mark, which signals that it can irritate skin and eyes. It doesn’t take much for a few drops to start burning or for a puff of dust to trigger sneezing. Once, I watched someone spill a tert-butyl ester on a gloveless hand. Immediate redness, a dash for the wash station, and the next few hours spent with a sore palm.
Eye protection matters. Standard lab specs or goggles block out splash risks. This isn’t just a recommendation—it stops a careless moment from turning into a medical emergency. Gloves need to be disposable nitrile or another chemical-resistant variety. Latex has a nasty habit of letting organics sneak through if you’re not careful. Nitrile stands up better against a splatter or a slow leak.
Air Quality and Storage Conditions
Fume hoods aren’t just there for show. I’ve noticed organic esters give off fumes, even without a strong smell. The gases can irritate nasal passageways and sometimes leave a taste that sticks for hours. Work with the chemical in a vented hood, especially during transfers or if heating becomes necessary. The risk isn’t about dramatic explosions—more about steadily accumulating irritation and potential allergic reactions over time.
Keep the bottle sealed, away from direct sunlight and heat. Room temperature works well, but dry conditions make a difference. Moisture—even a little—invites hydrolysis, which ruins your reagent and can start releasing acidic byproducts. I once cut corners on this front, and the stuff turned to sludge after a week in a steamy storeroom. Freshness matters, and an old batch can behave unpredictably on a project, costing days of troubleshooting.
Spills and Waste—Don’t Leave It to Happenstance
Spills are inevitable, even with steady hands. Absorb with an inert material and attach a proper label marking the waste as hazardous. Tossing this chemical into general waste just creates headache for the next person handling it. A dedicated solvent or chemical waste container works best. I still remember cleaning up after a colleague who poured organic waste down the sink “just this once”—which triggered a lab-wide inspection and days of paperwork. Regulations enforce themselves, and the fines get expensive fast.
Personal Experience: Small Habits Make a Big Difference
Working with esters like this one has convinced me that good habits prevent most accidents. Keeping clear standard operating procedures on benches, posting quick reference guides, and running annual safety refreshers set the tone. Encourage open talk about near-misses and lessons learned. In every lab I’ve joined, the safest teams acted on problems before they started, not after the fact. Good safety is contagious, and it pays off over a career.
Looking Ahead
Steady handling of N-Boc-4-Oxo-L-Proline Tert-Butyl Ester means combining basic safety gear, a reliable hood, and careful storage. With these, you can focus on the chemistry—without the distractions of avoidable burns or ruined experiments. Everyone benefits, from fresh students to seasoned employees. Good habits build trust, smooth research, and a lab where everyone gets to go home with ten fingers and two healthy eyes.
| Names | |
| Preferred IUPAC name | tert-butyl (2S)-2-[(tert-butoxycarbonyl)amino]-4-oxopyrrolidine-1-carboxylate |
| Other names |
tert-Butyl (2S)-1-(tert-butoxy)-4-oxo-2-pyrrolidinecarboxylate tert-Butyl N-Boc-4-oxo-L-prolinate |
| Pronunciation | /ɛn-bɒk-fɔːr-ˈɒksəʊ-ɛl-prəʊliːn tɜːt-ˈbjuːtɪl ˈɛstə/ |
| Identifiers | |
| CAS Number | 1256346-37-4 |
| Beilstein Reference | 15728606 |
| ChEBI | CHEBI:141529 |
| ChEMBL | CHEMBL3702077 |
| ChemSpider | 20568217 |
| DrugBank | DB08205 |
| ECHA InfoCard | 01-2120767900-52-XXXX |
| Gmelin Reference | 114715 |
| KEGG | C16612 |
| MeSH | D017259 |
| PubChem CID | 16026757 |
| RTECS number | WH2038756 |
| UNII | L2MT978Q8E |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | EPA CompTox Dashboard: "DTXSID109195936 |
| Properties | |
| Chemical formula | C13H21NO5 |
| Molar mass | 287.33 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.2 g/cm³ |
| Solubility in water | Insoluble |
| log P | 0.09 |
| Acidity (pKa) | 8.21 |
| Basicity (pKb) | 7.6 |
| Magnetic susceptibility (χ) | N-Boc-4-Oxo-L-Proline Tert-Butyl Ester: χ = -73.1 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.470 |
| Viscosity | Viscous oil |
| Dipole moment | 3.27 D |
| Hazards | |
| Main hazards | H302, H315, H319, H335 |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P264, P270, P280, P301+P312, P305+P351+P338, P330, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: -- |
| Flash point | > 108.5 °C |
| PEL (Permissible) | Not established |
| REL (Recommended) | 250 mg |
| Related compounds | |
| Related compounds |
N-Boc-L-Proline L-Proline N-Boc-4-Hydroxy-L-Proline 4-Oxo-L-Proline Tert-Butyl L-Prolinate N-Boc-4-Oxo-D-Proline Tert-Butyl Ester N-Boc-L-Proline Tert-Butyl Ester |

