N-Acetyl-P-Phenylenediamine: From Its Roots to Modern Science
Historical Development
The roots of N-Acetyl-P-Phenylenediamine trace back to the early 20th century, a period brimming with discoveries in synthetic chemistry. Chemists seeking to tame the complexity of aromatic amines experimented widely with acetylation reactions, making breakthroughs that still echo today. The compound, known for its distinctive structure and reactivity, emerged from a push to create more stable, less allergenic derivatives of p-phenylenediamine. Historical records tell stories of its earliest uses as both a laboratory reagent and a building block for dyes, underpinning the textile revolution. Its introduction followed demand for less caustic chemicals in personal care products, and the shift away from crude coal tar intermediates. This compound gained a reputation for offering a lower toxicity profile than unmodified phenylenediamine, drawing the attention of both industrial chemists and toxicologists.
Product Overview
N-Acetyl-P-Phenylenediamine stands as a product of acetylation—swapping hydrogen with an acetyl group on the parent p-phenylenediamine molecule. The structure features a benzene ring flanked by two amino groups, with one bearing the acetyl moiety. Chemists have relied on it as a key intermediate in producing advanced polymers, pharmaceuticals, and specialty dyes. Beyond chemical synthesis, the product often finds its way into labs focused on antioxidant research or analytical chemistry, where its stability and reactivity come into play. Its reliable purity and defined behavior in reactions grant it firm ground in quality assurance setups and R&D labs.
Physical & Chemical Properties
At room temperature, N-Acetyl-P-Phenylenediamine appears as off-white or beige crystals, revealing a characteristic, faint odor. The molecular formula is C8H10N2O, and its molar mass hovers around 150.18 g/mol. The melting point lies between 132°C and 137°C, reflecting its stability compared to its parent compound. It dissolves moderately in water but shows a greater affinity for polar organic solvents—ethanol, DMSO, and acetone top the list. This solubility profile enables precise formulation work. Chemically, the electron-rich aromatic ring coupled with the acetylated amino group offers selective reactivity, enticing chemists exploring oxidative coupling or polymer precursors.
Technical Specifications & Labeling
Manufacturers list N-Acetyl-P-Phenylenediamine under strict technical specifications, responding to calls for lot-to-lot consistency and regulatory compliance. Certificates of analysis usually report assay purity—often above 98%—as well as residual moisture, ash content, and heavy metal levels. Labels display hazard pictograms and warnings according to the Globally Harmonized System (GHS), as well as REACH and OSHA-aligned statements for workplace safety. Batch numbers, shelf life dates, and guidelines for storage—cool, dry, away from direct sunlight—come printed on each container, reaching back to the lessons drawn from earlier decades of chemical mishandling.
Preparation Method
Production leans on straightforward laboratory chemistry, usually initiated by acetylation of p-phenylenediamine. The base reactant combines with acetic anhydride in an aqueous or alcoholic medium, often in the presence of a gentle base like sodium acetate. This route limits the formation of unwanted by-products, trimming down purification steps. Once the acetylation completes, several washes with water and recrystallizations follow, yielding a product with a high degree of chemical uniformity. The process doesn’t demand exotic equipment, reflecting the democratization of synthetic protocols over time.
Chemical Reactions & Modifications
N-Acetyl-P-Phenylenediamine takes a starring role in oxidative chemistry. The acetyl group shields one amino position during reaction, granting synthetic chemists fine control when introducing additional groups to the aromatic system. Hydrogenation, halogenation, and diazotization reactions open up further modifications, a key factor in its popularity for dye and pharmaceutical R&D. The molecule withstands moderate acid and base conditions, which simplifies downstream work. Chemists often turn to this compound for constructing more elaborate molecules with electron-donating features, supporting advances in organic semiconductors and antioxidants.
Synonyms & Product Names
N-Acetyl-P-Phenylenediamine carries a parade of synonyms in commercial and scientific circles: 4-Aminoacetanilide, N-Acetyl-1,4-phenylenediamine, and para-acetylaminophenylenediamine being standouts. Some suppliers list it under trade names reflecting manufacturer brands or purified forms, creating a bit of confusion for those browsing catalogs for the first time. Checking CAS numbers helps avoid mix-ups, as names fluctuate between language conventions and regional regulations. This underscores the ongoing challenge of harmonizing chemical nomenclature worldwide.
Safety & Operational Standards
For handlers, the compound demands respect. While it addresses some allergy risks linked to p-phenylenediamine, it still carries its own hazards—risk of eye, skin, and respiratory irritation, and potential sensitization in susceptible individuals. Workflows keep exposure to a minimum: gloves, goggles, and ventilated hoods serve as standard PPE in labs and factories. Chemists stick to well-ventilated areas and handle even minor spills with caution, sourcing lessons from incidents recorded in old industrial safety bulletins. Environmental standards push companies to treat effluent with care, preventing contamination downstream. Site-specific training and emergency protocols round out the safety net for all personnel touching this compound.
Application Area
Industry finds many uses for N-Acetyl-P-Phenylenediamine. In dye and pigment manufacturing, its role as an intermediate offers pathways to vibrant colors for textiles and inks. Pharmaceutical developers value its stability and the way the acetyl group tempers reactivity, streamlining the creation of prodrugs and metabolites for testing. Analytical chemists rely on it as a reagent for assays where its colorimetric changes under specific conditions flag the presence of oxidizing agents. Outside the lab, some hair dye formulations tap into its chemistry, seeking alternatives to harsher amines. My own experience handling material safety evaluations highlights the compound’s reliability and the trust placed on its consistent behavior during formulation.
Research & Development
Research teams keep N-Acetyl-P-Phenylenediamine at arm’s reach when pushing into antioxidant chemistry and bioconjugation. Publications trace exploratory work in developing more stable anti-aging actives and pharmaceutical candidates that exploit the protected amino group. Leading innovation labs combine it with transition metal catalysts, seeking breakthroughs in polymer development for electronics. Efforts in analytical methodology use the compound’s distinctive UV-vis absorption profile to benchmark detection methods, feeding into improvements in food and environmental safety analysis.
Toxicity Research
Toxicologists examine N-Acetyl-P-Phenylenediamine for both acute and chronic health risks. Animal studies suggest lower sensitization rates than plain p-phenylenediamine, though not to a level that permits unrestricted use. Skin patch testing in cosmetics verifies irritation thresholds, guiding regulatory approvals for leave-on and rinse-off applications. Environmental toxicology probes breakdown pathways and the persistence of metabolites, since regulatory agencies demand proof compounds won’t linger or bioaccumulate in aquatic systems. In my years following product safety dossiers, the consensus leans toward treat-with-caution, keeping stakeholders on guard until longer-term data come in.
Future Prospects
Eyes watch advances in green synthesis of N-Acetyl-P-Phenylenediamine, with academic and industry labs ramping up bio-based acetylation methods to minimize waste. Digital chemistry finds traction here, using molecular simulation to predict new derivatives that might unlock further use—improved pharmaceuticals or novel biosensors top researcher wishlists. Tougher chemical regulations spur efforts in life-cycle assessment and end-of-life management, as both producers and end-users work to lower environmental impacts. Materials science draws on the compound’s adaptability, seeking to bridge legacy dye and modern sensor needs in a single, scalable molecule. I see peers in pharma and analytical labs banking on new patent filings, proof investment and curiosity around this old yet ever-relevant compound persist.
What is N-Acetyl-P-Phenylenediamine used for?
A Common Ingredient with Hidden Reach
N-Acetyl-P-Phenylenediamine doesn’t show up on grocery lists, but most people have interacted with products that rely on it. Its most recognized role comes from the world of hair color. This chemical sits in the toolkits of many hair professionals and shows up on the ingredient lists of at-home coloring kits. N-Acetyl-P-Phenylenediamine works as an intermediate, meaning it helps create the deeper browns, rich blacks, and even auburns many people want. Coloring hair goes well beyond vanity; it often connects to self-confidence, identity, and cultural expression.
Through my work covering the beauty industry, I’ve noticed how important safety becomes when chemicals touch skin and hair. Dermatologists often talk about allergic reactions tied to para-phenylenediamine (PPD), a related compound used in permanent dyes. N-Acetyl-P-Phenylenediamine aims to lower some of those risks. Its chemical tweaks may reduce the chances of sensitivity in some users, making hair coloring less of a gamble. Lab studies back this up, showing a milder reaction profile compared to traditional PPD. This matters for people who have had bad experiences with dyes but aren’t ready to give up coloring. Safer alternatives help more people maintain their personal style without discomfort or social anxiety.
Role in Diagnostics and Research
Beyond hair products, N-Acetyl-P-Phenylenediamine finds work in laboratory settings. Clinical labs use derivatives of this compound to test for certain bacteria. Its ability to change color under specific conditions helps microbiologists identify pathogens with more accuracy. Spotting infections early saves lives, especially in high-risk hospital environments. Medical journals highlight this utility in testing for organisms like Neisseria gonorrhoeae, which requires fast, dependable identification to prevent outbreaks.
This chemical also supports innovation in chemistry labs around the world. N-Acetyl-P-Phenylenediamine acts as a starting point for synthesizing complex molecules. These building blocks allow researchers to develop new drugs, industrial materials, and scientific probes. The incremental progress in labs often translates to advances in treatments for diseases, better quality control in manufacturing, and environmental monitoring technologies. Science relies on workhorse compounds like this—often unsung, always essential.
Sustainability, Safety, and the Path Ahead
N-Acetyl-P-Phenylenediamine’s versatility brings both opportunities and responsibilities. Its wide use has led to regulatory discussions. European agencies like the SCCS (Scientific Committee on Consumer Safety) regularly assess its health impact. They look at exposure data and toxicology reports, aiming to find the right balance between meeting consumer demand and ensuring public health. Regulation demands transparency from cosmetic companies, who must test for impurities and provide clear labels. This builds trust and keeps harmful effects in check.
From what I’ve seen, education holds the key. Many people don’t recognize long ingredient names. Salons, schools, and drugstores could share more about potential reactions and patch testing. Training hairdressers to spot allergies early has already made a difference in some communities. On the science side, ongoing research into safer dye molecules matters just as much. Green chemistry methods, which cut down on hazardous by-products, give further momentum to safe innovation.
N-Acetyl-P-Phenylenediamine fits quietly into many routines—personal care, medical safety, and scientific research. Value doesn’t always mean visibility. Staying informed about what goes into products and what happens in labs helps protect people and drive smarter choices throughout society.
Is N-Acetyl-P-Phenylenediamine safe to use?
What Is N-Acetyl-P-Phenylenediamine?
N-Acetyl-P-Phenylenediamine often pops up in hair dyes, research labs, and industry settings. Its main appeal comes from the way it helps develop color. Anyone who’s ever mixed up a box of permanent hair color might have applied something closely related to this chemical.
Sifting through the Safety Concerns
The question of safety isn’t an academic exercise—people care about the stuff they put on their skin or breathe in at work. N-Acetyl-P-Phenylenediamine belongs to the larger family of aromatic amines. Some in this group have a sketchy reputation. Take para-phenylenediamine (PPD); it’s known to cause allergic reactions in some folks, leading to skin rashes, redness, and irritation. Acetylation, the process of adding an acetyl group, changes how the chemical acts, and some studies suggest that the reaction may reduce its allergenic punch.
What Science Says So Far
A few years back, researchers examined the allergic effect of N-Acetyl-P-Phenylenediamine by patch testing it on people sensitive to PPD. The overwhelming majority did not react. One study in Contact Dermatitis saw only a small fraction of PPD-allergic patients reacting to the acetylated version. That gives some hope to anyone who wants to avoid a rashy mess.
Still, sitting on a few studies doesn’t close the case. The European Commission’s Scientific Committee on Consumer Safety looked at the available evidence and put the ball in industry’s court, saying more data would help clear things up. This tells me that while current evidence points to relatively low risk, absolute certainty is in short supply.
Real-Life Exposure
Personal experience shapes how I look at these things. My mother colored her hair at home for decades. She never broke out in hives, but I remember reading the warning labels out loud before she started, making sure to follow the instructions to the letter. I’ve also met several people who developed itchy scalps and blistering from some dyes, especially those containing PPD. Even if N-Acetyl-P-Phenylenediamine offers a gentler approach, a patch test before full use never hurt anyone.
Working with Chemicals at Home and at Work
People sometimes forget that workplace exposure looks different from home use. Someone who mixes dyes daily in a hair salon faces repeated contact, and the risk multiplies. The Occupational Safety and Health Administration (OSHA) encourages gloves, good ventilation, and regular cleaning of work surfaces. This advice doesn’t just apply to PPD; it covers related chemicals, too.
Paths Forward: Staying Informed and Cautious
Nobody wants to play guinea pig, especially with something as personal as hair or skin. Patch testing remains a simple, practical step. If reactions show up, it’s time to avoid the product and tell a doctor. Reading ingredient lists carefully, choosing alternatives if sensitive, and talking with knowledgeable pharmacists or healthcare professionals can make a difference.
Brands and manufacturers have a duty to stay transparent and invest in more safety studies. With data guiding decisions, people can trust the products they buy. Until science gives a clearer answer, using N-Acetyl-P-Phenylenediamine means balancing personal comfort with the latest research and advice from qualified experts.
What are the storage conditions for N-Acetyl-P-Phenylenediamine?
Looking at What N-Acetyl-P-Phenylenediamine Needs
N-Acetyl-P-Phenylenediamine is no stranger to folks in labs or factories dealing with dyes, pigments, and chemical analysis. Like a lot of small organic molecules, it brings both promise and risk. Letting it sit on a shelf, or placing it somewhere warm, can turn it unreliable. Heat, light, and moisture turn this compound into a different beast—a health risk and a scientific headache. Years of working near chemical storerooms tell me a bottle only gives as good as the care it gets.
Why Environmental Control Matters
You don’t need high-tech gadgets to treat this chemical with respect. In practice, the most common problem comes from sunlight and fluctuating temperatures. Bright light breaks down the molecule, which ruins both purity and consistency. I’ve seen expired samples go brown and start caking up, making a solid mess for everyone involved. Dry conditions keep it steady, and avoiding humidity stops chunks from forming, which saves time and resources. In fact, the United States Pharmacopeia points out that moisture encourages decomposition and contamination, so dry storage prevents wasted product and failed experiments.
Sensible Storage Strategies
Any chemist will tell you the golden rule: store it in a tightly sealed container, away from sunlight, away from moisture, and at stable room temperature. This doesn’t mean a high-end freezer, but a storage cabinet away from heaters and windows does the trick. Try to keep humidity below 60%. If you live somewhere humid, desiccants inside the storage box help a lot. No magic required—just keep the lid closed and don’t let the bottle sit open on the bench after use.
Health and Safety Take Priority
Direct contact with N-Acetyl-P-Phenylenediamine can cause allergic reactions or more serious health issues. My early mistakes working in shared chemistry spaces taught me that even small spills, especially if unnoticed, stick around. That’s one reason safety data sheets tell you not just to store well, but to keep everything labeled, and always use gloves. Always treat every bottle like it matters for the next person’s safety too.
Avoiding Accidents and Wasting Resources
Poor storage isn’t just a laboratory issue; it also wastes money. Degraded chemicals throw off test results and set back production timelines for dyes and related products. I’ve seen whole batches of product tossed out because one ingredient, forgotten on a shelf in the sun, spoiled the lot. Inventory control keeps track of dates, but even the oldest chemical holds up if you keep light, air, and water out.
Making Improvement Easy for Everyone
It helps to build habits everyone on the team follows, like checking containers for cracks every few months and rotating older stock forward. A checklist on the door of the chemical cupboard, and easy access to updated safety sheets, works wonders. Showing new folks what happens to poorly kept N-Acetyl-P-Phenylenediamine means fewer replacements and safer labs.
Responsible Storage Practices Build Trust
Good storage isn’t rocket science, but it’s all about respect for the process, the material, and your coworkers. If you treat N-Acetyl-P-Phenylenediamine right, it won’t let you down. That’s been true for every lab, big or small, that I’ve ever worked in.
How should N-Acetyl-P-Phenylenediamine be handled and disposed of?
What N-Acetyl-P-Phenylenediamine Is—and Why Handling Matters
N-Acetyl-P-Phenylenediamine sounds technical, but you can find it in places that surprise you. Laboratories test it for pharmaceuticals and chemical processes. Most folks won’t recognize the name, but the dangers are real for people who work with it. It’s sensitive to air, moisture, and heat and can cause issues to eyes, skin, and lungs. Mishandling leaves workers at risk and creates headaches for the next shift. I’ve seen what happens when a lab takes shortcuts – it hurts morale, costs money, and threatens safety. Respect is the right word here, not fear.
Personal Protective Equipment Isn’t Optional
One thing I always tell new lab members: PPE only seems inconvenient when nothing goes wrong. Gloves, safety goggles, and lab coats are standard for a reason. Accidental skin contact burns and inhalation risks get minimized with proper gear. Nobody wants to scrub down a spill in shorts and a t-shirt. Proper ventilation changes the game, not just for preventing fumes, but also for keeping contamination out of common areas. Running a fume hood is always better than wishing for one later.
Chemical Storage Prevents Small Problems From Becoming Big Ones
Steady storage keeps this chemical away from heat and moisture. I’ve seen careless storage decisions lead to ruined batches and expensive investigations. Sealed containers, well-labeled, go on shelves away from direct sunlight. Stacking things to save space can end in disaster if something leaks. Spill kits and accessible data sheets mean teams act quickly and know what to expect. Training shouldn’t get skipped – everyone on duty ought to run through response steps, at least twice a year.
Disposal: No Shortcuts and No Sink Drains
Pouring leftover chemicals down the drain can poison water, corrode pipes, and end up with hefty fines. No one wants to read their own lab cited in a news report. Collection in proper containers, labeled for hazardous waste, sets the standard. In my experience, phone calls to waste management can save hours of confusion. Municipal guidelines usually require professional removal; it costs time and money, but the alternative is far worse. Separate the compound from ordinary trash. Double check the schedule for waste pickup and keep clear records. Good habits save more than just face – they protect everyone in the chain of disposal.
What’s at Stake?
Safe chemical practices keep workers healthy, sites operating, and communities free from extra pollution. I’ve seen how training and respect for rules make teams confident—and accidents rare. It’s about watching out for neighbors, colleagues, and the environment, even if it means taking a few extra minutes. By following protocols, you prevent environmental contamination and stay on the right side of the law. Safe handlers treat these compounds not as routine but as serious business. Responsible choices pay off over time, in safety and trust.
What is the chemical structure of N-Acetyl-P-Phenylenediamine?
Breaking Down the Chemical Structure
N-Acetyl-P-Phenylenediamine, known in chemistry circles as 4-acetylaminophenylenediamine, features a benzene ring at its core. This ring carries two amino groups (-NH2) at positions 1 and 4, often called the para positions. An acetyl group (-COCH3) hangs on one of the amino groups, turning it into an acetamide. The full chemical formula: C8H10N2O. The acetyl group transforms the nature of this molecule, giving it new traits compared to plain p-phenylenediamine.
Structure and How It Connects to Use
In practical terms, you find subtle changes from a quick tweak in structure, especially in the world of dyes and colorants. That acetyl group acts like a shield, moderating the tendency of the free amine group to react. Why does this matter? Reactive chemicals often trigger allergic responses and skin sensitivities. Acetylation softens the blow, making this molecule easier to handle and less likely to spark irritation for people who use hair colorants or textiles.
In the lab, the look of this molecule lines up with its behavior. The amino groups at the ends of the ring allow for hydrogen bonding, which affects how easily the compound can dissolve in water or bond with other chemical partners. The acetyl group, with its carbonyl and methyl piece, changes that affinity a bit, away from full-on polarity but not into the greasy, non-polar world. This kind of balance guides people working on product formulations to choose N-Acetyl-P-Phenylenediamine carefully.
The Value in Day-to-Day Products
In the salon, you wouldn’t see this name on a bottle. Still, anyone mixing permanent hair color is likely crossing paths with this compound’s relatives. Regular p-phenylenediamine made its reputation for strong coloration but also caused allergic contact dermatitis in a share of users. N-Acetyl-P-Phenylenediamine serves up less risk because that acetyl group tamps down the molecule’s reactivity, calming allergic reactions in many cases. Scientists ran patch tests and found lower rates of sensitivity—still present, but meaningfully reduced. Safety for both users and workers in dye manufacturing matters, and choosing safer molecules makes a difference in real outcomes.
Environmental and Health Questions
Workplaces and the environment can’t be separated from good chemistry. Compounds related to phenylenediamine linger in wastewater and raise toxicology red flags for fish or amphibians. Substituting or modifying these compounds—making them less reactive, less likely to break down into harsher chemicals—lowers environmental risks. There’s a social push to keep chemistries as safe as possible without trading performance. I remember vigilant coworkers in labs pushing every supplier for ingredient breakdowns; the demand for safety is not just a compliance box anymore.
People still want bright, lasting results in dyes, but they also expect less risk. N-Acetyl-P-Phenylenediamine points to the middle ground: pickier chemical tailoring that balances performance with real-world safety. The acetyl group is a tool in the chemist’s kit—small by atomic count, influential by impact—proof that the structure of a molecule shapes both human health and industrial possibility.
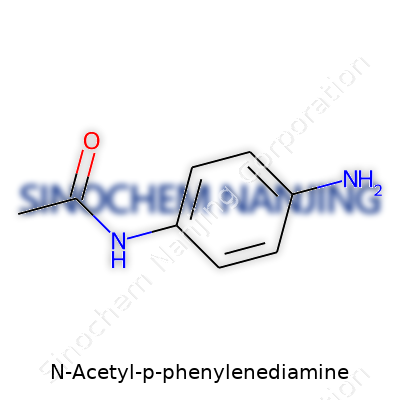
| Names | |
| Preferred IUPAC name | N-(4-aminophenyl)acetamide |
| Other names |
N-Acetyl-1,4-phenylenediamine 4-Acetamidophenylenediamine N-Acetyl-pphenylenediamine |
| Pronunciation | /ˌɛn əˈsiːtəl pi ˌfiːnɪliˈndaɪəmiːn/ |
| Identifiers | |
| CAS Number | 101-01-9 |
| 3D model (JSmol) | `3D model (JSmol)` string for **N-Acetyl-p-phenylenediamine** (also known as N-acetyl-1,4-phenylenediamine) is the **SMILES** string: ``` CC(=O)Nc1ccc(N)cc1 ``` This string can be used in JSmol or other molecular visualization tools. |
| Beilstein Reference | 822930 |
| ChEBI | CHEBI:28218 |
| ChEMBL | CHEMBL1386 |
| ChemSpider | 5403 |
| DrugBank | DB14606 |
| ECHA InfoCard | 03ba2b0c-4be5-4c8d-a2e9-6e2c46d8cee3 |
| EC Number | 1.14.13.7 |
| Gmelin Reference | 82466 |
| KEGG | C02341 |
| MeSH | D010312 |
| PubChem CID | 8469 |
| RTECS number | SG1575000 |
| UNII | BG9L1Y7LL5 |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C8H10N2O |
| Molar mass | 164.20 g/mol |
| Appearance | White to light brown crystalline powder |
| Odor | Odorless |
| Density | 1.2 g/cm³ |
| Solubility in water | Slightly soluble in water |
| log P | 0.08 |
| Vapor pressure | <0.01 mmHg (25°C) |
| Acidity (pKa) | 5.5 |
| Basicity (pKb) | 11.08 |
| Magnetic susceptibility (χ) | -72.0e-6 cm³/mol |
| Refractive index (nD) | 1.618 |
| Viscosity | 18.64 cP |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -49.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2762 kJ/mol |
| Pharmacology | |
| ATC code | D02BB01 |
| Hazards | |
| Main hazards | Harmful if swallowed, inhaled, or absorbed through skin; may cause allergic skin reactions; causes eye, skin, and respiratory tract irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H315, H317, H319 |
| Precautionary statements | Precautionary statements: P264, P270, P273, P280, P301+P312, P302+P352, P305+P351+P338, P310, P330, P337+P313, P362+P364 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 104°C |
| Autoignition temperature | Autoignition temperature: 482°C (900°F) |
| Lethal dose or concentration | LD50 oral rat 237 mg/kg |
| LD50 (median dose) | LD50 (median dose): **1790 mg/kg (rat, oral)** |
| NIOSH | ST0185000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.1 mg/m3 |
| Related compounds | |
| Related compounds |
p-Phenylenediamine Acetanilide 4-Nitroacetanilide N,N-Dimethyl-p-phenylenediamine Benzidine |

