N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide: Scrutinizing a Chemical’s Role, Risks, and Future
The Chemical’s Trail: History and Origins
N-(2-Hydroxyethyl)-N-methylperfluorooctanesulfonamide has roots in the golden age of fluorine chemistry, with development ramping up in the latter half of the 20th century. Large manufacturers spotted the unique value of perfluorinated compounds in applications demanding resilience, water resistance, and chemical stability. Early research into sulfonamides pointed to their ability to impart those same properties to textiles, paper products, and firefighting foams. As public consciousness about environmental safety lagged behind industrial progress, this compound entered widespread use before people questioned persistence or toxicity. Now, the historical momentum surrounding legacy perfluorinated substances gives way to sharp scrutiny, even as these materials remain difficult to replace because competitors struggle to match their exact blend of performance and endurance.
Physical and Chemical Identity
Looking at N-(2-Hydroxyethyl)-N-methylperfluorooctanesulfonamide up close, the long perfluorinated tail grants it powerful repellency to both oil and water, helping treatments cling to surfaces and resist stains. It comes to the user most often as a clear, viscous liquid, with a mild, sometimes odd odor due to its sulfonamide structure. Its molecular structure resists heat, acids, and bases, giving it a stubbornness few other compounds can claim. A compound this robust heads into industrial settings that demand absolute reliability, yet the same properties that keep it from breaking down in a rainstorm or washing machine also stop environmental breakdown—once out in the world, nature doesn’t break this molecule’s backbone easily.
Technical Specifications and Labeling
Industry labels tend to reference both the IUPAC and the common name, but the mouthful never really tells outsiders much about the implications of handling this substance. Most chemists recognize the perfluorooctyl part, which hints at both its lasting power and its concern for environmental health. The technical community has long measured purity levels, moisture content, and potential cross-contamination with related perfluorinated substances, since a batch with impurities can create regulatory headaches or introduce unpredictable hazards in downstream uses. Labeling for shipments often includes warnings about the need for gloves, eye protection, and ventilation, reflecting recognition that casual handling isn’t wise.
Production and Synthesis: Crafting a Stubborn Molecule
Setting up a synthesis line for this compound asks for specialty reactors and careful temperature control. Perfluorooctanesulfonyl fluoride, a key building block, reacts with methylamine and ethanolamine in a series of stages, producing the sulfonamide that’s so prized in manufacturing circles. Operators must monitor conditions closely, since side reactions can generate unwanted byproducts or result in unreacted starting materials that complicate separation and disposal. The manufacturing challenge isn’t just technical—runoff or emissions containing even small amounts of perfluorinated substances can persist in waterways and soil, reminding us that great chemicals for industry aren’t without environmental baggage.
Chemical Reactions and Modification Potential
From a chemist’s perspective, this molecule stands out for how little it wants to react—a trait that attracted so many industries but now sparks concern among environmental scientists. The perfluorinated chain defies most forms of degradation, including exposure to strong acids, bases, or high temperatures. Some researchers have tried oxidation or advanced photolysis methods to break it down, but none seem ready to scale or cost competitive for real-world scenarios. Chemical modification usually targets the sulfonamide group, which can attach to larger polymers or enable compatibility with other additives during product formulation. The flipside is a molecule so sturdy that once released, it clings to the environment, and only the most aggressive laboratory methods make a dent in its structure.
Synonyms and Industry Naming Conventions
N-(2-Hydroxyethyl)-N-methylperfluorooctanesulfonamide pops up under a variety of labels. Chemical trade often abbreviates lengthy names, calling it by shorter versions, or simply referencing it by its function—surfactant, repellent additive, or precursor to certain fluorinated polymers. Any scientist or regulator stepping into the literature must wrestle with these variations, which can complicate tracing usage patterns across regions or decades. The result is a web of regulatory loopholes, as enforcement agencies struggle to pin down exactly what’s moving through supply chains and where it actually ends up.
Safety and Operational Rules
Folks working with this compound know personal protective gear isn’t optional. Direct contact can irritate skin, and inhalation of mist or vapor challenges respiratory health. Regulatory focus has shifted from acute hazards to chronic, with growing evidence of perfluorinated substances building up in blood and tissues over time. While a product label might suggest routine care—ventilation, gloves, goggles—the real risk comes not from one mishap on the job but from low-level, long-term exposure in manufacturing neighborhoods or the wider community as the compound escapes into water or dust. Legislation keeps tightening, but workplace safety often lags behind new findings from epidemiology and wildlife studies.
Where It’s Used: Applications Past and Present
This sulfonamide once powered a silent revolution in stain repellency. Textiles, carpets, and paper relied on it to keep water, oil, and dirt from soaking in. Firefighting foams used its wetting properties to beat down blazes faster, while industrial cleaners banked on its stability. Coatings on cookware and electronics kept grime at bay using related chemistries. Over years of use, environmental watchdogs sounded an alarm: what makes it cling to fabrics also lets it slip into soil and groundwater, then move up the food chain. A handful of alternatives now attempt to take its place, but performance lags behind, and the switch to something safer asks for both technical ingenuity and regulatory resolve. Observing this shift, it’s clear product designers must weigh long-term impacts over short-term benefit, especially with persistent chemicals.
Research and Development: Pushing for Answers
Scientists enter this field split. On one side, industry chemists refine production and hunt for tweaks that keep performance high but limit environmental impact. On the other, toxicologists and environmental scientists draw clearer links between exposure and health risks, using blood serum surveys, animal models, and wider ecological assessments. Despite heavy investment, emerging “alternatives” don’t always deliver a clean slate. Many new compounds stay under patent umbrellas or proprietary secrecy, raising concerns about repeating mistakes of the past by simply swapping one persistent, mysterious molecule for another. The pressure is on for clear, public data—and longtime observers see collaboration as the only path forward, with industry, regulators, and communities sharing research and monitoring results.
Toxicity and Health Hazards: The Long Shadow
A growing pile of peer-reviewed studies links perfluorinated substances, including N-(2-hydroxyethyl)-N-methylperfluorooctanesulfonamide and its close relatives, to real and lasting health concerns. Animal experiments reveal bioaccumulation that doesn’t quit, with effects on liver, immune function, and development. Human studies are trickier, with results pointing to increased cholesterol, altered immune response, and potential cancer risks, but confirmation takes more time and data. Personal experience working with industrial production teams showed deep worry when groundwater tests near old manufacturing sites kept finding perfluorinated residues years after production stopped. This compound’s refusal to break down means communities pay the price long after the makers close up shop—a sobering reminder that chemical durability stretches the definition of safety.
Future Prospects and Solutions
Calls for stewardship ring louder with each new study. New frameworks demand producers take lifelong responsibility for the molecules they release, from manufacture to disposal. Innovations in green chemistry push for shorter carbon chains or even non-fluorinated alternatives, though few match old benchmarks for stain resistance or chemical inertness. Environmental monitoring networks expand, with real-time tracking tools now deployed around wastewater outlets and landfills. In personal conversations with environmental engineers and policy experts, frustration bubbles up—while solutions develop in labs, regulators and communities must stay vigilant, refusing easy endorsements for “next-generation” repellents without evidence of safety. The path forward requires patience, steady investment, and honest communication about the limits of current technology and the shared cost of persistence. Localized cleanup methods, community engagement, and transparency in ingredient disclosures mark the way toward responsible use, even as true substitutes continue to lag. The future likely belongs to safer, simpler chemistry—but society must not look away from the legacy left by robust, persistent molecules like N-(2-hydroxyethyl)-N-methylperfluorooctanesulfonamide.
What are the primary applications of N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide?
Understanding the Role in Manufacturing
Factories have relied on N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide, often known as a fluorinated surfactant, for decades. It delivers a unique surface tension reduction, which makes it attractive for products that need to repel water, resist oil stains, and stand up to extreme environments. In textile production, for example, this compound finds its way into water-repellent outerwear. The classic raincoat or treated hiking gear owes its bead-up effect to compounds like this one. You spill coffee or mud, it just glides off.
Firefighting and Electronics
Firefighters have fought tough blazes with aqueous film-forming foams, and many of these foams incorporate N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide. The stuff creates a thin film that cuts off oxygen, knocking down fuel fires fast. Decades of demonstration have shown its effectiveness during emergencies at airports and oil depots—places where ordinary water just won’t cut it. Electronics producers also turn to this compound for precision etching and cleaning. Circuit boards can’t handle contamination, and this surfactant cleans without lacing residues behind or corroding delicate parts.
Paper and Packaging
Paper manufacturers have included this ingredient in coatings that keep pizza boxes and fast-food wrappers from soaking through. The fast-food industry bet on quick service and clean hands, and the compound’s barrier properties helped make that a reliable bet. The grease doesn’t seep, and stains don’t bleed through. It’s something a lot of people never see, but appreciate in action—think about how often you’ve grabbed a sandwich on the run.
Wider Effects and Growing Scrutiny
Despite its undeniable function across markets, concerns have grown. Perfluorinated substances, which include this compound, linger in the environment for generations. Studies from agencies like the U.S. Environmental Protection Agency and peer-reviewed journals describe a buildup in water, soil, and even livestock. Farmers near industrial sites talk about contaminated wells, and biologists keep finding these chemicals in wild fish.
Communities have called for tighter limits. Many countries now restrict some uses of perfluorinated substances, and big brands started searching for alternative solutions. Replacing something with proven performance hasn’t been easy. Some companies lean toward less persistent chemicals, while others research plant-based barriers or other types of surfactants. Each swap triggers new questions: Will these new options last as long? Do they come with surprising health risks? My own background in environmental advocacy reminds me that innovation always bumps up against the law of unintended consequences. That’s why I see real value in thorough safety testing before any new chemistry rolls out at scale.
Balancing Practicality with Responsibility
No one likes rain-soaked jackets or kitchen oil fires, but cleaner, smarter chemistry must run the show now. Government policy has started nudging manufacturers in that direction—public pressure isn’t going to let up. Watching this transition, I keep hoping for real talk between industry, public health experts, regulators, and the communities living downstream. We all use these products in some way, even if we rarely see the science behind them. Using safer alternatives could open up new paths for those of us who care just as much about what ends up in our rivers as what keeps our takeout tidy.
Is N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide hazardous to health and the environment?
What is N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide?
You might not see its full name printed at the grocery store, but N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide (HFPOS or FOSA) turns up in several products. This chemical belongs to the PFAS family, a group known for resisting grease, water, and stains. Scientists engineered these molecules for their staying power—not just in products, but unfortunately, in our world.
Persistence and Bioaccumulation
The strength of HFPOS lies in its carbon-fluorine bonds. Once it leaks or spills, it doesn’t just go away—rain, sunlight, and bacteria barely touch it. I’ve followed studies from Minnesota to Japan showing PFAS compounds sticking around in rivers, soils, and even fish. The build-up doesn’t stop there: many researchers report these chemicals gradually climbing up the food chain, collecting in predators and, finally, in people.
Potential Health Problems
Scientists have measured HFPOS and similar PFASs in blood samples from adults and children across the world. No one can ignore that. Research from Harvard, the CDC, and international agencies draws a connecting line between these persistent chemicals and possible health risks—lower birth weights, thyroid issues, cholesterol disruption, and certain cancers. Parents want answers. Communities living near airports and manufacturing sites report higher levels in their drinking water, causing anxiety and legitimate concern for future generations.
Impact on Wildlife and Water
Water tests pick up PFASs in everything from city rivers to mountain lakes. Fish exposed to these substances end up with them in their tissues, passing the exposure up the food web. I’ve read firsthand accounts from biologists who track PFAS levels in eagles, otters, and even polar bears, surprised by how far these chemicals travel. Animals, just like people, struggle with immune disruption and reproductive effects from long-term exposure.
Tackling the PFAS Problem
Communities and policymakers want to cut off contamination at the source. I’ve spoken to neighbors installing whole-house carbon filters after learning PFAS levels in tap water exceeded the EPA’s new advisory limit. In Denmark, entire countries have forced chemical makers to phase out long-chain PFAS in food packaging and industrial processes. Municipal water agencies scramble for better cleanup technology. Laboratories keep trying new filtration methods—activated carbon, ion exchange, and high-tech membranes—to tackle what some folks call “forever chemicals.”
It’s no longer enough for companies to call these compounds safe just because they stand up to heat or grease. Consumers and regulators are demanding transparency about where PFASs show up and whether safer options exist. Honest labeling and independent safety testing can push industries toward less hazardous replacements. More than just talk, putting research dollars into finding—and safely using—alternatives remains key.
Living with Legacy Chemicals
FOSA and similar PFASs highlight a dilemma: solutions are complicated, but inaction isn’t an option. Every town with a contaminated well, every scientist tracking PFAS in polar snow, and every parent searching for safe baby gear shows that health and environmental safety matter on a daily level. Real change calls for better oversight, smarter choices in manufacturing, and a shared willingness to put health over convenience.
What are the recommended safety precautions when handling this product?
Why Safety Precautions Matter
Eyeballing a product label loaded with warnings doesn’t always prompt an immediate sense of risk. In reality, small missteps can leave a lasting impact. Chemicals, industrial agents, and even household supplies all demand respect — one careless move can cause burns, breathing trouble, or worse. Growing up around a garage full of solvents and automotive fluids drove that point home for me the hard way, after a simple cleaner splashed onto my skin. I’ve learned to value the hard lessons experience brings a lot more than fancy authority statements.
Reading and Understanding Labels
Product labels serve as the first line of information. Most lay out key dangers with symbols: a skull for toxicity, a flame for flammability, and so forth. These aren’t just for show. Even if the symbol looks dramatic, they give a clear message, like “Keep away from sparks” or “Open windows before use.” The label isn’t just for legal coverage — it’s often the only thing between you and an emergency room visit.
The Role of Personal Protective Equipment
Gloves and goggles change the game. Chemical-resistant gloves help keep caustic or irritant substances away from your skin. Splashing household bleach into an open cut can teach almost anyone to suit up. Eye protection keeps sprays and vapors away from one of the most sensitive areas of the body. A dust mask or true respirator is necessary wherever fumes escape. I’ve stood in rooms thick with paint vapors and felt dizzy — now, a mask is as important as the brush in my hand.
Safe Handling Techniques
Pour and mix only in well-ventilated places. Fumes can creep up slowly, then suddenly knock you off your feet. Work outside, or near an open window with a fan aimed out to keep the air moving. Mixing chemicals creates even bigger risks. Some combinations generate dangerous gases in seconds. For example, bleach and ammonia together produce toxic chloramine gases. Even with common cleaners, keep them separate unless the label says otherwise.
Keep your hands away from your face when working. I habitually wiped sweat from my brow before, but after a stinging reminder from an adhesive, I learned to keep a wiping cloth and wash hands before touching skin or eating.
Storing and Disposing Safely
You can’t just toss chemical-laden products under the sink. Keep them tightly sealed in a cool, dry place, away from children and pets. Some substances react dangerously if exposed to heat or sunlight. Used or leftover material needs careful disposal. Pouring old paint thinner down the drain damages pipes and contaminates groundwater. Most towns host hazardous waste drop-off events — a little effort goes a long way for the wider community.
Dealing With Accidents
Accidents still happen, even after taking every step. Immediate action can cut down the damage. Rinse skin or eyes with lots of water fast, then seek medical help if burning or redness persists. If vapors make you cough or feel faint, get outside right away. Keep emergency contacts posted and know where to find nearby eyewash stations if working in a workshop.
Building Good Habits Keeps Everyone Safer
Safety is not just about ticking boxes — it grows from habits and a little respect for the dangers hiding in everyday jobs. A splash of careful planning and sturdy equipment keeps hands, eyes, and lungs intact. Familiarizing yourself with common risks and acting on them every single time means you’re not just following the rules, you’re setting yourself up to avoid the mistakes that catch others off guard.
How should N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide be stored and transported?
Understanding the Risks
Working around chemicals like N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide, usually known as N-EtFOSE, means you have to keep your head in the game. Folks who handle these substances start to notice patterns, especially after reading about contamination cases in the news or coming across agency warnings. This compound shows up in a lot of industrial and commercial products, but the health effects linked to PFAS chemicals are real. They have the sort of persistence that worries experts, leaving traces in the water, in the soil, and sometimes in people.
What Storage Looks Like in Practice
Locking up N-EtFOSE begins with a close look at the chemical’s label and the safety data sheet. Some industry veterans keep these right on the wall. This approach beats relying only on memory. The storage area needs a solid, well-ventilated space where leaks drain toward sealed catchments—not into floor grates or stormwater. Metal shelving with spill trays, clear labeling on every container, and a written log keep mistakes to a minimum. No one wants a shelf collapse or cross-contamination, especially after hours when fewer people are around.
Temperature matters. The compound does not handle extreme heat well, so a cool, dry room fits the requirement. Some folks use climate-controlled cabinets with a lock, mainly because PFAS-class chemicals can break down and release fumes over time. These fumes do more than just stink; a single mistake can trip a facility's alarms or, worse, harm a worker. Placing N-EtFOSE away from acids, oxidizers, and anything flammable helps cut back on fire and reactivity issues, according to guidelines from OSHA and the EPA.
Real-Life Hazards on the Road
Moving chemicals always makes people uneasy, even with regulations. The Department of Transportation posts regular updates about what needs placards, which carriers have violations, and where spills have polluted the land. After seeing tankers tip over or packaging fail, it becomes clear that packaging—real, sturdy UN-rated containers—matters as much as the label. Drivers who go through training and carry spill kits in the cab make a big difference. They know not to park near storm drains or leave containers in the sun.
Transport rules say to keep chemicals upright, secured so nothing slides if a truck takes a sharp corner. Drivers avoid rough roads when they can, because vibrations can split seams or crack caps. Companies who care about safety run double checks at the loading dock: check paperwork, check seals, check compatibility. Each step feels routine until the one time someone skips it—and a problem follows. Gauges on tanks and real-time tracking on trucks give supervisors peace of mind. They can spot issues mid-route and step in before a minor incident turns serious.
The Path Forward
Chemical safety feels like chasing a moving target. Engineers and safety managers keep pushing for new best practices because science keeps showing us how persistent and risky PFAS chemicals can be. Facilities now invest in corrosion-resistant containers and databases tracking quantities in real time. Strong vent fans and regular air quality checks are part of the routine. Through skill-building and good policy, workers can do their jobs without putting the environment or community in harm’s way. These options, from better labeling to better packaging, give everyone more control and peace of mind.
Are there any regulatory restrictions on using or importing N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide?
The Backstory Behind This Chemical
N-(2-Hydroxyethyl)-N-Methylperfluorooctanesulfonamide, sometimes called by the acronym N-MeFOSE, plays a niche role in industrial chemistry. It shows up in specialty coatings, firefighting foams, and textile treatments. This compound links directly to the family of per- and polyfluoroalkyl substances—PFAS for short. These substances grab headlines for sticking persistently in both the environment and our bodies. Anyone watching public health news will recognize the increased attention regulators have given to such chemicals due to emerging health studies.
Why Regulatory Scrutiny Hits Hard
Looking globally, several regions clamp down on PFAS, and N-MeFOSE rarely escapes that attention. The United States Environmental Protection Agency (EPA) and the European Chemicals Agency (ECHA) both track and restrict these substances. Some perfluoroalkyls have even ended up on the Stockholm Convention's list of banned persistent organic pollutants. Canada’s Prohibition of Certain Toxic Substances Regulations includes perfluorooctane sulfonate (PFOS) and its derivatives, with N-MeFOSE directly referenced as a restricted substance for most uses.
Researchers have linked PFAS like N-MeFOSE to liver enzyme changes, increased cholesterol, and certain cancers in animal studies. These findings put them under the microscope. The real kicker? These chemicals don’t easily break down. They leach into soil and water, moving through living things up the food chain. Even factories that stick to the rules can struggle to keep these molecules out of nearby streams and drinking water sources. That persistence means tighter import and use rules often come down the pipeline after new data lands.
What Restrictions Look Like in Practice
In the United States, importers meet a tangled web of reporting requirements. Anyone shipping this compound must keep up with the EPA’s Toxic Substances Control Act (TSCA) regulations, which have grown in scope for PFAS in recent years. TSCA’s Significant New Use Rule covers a wide range of PFAS, and importers must file paperwork before bringing them in for new types of applications. Violations can mean steep penalties—think serious fines or even criminal charges for willful non-compliance.
The European Union approaches the issue through the Registration, Evaluation, Authorisation and Restriction of Chemicals (REACH) regulation. Manufacturers or importers face strict notification and testing demands. Using such substances without proper authorization can shut down operations or spark costly recalls. Many companies voluntarily drop these ingredients, shifting to alternatives, to avoid regulatory heat and manage future risks.
Finding Healthier and Safer Paths Forward
Industry groups and consumer advocates both push for safer substitutes. As a parent, I feel a certain relief knowing companies scramble to cut PFAS from common household items. The evidence keeps piling up—some clouds aren’t worth the silver lining they bring. Green chemistry approaches open doors for functionally similar fire retardants and stain barriers, without the environmental baggage. Investing in pollution control upgrades also cuts accidental leaks and later cleanup costs.
Public databases, like the EPA’s PFAS Action Plan and Europe’s Substances of Very High Concern (SVHC) list, simplify the job for researchers, importers, and regulators. They set a clear bar for health and safety. At-home testing and community pushback add even more pressure. Shoppers increasingly demand full disclosure about chemical footprints. In my neighborhood, parents trade information on safe brands for rain gear and kitchenware, no longer just trusting labels. That grassroots energy spurs companies to act faster than laws alone ever could. Tracking and sharing real-world outcomes for substitute chemicals gives everyone more solid ground to stand on. No one wants to trade one risky shortcut for another down the line.
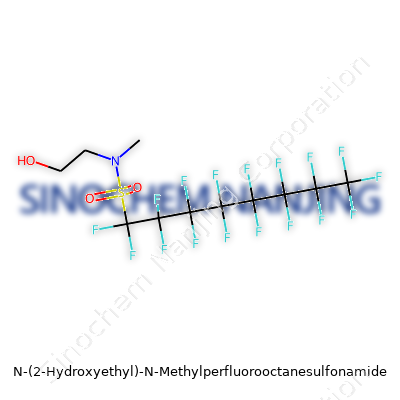
| Names | |
| Preferred IUPAC name | N-methyl-N-(2-hydroxyethyl)perfluorooctane-1-sulfonamide |
| Other names |
N-Ethyl-N-methylperfluorooctanesulfonamide NMEA MeFOSE N-Methyl perfluorooctane sulfonamidoethanol |
| Pronunciation | /ɛn tuː haɪˌdrɒk.siˌɛθ.ɪl ɛn ˈmɛθ.ɪl pɜːrˌfluː.rəʊˌɒk.teɪnˌsʌlˈfɒn.ə.maɪd/ |
| Identifiers | |
| CAS Number | **2991-51-7** |
| Beilstein Reference | 6831464 |
| ChEBI | CHEBI:88205 |
| ChEMBL | CHEMBL1802823 |
| ChemSpider | 406694 |
| DrugBank | DB02939 |
| ECHA InfoCard | 03ab725d-1bd1-4eeb-81bb-4e51ab833ff0 |
| Gmelin Reference | 1310557 |
| KEGG | C18096 |
| MeSH | D020127 |
| PubChem CID | 2735004 |
| RTECS number | KT1575000 |
| UNII | K7O847912G |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID3047352 |
| Properties | |
| Chemical formula | C11H20F17NO3S |
| Molar mass | 549.248 g/mol |
| Appearance | Colorless to yellowish liquid |
| Odor | Odorless |
| Density | 1.7 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 3.68 |
| Vapor pressure | <0.01 mmHg (20 °C) |
| Acidity (pKa) | 8.0 |
| Basicity (pKb) | 5.3 |
| Magnetic susceptibility (χ) | -1.92×10⁻⁶ |
| Refractive index (nD) | 1.355 |
| Viscosity | 66 cP (25°C) |
| Dipole moment | 2.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 527.6 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1554.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -7211 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | S01XA18 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause damage to organs through prolonged or repeated exposure. |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS08 |
| Signal word | Warning |
| Hazard statements | H302-Harmful if swallowed. H315-Causes skin irritation. H319-Causes serious eye irritation. H332-Harmful if inhaled. |
| Precautionary statements | P261, P273, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | > 129°C (264°F) |
| Autoignition temperature | 275 °C |
| Lethal dose or concentration | LD50 Oral - rat - > 2,000 mg/kg |
| LD50 (median dose) | LD50 (rat): > 5,000 mg/kg |
| NIOSH | RN1051 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.005 mg/m³ |
| Related compounds | |
| Related compounds |
Perfluorooctanesulfonamide N-Methylperfluorooctanesulfonamide N-Ethylperfluorooctanesulfonamide N-(2-Hydroxyethyl)perfluorooctanesulfonamide Perfluorooctanesulfonic acid |

