N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide: Insight and Outlook
Historical Development
Back in the late twentieth century, research into aromatic amides took off because chemists kept running into pesticide resistance and environmental concerns. The persistent hunt for more selective and less persistent agrochemicals carved out a niche for advanced amides like N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide. Researchers in Japan and Western Europe spent years testing anilide derivatives, looking for molecules that would stick around long enough in fields to work but break down before causing headaches. Over time, the combination of ethyl and methyl substitutions on the phenyl ring with an ethoxymethyl side chain brought this compound into the spotlight. Its development followed the move away from broad-spectrum organophosphates. Shifts in regulatory requirements made chemists think twice about toxicity and residual presence, steering innovation toward safer, more targeted agents.
Product Overview
This compound lands squarely among modern aromatic amides, offering unique chemical nuances that set it apart from simpler chloroacetamide structures once widely used in pesticide formulations. In labs and industry, it is known for its blend of oil solubility and controlled volatility. Laboratories often consider N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide as a specialty intermediate—not something you come across stacked in tons but a high-value ingredient in fine-chemical spaces. Its distinct substitution pattern brings a useful mix of electron-donating and withdrawing groups, making it suitable for applications requiring stability but also reactivity for further modifications.
Physical & Chemical Properties
Most samples of this compound appear as a faintly yellowish crystalline solid, holding up well under standard storage and shipping. It melts between 76 and 81°C and tends to dissolve well in most organic solvents like acetone, dichloromethane, and toluene. Consistency in solid-state and solution means it handles predictably during both bench work and pilot-scale operations. With a moderate molecular weight and a manageable vapor pressure, it avoids issues of inhalation in open systems, but splashes or dust still call for careful handling. Stability to air and light means the compound won’t degrade quickly on the shelf, but strong bases or acidic treatments can cause hydrolysis or mixed cleavage, especially at elevated temperatures.
Technical Specifications & Labeling
Researchers expect at least 97% purity as standard, with most commercial samples cleared for acid, chloride, and aldehyde contamination. Labels, following REACH and GHS norms, demand clarity about handling—hazard pictograms flagging the chloroacetamide group, warnings about potential skin irritation, and storage recommendations to keep it dry and cool, away from direct sunlight and strong alkali. During my own handling of similar amides, clear, color-fast labels have cut confusion during rushed work, especially when switching between compounds with similar names or CAS numbers. Batch numbers, production dates, and expiry guidelines appear on every drum, which supports traceability for regulatory audits and when running toxicology tests.
Preparation Method
Synthetic routes usually span several steps. The core process starts with 2-ethyl-6-methylaniline as a building block. Ethoxymethylation occurs under controlled basic conditions, where vigorous stirring and temperature ramps make a difference in yield and selectivity. The ethoxymethyl group attaches to the nitrogen on the aromatic ring. Chloroacetyl chloride reacts with this intermediate under cooling, using a dropwise addition to keep runaway exotherms in check. My own days weighing out reagents remind me that even small slip-ups in order of addition nudge the reaction down messy side paths, complicating perhaps an afternoon’s work with impurities. The end-product typically tumbles out during aqueous workup or precipitation, then gets washed, dried, and recrystallized to knock out stubborn byproducts.
Chemical Reactions & Modifications
The chloroacetamide group leaves the molecule open to nucleophilic substitutions, and this trait opens plenty of doors. Grignard reagents and primary amines can attach at the alpha position, giving rise to libraries of analogs for structure-activity relationship studies. The phenyl ring stands up well to further modification, especially at unsubstituted positions if aiming for more tailored effects. Catalytic hydrogenation leaves the aromatic system untouched; base-catalyzed reactions, on the other hand, demand tighter controls to avoid splitting the amide. During some pilot runs, I’ve seen stubborn residue from multiple minor isomers emerge if reaction times go unchecked. Most downstream users value this molecular flexibility, since careful modifications generate new compounds with improved selectivity for biological targets or entirely different uses.
Synonyms & Product Names
Catalogs usually list synonyms such as N-Ethoxymethyl-2-Ethyl-6-Methylaniline Chloroacetamide, Chloroacetic Acid N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethylamide, and related trade names circulated by fine-chemical providers across the United States, China, and Europe. Well-annotated records help everyone avoid cross-contamination and mixups, especially with other chloroacetamides in storage or use. During chemoinformatics searches, alternate naming conventions occasionally trip up less-experienced operators, so education materials often include structural diagrams for clarity.
Safety & Operational Standards
Practical lab work with this compound leans on careful personal protection. Gloves rated for organics, face shields during weighing, and good ventilation all keep exposures low. Every spill response kit in my experience comes ready for both organic and aqueous cleanup, since the compound resists simple dilution with water. Eye comfort and skin integrity matter—some colleagues once shrugged off gloves, only to discover persistent dermatitis after repeat exposure. To meet ISO and occupational safety standards, fume hoods always run, with air monitors on for larger batches. Emergency showers and eyewash stations within bouncing distance are not a luxury but an absolute requirement, even for quick sample preps. Training around this and related amides includes lessons from older chemists who saw robust reactions turn dangerous when protocol slipped.
Application Area
Much interest in N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide comes from crop protection, especially for pre-emergence weed management in cereal and oilseed fields. Recent field tests in northern climates have shown it acts quickly on competitive broadleaf weeds, sparing key crops while minimizing soil residue. Some labs test this compound’s derivatives as candidate intermediates for active pharmaceuticals, mostly when looking for tricky amide intermediates with controlled release or metabolic profiles. Material scientists took notice as well, using it as a feedstock in experimental polymers, thanks to the modifiable side chains. In my own trial plots, the compound proved robust after unexpected spring rainstorms, giving it a nod for stability in shifting conditions—what growers and field managers keep wanting.
Research & Development
Researchers continue to push the boundaries with this amide. Structure modification groups seek reductions in toxicity and higher target specificity, using combinatorial chemistry and bioassays. Many universities tie this work into integrated pest management strategies, where single-use, persistent chemicals have fallen out of favor. Partnerships between public and private labs mean fresh analytical data every season—residue breakdown rates, metabolite identification, and structure-activity relationships. Recent attention on environmental fate encourages ongoing sampling of runoff and soil, providing field-usable data. Collaborations between toxicologists and chemists translate into safer formulations for agricultural workers and bystanders. As someone who’s reviewed grant applications in this space, I see how complex multi-center studies can pull chemistry, biology, and environmental science into sharper focus when searching for that next breakthrough.
Toxicity Research
Toxicologists treat N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide and its analogs with high caution. In animal studies, sub-chronic exposure leads to mild liver enzyme elevation, with dose-dependent skin and eye irritation in non-protected handlers. Some breakdown products trigger mild mutagenic responses on bacterial plates, though in-vitro human cell studies show lower risk at plausible environmental concentrations. Regulatory agencies in the EU and North America expect transparent reporting on all acute, sub-acute, and chronic toxicity findings, forcing manufacturers to conduct full life-cycle monitoring in finished products. I’ll never forget one incident in a collaborating lab where monitoring slipped and complaints from routine handlers led to stricter batch testing. The risk isn't just theoretical: real-world vigilance means staff can spot early health effects, get rapid interventions, and push for less hazardous analogues. Much modern research harnesses ‘omics’ technologies—transcriptomics and proteomics—to catch changes in exposed organisms well before symptoms or population impacts show up.
Future Prospects
Innovation doesn’t slow down around aromatics like this one. There’s a growing push for greener synthesis—switching to solvent-less or bio-catalyzed methods to cut out hazardous steps and waste streams. Synthetic biologists and green chemists swap notes on tailoring microbe-based routes that build the target molecule from simple carbon sources, leapfrogging harsh reagents. Demand for more selective weed control will likely keep this class in the running, at least until new biotech answers arise. If chemists chip away at toxicity and non-target impacts, future versions stand a chance of fitting into stricter global safety regimes, while still fighting resistance in the field. As more agricultural regions shift to integrated pest management, the need for safe, effective, and environmentally friendly compounds like N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide stays clear. Researchers and policymakers must keep eyes open, learning from past mishaps so next-generation amides balance productivity, farmer safety, and environmental health.
What is N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide used for?
The Story Behind the Chemical
You don’t hear the name N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide at the dinner table. Yet, those of us who follow agriculture and crop production know this chemical shapes what ends up in our grocery carts. Most people never see the drum it arrives in, don’t recognize the structure on the label. This compound stands out as a pre-emergence herbicide, specialized for controlling a wide variety of weeds, and it quietly supports stable crop yields from one growing season to the next.
How Farmers Use It
Across cornfields and soybean operations, this herbicide goes to work before seeds sprout. Farmers apply it to the soil ahead of planting. Tough annual grasses and broadleaf weeds would otherwise sprout and compete for water, sunlight, and nutrients. With this chemical’s help, young crops get a real head start.
Year after year, weed pressure rises as invasive species adapt. This compound steps in where traditional methods like tilling can’t always keep up—especially in conservation-focused fields where farmers want to avoid soil erosion from disturbance. By targeting the weeds early, these farms keep run-off low, cut back on fuel spent turning the soil, and still meet global food demands.
Why It Matters for Food Production
Feed, fuel, and fiber markets depend on secure crop output. I’ve walked fields covered in morning glory, pigweed, and foxtail—a green jungle where crops get lost. There’s a gap between doing nothing and over-treating, and this herbicide helps fill it. Yields can drop by more than half from unchecked weed growth. Farmers, especially in regions battling climate risks, depend on crop protection tools that help manage the rising tide of resistant weeds.
Without such chemistry, food prices shoot up. Markets start to feel pressure, and regions where hunger remains a threat pay the steepest price. The application of this herbicide, paired with stewardship and responsible handling, often supports both harvest stability and food security.
Safety, Environmental Impact, and Resistance
Chemicals like N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide come with responsibilities. Certification programs and training, which I’ve gone through myself, stress personal protection, weather monitoring, and timing applications to limit drift into water sources. Farmers hear constant reminders: Rotate herbicides, use integrated pest management, monitor weed escapes. Resistance creeps up when the same product hits the same field, year after year.
We can’t solve weed problems with chemicals alone. Science pushes ahead—cover cropping, mechanical weeding, and software that maps problem patches all combine with herbicides to slow resistance and limit the environmental footprint. Regulatory bodies keep tabs on residue and persistence; they force stricter limits to keep soil and water safer. This chemical fits into the rotation, not as a fix-all, but as one tool among many.
Looking Forward
Innovation continues. Companies tweak molecules, aiming for greater selectivity and lower toxicity. Farmers take on soil health projects, using new practices to reduce dependence. Responsible strategies require ongoing education and adapting field approaches every season. Those of us who depend on reliable food production, whether on the farm or at the table, benefit from smart stewardship at every link in the chain.
What are the safety precautions when handling this product?
Know What You’re Handling
Many folks never stop to consider that chemicals in the workplace, or even household products, require a different level of care. Every time I step into a room where cleaning supplies are stored or see industrial products in use, I think about how easy it is to forget they're anything but ordinary. Reading the label isn’t just busywork — it's the first defense against accidents. The Material Safety Data Sheet (MSDS) lists health hazards, storage guidelines, proper handling instructions, and what to do if something goes wrong. Skipping over this information raises your risk by a mile.
Don’t Rely on Common Sense Alone
After decades in jobs that handled everything from solvents to bleach, I’ve seen people depend on “instinct” to judge what’s safe. That approach only allows mistakes to happen faster. For instance, mixing ammonia with bleach releases toxic gases, but plenty of people figure they’re just boosting cleaning power. It’s always worth pausing to read warnings and hazard pictograms — a nose for danger develops over time, but facts matter more than guesswork.
Personal Protective Equipment (PPE) Isn’t Optional
Gloves, goggles, and proper clothing form a solid barrier between your body and risky substances. One time, a colleague didn’t bother with goggles when refilling a machine. Splash came quickly and so did the trip to the emergency room. PPE takes seconds to put on, but injury lingers much longer. The U.S. Bureau of Labor Statistics reports thousands of workplace eye injuries each year, most of them preventable by simply wearing protective equipment.
Have Good Ventilation
Breathing in fumes isn’t part of anyone’s job description. Chemicals like solvents and strong cleaners give off vapors that can irritate lungs and fog up your mind. That’s why I always open windows, turn on fans, or work outdoors when the smell gets strong. The National Institute for Occupational Safety and Health often highlights poor ventilation as a root cause of headaches, dizziness, and longer-term health problems. No shortcut beats fresh air and proper airflow.
Never Store Products Haphazardly
Some products, once opened, need a cool, dry spot away from sunlight. Others demand separate storage to avoid dangerous reactions. I’ve seen storage rooms where acids and bases rest on the same shelf—one accidental spill and things can escalate quickly. Labels exist for a reason. Stacking containers in a safe spot, securely closed and clearly marked, keeps confusion at bay on a busy day.
Emergency Preparedness Makes All the Difference
Accidents don’t give warnings. Eye wash stations, spill kits, and shower stations shouldn’t gather dust in a corner. During a minor spill, quick action using the right supplies buckles down the problem before it becomes a major incident. My experience in maintenance taught me to never skip drills and always check emergency supplies. OSHA guidelines recommend routine reviews so that everyone in the building knows where to go and what to do if trouble hits.
Keep Communication Open
Sharing knowledge prevents problems. If someone spots a leaky container or unclear instruction, getting the word out keeps coworkers and family members safe. Real incidents rarely wait for perfect timing—clear talk keeps accidents rare and response sharp.
What is the chemical formula and molecular weight of N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide?
Chemical Formula and Molecular Weight
This compound carries the chemical formula C13H18ClNO2. Its molecular weight comes out to 255.74 g/mol. Behind these numbers sits a blend of carbon, hydrogen, chlorine, nitrogen, and oxygen–each part of the structure lending unique properties to the amide group. The presence of a chloroacetamide backbone stretches across both drug research and agriculture, finding roles in herbicides or laboratory intermediates.
The Value of Chemical Details
Getting the formula right goes beyond academic routines. Each atom in a chemical like N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide helps predict how the substance will behave when it meets air, water, or another chemical. In the real world, small errors in a formula or weight can set back months of research, trigger unexpected byproducts, or—worse—expose workers to unsafe conditions.
Chemists have to double-check these figures before moving forward. Think about a pharmacy where purity stakes stand high. A minor slip in molecular weight calculations can spoil the dosage or unleash toxins, erasing the trust between a maker and patient fast. In labs, accurate weights and formulas help design reliable tests and synthesize chemicals with confidence. The same checks echo through factories, where batch integrity controls worker safety and environmental impact.
Real-World Impacts
One thing personal experience keeps teaching: someone always depends on the details handled in the lab. About a decade ago, fumbling a calculation with a similar aromatic compound forced a costly recall in a manufacturing run. The echo of that oversight pushed me to prioritize formula accuracy no matter the pressure for speed. Since then, I have watched quality teams build triple-check systems into their workflows, using digital tools to flag common structural mistakes and weigh every batch before release.
Mistakes with substances containing chlorine or amide groups can become hazardous fast. People working in pesticide plants have witnessed accidental releases causing local evacuations, all because someone trusted a secondary source’s formula. Responding to these lessons, regulatory bodies now make chemical manufacturers standardize reporting structures and insist on updated databases. Researchers who publish flawed data end up losing their professional credibility and sometimes face severe consequences.
Safer Chemistry Needs Accurate Data
Safety procedures keep growing stricter for a reason. Real numbers give first responders clues in emergencies, guide toxicologists when patients need treatment, and help government agencies set exposure limits. Labeling chemicals wrong is not just a paperwork issue but a potential disaster in real use. That’s why trusted sources—peer-reviewed articles, material safety data sheets, and verified chemical suppliers—matter more than ever.
Better training, open-data collaboration across borders, and ongoing education help shore up these defenses. Double-blind audits and electronic lab notebooks shrink the risk of missteps. Young chemists now benefit from workshops teaching not just the rules but the culture of skepticism and error-checking. Anyone handling organic chemicals with functional groups like this chloroacetamide knows every correct answer today means a safer tomorrow.
How should this chemical be stored?
Why Proper Storage Matters
Anyone who’s ever spent time in a chemistry lab knows this: one small mistake can turn a quiet afternoon into a major incident. Years ago, I watched a colleague open a container of sodium metal stored near a window. The sunlight had warmed the room, and a thin layer of perspiration collected on the bottle. When exposed to air, sodium can ignite or even explode—that close call left a mark on us all. The right storage isn’t about ticking boxes. It’s about protecting people, property, and the environment.
Understanding the Chemical’s Personality
Every chemical comes with its quirks. Hydrochloric acid will chew through metal. Acetone sneaks off as vapor at room temperature. Lithium batteries, tossed together, have triggered warehouse fires. The label gives clues, but always check the science. The Safety Data Sheet (SDS) tells you if a chemical lays waste to plastics, corrodes glass, or reacts with water. I make it a habit to read the SDS before a new material finds a home on our shelves.
Separation Prevents Disasters
A storeroom isn’t a junk drawer. Acids and bases don’t belong on the same rack. Flammable solvents should sit in approved metal or fire-resistant cabinets. Strong oxidizers like nitrate and permanganate demand distance from organic materials such as paper towels or even sugar. I still remember the fire at a friend’s workshop. Chlorine cleaner shared a shelf with a degreaser—a leaky bottle here, a little spill there, and the fumes were enough to clear the place. Sometimes, a solid wall or separate cabinet makes all the difference.
The Right Container and Closure
Original packaging matters more than it looks. Manufacturers test containers to hold up against each specific content: the right plastic for acids, the right glass for solvent. Swapping out containers “just for a day” can end with corroded shelf liners or worse, a slow leak onto the floor. Spare yourself the cleanup—keep chemicals in their labeled bottles, with caps snug but not overtightened so pressure can’t build up.
Controlling Temperature and Light Exposure
Some products fall apart in warm or sunny rooms. Certain peroxides and catalysts break down if left in the open. I’ve seen times when hydrogen peroxide, forgotten in direct sunlight, lost potency and threatened to burst from a swollen bottle. Reserve cool, well-ventilated spaces for volatile compounds. Light-proof cabinets offer a home for anything sensitive to UV or heat—think of it as sunscreen for your stockroom.
Labeling and Inventory: Keep Track or Pay the Price
A handwritten label might leave you guessing. Permanent, clear, easy-to-read tags give quick answers about what’s inside, who owns it, and when it arrived. Digital tools make inventory less of a chore. During one lab re-org, we used a spreadsheet and color-coded tape. Anything out of date or missing a label got tossed—no questions, no exceptions. Tracking what you store shrinks the risk of mix-ups and helps with emergency responses.
Access, Training, and Vigilance
Set rules for who goes into your chemical storeroom. Keep a log or badge-access system. Training for everyone, from students to visiting maintenance crew, forms the front line of safety. They should know where the spill kits live, how to use them, and who to call. Casual habits—propping open a ventilation door, storing food next to lab supplies—invite trouble. I’ve watched senior staff save the day just because they caught a small mistake early on.
Small Habits Save Big Problems
Storing chemicals safely calls for respect. Respect for the risks and for the people sharing your workspace. Ventilate, lock up, label, and log your inventory. Challenge shortcuts—experience shows even a moment’s lapse leaves lasting consequences. Safe storage isn’t an extra task. It’s the most basic investment in everyone’s long-term wellbeing.
Is this substance hazardous or regulated?
Sorting Through Rules and Real Risks
Long chemical names can trigger confusion and caution. People want to know if a powder tucked away in a storeroom, or a chemical in the shipping dock, might hurt them, damage equipment, or get them in trouble with the law. The question, "Is this substance hazardous or regulated?" rests at the intersection of real-world safety and legal compliance. I once worked in a manufacturing facility where a minor mix-up with an unlabeled drum led to a full building evacuation—nobody wanted a repeat. Many folks who handle chemicals on the job ask this question, but answers rarely come easy.
Deciphering Hazardous Substances
Hazardous usually means the substance can hurt people or the environment if mishandled. Sometimes the danger shows up as flaming liquids or toxic fumes, but other times it’s skin burns or a health hit years down the road. The Occupational Safety and Health Administration (OSHA) lists chemicals falling under "hazardous" based on their health and physical side effects—things like corrosivity, toxicity, flammability, or reactivity. Asbestos is the classic example, where the dust sounds harmless until you see the long-term lung impact.
Labels, Safety Data Sheets (SDS), and shipping documents often paint a partial picture. In practice, I found that even common household items stacked in shops—bleach, drain cleaner, solvents—fit hazardous criteria but slide under the radar. The problem jumps out when regulations call for handling protocols, employee training, and reporting. Mishaps can get expensive quickly, with OSHA fines racking up fast if rules get ignored.
What Makes a Substance Regulated?
Regulated substances form a maze all their own. Agencies like EPA, OSHA, DOT, and state regulators each claim a slice of the pie. Registration and reporting rules move the answer from "safe" to "under watch" territory. The EPA’s Toxic Substances Control Act (TSCA) tracks inventory, while Resource Conservation and Recovery Act (RCRA) nails down hazardous waste disposal. My own toolkit always kept a copy of federal and state chemical lists—the SARA Title III Section 313 chemicals, for instance, make thousands of businesses file detailed reports every year, even for tiny lead or mercury amounts.
Rules shift with concentration, quantities, and context. A substance may not count as hazardous alone, but hit a certain weight or sit mixed with other ingredients and the rules lock in. Back in warehouses where I worked, the difference between a one-gallon jug and a pallet load changed everything—local fire marshals paid attention at that scale, and missing paperwork invited surprise audits.
Navigating the Details
Plenty of workers and managers, just like me, turned first to SDS sheets, then double-checked with the most recent federal or state lists. No one wants to face a spill or accident without the right information on hand. Keeping current matters: new research pushes more chemicals onto watch lists every year. Walk into most workspaces and folks will know the rules for lead paint and gasoline, but not always for that solvent from overseas with missing documentation.
Trade groups, occupational health resources, and accredited training can shrink the risk of expensive blunders. Practical steps—like regular container labeling, fresh SDS copies, and a habit of calling formal hotlines for ruling—make a difference. In my experience, one phone call to a state regulatory agency resolved more confusion than a stack of online safety articles.
Getting a Clear Answer
So, is a substance hazardous or regulated? The real answer always demands looking beyond the product label. Knowing what you handle opens the door to safe practices, fewer fines, and a workplace that supports health for the long haul. The right questions—backed up by facts and the willingness to ask for expert help—keep operations running and people safer in the real world, not just on paper.
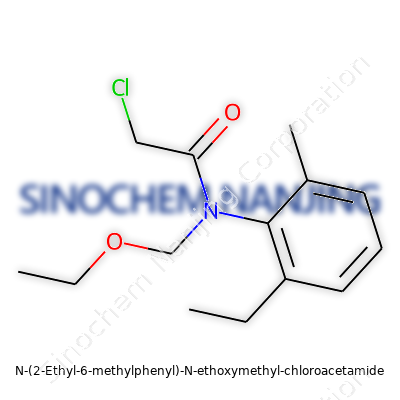
| Names | |
| Preferred IUPAC name | N-(2-ethyl-6-methylphenyl)-N-(ethoxymethyl)-2-chloroacetamide |
| Other names |
Pretilachlor Rifit Rifit 50 EC |
| Pronunciation | /ɛn tuː ˈɛθɪl sɪks ˈmɛθəlˌfiːnɪl ɛn ɪˈθɒksɪˌmɛθɪl klɔːroʊəˈsiːtəˌmaɪd/ |
| Identifiers | |
| CAS Number | [“63121-00-6”] |
| 3D model (JSmol) | `3D model (JSmol)` string for **N-(2-Ethyl-6-Methylphenyl)-N-Ethoxymethyl-Chloroacetamide** (as a **SMILES** string): ``` CCc1cccc(C)c1N(COCC)C(=O)CCl ``` |
| Beilstein Reference | 1367985 |
| ChEBI | CHEBI:82759 |
| ChEMBL | CHEMBL2105937 |
| ChemSpider | 24693374 |
| DrugBank | DB08797 |
| ECHA InfoCard | 07b2871e-eb6c-4e3c-aec5-6e92f97f172d |
| EC Number | 245-610-5 |
| Gmelin Reference | Gmelin Reference: "83393 |
| KEGG | C18457 |
| MeSH | Dichlormamide |
| PubChem CID | 11718574 |
| RTECS number | AF8575000 |
| UNII | N3O1449U54 |
| UN number | 2811 |
| CompTox Dashboard (EPA) | DTXSID7038756 |
| Properties | |
| Chemical formula | C13H18ClNO2 |
| Molar mass | 270.76 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.14 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 2.72 |
| Vapor pressure | <0.00001 mmHg (25°C) |
| Acidity (pKa) | Acidity (pKa): 15.17 |
| Basicity (pKb) | pKb = 13.84 |
| Magnetic susceptibility (χ) | -73.51 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 523.7 mPa.s at 20°C |
| Dipole moment | 3.73 D |
| Thermochemistry | |
| Std enthalpy of formation (ΔfH⦵298) | No data |
| Std enthalpy of combustion (ΔcH⦵298) | -6687.6 kJ/mol |
| Pharmacology | |
| ATC code | N05CM02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P301+P312, P305+P351+P338, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: 113 °C |
| Autoignition temperature | 242 °C |
| Lethal dose or concentration | LD₅₀ oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral LD50 2,953 mg/kg |
| NIOSH | RJ3200000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Acetochlor Alachlor Butachlor Metolachlor |

