N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide: Beyond the Lab Bench
Historical Development
N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide didn’t pop up overnight. Its path traces through decades of agrochemical innovation, particularly at a point in history when chemists scoured benzene rings for new ways to win the battle against tough weeds and pests. I remember reading old journals where researchers, credit cards in hand at chemical suppliers, obsessed over fine-tuning amide derivatives for reliability in variable climates. By the 1980s, chemical modifications like the shift from methoxymethyl groups to different acyl components became the key tools for tuning both toxicity and persistence in fields. The journey has been long, marked by regulatory pushback, trials with ever-stricter environmental standards, and a relentless demand for safer, more targeted compounds.
Product Overview
This compound stands out because it promises selective action. The N-(2,6-diethylphenyl) backbone paired with the methoxymethyl group creates a shield of sorts against fast metabolism in plants. Its practical roots take hold in herbicidal formulation, especially where seasonal cycles drive waves of unwanted plants threatening food supply and farm income. Researchers leaned toward this structure because of its unique absorption profile—getting just enough action on weeds without burning everything else. Commercial chemists found that the addition of a chloroacetamide fragment locked in the right balance of activity and environmental breakdown, pushing it ahead of simpler amides still used in some markets.
Physical & Chemical Properties
Anyone who’s handled it knows: white to off-white appearance, faint organic odor, stable under dry, dark storage conditions. Once exposed to light and humidity, degradation picks up—something that keeps both warehouse managers and field handlers on their toes. Its moderate solubility in polar organic solvents offers challenges and advantages, depending on your formulation process. I recall talking to an older process engineer who admired how a tightly bound phenyl ring and the bulky ethyl groups cut down on unwanted byproducts during synthesis, saving loads on post-reaction cleanup.
Technical Specifications & Labeling
Regulators expect clear labeling that spells out hazards without jargon—no blurred lines on this front. Labels focus on skin and eye irritation risk and remind handlers to wear gloves and goggles, lessons learned from years of cuts and rashes documented in farmworker safety reports. Specifications demand purity levels above 98% for most applications, and reliable certificates of analysis back up each lot. From my own time in a field application crew, the difference between clear, honest labeling and some vague sticker often meant the difference between a good shift and a ride to occupational health.
Preparation Method
Synthesis routes center on acylation of an amine with a chloroacetyl chloride after prior methoxymethylation of the phenyl amine. Not the sort of reaction mix you’d want to scale carelessly—a point made all too clear by accident accounts in older factories. Labs built out proper venting and temperature controls only after tough lessons with runaway reactions. This path offers high yield under careful control, but always brings lingering questions about green chemistry: Is there a safer, cleaner alternative that avoids tough reagents? That question still drives most of the greener R&D efforts today.
Chemical Reactions & Modifications
Once made, chemists tweak structural components through hydrolysis, oxidation, or further amide functionalization. The backbone is strong, but side chain manipulation brings big performance changes. Substituting other alkyl groups or varying the halide sometimes unlocks new fields of application or improves breakdown rates for better environmental outcomes. Still, every modification draws a new wave of regulatory scrutiny, giving formulating chemists headaches over the pile of paperwork that follows even a small change. In my experience, minor changes can lead to big upsets in field trials—a reminder that lab data doesn’t always travel well to the real world.
Synonyms & Product Names
Chemists, traders, and farmers often use shorthand, swapping full IUPAC names for simpler tags or code names. This can lead to mix-ups, especially when similar amides crowd the same catalog and market labels change with region or supplier. In technical publications, I often see references to its chloroacetamide class, though trade names pop up in order forms and seed store shelves. As new formulations hit the market, the list of synonyms grows, complicating recalls and exposé reporting on safety—another reason why harmonized naming in international databases matters for anyone tracking usage and risk.
Safety & Operational Standards
Widespread training now makes PPE standard: gloves, coveralls, face shields for mixing, plus ventilation protocols in large batch operations. Incidents in the field have forced a strict “no-exceptions” culture, since even small exposures can irritate or sensitize users, and larger volumes pose risks in spill events. Disposal rules aim to keep runoff away from waterways where aquatic life can suffer low-dose effects, based on long-term water quality studies that showed clear negative impacts. There’s no substitute for clear, repetitive safety dialogue—something I’ve witnessed firsthand saving junior team members from real harm on fast-paced jobs.
Application Area
The go-to use lands in weed management for row crops, with special preference in plots facing resistance to simpler herbicides. Application timing becomes everything. Farmers watch weather and growth cycles closely to thread the needle between maximum weed vulnerability and minimum crop stress. Some custom blends use it with other agents for multi-pronged weed management. Handlers in the field prefer water-based dispersions for coverage. Nonetheless, restrictions grow tougher each year, with increasing bans in sensitive watersheds.
Research & Development
R&D teams chase more than just higher efficacy; they’re looking for smarter, more sustainable action. Many shift attention to breakdown profiles and the impact on non-target species. Analytical chemists invest time in field residue studies, using LC-MS and GC-MS to track minute residues in soil and runoff. Green chemistry efforts explore enzymatic transformations or renewable feedstocks, but production costs and uncertain yields stall wider adoption. Collaboration with ecological researchers could help close the lab-field gap, and it wouldn’t hurt to give farmworkers and community groups more voice in setting research priorities.
Toxicity Research
Studies began with acute toxicity screens—mice and rabbits in cages—and branched into chronic exposure data by the late 1990s. Sensitive aquatic species showed trouble at lower doses than warm-blooded test animals, shifting regulatory focus to runoff and groundwater persistence. Farmworker studies brought new urgency; exposure events in the field correlated with skin and respiratory irritation. Modern studies push for better biomarkers to track and predict human impact, but funding gaps and industry pushback slow progress. Seeing the data firsthand reminds one that real harm isn’t a hypothetical—public and occupational health teams need more support across the board.
Future Prospects
Demand for safer, more precise agrochemicals keeps the spotlight on compounds like N-(2,6-diethylphenyl)-N-methoxymethyl-chloroacetamide, but pressure mounts for alternatives as public concern drives regulatory reviews. Advances in application technology, like drone spraying and seed coatings, may cut environmental drift, but no gadget can erase toxicity at the molecular level. The best hope likely lies in continuing to fund open, peer-reviewed research, keeping regulatory bodies independent, and listening more to frontline workers and communities than trade lobbyists or quarterly sales sheets. Progress isn’t about perfect chemistry alone—it’s about a smarter farm ecosystem where innovation serves both yield and safety, with all stakeholders getting their say.
What is the primary use of N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide?
How This Unusual Compound Shapes Everyday Farming
Few people outside of agriculture give much thought to complicated-sounding chemicals like N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide. Its name hardly rolls off the tongue, but this compound holds a major place in a farmer’s toolkit. Many know it better by its trade association with pre-emergent herbicides, most notably acetochlor. Companies blend this chemical into weed control products used in row crops—think corn, soybeans, and cotton across sprawling Midwestern fields. The whole goal: knock out weeds before they start, helping crops grow without competition that drains water, nutrients, and sunlight.
Weed pressure chews up billions of dollars in lost yields each year. Chemical weed control jumped to the forefront as traditional tilling started causing soil problems—erosion, nutrient loss, frustrated yields. Acetochlor’s active ingredient, derived from N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide, fills this gap by targeting weed seeds and young shoots before they surface. Chemists shaped its properties for just that: once in the ground, it blocks enzymes weeds use for growth, but leaves established crops alone, provided timing and application rates follow science, not guesswork.
Why Responsible Use Matters
No shortcut exists for good weed control. Chemical solutions like this have fueled leaps in corn and soybean efficiency. Even so, misuse spills into land management headaches. Waterways pick up traces that run off fields after heavy rain, stirring fears about wildlife effects and drinking water safety. The U.S. Environmental Protection Agency keeps tight reins on application, setting clear-cut rules to limit impacts away from target areas. Some regions require buffer zones or reporting, especially near streams or sensitive habitats.
What stands out isn’t just the molecule’s power—it’s the balancing act between food production and environmental stewardship. Overreliance breeds resistant weeds, meaning fields see more and more types of these compounds, chasing short-term fixes over long-term planning. Integrated weed management builds a tougher shield, blending crop rotation, cover crops, smart tillage, and limited herbicide use. Farmers who cycle modes of action cut down resistance problems and help preserve the tools they’ve got.
The Path Forward—Science on the Field
Farming rarely deals in certainties. Those who trust N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide as part of their weed plan see the benefits quick—a cleaner field, more grain per acre, less hand-weeding or extra tractor passes. But nobody can ignore the tightrope that stretches between productivity and ecosystem health. Researchers keep working to refine dose guidelines, map out safe water thresholds, and breed crops that need fewer chemical supports.
Farmers and agronomists learn from each season. Every application cycles through new weather, new weeds, new economic pressures. Responsible herbicide practices rest on reliable information—scouting, soil science, weather models, and local experience—rather than blanket coverage or habit. The right use of N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide keeps modern agriculture thriving, but only with eyes open to everything happening outside the field edge.
What are the safety precautions when handling this compound?
Being Proactive Beats Being Sorry
Some chemicals can surprise you even if you think you know them. Getting too comfortable around hazardous substances often leads to close calls, or worse. I learned early on how easy it is to skip gloves or forget goggles during a hectic lab day, only to regret it once a splash or whiff of fumes ruins your afternoon. Real trust in a chemical starts with what you know about it, not what you hope won’t happen.
Why Labels Matter More Than You Think
Reading the label and checking the Safety Data Sheet isn’t a pointless ritual—it’s your best shot at staying safe. Let’s say a bottle carries a corrosive symbol or warns “toxic if inhaled.” That red diamond, that line of text, tells you exactly where trouble could start: skin contact, breathing in dust, or even something as sneaky as vapors you can’t see. According to the National Institute for Occupational Safety and Health, half of lab accidents link back to misunderstood or ignored hazard warnings.
Personal Equipment Isn’t Optional
No one actually looks cool in safety goggles and a lab coat, but both block accidents from becoming injuries. Eye protection shields against splashes, while gloves keep dangerous stuff off your hands. In my own work preparing acids, I added a face shield after a classmate got splashed and needed weeks to recover. Staying covered up with long sleeves and closed shoes can save you from chemical burns or allergic reactions.
Good Habits Make Hard Work Safe
Never eat, drink, or touch your face if you’re near a hazardous compound. Open bottles under a fume hood to pull dangerous fumes away from your lungs. If fumes can’t be captured, switch to a less risky substitute, or rethink whether the experiment is worth it. The Chemical Safety Board reports that skipping fume hoods is behind hundreds of annual incidents, many involving students who decided it’d “just take a second.” No shortcut pays off if you land in the ER.
Knowing What to Do If Something Goes Wrong
Even with the best habits, accidents still find a way. It makes sense to keep an eyewash station and safety shower close by. Training should cover what to do if you spill something on your skin or clothes. I once watched a peer hesitate after a spill, losing precious time. Drilling emergency response routines means muscle memory can take over when thinking gets hard.
Storage and Disposal Make a Difference
Careless storage can turn a shelf into a hazard zone. Keep incompatible chemicals apart: acids stay away from bases, oxidizers away from organics. Tightly sealed bottles stop leaks and fumes from escaping. Used compounds belong in designated waste containers. Sometimes the temptation is strong to pour leftovers down a sink, but that puts water systems and people at risk. The Environmental Protection Agency stresses that following disposal instructions avoids a chain reaction of bigger problems outside the lab.
Better Safe Than Lucky
Being careful isn’t about fear. It’s about respect for things we use to get a job done. Safety culture grows stronger when people talk about near-misses and support each other in staying alert. Leadership that rewards speaking up sets a tone that values every team member. Taking small but steady steps beats making excuses later on.
Stay stubborn about your safety. You only get one set of eyes, and you want to keep all ten fingers.What is the chemical structure or formula of this product?
Why Chemical Structure Matters
Chemical structure turns up in every product we use, from simple table salt to the most complex pain relievers on the pharmacy shelf. Some might ask why they should care about molecules and formulas. For me, digging into chemical structure showed just how different one version of a product can be from the next. In college, when I studied acetaminophen and ibuprofen, I learned both treat headaches, but one can wreck your liver if overused, while the other is tough on your stomach. Their chemical structures tell the full story.
How Structure Shapes Function
Take table salt—sodium chloride, NaCl. This compound forms by pairing one sodium atom with a single chlorine atom. Its simple structure turns sodium—dangerous on its own—and chlorine—used in bleach—into a safe seasoning most kitchens use every day. That transformation blows my mind each time I sprinkle salt on food. The lesson: structure determines function.
For something more complicated, think about aspirin. Its chemical name is acetylsalicylic acid. The molecular formula is C9H8O4. Changing just a small part of that molecule produces different effects. For instance, swapping parts of its structure led to the development of drugs like acetaminophen and ibuprofen, each with a special profile for pain relief or inflammation.
Why Transparency Matters
Companies that share chemical structure information embrace public trust. Food allergies taught me to look close at ingredient lists. One wrong molecule can mean a hospital trip for a friend of mine allergic to peanuts. The same holds true for people sensitive to certain preservatives or color additives.
Historically, a lack of transparency has caused health scares. The 2008 melamine tainting crisis in China endangered children because people did not know melamine’s structure or its dangers. Full chemical information would have sparked faster testing and bans.
The Role of E-E-A-T: Trust, Quality, and Expertise
Doctors and scientists keep up with chemical structures to recommend safe products. The recent COVID-19 vaccine rollout showed how important expertise and clear information are. Each vaccine formula, like mRNA or viral vector, works due to its structure. Misinformation and hesitation spread when these details get lost or distorted.
Sharing verified chemical structures, backed by peer-reviewed evidence, lets anyone make better choices. It gives consumers the power to weigh risks and benefits of substances they eat, use, or put on their skin. The more credible experts participate in conversations about formulas, the stronger the trust in products that shape everyday life.
Real Solutions for Access and Understanding
Clear labeling standards and open access databases go a long way in solving confusion. A few useful resources include PubChem and ChemSpider. They let users search for any chemical and view its structure, toxicity, and applications. Schools that teach students how to read simple chemical diagrams help children build lifelong health and science skills.
Companies that offer QR codes or web links to chemical information build stronger reputations. This helps parents, caregivers, and even curious teens check substances before buying. Honest discussion, accessible technology, and greater involvement from experts give everyone a stake in safer products.
How should N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide be stored?
Looking Closely at a Sensitive Chemical
Working in a lab for several years, I’ve learned that certain compounds require more than just a quiet corner on a shelf. N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide falls into that wary group. Its structure means stability can’t be taken for granted, and proper storage isn't something to shrug off. Mishandling can lead not only to spoiled samples but also threaten health and safety.
Why Proper Storage Matters
Chemical safety isn’t just a regulation box to tick. Several accidents trace back to storing organic compounds alongside flammable solvents or exposing them to light and air. Compounds with a chloroacetamide structure, for example, often break down or turn hazardous under poor conditions. The molecule’s reactive functional groups can interact with moisture or heat, setting off unwanted reactions or making toxic byproducts.
The Essentials for Storing N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide
A good storage method starts with temperature control. High heat speeds up decomposition, while cooler conditions slow it down and help preserve chemical integrity. I always look for a dedicated chemical refrigerator with solid seals and stable temperatures (usually 2–8°C works well). Room temperature can sometimes work for short periods, but cooler is safer for this molecule in the long run.
Humidity poses another risk. It’s best to seal this compound in an airtight container with a gasketed cap, preferably made from materials that resist corrosion from organochlorines. Tightly sealed bottles keep out moisture and limit air exposure, both of which can shorten shelf life. For extra protection, adding a desiccant packet inside the storage container can absorb stray humidity. This trick helps, especially in labs where the air tends to run damp.
Think Beyond the Flask: Safety for People and the Environment
Years of handling similar compounds have shown me the headaches that come from inadequate labeling. So, each container deserves a clear, weather-resistant label carrying the chemical name, hazard symbols, and date of receipt. This keeps mistakes at bay and gives anyone in the lab the information needed to handle emergencies. Strong labeling and use logs help prevent accidental mixing with other substances—a recipe for spills or unexpected reactions.
Beyond the container, storage cabinets should be reserved for incompatible chemicals. Keeping N-(2,6-Diethylphenyl)-N-Methoxymethyl-Chloroacetamide away from strong acids, oxidizers, and anything reactive cuts down fire and neutralization hazards. Sometimes, labs work under local or institutional rules for hazardous waste—being familiar saves a lot of trouble down the road.
Improving Storage: Real-World Solutions
A strong lab culture always puts safety talks front and center. Training for new staff and refreshers for experienced hands demystify these steps. Routine audits can catch gaps: are containers dated, are seals intact, does the logbook match what's on the shelves? I’ve seen organizations introduce inventory management software to track chemical stocks and expiry dates, which works wonders for minimizing forgotten or expired chemicals.
Innovation can make a difference. Improved packaging, like double-walled bottles and smart caps that indicate exposure to air, offers fresh ideas for extending shelf life. Always checking Safety Data Sheets and staying connected with suppliers brings in the latest recommendations, since new research can shift what we thought we knew about safe storage.
Is this product available in different purity grades or packaging sizes?
Getting Clear on What’s Really on Offer
Questions about product purity grades and package sizes pop up all the time. Some people shrug and grab the first thing they see, but others know that these details can make all the difference. Maybe you need chemical reagents for a research lab, or maybe you run a local bakery that needs stabilizers in bulk. Either way, it pays to stop and dig into product listings.
Experience at the Lab Bench and the Work Floor
Anyone who has worked in a lab, especially with volatile or sensitive materials, will tell you: purity isn’t just some mysterious metric on a label. Reagent-grade means you won’t introduce random unknowns to your reaction. Years ago, I mixed up a subpar solvent with a trace contaminant, and my whole day’s results landed in the waste. Purity grades save money and avoid headaches since skipping on them can cost hours and even ruin expensive batches.
On the other hand, folks working with cleaning agents for facilities or minerals for construction sites don’t always need ultra-fine, pricey grades. There’s such a thing as overkill — you wouldn’t cook dinner with food-grade gold leaf every night. Companies and buyers need access to different purity grades to match each application. Sometimes it’s just a matter of cost savings, other times it’s about safety or even regulatory compliance.
The Wild World of Sizing
Packaging sizes affect everyone. I’ve worked places where a couple of grams in a glass vial made sense, and at others where sacks stacked to the ceiling were standard. Why does it matter? There’s storage. Small packs help smaller teams avoid waste. You open just what you need, keep the rest sealed up, and the product stays fresh. Bulk suppliers cater to bigger outfits where moving several kilos in a week is just routine.
Take a popular research chemical. If suppliers only sell it in one-kilo drums, students or smaller labs are left scrambling, splitting costs with other groups, or—worse—risking poor storage and early spoilage. Smaller bottles allow safer handling and more accurate measurement. On the flip side, too much focus on tiny packs annoys manufacturers who don’t want to open hundreds of containers for a job that one drum would cover.
Balance, Transparency, and Trust
Brand trust comes from transparency. A straightforward catalog or website should clearly state grade and available sizes. Companies that play coy or hide this info slow down production almost as much as a missing delivery. As a customer, seeing full details makes me more likely to stick with a supplier. The Association of Official Analytical Collaboration (AOAC) and ISO have long pushed for strong labeling practices, and it’s easy to see why.
Flexible options also encourage sustainability. Less waste, fewer returns, and less disposal of expired product happen when you can buy only what fits your use case. Smaller packs mean lower risk in transport and handling. In some industries, like pharmaceuticals, the law demands strict adherence to batch size and source information. Being able to select the right purity and size isn’t just helpful—it’s a requirement for compliance and safety.
Paths Forward from Both Sides
Suppliers could keep lines open for feedback. Regular surveys and direct lines to technical teams build trust. Customers, for their part, should push for detailed product info before buying. That basic back-and-forth doesn’t just improve business; it keeps day-to-day work flowing, keeps budgets under control, and ensures safe handling. It’s less about trying to tick a box, and more about building a world where the right product finds the right hands, every time.
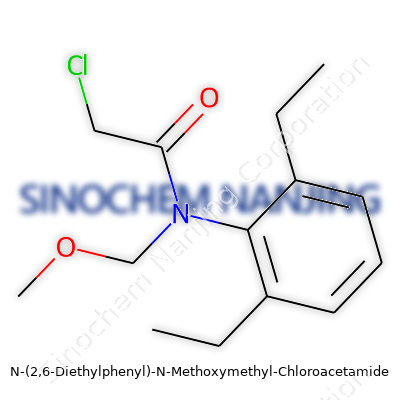
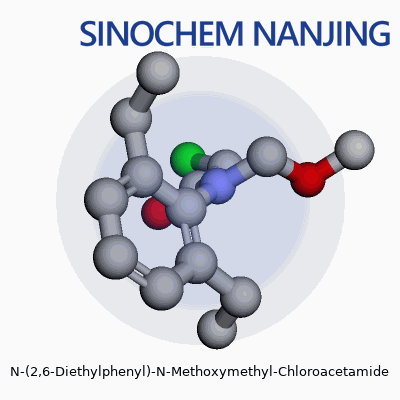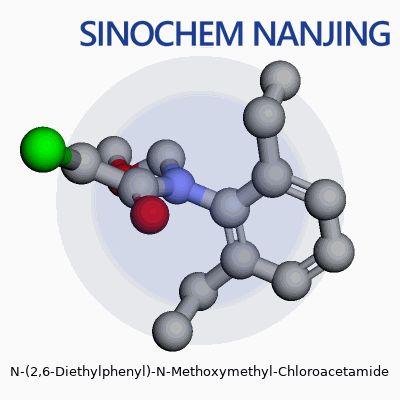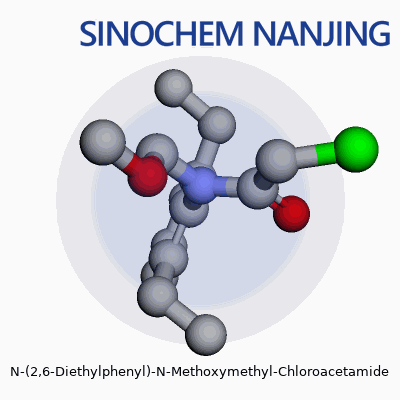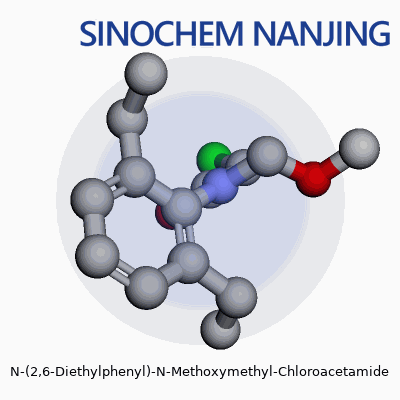
| Names | |
| Preferred IUPAC name | N-[(2,6-diethylphenyl)-(methoxy)methyl]2-chloroacetamide |
| Other names |
Acetamide, N-(2,6-diethylphenyl)-N-(methoxymethyl)-2-chloro- Chloroacetic acid N-(2,6-diethylphenyl)-N-(methoxymethyl)amide Chloroacetamide, N-(2,6-diethylphenyl)-N-(methoxymethyl)- |
| Pronunciation | /ɛn tuː sɪks daɪˈɛθəlˌfɛnəl ɛn ˌmiːˈθɒksiˌmɛθəl klɔːrˈoʊæsɪtəmaɪd/ |
| Identifiers | |
| CAS Number | “733742-47-7” |
| 3D model (JSmol) | `3D model (JSmol) string:` `CCc1cccc(CC)c1N(COC)C(=O)CCl` |
| Beilstein Reference | 2778734 |
| ChEBI | CHEBI:91585 |
| ChEMBL | CHEMBL2103837 |
| ChemSpider | 21580906 |
| DrugBank | DB08439 |
| ECHA InfoCard | 03e2b130-7d9a-4a4c-9d71-f2f9a94828dd |
| EC Number | 3.5.1.73 |
| Gmelin Reference | 89841 |
| KEGG | C10757 |
| MeSH | D008099 |
| PubChem CID | 21720945 |
| RTECS number | AT6825000 |
| UNII | 5H31GI9502 |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID8035065 |
| Properties | |
| Chemical formula | C13H18ClNO2 |
| Molar mass | 313.83 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.11 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 1.98 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 14.7 |
| Basicity (pKb) | pKb ≈ 13.4 |
| Magnetic susceptibility (χ) | -63.87 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.563 |
| Viscosity | 502 cP |
| Dipole moment | 3.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 489.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -6172 kJ/mol |
| Pharmacology | |
| ATC code | N01XA04 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS08, GHS09, Warning, H302, H315, H319, H332, H373, H411 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P363 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | Flash point: 110°C |
| Lethal dose or concentration | LD50 oral (rat): 900 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1010 mg/kg (rat, oral) |
| NIOSH | PB6742000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 6 mg/L |
| Related compounds | |
| Related compounds |
Chloroacetamide N-Methoxymethylchloroacetamide N-(2,6-Diethylphenyl)chloroacetamide N-(2,6-Diethylphenyl)-N-methoxymethylacetamide |

