Monomethyl Succinate: A Closer Look at Its Science, Applications, and Future
Historical Development of Monomethyl Succinate
Chemists in the early twentieth century started exploring the value of small, functional carboxylic acid derivatives for industrial and pharmaceutical use. Monomethyl succinate entered the scene as one of these promising compounds, building on the backbone of succinic acid. Early laboratory preparations usually involved straightforward esterification, serving both as a proof of concept for organic synthesis classes and as groundwork for more elaborate derivatives. Interest picked up when industries started to seek safer alternatives to harsher solvents and less benign intermediates, opening a door for simple molecules like monomethyl succinate to step in. Years of incremental process improvements eventually allowed for scalable, reliable production. As the digital age spread information, processes became more refined, purity increased, and regulatory standards began shaping its commercial life.
Product Overview
Monomethyl succinate falls under the category of monoesters of dicarboxylic acids, essentially serving as a bridge compound between succinic acid and dimethyl succinate. Its structure features a carboxylic acid on one end and an ester on the other, lending it characteristics important across several industries: polymers, pharmaceuticals, flavorings, and specialty chemicals. People who work with this molecule often mention its balanced reactivity and ease of handling, qualities that set it apart from more temperamental esters. Depending on the scale and the application, it's sold as a clear liquid or crystalline solid, with purity grades tailored toward downstream needs.
Physical and Chemical Properties
Monomethyl succinate presents as a colorless, sometimes slightly viscous liquid or a crystalline substance at lower temperatures. Its melting point hovers around 17°C, and boiling points rise to the vicinity of 196–198°C under ordinary pressure. Water solubility keeps it relevant where hydrolysis and aqueous reactivity matter. Its molecular formula, C5H8O4, helps reveal its moderate polarity, making it miscible with common solvents like methanol, ethanol, and acetone. Unlike bulkier esters, this molecule keeps a low molecular weight, which translates to reasonably quick reaction times and manageable volatility in open lab settings. Thanks to its combination of acid and ester groups, it serves dual purposes—participating as either a nucleophile or an electrophile, depending on the reaction setup.
Technical Specifications and Labeling
Commercial monomethyl succinate gets packaged with clear technical guidance. Typical bottles arrive with purity stamped above 98%, water content measured to tenths of a percent, and residual acid levels tightly controlled. Labels always include information like batch number, storage recommendations (keep sealed, avoid humidity, protect from sunlight), and expiry dates set based on stability data. Suppliers provide safety data sheets with hazard pictograms, recommended PPE, and emergency measures. It’s not uncommon to see product certifications tied to major standards, like ISO or REACH compliance, reassuring buyers that the supply chain upholds modern standards.
Preparation Method
Production often relies on simple esterification: succinic acid reacts with methanol, using an acid catalyst—sulfuric acid remains a favorite, though other mineral acids can do the trick in a pinch. The reaction proceeds at moderate temperatures; methanol acts as both solvent and reagent, pushing the equilibrium along. After the chemistry, purification steps swing into action: extractive washes remove excess methanol and unwanted catalyst, followed by distillation or crystallization depending on product form. Some green chemistry approaches skip harsh acids, favoring solid-state or enzyme-catalyzed syntheses, but these remain less common in high-volume applications. Staying vigilant with stoichiometry and impurity control ensures a consistent product—important for pharmaceutical or food ingredient supply.
Chemical Reactions and Modifications
Monomethyl succinate’s greatest strength comes from its chemical flexibility. Synthetic chemists view it as an access point for making more complex molecules. The free carboxyl group opens up routes like amidation, salt formation, or further esterification. On the ester side, hydrolysis reverts it back to succinic acid under base- or acid-catalyzed conditions. Reduction yields monomethyl succinate alcohols, useful in flavor syntheses or certain active pharmaceutical ingredient (API) routes. Alkylation, acylation, and transesterification processes extend its utility further, finding roles in fragrance chemistry, biodegradable polymer synthesis, and even ionic liquid development. For those working in research labs, its stable yet sensitive profile means scale-up seldom throws surprises regarding side-product formation.
Synonyms and Product Names
Depending on context and supplier, monomethyl succinate might appear on labels by a handful of synonyms: methyl hydrogen succinate, succinic acid monomethyl ester, monomethyl ester of succinic acid, or simply methyl succinate (not to be confused with dimethyl succinate). CAS registry numbers and systematic names usually settle identification disputes, but reading labels closely steers buyers clear of mix-ups with dimethyl or diethyl succinates. Some catalogs use manufacturer designations tied to in-house codes, making international trade rely heavily on accurate translation and regulatory filings.
Safety and Operational Standards
Experience in process chemistry reveals a manageable safety profile for monomethyl succinate—milder compared to halogenated or aromatic esters, but not entirely risk-free. Its principal hazard stems from irritant properties: direct contact can cause redness on skin or mild conjunctival irritation if splashed in the eyes. Inhalation doesn’t typically trigger acute effects, thanks to modest volatility, but standard ventilation and PPE—gloves, goggles, lab coats—always get the nod. Regulatory reviews place the compound outside most lists of highly toxic or carcinogenic materials, yet industry best practice dictates robust engineering controls, consistent training, and spill management plans. Transport and storage standards insist on proper secondary containment, correct container labeling, and periodic inspection for leaks or degradation.
Application Area
End users find monomethyl succinate a problem-solver in a range of settings. In pharmaceutical synthesis, it steps in as a precursor for active agents and intermediates. Chemical manufacturers use it to build block copolymers or as a biodegradable plasticizer—the monoester group helps regulate flexibility in polymers for medical or packaging use. The food and fragrance sectors apply it in controlled flavors, where the mild ester note blends into fruit or cream profiles without introducing harsh or persistent aftertastes. In textile finishing and surface treatment, its role as a reactive solvent or dispersant offers predictable evaporation rates and safe handling compared to legacy solvents. Recent interest in green chemistry has scientists studying it as a renewable building block, owing to the push for bio-based feedstocks and sustainable supply chains.
Research & Development
Universities and R&D arms of chemical companies keep monomethyl succinate on hand for both exploratory and pilot-scale studies. New catalyst systems get bench-tested for selectivity using monoesters like this one, as they strike a balance between volatility and reactivity. Bio-based routes, leveraging genetically engineered microorganisms, currently attract significant funding. Investigators want to bypass fossil inputs and minimize waste, and some startups claim pilot plants capable of producing monomethyl succinate from agricultural side streams or sugar beet waste. Combination studies with novel enzymes have started to show promise, achieving yield figures that push toward commercial thresholds. Downstream, interest keeps growing in coupling monomethyl succinate with green oxidants or photochemical reagents to unlock next-generation functional groups.
Toxicity Research
Animal models and in vitro cell cultures have played a major role in clarifying the safety profile of monomethyl succinate. Results point toward low acute oral and dermal toxicity; repeated dose studies, at concentrations present in typical handling environments, show minimal concerns for bioaccumulation or chronic harm. Metabolic breakdown products match those of succinic acid and methanol—both rapidly processed in mammalian systems. Inhalation exposure under experimental conditions doesn’t provoke organ-specific toxicity or mutagenic effects. Regulatory agencies continue to collect environmental fate data, measuring aquatic toxicity and persistence to calibrate wastewater controls. Toxicologists track occupational exposures, but existing limits seldom get approached outside accident scenarios.
Future Prospects
The chemical and allied industries face mounting pressures—heightened regulations, consumer demand for safer alternatives, global supply chain shifts. Monomethyl succinate is well-positioned to see broader use as manufacturers pivot to bio-based, less hazardous intermediates. Ongoing work in catalytic route optimization promises better atom economy and lower emissions, cutting both costs and environmental footprints. Application engineers eye new uses in advanced materials: hydrogels, specialty elastomers, responsive coatings, and slow-release formulations. Academic collaborations stretch into the design of next-generation green solvents and tailor-made drug carrier systems. As downstream sectors prioritize carbon neutral processes, demand for renewable monomethyl succinate will likely expand, spurring investment in efficient, scalable production routes that don’t sacrifice performance or purity.
What is Monomethyl Succinate used for?
Spotlighting a Lesser-Known Chemical
Monomethyl succinate rarely grabs headlines or sparks debates at conferences, but it threads its way through factories and laboratories that shape the products people rely on every day. Out in the wild world of chemical manufacturing, this compound plays a sturdy supporting role, even if its name doesn’t light up billboards. My years dabbling both in industrial environments and behind academic desks showed me one thing: often, it’s the unglamorous compounds like this making things tick in the background.
Helping Hand in Manufacturing
Factories use monomethyl succinate in the production of polymers and resins. It slips into the process as an intermediate—think of it as a step in a much bigger dance. The chemical’s structure, rooted in the succinic acid family, lends a hand to manufacturers trying to build strong, flexible plastics. Producers value its consistency, which means finished materials don’t surprise anyone with sudden cracking or other quality issues. My visit to a coatings plant in Ohio showed how a dependable compound like this helps stabilize finished mixtures—coating everything from furniture to wind turbines.
Beyond the Factory: Everyday Products
Consumers might not recognize the name, but they sometimes touch or see goods made with monomethyl succinate. For example, manufacturers drawing up a new paint or adhesive formula will often call for safe, stable intermediates. Chemical engineers in the paints sector use monomethyl succinate to achieve a certain balance: a sticky solution that handles moisture and heat while meeting environmental benchmarks. I remember speaking to a formulator at a small adhesives company in the Midwest who praised this chemical for steering clear of harsh byproducts.
The Push for Greener Chemistry
No company wants backlash over toxic byproducts or risky chemical waste. Here’s where monomethyl succinate brings something extra to the table. It doesn’t leave manufacturers wrangling with mountains of hazardous leftovers. The chemical breaks down into benign substances, so treating waste water or emissions from the factory often presents less of a headache. This biodegradable profile fits into broader pushes for safer chemistry and environmental accountability. When California tightened environmental rules on volatile chemicals, a handful of companies saw monomethyl succinate as a step toward compliance.
Safety and Handling
Experience in the lab and on the shop floor shows me that easy-to-handle chemicals cut down on accidents or costly training. Monomethyl succinate doesn’t hit the panic button among safety officers. You don’t see operators kitted out like astronauts just to pour a batch into the mixing tank. Simple gloves and goggles do the job. That kind of routine means less downtime, more time focused on product quality, and lower overall costs.
What the Future Holds
Professional circles talk more and more about sustainable supply chains. As demand rises for safer and ‘greener’ inputs in manufacturing, monomethyl succinate stands out as a practical option. Spearheading new research into polymers that balance high strength and low environmental impact, labs often reach for this compound. The shift toward responsible chemistry won’t happen overnight, but chemicals like monomethyl succinate show that real progress often hides in plain sight.
Is Monomethyl Succinate safe for cosmetic formulations?
Understanding Monomethyl Succinate
Monomethyl Succinate pops up on ingredient lists in a range of personal care items. Formulators turn to it for its role as a gentle pH adjuster and skin conditioning agent. If you’ve ever scanned the back of a moisturizer, or tried out a new serum, you probably brushed across this name among the unfamiliar ones. There’s a growing urge from everyday users to dig past pretty packaging and ask, “Can I trust this stuff on my skin?”
What the Science Says
Safety has taken center stage as customers grow more ingredient-savvy. Monomethyl Succinate comes from succinic acid, a molecule you also find in foods and in our own cells. Most cosmetic chemistry databases—the ones that manufacturers and regulators use—agree this ingredient ranks low on skin irritation and toxicity levels. Animal testing and patch studies back this up. The Environmental Working Group puts Monomethyl Succinate in its lowest hazard bands. Europe’s Scientific Committee on Consumer Safety has not flagged it as concerning either.
Some folks, usually those with sensitive skin or a history of allergies, ask about risk of irritation. Every compound carries potential for someone, somewhere, to react, but large-scale clinical use hasn't produced red flags for Monomethyl Succinate. Dermatologists and toxicologists, including teams I've spoken to at trade shows, consider it “unremarkable” in terms of its risk profile. In other words, it doesn’t stand out as a worry in the lab or the dermatologist’s office—especially used at levels set by product guidelines.
Why This Matters for People Using Cosmetics
Trust matters, especially with products soaking into your skin day after day. People depend on the watchdogs: FDA in the US, European Commission abroad, and independent panels like CIR (Cosmetic Ingredient Review). They pore over animal studies, in-vitro data, and human patch tests. History teaches us never to assume an ingredient’s safety from one good study, but when low concern remains steady after years of review, customers gain peace of mind.
Stress and pollution leave skin more reactive for many people, so reassurance about core ingredients grows more important. Seeing an ingredient with both a solid regulatory record and low allergenicity does help me pick a product off the shelf with more confidence. It’s not just about danger avoidance; it’s about making healthy, informed choices.
How the Industry Can Support Consumer Safety
Transparency remains essential. Companies can help by giving plain-language explanations for why each ingredient shows up in a formula. Monomethyl Succinate, for example, improves product feel and maintains stability, making creamy lotions glide better and last on the shelf. Honest labeling, open safety dossiers, and rapid response to new data keep consumers protected and informed.
Innovation teams should stick close to evolving research. Open dialogue with dermatologists and toxicologists helps brands adapt if any new risks show up in future. If a customer has concerns or shows sensitivity, companies should make it easy to contact support, report an issue, or find alternatives—this doesn’t hurt business, it builds trust.
Ingredients are more than names on a bottle. The choices companies make shape not just how skin looks, but how safe and cared for customers feel. With Monomethyl Succinate, current evidence gives little reason for worry. Paying attention, asking questions, and staying curious about chemical safety will always make the market better for everyone using these products every day.
What are the physical and chemical properties of Monomethyl Succinate?
Monomethyl Succinate at a Glance
Monomethyl succinate steps out of the crowd as a straightforward chemical built from the backbone of succinic acid. It carries a simple structure—just one of the two carboxylic acid groups in succinic acid has been transformed into a methyl ester. This single tweak shifts the way the molecule behaves and what it can do in manufacturing or daily use.
The Physical Side: Color, Feel, and Form
This compound shows up as a colorless, sometimes faintly yellow liquid. Its clarity and lack of strong color make it easy to spot when even a tiny bit of contamination sneaks in. Touching it, the material feels slick and a bit oily; it won’t evaporate off your hands quickly. It gives off a mild, almost undetectable scent—nothing like the strong punch of some solvents. Storing it, you want to keep containers tight since it doesn’t love open air for long stretches.
Boiling starts happening around 191 °C at standard atmospheric pressure. This means Monomethyl succinate stands up to heat better than many lighter esters. You’re not likely to see it catch fire under regular handling, with its flash point sitting near 83 °C—a level where spill management still matters, especially in crowded labs or warehouses.
Its density hovers about 1.17 grams per cubic centimeter, so it settles below the surface of water. Talking solubility, it’s not eager to dissolve in water, but brings good compatibility with alcohols and ethers, which matters for those mixing chemicals batch by batch.
The Chemical Game: How It Reacts
Monomethyl succinate holds its own as a small ester. Under the right conditions—and with a touch of acid or base—it jumps into hydrolysis, splitting into methanol and succinic acid. This reaction lines it up as a starter for other chemical transformations, both in labs and in industrial-scale reactors. Stable at room temperatures and absent strong acids or alkalis, this molecule doesn’t give people too many surprises in day-to-day handling.
Oxygen in the air can push esters towards slow degradation, but Monomethyl succinate handles shelf-life better than many ether or amine counterparts. Still, manufacturers with any skin in the game store it sealed up, away from sunlight and heat. Otherwise, the material’s performance can start to drift, sometimes without clear warning signs.
Health and Safety—Know What You’re Handling
It’s easy to skip reading labels after years on the job, but Monomethyl succinate calls for gloves and eye protection any time you’re pouring or mixing. It can irritate the skin and eyes if splashed. Breathing in heavy fumes might upset sensitive airways, though this ester rarely gives off enough vapor to pose a bigger threat in well-ventilated spaces.
No animal studies suggest alarming toxicity, but long-term exposure hasn't been tested to the standards seen with pharmaceuticals or food additives. Responsible handling matters, and so does a spill kit nearby. Good ventilation and sensible use of personal gear answer most safety concerns before they even start.
Looking Ahead: Smarter Handling and Applications
Industry teams prize Monomethyl succinate because it steps in as a building block for fine chemicals, resins, and coatings. Waste disposal rules stay strict, especially since this chemical ties back to compounds found in living things. Recycling or re-purposing excess material can save money and cut down on waste—efforts paying off for companies working toward greener operations. More research on its biodegradation may open the door for new uses and safer environmental management down the road.
How should Monomethyl Succinate be stored and handled?
Why Storage Matters
Monomethyl succinate pops up in the lab for a lot of reasons, usually as an intermediate for synthesis work or a chemical building block fed into larger processes. At first glance, it may seem just another white powder or crystalline solid destined for the back shelf. Past experience shows that small mistakes with something this common can lead to big setbacks—lost product, wasted money, even safety scares.
Good Practice Starts with Temperature
Placing containers in a cool, dry place isn’t just an old chemistry cliché. Monomethyl succinate reacts poorly to moisture. Damp air, especially in storage rooms with inconsistent ventilation, causes caking and lumps inside the container. This mess complicates every step: weighing, scooping, and even mixing into solutions. Temperature swings accelerate this effect. For example, I once saw a solid batch of monomethyl succinate start sticking together after a few weeks in an overheated storeroom. It slows down work and risks altered reactivity.
Sealing Out Trouble
I always trust screw-cap glass bottles for anything sensitive to air or humidity. Containers made of polyethylene or polypropylene seem sturdy enough, but glass or high-density plastic with tight lids keeps out both moisture and any sneaky vapors. Labels peel off annoyingly fast from cold surfaces, so use permanent markers for clear, readable warnings. Some labs slap on hazard icons without hesitation, but knowing exactly what’s inside prevents both confusion and cross-contamination.
Safe Handling Isn’t Just Regulations
Goggles and gloves are a given—skin contact leads to mild irritation for most people, and nobody forgets the burning sensation after splashes. Many experienced lab techs don’t just grab the nearest gloves. Nitrile gloves work better than latex against chemicals like monomethyl succinate because they hold up longer, especially if any accidental spills happen.
Working under a fume hood, not open bench, keeps vapors and fine dust out of the breathing zone. Some look at small quantities and brush off these rules, yet inhaling even low levels of organic dust does lasting harm. Chemical safety data points out the risks, but as someone who’s helped clean up unexpected spills from rushed colleagues, I know speed does not equal safety.
Fire and Waste: Not Just Someone Else’s Problem
Monomethyl succinate doesn’t explode easily, but it burns. Failing to keep storage away from ignition sources invites disaster. Always keep fire extinguishers close, clearly labeled for chemical use (Class B and C). Even a small spark from static or a neglected heating plate could set off a chain reaction that no one wants.
Once containers empty, rinsing thoroughly before tossing them in the chemical waste stream prevents leftover solids from reacting with other discarded chemicals. Skipping this step sends unsafe compounds through waste treatment—bad news for both people and the environment. Waste labels should list the actual content and be dated, so there’s no guessing game for disposal managers.
Room for Improvement
Routine audits easily catch early signs of improper storage—sticky shelves, damaged labels, or old stock. Some organizations encourage regular walkthroughs and reward safe behavior; others rely on protocols buried in old PDFs. Investing in better shelving, clearer signage, and basic training for anyone handling chemicals provides payback in fewer incidents and less downtime.
Real safety doesn’t come from the number of rules posted in the hallway, but from habits built up by people who genuinely care about their work and the wellbeing of everyone in the building. Monomethyl succinate might look harmless, yet good habits make all the difference.
What is the recommended usage level of Monomethyl Succinate in products?
Understanding Where Monomethyl Succinate Sits in Formulation
Monomethyl succinate turns up in all sorts of household and personal care products. Companies use it mainly as a solvent, a skin conditioning ingredient, or as a building block for more complex molecules. It’s popular for good reason: it brings gentleness and a sense of safety compared to harsher chemicals. This alone matters to many families aiming to avoid skin irritation or exposure to volatile ingredients. Over time, regulators and trade groups studied its use inside and out, especially since it often ends up on the skin or in contact with food containers.
Most formulators rely on published toxicology studies and guidance from authorities like the European Chemicals Agency (ECHA) or the US Environmental Protection Agency (EPA). For Monomethyl Succinate, several safety assessments point to a general usage range of up to 5%. A lot of mainstream skincare products with this ingredient land somewhere between 0.1% and 2%, which usually brings the desired performance without causing irritation or overloading the formula. Of course, the exact amount depends on the product’s end use. Nobody wants more ingredients than necessary, especially if lower levels get the job done just as well.
Digging Into the Science
Scientific reviews underscore that this compound breaks down easily in the environment and doesn’t tend to accumulate in human tissue. Reports from the Cosmetic Ingredient Review (CIR) and other bodies flag only minor concerns at high concentrations. In patch testing, it rarely caused reactions. Still, anything can become a problem in heavy doses, so formulators keep an eye on total exposure from all sources.
Experience shows it works well in creams and lotions where it acts as a mild moisturizer without a greasy feel. In cleaning sprays, it gives a solvent kick to help lift dirt without adding much odor. Here too, keeping levels below 5% gives a balance of safety and cleaning heft, even if skin stays in direct contact—think of products for wiping counters or toys. Guidance always ties recommended use levels to total consumer exposure, so brands run risk assessments based on the intended application, not just the ingredient in isolation.
Practical Solutions for Product Makers
Every product team faces a question: how do you pick a level that balances safety, cost, and performance? Here’s where professional advice and published studies come into play. I once worked with a natural body wash maker who worried about skin sensitivity among their youngest customers. By referring to CIR guidelines and real-world patch test feedback, the final recipe capped Monomethyl Succinate at 1.5%. After launch, they saw plenty of positive reviews for mildness and effectiveness, which matched what studies suggested.
Brands should always test their unique formulations, check for allergy data, and follow legal limits in different countries. Labels and safety data sheets matter, too. If guidance updates as science grows, manufacturers adapt quickly—nobody wants a recall or lawsuits over ingredient safety. Staying informed and transparent gives both makers and customers greater trust in what ends up in their homes.
Moving Forward With Confidence
For anyone mixing up formulas—whether for skin, surfaces, or beyond—Monomethyl Succinate has its place. Stick within documented safe ranges, keep customers' real-life use in mind, and don’t hesitate to reach out to experts when things look uncertain. The industry already knows a lot about this ingredient, and with proper care, it keeps showing up as a useful, reliable choice for a cleaner, gentler world.
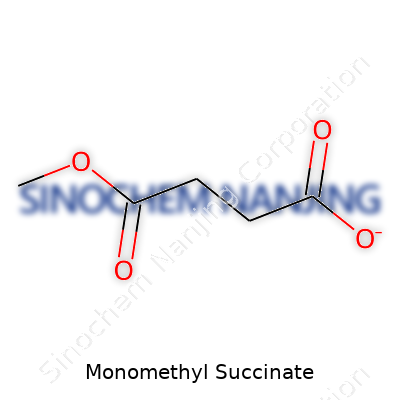
| Names | |
| Preferred IUPAC name | Methyl 3-carboxypropanoate |
| Other names |
Methyl hydrogen succinate Monomethyl ester of succinic acid Succinic acid monomethyl ester Monomethylsuccinic acid |
| Pronunciation | /ˌmɒnoʊˈmɛθɪl səksɪˌneɪt/ |
| Identifiers | |
| CAS Number | [3784-30-3] |
| 3D model (JSmol) | `3DModel:JSmol:C[C@@H](C(=O)O)C(=O)O` |
| Beilstein Reference | 1720862 |
| ChEBI | CHEBI:32312 |
| ChEMBL | CHEMBL565303 |
| ChemSpider | 24607242 |
| DrugBank | DB13933 |
| ECHA InfoCard | 13-771891585 |
| EC Number | 246-807-3 |
| Gmelin Reference | 85777 |
| KEGG | C06166 |
| MeSH | D017060 |
| PubChem CID | 13278 |
| RTECS number | WS6500000 |
| UNII | OI1E22F4V8 |
| UN number | UN3229 |
| Properties | |
| Chemical formula | C5H8O4 |
| Molar mass | 146.14 g/mol |
| Appearance | white crystalline powder |
| Odor | Odorless |
| Density | 1.27 g/cm3 |
| Solubility in water | soluble in water |
| log P | -0.46 |
| Vapor pressure | 0.03 hPa (20 °C) |
| Acidity (pKa) | pKa = 3.63 |
| Basicity (pKb) | 8.12 |
| Magnetic susceptibility (χ) | -45.6 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 7.3 mPa·s (25 °C) |
| Dipole moment | 3.07 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -854.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1549.7 kJ/mol |
| Pharmacology | |
| ATC code | A16AX30 |
| Hazards | |
| GHS labelling | GHS labelling: "Warning; H319 - Causes serious eye irritation; P264, P280, P305+P351+P338, P337+P313 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| Flash point | 126°C |
| Autoignition temperature | > 287°C (549°F) |
| Lethal dose or concentration | LD50 (oral, rat) > 5000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 5000 mg/kg |
| NIOSH | NIOSH: ESU |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2 mg/m³ |
| Related compounds | |
| Related compounds |
Dimethyl succinate Succinic acid Monomethyl fumarate Monomethyl maleate |

