Monohydrate Glucose: Insight into a Cornerstone of Industry and Science
Historical Development
Glucose monohydrate stands as one of those time-tested mainstays in both chemistry and daily life. In the early days, people recognized sweet plant extracts but knew little about their chemical basis. Over centuries, chemists found ways to isolate sugars and learned to describe their structures. Glucose monohydrate itself emerged through careful crystallization out of aqueous solutions—a discovery that shaped new opportunities in medicine, food, and more. Chemists in nineteenth-century Europe like Dumas and Liebig took glucose from an exotic laboratory curiosity to an ingredient everyone recognizes. By the twentieth century, sugar factories made it in bulk, opening the door for newcomers in food technology and pharmaceuticals. My first experience handling glucose monohydrate in a university lab echoed the historic thrill: a substance with roots in old-school science but firmly embedded in modern routines.
Product Overview
Monohydrate glucose typically presents as small, colorless or white, crystalline grains. Its key draw is its fine control of sweetness and ready solubility. People reach for it where bland sugars just won’t do—formulating energy supplements, IV solutions, and delicate confections that demand precision. The monohydrate form brings an edge of stability. It resists caking, pours smoothly, and withstands storage shifts with fewer headaches than its anhydrous cousin. The food industry values it for more than its taste: the structure supports reliable shelf life and supports clean labeling—an expectation I’ve seen become non-negotiable in both artisan and mass-produced goods.
Physical & Chemical Properties
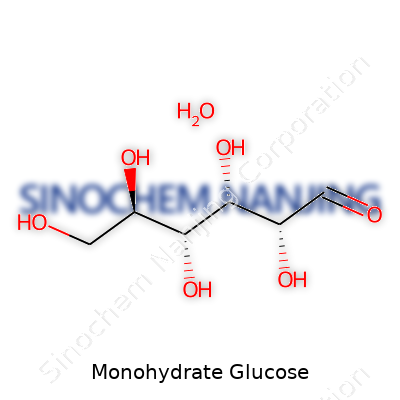
Chemically, this sugar arrives as C6H12O6·H2O. Each molecule latches onto one water molecule, which matters for how it dissolves and behaves in mixing. Melting comes around 146°C, and the crystals keep their cool under normal air. Its sweet flavor clocks in at about three-quarters that of table sugar, and it swings optical rotation to the right—a quality prized in labs that track raw materials. Glucose monohydrate dissolves in water far more readily than other carbs—an attribute that makes it near-essential for quick-mix applications. That telltale sweet taste and smooth mouthfeel regularly get harnessed in healthcare when gentle delivery is vital for patients and consumers alike.
Technical Specifications & Labeling
Anyone sourcing glucose monohydrate for high-stakes work keeps a close eye on technical parameters: purity, heavy metal content, microbial load, and particulate matter stand at the front of quality-checks. European Pharmacopoeia and USP monographs both spell out acceptable ranges for glucose content and lay down strictures on contaminants. You won’t find dyes, foreign matter, or strange odors—standards demand a squeaky-clean product. Labels must declare water content, batch number, and in medical settings the glucose source: wheat, corn, or potatoes, to settle allergy and dietary questions. Retail and industrial bags often declare origin, granulation, and expiry details, helping keep operators accountable from warehouse to end-user.
Preparation Method
Large-scale production usually starts with starch, tapped from corn, wheat, or potatoes. The raw starch faces acid hydrolysis or controlled enzyme action—think glucoamylase and alpha-amylase—until chains split down into single glucose molecules. From the resulting syrup, crystallization begins: cooling the mix leads glucose to settle out, trapping a single water molecule. This step calls for clean process water and careful temperature control—too much heat strips water, too little triggers unwanted byproducts. The art here isn't just science; it's about balancing cost and purity in a non-stop operation, with less waste and higher yields year after year.
Chemical Reactions & Modifications
Under the hood, glucose monohydrate behaves as a strong reducing sugar. Thanks to its free aldehyde group, it reacts with various chemicals, making it a star in medical tests—like Fehling's or Benedict's for simple detection of glucose in fluids. Glucose turns up as a launching pad for building other compounds: scientists oxidize it to produce gluconic acid or ferment it to churn out bioethanol and lactic acid. Under acid or alkaline conditions, it breaks down further, releasing a medley of carbon compounds valued in food flavoring and industrial solvents. Even when modified, the underlying chemistry means that safety and traceability need to remain at the top of the checklist, based on my time troubleshooting issues in both lab and manufacturing environments.
Synonyms & Product Names
Across countries, people might call glucose monohydrate "dextrose monohydrate," "grape sugar," or just plain "dextrose." Pharmaceutical circles mention "D-glucose hydrate." Food technologists sometimes shorten it to "corn sugar" based on how it’s sourced. Ingredient panels in North America often go with "dextrose," while European packaging leans toward "glucose." In lab supply catalogs, the substance pops up as "GlucoPure" or some other short-form branding, but at the molecular level, it’s all the same staple.
Safety & Operational Standards
Lab and plant users follow clear safety guidance: maintain dry storage, safeguard against airborne dust, and avoid cross-contamination with allergens like gluten when sourcing from wheat. Direct handling presents low toxicity—occasional dust might trigger mild respiratory or skin irritation, but nothing alarming based on decades of toxicology research. Workers rely on gloves, masks, and general hygiene, especially to maintain pharmaceutical and food-grade integrity. Globally, manufacturers align with ISO 9001 or ICH Q7 standards, putting documentation and traceability at the heart of responsible supply. My own experience in enforcing safety protocols reinforces how lapses at this step can undo the tightest manufacturing practices downstream.
Application Area
No other sugar slips so seamlessly between the worlds of medicine, food, and industry. Pharmacists rely on glucose monohydrate for IV infusions to stabilize energy levels in patients—paramedics deploy it to reverse diabetic hypoglycemia fast. The confectionery world values its quick-dissolving crystals to drive chewy candies and energy bars. Sports nutritionists depend on its rapid absorption; it sits in many gels and drinks used for performance boosts. Food preservation and fermentation both draw on glucose for yeast feed—a backbone ingredient for bread, beer, and pickles. Chemical manufacturers count on its reducing power for specialty syntheses or as a basic building block for downstream bioproducts.
Research & Development
Research keeps the story of glucose monohydrate alive. Newer studies in the nutraceuticals field search for ways to tailor release profiles, bind with trace minerals, or reformulate with fewer side-effects. Plant biotechnologists fine-tune starch extraction and enzymatic conversion to push down the environmental footprint. Clinical researchers follow how the body handles different carbohydrate forms, using labeled glucose to trace metabolic pathways in health and disease. My own time working on rapid diagnostics proved how tightly the science and supply of pure glucose intertwine in device reliability and patient trust.
Toxicity Research
Long-term safety research on glucose monohydrate anchors its widespread use. Years of ingestion and injection studies reveal a high safety threshold; the main real-world concern remains excessive intake leading to metabolic overload or, for vulnerable groups, spikes in blood sugar. Inhaled or splashed, the substance causes little more than mild irritation, and allergies tied directly to glucose remain vanishingly rare in published medical accounts. Toxicology panels still keep an eye on impurities: lead, arsenic, and microbial byproducts receive strict control before product shipment. That's the price of trust in both medical and food supply chains today.
Future Prospects
Looking ahead, glucose monohydrate finds itself right at the crossroads of sustainable chemistry and precision healthcare. Demand for plant-based, less-intensive sources keeps rising, driving enzyme engineering and fermentation science to the forefront. Bioplastic innovators look to glucose as a starter for carbon-neutral polymers. Personal health trackers and diagnostics might soon rely on more precise glucose derivatives to monitor blood sugar in real time, tailored for people with different metabolic profiles. For all the heady progress, the fundamentals remain unchanged: purity, consistency, and safety will shape the next chapter—qualities that have stood the test of time in my own work with this essential chemical.
What is Monohydrate Glucose used for?
Sugar That Powers More Than You Think
Walk through the aisles of any pharmacy, and you’ll probably find glucose monohydrate tucked away in more products than you realize. Growing up, I had my fair share of science classes, but I never thought twice about what went into an electrolyte drink or an IV solution at the hospital. Years later, after some time spent volunteering in clinics and reading up on nutrition, I’ve come to realize that this simple sugar is the backbone in many essential uses.
Healthcare’s Reliable Energy Source
In hospital settings, monohydrate glucose keeps patients going when their bodies can’t. Doctors often turn to it for intravenous infusions. If someone has low blood sugar or struggles to eat after surgery, a glucose drip stabilizes energy and helps prevent dangerous drops. Diabetics rely on it during hypoglycemic emergencies. Medical staff trust its purity and consistency to deliver quick relief. The body knows how to handle glucose, making it one of the safest go-tos for a quick metabolic pick-me-up.
Sports, Supplements, and Recovery
I’ve watched athletes chug glucose-based drinks after tough matches and gym sessions. Glucose monohydrate ends up in many of these rehydration powders, offering a fast-absorbing carbohydrate for muscles and minds running on empty. Rapid absorption matters in recovery, and this sugar handles the job. With the World Health Organization including glucose in their oral rehydration solutions, it isn’t just for athletes—children battling dehydration from stomach bugs around the globe also benefit from this blend.
Food Industry: More Than Just Sweetness
Baking and candy makers have long understood glucose monohydrate’s value. It prevents crystallization in confections and balances texture in baked goods. In my kitchen, I’ve found it helps keep preserves and sauces smooth, stopping dreaded graininess. Many processed foods use it not just for its sweet taste, but for its effect on mouthfeel and shelf life. Its ability to stabilize flavors and colors—something I learned from talking to a small bakery owner—means fewer surprises for the cooks and bakers who use it every day.
Tablets and Capsules in Pharma
The pharmaceutical world counts on glucose for more than infusions. Tablet makers often use it as a binder and filler. A good pill should dissolve easily and not fall apart in your hand. Glucose monohydrate enables consistent dosage and helps active ingredients reach the bloodstream efficiently. During my time reading quality assurance reports from a supplement factory, I saw how even a tiny percentage of the formulation can make or break a batch’s performance.
Expanding Into Animal Health and Biotechnology
Nutrition isn’t just for humans. Veterinarians reach for glucose monohydrate to help animals bounce back from illness. It flows into feed mixes, supporting growth in young livestock and recovery in sick pets. The biotech industry uses it as food for cells growing in petri dishes, fueling research into new medicines and vaccines. Pure, predictable energy is a big deal whether you’re caring for a person or producing high-tech medicines.
Solving Shortages and Trusting Quality
As demand rises, shortages and price jumps threaten both clinics and communities. Governments and suppliers must invest in reliable supply chains and quality testing. In a world where counterfeits threaten safety, strong regulation keeps patients safe. From hospitals to home kitchens, a trustworthy source of glucose monohydrate means fewer emergencies and better outcomes for everyone who depends on it.
Is Monohydrate Glucose safe for consumption?
What Exactly Is Monohydrate Glucose?
Monohydrate glucose goes by a simpler name in the kitchen: glucose or dextrose. It’s a simple sugar and pops up everywhere, from bakery shelves to intravenous drips at the hospital. The food industry uses it in candies, baked goods, and drinks, mostly because it dissolves well and provides instant sweetness. Science types know it as glucose with a bit of water attached, and your body instantly recognizes it the moment it hits your bloodstream.
Digestion and Health
Every time you eat bread, fruit, or potatoes, your digestive system breaks the starches down into basic sugars like glucose. Monohydrate glucose works just like this, so the body uses it for quick energy. Diabetics know all about glucose because their bodies have a harder time controlling blood sugar. For most people, eating foods with glucose doesn’t cause trouble unless consumed in very high amounts, the kind that overwhelms your body’s ability to keep blood sugar at healthy levels.
Sourcing and Quality Concerns
If you’ve ever wondered where this ingredient comes from, manufacturers typically extract it from corn or sometimes wheat. High-quality production matters because nobody wants contaminants in what they eat. Reliable food standards exist in most countries, and foods carrying glucose monohydrate pass through safety checks. The United States Food and Drug Administration (FDA) and the European Food Safety Authority (EFSA) list it as safe for direct use. Reports show that proper purification and controlled manufacturing ease concerns about impurities such as heavy metals or solvents.
Scientific Perspective
Medical researchers spent decades studying glucose, both as a dietary ingredient and medical supplement. For emergency cases, paramedics give glucose solutions to people with dangerously low blood sugar. Hospitals rely on this same substance, sometimes mixed in IV fluids, to support patients who can’t take solid foods. In the context of sports and recovery, glucose-based drinks help replenish lost energy. The risks tied to this ingredient have more to do with excessive intake than the molecule itself. Frequent overconsumption increases the chance of weight gain, insulin resistance, and other metabolic issues, much like eating excess sugar of any kind.
Label-Reading and Practical Advice
Reading food labels for sugar content matters a lot, especially for those with diabetes or trying to avoid added sugars. Monohydrate glucose can sneak into processed snacks, soft drinks, or ready meals, making daily sugar totals climb without much notice. Keeping consumption moderate fits expert guidelines. The American Heart Association encourages adults to limit added sugars to help prevent chronic disease. Habit makes a difference: choosing fresh fruits, whole foods, and natural sources of sweetness lowers risk and improves well-being. There’s nothing wrong with using a glucose supplement when needed, but using common sense and reading up on what’s in packaged foods goes a long way toward better health.
Exploring Alternatives
Many people searching for ways to reduce sugar intake experiment with stevia, monk fruit, and other sweeteners. Some alternatives cut calories, while others mimic the taste of sugar. These substitutes may offer solutions, but they won’t entirely erase the need for moderation. Tastes can shift over time, and focusing on a balanced diet with fewer processed foods keeps things simple and safe.
What is the shelf life of Monohydrate Glucose?
Understanding Monohydrate Glucose isn’t Just Science—It’s Safety
Glucose monohydrate might seem like another chemical name out of a textbook, but people rely on it every day. Whether it’s in hospitals for fluid therapy, athletes seeking energy boosts, or the food industry adding sweetness, its shelf life can end up deciding if someone feels better or if a product does what it’s supposed to. Let’s get right to it: unopened monohydrate glucose stored in a sealed container can last about three to five years. That timeframe depends heavily on the environment—temperature shifts, moisture sneaking in, sunlight exposure, or poking into the container before it’s needed will speed up its decline.
What Kills Shelf Life?
Sitting in a pharmacy for years, I saw staff treat glucose monohydrate like a rock: stable, reliable. Laboratory-grade containers sit on cool dry shelves and usually hit their expiration dates without noticeable change. But in a hot, humid dispensary or a kitchen, those same containers start clumping, hardening, and growing off smells. Glucose pulls water out of the air—leave the lid loose and the powder gets sticky or forms solid chunks. These changes mean it might deliver less predictable results, especially in a hospital where accuracy isn’t a suggestion, it’s the rule.
Humidity is enemy one. At over 60% relative humidity, glucose soaks up moisture and can start to break down. Heat isn’t far behind. Temperatures above 25°C (77°F) speed up chemical changes, leading to yellowing and degradation. Even tiny bits of contamination from improper handling, dirty scoops, or unwashed hands start a slow spiral: mold growth, microbial contamination, and reduced safety. Every time someone dips a scoop or finger into the container, it’s another invitation for spoilage.
Why Shelf Life Should Matter to Everyone
Medication and supplement recalls pop up for many reasons. Inconsistent shelf life might seem small, but it can cost lives. A hospital relying on old glucose powder for IV drips risks patient safety if the powder delivers less than what’s printed on the label. Athletes using expired glucose supplements for energy may not get the boost they expect—and could face stomach upsets or worse if mold or bacteria have moved in.
Food producers have another headache. The food industry leans on trust—if customers open a box of sweets and see clumped sugar or taste something off, they call customer service or stop buying. Baked goods and candies depend on the sugar being free-flowing and tasting right, so the shelf life of glucose monohydrate means lost money if suppliers or end users don’t respect it.
Tried-and-True Ways to Maximize Shelf Life
Storing glucose monohydrate seems easy—just keep it in a cool, dry, sealed container away from light. But reality gets in the way. Storage rooms heat up. Humidity inches up in rainy seasons. Even the best facilities can get sloppy with buckets and scoops. Making it policy to use airtight containers, silica desiccant packs, and strict “first in, first out” inventory helps. Training every staff member handling glucose on proper scoop hygiene sounds simple, but saves far bigger headaches than you’d expect. Digital tracking of batch numbers and expiration dates beats sticky notes or memory games any day—especially as regulations tighten around traceability.
Glucose monohydrate isn’t kryptonite. It’s safe, stable, and widely trusted when treated with respect. The lesson is simple: shelf life isn’t only about the date stamped on the label, but the discipline behind storage and use every single day.
How should Monohydrate Glucose be stored?
Why Proper Storage Actually Matters
People might think storing a common lab or food ingredient like monohydrate glucose can’t be too complicated. Here’s where experience steps in: even the most routine chemical turns into a headache if left in the wrong spot. I saw a bakery scramble once after their supply went clumpy from humidity, and staff spent an hour pounding it back into usable form. Wasted time, wasted money, and off-quality products. Whether you're running a kitchen, a lab, or a warehouse, poor storage choices hit hard in daily life. A little forethought can prevent ruined ingredients and lost money.
Direct Approach: Cool, Dry, and Sealed Makes All the Difference
Glucose monohydrate likes a predictable environment. I’ve worked in places where they pushed pallets near open doors or stashed product above ovens—never a good plan. I learned the hard way that even a little extra moisture in the air turns that fine powder or those neat crystals into a sticky mess. So, I always look for storage in an area well below 30°C. Cooler is better, especially during humid months. Dry means dry: a room with a dehumidifier, if possible, or at least shelving that stays well away from splashes, leaks, or steam. Airtight containers save a lot of disappointment. Once, a snapped lid meant the last scoop of glucose was nearly solid. Manufacturers are right to pack this stuff in thick bags or sturdy drums with tight seals.
Don’t Ignore the Details: Cleanliness and Labeling
A clean shelf is an underrated hero. More than once, cross-contamination crept in when someone stored glucose next to strong-smelling acids or left powder residue from another project. Good habits, like wiping down shelves and not repurposing containers without a wash, stop a lot of mistakes. Clear labeling shaves minutes off every search, especially if you juggle multiple sugars—no more sniff tests or sticky hands. Rotating older stock up front keeps everything fresh, with less risk of caked powders or odd odors.
Safety and Compliance Count
Dust isn’t just a cleaning chore; too much floating glucose powder sets the stage for respiratory irritation and, in extreme cases, can even contribute to rare dust explosions. From my experience, decent ventilation and regular sweeping beat drama and paperwork later. Safety data sheets give storage bullet points for a reason. Compliance isn’t red tape. Skipping the recommended storage temperature or humidity exposes companies and schools to all sorts of trouble—from product recalls to spoiled experiments.
Getting the Most Value from Your Supply
Keeping a close eye on temperature, humidity, container condition, and shelf hygiene stretches the life of every kilo. Some places invest in climate-controlled rooms; others make do with closet-sized storage and good old-fashioned vigilance. In either case, thinking ahead stops unnecessary spoilage and saves cash. Within the food industry, and certainly in labs, documentation on storage conditions means you catch small problems before a full batch gets ruined. In my work, I’ve seen colleagues jot a quick log entry whenever they spot condensation or humidity spikes, and it catches issues early.
Simple Steps, Better Results
Storing monohydrate glucose doesn’t need a science degree, just some attention and care. Stick with cool, dry spots, keep everything airtight, and keep labels and logs up-to-date. These habits build quality and safety into every batch you use. I’ve watched teams who follow these basics consistently outshine competitors, avoid rework, and boost their bottom line. A good supply of glucose is only as good as where and how it’s stored—and the daily habits you put in place matter more than most realize.
Are there any side effects of Monohydrate Glucose?
Looking Past the Sweetness: Monohydrate Glucose in Daily Life
Monohydrate glucose, often just called glucose, lands in everything from medical treatments to sports supplements. It shows up in hospitals as a staple for energy during IV infusions. Bakers use it for texture in foods; athletes reach for it to refuel muscles fast. The body knows this sugar well. In fact, it treats glucose as a go-to energy source in every cell.
Short-Term Side Effects: What Happens Right After Consumption
Most healthy people notice little after eating or drinking something with glucose monohydrate, unless the dose is far above a typical serving. Climbing glucose levels can bring a wave of thirst, dry mouth, or a burst of energy followed by fatigue. Some people complain about bloating, gas, or a bit of nausea if they take it on an empty stomach. People with diabetes need to pay special attention, since rapid rises in blood sugar open the door to much more than mild discomfort.
Why Blood Sugar Spikes Matter
Regular use of monohydrate glucose shakes up the balance the body works so hard to maintain. After glucose intake, insulin jumps in to move sugar out of the blood and into the cells. Healthy adults hardly notice this dance. Blood sugar comes up, falls back down, and life goes on. For people with insulin resistance or diabetes, this balancing act feels much riskier. As blood sugar climbs and insulin struggles to catch up, side effects like blurred vision, trouble focusing, fatigue, and headaches can follow. Chronic overexposure pushes the metabolism toward trouble: weight gain, triglyceride spikes, and other changes that often slip under the radar.
Allergic Reactions and Rare Cases
Monohydrate glucose itself rarely triggers allergies. The body deals with it just like homemade sugar. Filler ingredients in tablets or supplements, though, can spark unexpected reactions in sensitive folks. I’ve seen patients chalk up stomach cramps or hives to glucose, only to find some coloring, stabilizer, or preservative hiding in the blend. Read ingredient lists with care if you’ve got food allergies or sensitivities.
Looking at Long-Term Consumption
In the long run, a diet loaded with simple sugars—glucose included—chips away at metabolic health. The American Heart Association warns about high sugar intake for a reason. More sugar means more work for the pancreas, more insulin pumped out, and an extra push toward high blood pressure and fat in the liver. I recall counseling a young athlete who thought daily glucose drinks fueled better workouts. Over time, blood tests told a different story: inflammation rising, blood sugar creeping up. Cutting back paid off, and health markers improved.
How to Avoid Problems
Pay attention to labels on supplements and processed foods. Stick to moderate amounts, especially outside of exercise or recovery from illness. People with diabetes or prediabetes need to track carbohydrate sources closely. I’ve found registered dietitians and certified diabetes educators to be great partners for patients wanting smart, safe plans. Anyone who faces nausea, headache, or a racing heart after taking glucose should talk to a healthcare professional.
Simple Choices for Better Outcomes
Safe and sensible use of monohydrate glucose starts with recognizing how your body handles sugar. A healthy adult with no blood sugar issues can tolerate moderate use with little risk. People with metabolic conditions benefit from a more cautious approach. Nobody needs to cut glucose entirely, but respecting its effects goes a long way toward long-term health.
| Names | |
| Preferred IUPAC name | D-glucopyranose monohydrate |
| Other names |
Dextrose monohydrate Glucose monohydrate Corn sugar Grape sugar Blood sugar D-Glucose monohydrate |
| Pronunciation | /ˈmɒn.oʊ.haɪ.dreɪt ˈɡluːkoʊs/ |
| Identifiers | |
| CAS Number | 5996-10-1 |
| Beilstein Reference | 1724621 |
| ChEBI | CHEBI:17234 |
| ChEMBL | CHEMBL1239811 |
| ChemSpider | 5793 |
| DrugBank | DB09530 |
| ECHA InfoCard | 03f843932ac5-49e8-bbb0-57add1ae72ba |
| EC Number | 200-075-1 |
| Gmelin Reference | **63252** |
| KEGG | C00221 |
| MeSH | D006023 |
| PubChem CID | 5793 |
| RTECS number | LC0700000 |
| UNII | 5SL0G7R0OK |
| UN number | UN 3245 |
| CompTox Dashboard (EPA) | DTXSID2022222 |
| Properties | |
| Chemical formula | C6H12O6·H2O |
| Molar mass | 198.17 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.54 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.24 |
| Vapor pressure | Vapor pressure: <0.01 mmHg (25°C) |
| Acidity (pKa) | 12.28 |
| Basicity (pKb) | 12.62 |
| Magnetic susceptibility (χ) | -6.1×10⁻⁷ |
| Refractive index (nD) | 1.333 |
| Viscosity | 800-1200 cP |
| Dipole moment | 6.5 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.6 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -1274 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2805 kJ/mol |
| Pharmacology | |
| ATC code | A11GA01 |
| Hazards | |
| Main hazards | No significant hazards. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Not a hazardous substance or mixture. |
| Precautionary statements | Precautionary statements: P264, P270, P301+P312, P501 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Autoignition temperature | 440 °C |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 25,800 mg/kg |
| LD50 (median dose) | > 25,800 mg/kg (rat, oral) |
| NIOSH | GM3675000 |
| PEL (Permissible) | 15 mg/m³ |
| REL (Recommended) | 300 mg/kg bw |
| Related compounds | |
| Related compounds |
Anhydrous glucose Fructose Galactose Mannose Sucrose Lactose Maltose Dextrose |

