Methylphenyldichlorosilane: Progress, Promise, and Caution in a Tiny Molecule
Historical Development
Innovation moves quickest where chemistry and industry meet, and methylphenyldichlorosilane deserves its place in that story. Early work with organosilicon compounds showed both ambition and some guesswork. People needed compounds that were neither as fragile as early plastics nor as limited as simple solvents. Chemists in the mid-20th century looked for molecules that could improve stability and diversify the range of available synthetic polymers. Methylphenyldichlorosilane, with its mixture of methyl and phenyl groups clinging to a silicon atom and two reactive chlorines, popped up as a practical answer. Key gains in silicon chemistry before World War II led to industrial applications, moving organosilanes from laboratory curiosities to building blocks for everything from fancy surface coatings to advanced electronics. Methylphenyldichlorosilane joined this lineup as both a promising monomer and as an intermediate—one of those odd, slightly dangerous, quietly important chemicals found far from household shelves, but fundamental for modern industry.
Product Overview
Nobody keeps a bottle of methylphenyldichlorosilane on the kitchen counter. For folks in materials science, polymer chemistry, and electronics manufacturing, it has become a staple. The molecule offers both a handle (the reactive Si-Cl bonds) and a unique property set thanks to its phenyl and methyl attachments. It resists hydrolysis in controlled settings, but reacts eagerly with moisture in the open air, releasing hydrogen chloride fumes—a memorable smell and a nasty irritant. Its usefulness comes from the ability to introduce both rigidity (phenyl) and flexibility (methyl) into the silicon-oxygen backbone that defines silicones. Blending structure and reactivity, methylphenyldichlorosilane supports custom silicone materials, water-repellent coatings, and specialized adhesives.
Physical & Chemical Properties
At room temperature, methylphenyldichlorosilane looks like a clear, colorless liquid with a sharp, biting odor. Physically, its boiling point hovers not too far above water's, so it readily vaporizes—a point to remember, since inhalation can cause marked discomfort. It dissolves in certain organic solvents, not water. Chemically, its defining trait lies in those two chlorine atoms bonded to silicon. Contact with water breaks those bonds and releases hydrogen chloride, while the core Si-C connections (to the methyl and phenyl groups) provide resilience and compatibility with many organic reaction partners. Understanding these traits reduces surprise and keeps those who work with the compound out of the emergency room.
Technical Specifications & Labeling
Factories receiving methylphenyldichlorosilane don’t trust luck; they look for high purity, meaning as few contaminating silanes and chlorinated byproducts as possible. Labeling must call out the fuming, corrosive nature of the liquid, and containers must seal tightly. Shipping and storage often demand metal, glass, or high-grade polymers to handle both the flammable and corrosive risks. As someone who’s handled silanes (with a respirator, gloves, and plenty of respect), I’ve seen teams take every detail seriously, even if the paperwork weighs heavy. Mishandling means ruined batches and potential injury, so attention at this stage never gets old.
Preparation Method
Chemists typically start with methylphenylsilane and bombard it with chlorinating agents like chlorine gas or thionyl chloride. In industry, careful temperature control and moisture exclusion become priorities. Each batch involves process control—too much heat or oxidizing power risks side reactions. Those who work in fine chemical production see this challenge up close. Some facilities now seek less hazardous, more sustainable synthetic methods, pushing for greener reagents or closed-loop chlorine management. Even as overall volumes remain small, the drive for cleaner manufacturing keeps pushing forward.
Chemical Reactions & Modifications
Methylphenyldichlorosilane’s main strength comes from transformation, not stasis. The Si-Cl bonds readily yield to water, alcohols, or amines, opening up routes to silanols, alkoxysilanes, and cross-linking agents. Siloxane polymerizations turn this molecule into flexible, heat-resistant materials or hard, glass-like networks, depending on the blend of co-monomers. It acts as a building block for hybrid organic-inorganic networks seen in advanced composites, protective finishes, and even certain optoelectronic devices. To get the best results, chemists experiment with reaction times, catalysts, and purification, learning with each run what tweaks get the desired product.
Synonyms & Product Names
“Methylphenyldichlorosilane” carries alternate names depending on labeling standards and local chemistry culture. You sometimes see “Phenyldichloromethylsilane” or “Dichloromethyldiphenylsilane” (though that last one really means the diphenyl version). Product literature in North America often abbreviates to “MPhDCS.” Synonyms pop up in catalogues and research papers, confusing those new to silicon chemistry. Consistency matters in the lab notebook, so I always double-check identifiers before ordering—or risking the wrong bottle for a late-night reaction.
Safety & Operational Standards
Cracked skin and streaming eyes teach quick lessons about methylphenyldichlorosilane’s hazards. Strict fume hood work, chemical-resistant gloves, and face protection aren’t up for debate. Spills demand neutralization and thorough ventilation, not improvisation; policy evolved after plenty of avoidable accidents. Institutional standards follow guidelines from agencies like OSHA and the European Union’s REACH—containment, emergency ventilation, and rapid spill response help protect both workers and the planet. Training saves lives, and ongoing drills ensure people remember that seriousness at all levels prevents the unthinkable.
Application Area
Few consumers see this compound directly, but its legacy runs deep. Silicone manufacturing, electronic coatings, rubber enhancement, and even anti-graffiti films all draw on its custom-tailoring ability. Researchers use it to craft new organosilicon molecules that bridge the gap between hard electronic substrates and soft, bendable sensors. Demand comes from industries building microchips as much as from those finishing automotive components.
Research & Development
Labs chasing the next best material keep methylphenyldichlorosilane in the toolkit. Its mixture of organic and inorganic traits sparks curiosity among polymer scientists striving for stable, shape-memory devices, medical implants, or flame-retardant polymers that outperform older formulas. Active research studies how tweaking the ratios of phenyl, methyl, and other substituents shapes electronic, mechanical, and surface properties. Environmental and health-conscious production remains a driving force, with some projects focusing hard on waste minimization and solvent recovery. Scientific journals routinely publish improved synthesis strategies, greener functionalizations, and fresh data on lifetime performance in end-use applications.
Toxicity Research
Early reports flagged methylphenyldichlorosilane as corrosive to eyes, skin, and mucous membranes, and more focused studies confirmed its hazards. Inhalation triggers coughing and lung irritation, while exposure to hydrolysis products—hydrogen chloride gas and silanols—can escalate the risks. There’s ongoing research into chronic effects, as workers seek safer means of exposure monitoring and the chemical community pushes for more thorough risk assessment. Recent data highlight the need for tighter personal protection and better engineering controls. Some groups explore containment and recovery, both to shield staff and to catch escapees before environmental release.
Future Prospects
Expect methylphenyldichlorosilane to stay behind the scenes, powering progress in silicone diversification and high-tech coatings. Green chemistry will shape its manufacture as industry faces tougher regulations and higher expectations. Researchers hope for less hazardous derivatives, more recyclable product lifecycles, and perhaps next-generation silicones that combine performance with a lower ecological footprint. Investment in on-site treatment and safer synthesis routes looks set to grow. Professionals and researchers keep one eye on short-term performance and another on long-term responsibility, knowing that every gain in safety and efficiency has ripple effects across the chemical landscape. Methylphenyldichlorosilane’s future promises both challenge and opportunity for anyone building the materials of tomorrow.
What are the main applications of Methylphenyldichlorosilane?
Chemical Industry as the Foundation
You can’t spend much time around a chemistry lab or a manufacturing plant without running into methylphenyldichlorosilane. It’s a mouthful, yet it plays a big role behind the scenes. The compound brings together silicon, chlorine, a methyl group, and a phenyl group, turning it into a versatile building block for a lot of everyday products.
Silicones: More Than Just Sealants
Silicones show up in kitchens, construction sites, electronics, and even personal care products. The path to those long, heat-resistant polymers often starts with methylphenyldichlorosilane. Factories rely on it as a starting material to create specialty silicones. Mixing methyl and phenyl groups into the siloxane backbone doesn’t just sound like a clever tweak; it actually lends better temperature resistance and flexibility to everything from gaskets to coated fabrics. High-performance silicone rubber, the kind that keeps car engines humming along in summer or winter, often owes some of its resilience to this compound.
Electronics Depend on Tailored Coatings
Every circuit board counts on insulation and protection. In semiconductor manufacturing, small improvements in chemical purity or thermal stability make a difference between a failed microchip and a product ready for market. Methylphenyldichlorosilane helps shape specialized silicones that can stand up to the demands of ever-smaller, more powerful devices. The phenyl group improves performance under high voltage and at extreme temperatures, giving engineers one more tool to push tech a little bit further.
Optics and Specialty Glass
Growing up, I always liked the clarity of a clean window or a polished lens. Most folks don’t realize chemistry makes a difference here, too. Some advanced glass coatings and optical elements count on methylphenyldichlorosilane as a surface modifier or as part of sol-gel processes. You end up with coatings that resist scratches, fog, and water spots—less wiping, longer-lasting clarity.
Water Repellency and Surface Protection
Rainproof jackets, protective sprays for shoes, and even treated building materials can trace some of their magic back to this compound. Manufacturers use it to develop agents that form a thin, water-repellent layer on textiles, stones, and ceramics. Instead of soaking up water or dirt, treated surfaces stay cleaner and last longer. This technology isn’t just for fancy outdoor gear. Think bridges, monuments, or even subway stations—surfaces hold up better against the weather and daily grime.
A Few Challenges, Some Responsible Solutions
Handling chemicals that include chlorine takes care and experience. Production and storage call for tight controls to avoid hazards. Companies now look to closed systems and improved ventilation to keep workers and neighbors safe. And as global regulations keep evolving, there’s more demand for green chemistry, waste treatment, and recycling where possible. By investing in education, responsible sourcing, and research into safer alternatives, the industry can both meet demand and reduce its footprint.
Looking Ahead
Methylphenyldichlorosilane isn’t a household name, but it touches a lot of the technology we rely on. Its continued use depends on a mix of chemistry know-how, safety culture, and responsible innovation—because as we keep pushing for stronger, lighter, and smarter materials, each ingredient and each process matters.
What are the safety precautions for handling Methylphenyldichlorosilane?
Seeing the Risks Clear
I’ve spent more time than I’d like in labs that smell like the edge of a swimming pool. Methylphenyldichlorosilane ranks high among those chemicals that demand respect—think of eyes stinging from chlorine, but far worse if you mess up handling it. This stuff can burn your skin, tear up your lungs, and reacts with water faster than you can blink, releasing hydrogen chloride gas.
Personal Protective Equipment Isn’t Optional
I have watched folks stroll into a chemical storage area wearing just a pair of goggles, thinking that’s enough. With methylphenyldichlorosilane, I always go for the full kit: chemical splash goggles, a face shield for good measure, nitrile gloves that lap my wrists, and a sturdy lab coat. I’ve seen old-timers insist on rubber aprons. For jobs involving bulk transfers or any risk of splash, rubber boots round out the outfit. It’s not being overly cautious—splashes find every gap if you give them the chance.
Know the Territory: Fume Hoods and Ventilation
Poor air movement turns a small spill into an emergency. Every gram handled goes straight under a functioning chemical fume hood. Never risk it in an open space—no project is too small to skip this step. I’ve seen rooms cleared because a colleague poured this compound too casually; after that, I swore to insist on ventilation, even for seemingly routine tasks.
No Room for Water: Dry Means Safe
I made the mistake once of storing moisture-reactive chemicals near a benchtop sink—never again. Even humidity in the air can trigger a nasty reaction. Silica gel packs in storage cabinets prevent accidental hydrolysis. When diluting or transferring, always use glass or appropriately rated plastic, fully dry, and keep emergency showers ready, but never plan for them to be your first line of defense.
Handling and Storage Habits
Hands touch doorknobs, pens, safety glasses, then the bottle—before you realize it, contamination trails through the whole lab. I started keeping clean and dirty gloves separated, making one way in and out of handling areas. Measure inside a tray, never over an open benchtop. Store containers tightly sealed with labels showing exact risks. This isn't about ticking boxes—it’s about knowing nobody touches a surprise spill because someone else got lazy.
Never Work Alone
Certain chemicals tolerate single work shifts. Methylphenyldichlorosilane doesn’t. Always have a partner or someone close enough to respond in case things go sideways. Every time I thought, “It’ll just take a moment, I’ve got this”—that’s precisely when I needed backup.
Spill Kits and Emergency Response
No substitute exists for a spill kit nearby. Not once have I met a person glad they had to use one, but everyone’s grateful it was close by when something spilled. I keep neutralizing solutions, proper absorbent materials, and clear instructions at arm's reach. Practice matters: knowing exactly what to do when your gloves get splashed keeps panic at bay.
Training Builds Safety Culture
Fatigue makes shortcuts tempting, and new hires skip steps out of ignorance, not malice. Regular refreshers—hands-on, not just slideshows—stop accidents before they begin. I’ve learned the most from walking through mock scenarios, with the pressure on, making mistakes where it’s safe to do so.
Small Habits, Lasting Impact
Working safely with methylphenyldichlorosilane comes down to doing the small things right, every time. The margin between a routine day and a dangerous one often shrinks to a single splash, roll of the dice, or skipped checklist. The habits I keep now go beyond one chemical—they’ve shaped how I approach every risk in the lab.
How should Methylphenyldichlorosilane be stored?
Looking Close at the Risks
Some chemicals invite extra worry the minute you see their name, and methylphenyldichlorosilane earns every cautionary glance. This substance, which plays a big part in the production of silicones, throws off strong fumes and reacts quickly with water. Those facts change the rules on storage. This isn’t a product you can tuck away in an all-purpose storeroom and forget.
Moisture: The Enemy
Every laboratory technician learns fast to run from leaks and drips when handling methylphenyldichlorosilane. Even a bit of moisture triggers white smoke and the quick release of hydrochloric acid. Damp air or an unnoticed drop of water means dealing with highly corrosive acid and a nasty cleanup. So, dry storage matters almost more than anything. A sealed, airtight drum or container—usually made of glass or high-grade stainless steel—shields against any contact with humidity.
Control Temperature, Control Danger
In hot summers, I’ve watched the wrong warehouse choice wreck tens of thousands’ worth of specialized silicones. Heat speeds up decomposition and kicks off gas buildup inside containers. Once, because of a faulty air conditioner, a barrel’s pressure valve failed, which called for a mid-day evacuation and a visit from the hazmat team. So, keeping methylphenyldichlorosilane at a cool, stable temperature—away from machinery and sunlight—stands at the core of safe handling. Industry consensus sets the upper recommended storage temperature below 35°C (95°F).
Keep It Separate
Some folks treat chemical compatibility charts as optional reading. Those people don’t last long in facilities handling chlorosilanes. Mixing up storage near other reactive chemicals or acids can trigger fire or release toxic fumes. To avoid that, you need a dedicated area, so accidental spills have nowhere to migrate. Make sure acids, strong bases, and oxidizing agents stay far from methylphenyldichlorosilane bins. Fireproof walls and proper labeling help, but real discipline means routine checks and secure barriers.
Ventilation and Safety Gear
People forget one whiff of methylphenyldichlorosilane fumes can scald the lungs and eyes. Every good storage area uses reliable fume-scrubbing ventilation. Even when barrels haven’t leaked, a worn gasket or cap can slowly release vapors. I keep a gas monitor clipped to my coat when entering the storage space. Regulators recommend chemical splash goggles, gloves, and flame-retardant lab coats be kept right by the storage entrance. Drills and real-world training keep new hires ready in case someone opens a valve by mistake.
Documentation: Life Saver, Not Just a Chore
It’s tempting to see paperwork as nothing more than a legal obligation, but in my own experience, lapses in tracking have led to mystery spills and nasty surprises. Recording the contents, dates, lot numbers, supplier details, and opening dates gives clear answers in an emergency. Up-to-date safety data sheets stay posted near the entrance.
Small Steps That Add Up
It’s the little things that keep disaster at bay. Simple actions like checking drum seals, ensuring fire extinguishers stay close at hand, storing only what’s needed, and rotating old stock out make every storage room safer. In chemical handling, no shortcut ever pays off in the end. Working with methylphenyldichlorosilane reminds me that the line between routine and emergency often comes down to storage choices made weeks—or years—ago.
What is the chemical formula and CAS number of Methylphenyldichlorosilane?
Understanding This Key Organosilicon Compound
Methylphenyldichlorosilane shows up often in chemical and manufacturing circles. Its chemical formula, C7H8Cl2Si, marks it out as a distinct member of the organosilicon family. The CAS number for this compound is 138-65-8, which serves as a unique identifier used by researchers and suppliers worldwide. Having these identifiers handy means researchers, students, and companies can avoid mix-ups and source the right compound for their needs.
Why Methylphenyldichlorosilane Gets Attention
Talk to anyone who spends time in a silicone lab, and they’ll mention how this compound plays a role in synthesizing specialty polymers and resins. Its two chloride groups make it a go-to reagent for modifications to silicon backbones or for introducing organic groups into silicon-based chains. Some resins produced from it are prized for boosting resistance to heat and keeping mechanical properties stable—qualities that matter for engineers producing coatings, adhesives, and specialty composites.
Its structure pairs a methyl group with a phenyl ring—simple, but that combo unlocks specific chemical reactivity that other silanes can’t match. Looking at industrial safety sheets, you’ll see why it draws respect. Methylphenyldichlorosilane reacts quickly with moisture, releasing hydrogen chloride gas, which can cause burns or breathing issues. Anyone storing or using it needs proper gear, ventilation, and training. I’ve seen firsthand the importance of paying attention to storage methods—glassware, dry atmosphere, and correct labeling reduce both accidents and wasted product.
Safety, Ethics, and Environmental Responsibility
Factories and labs turning out tons of this compound face a responsibility to protect workers and neighbors alike. Improper disposal sends hydrochloric acid and siloxane waste into waterways and soil. This isn’t just inconvenience—it's an ethical question. Qualified handlers must neutralize or recycle by-products and avoid dumping drains. Companies certified for environmental management know these routines well, but in smaller setups or newer operations, training and compliance need regular checks. If oversight slips, not only do health risks rise, but so do financial costs and community mistrust. My previous employer set up mandatory workshops and onsite spill drills. Participation wasn’t optional, and new hires learned early to respect these chemicals—more places should follow that model.
Boosting Access to Accurate Chemical Data
Getting the right formula and CAS number out into the world helps stop mistakes before they start. Students writing lab reports, buyers shopping for raw materials, and emergency responders all lean on this data to steer clear of costly or dangerous confusion. Many national chemical inventories—like the US EPA’s TSCA list or Europe’s REACH—build their regulations and tracking efforts around the CAS number. Transparency about hazards and supply chains is where science and public trust meet. Having good data systems, with open access for those who need them, cuts down on errors and gives the public confidence in lab and manufacturing activity.
Shaping a Better Future for Chemical Handling
With compounds like methylphenyldichlorosilane, careful stewardship can’t be an afterthought. If schools, businesses, and regulators pool expertise and share up-to-date safety information, the risks become manageable and the rewards—better materials, stronger industries—grow. Clear guidance, open channels of communication, and investment in professional development go further than glossy brochures ever will. Progress moves fastest when everyone can learn how and why certain formulas matter, and how easy access to data keeps people and the planet safer.
Is Methylphenyldichlorosilane compatible with other chemicals or solvents?
Why Some Chemicals Don’t Mix
Methylphenyldichlorosilane looks like a mouthful, but at its core it helps silicon-based industries make all sorts of advanced materials. The thing is, handling this compound isn’t as simple as pouring it into a beaker. I’ve seen labs rush through solvent selection, missing the fact that some combinations lead to ruined projects or even real danger. No chemist worth their salt would want to watch a flask fizz or emit toxic fumes—all because a solvent seemed harmless on paper. Methylphenyldichlorosilane reacts with water, alcohols, and even traces of moisture, releasing hydrochloric acid gas and heat. I’ve seen glassware etched by this stuff simply because someone forgot to dry a measuring cylinder.
Solvents and Unexpected Reactions
It’s tempting to toss every new chemical into a bottle and hope for the best. I once watched a synthetic pathway hit a wall because the chemist used methanol as a washing agent. What looked clean at first turned cloudy and smelly in minutes—classic exposure to the wrong solvent. Chlorosilanes like this one break down fast in protic solvents. That includes water, alcohols, glycols, even simple wet air. That reaction doesn’t just make a mess; it releases hydrochloric acid, which nobody wants in their lungs.
Laboratories and manufacturing outfits tend to stick with dry, aprotic solvents for methylphenyldichlorosilane. These include toluene, xylene, and dry hydrocarbons. Using these avoids the risk of violent hydrolysis. I remember how big the difference felt working with dry solvents compared to a humid one—a careful setup turns a jittery experience into something routine. That level of care matters because, beyond fumes, unplanned reactions drive up costs and slow down production lines.
Why Compatibility Testing Is Not Optional
Some companies think they can save time by skipping checks and running small-scale pilot runs, but poor compatibility puts workers at risk. Back in my early days, the temptation to “just try it” seemed harmless until a faulty test left a lingering acid smell around the fume hood for hours. Standard practice now starts with a review of all Material Safety Data Sheets (MSDS). Many list what not to use. Water and methylphenyldichlorosilane? Absolutely not. Even standard alcohols are dangerous here. The result of carelessness is corrosive vapors and wasted product. Product waste might sound minor, but disposal rules for halogenated waste really rack up budget pressures for university labs and startup companies.
Practical Solutions for Safe Use
Old-timers swear by color-coded solvent lines and moisture sensors. I think that’s a good place to start for anyone working in industry. Simple steps like nitrogen purging and running reactions in closed systems keep air and moisture away. If you need to clean glassware, rinse with dry toluene instead of an alcohol. Even using gloves rated for acid resistance makes a difference, since minor spills can etch skin before you even realize what happened. Proper storage also matters—keep this stuff away from sources of water and alcohol at all times.
One bright spot is the growing range of safer alternatives and newer processes. Some manufacturers now design modified silanes with less reactivity toward water, so the risk lowers some. The downside: often these alternatives cost more and behave differently in certain reactions, so chemists have to test every step. To me, that’s a fair trade-off. Reducing mishaps and protecting health always wins out over chasing small savings on solvents.
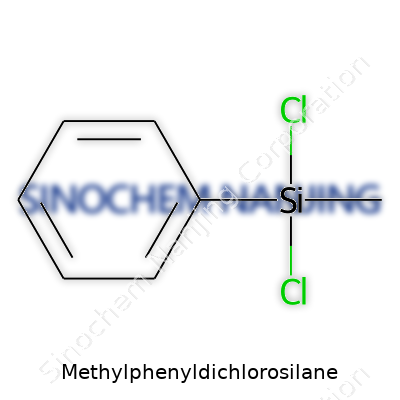
| Names | |
| Preferred IUPAC name | (chlorodichloro-λ⁴-silaneyl)benzene |
| Other names |
Dichloro(methyl)phenylsilane Dichlorophenylmethylsilane Methylphenylsilicon dichloride |
| Pronunciation | /ˌmɛθ.ɪlˌfiː.nɪl.daɪˌklɔː.roʊˈsaɪ.leɪn/ |
| Identifiers | |
| CAS Number | 80-10-4 |
| Beilstein Reference | 1209224 |
| ChEBI | CHEBI:34774 |
| ChEMBL | CHEMBL141619 |
| ChemSpider | 32593 |
| DrugBank | DB13879 |
| ECHA InfoCard | 100.020.068 |
| EC Number | 203-850-7 |
| Gmelin Reference | 69724 |
| KEGG | C18621 |
| MeSH | D008760 |
| PubChem CID | 66106 |
| RTECS number | TP4550000 |
| UNII | F4Y1KZ7212 |
| UN number | UN2481 |
| CompTox Dashboard (EPA) | urn:ss:C402932 |
| Properties | |
| Chemical formula | C7H8Cl2Si |
| Molar mass | 171.12 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Aromatic |
| Density | 1.174 g/mL at 25 °C |
| Solubility in water | Reacts |
| log P | 2.9 |
| Vapor pressure | 1 mmHg (25°C) |
| Acidity (pKa) | 12.6 |
| Basicity (pKb) | Basicity (pKb): 3.36 |
| Magnetic susceptibility (χ) | -54.0e-6 cm³/mol |
| Refractive index (nD) | 1.552 |
| Viscosity | 3.05 cP (25°C) |
| Dipole moment | 1.27 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.5 kJ/mol |
| Pharmacology | |
| ATC code | V09AX05 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS02,GHS05 |
| Signal word | Danger |
| Hazard statements | H314, H318, H331, H335, H411 |
| Precautionary statements | P210, P260, P262, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P308+P311, P363, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-1-W |
| Flash point | 67 °C |
| Autoignition temperature | 500°C |
| Lethal dose or concentration | LD50 oral rat 1620 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 1620 mg/kg |
| NIOSH | GV5950000 |
| PEL (Permissible) | PEL: 0.1 ppm |
| REL (Recommended) | 1 ppm (7 mg/m³) |
| IDLH (Immediate danger) | IDLH: 50 ppm |
| Related compounds | |
| Related compounds |
Phenyltrichlorosilane Diphenyldichlorosilane Methyltrichlorosilane Dimethyldichlorosilane |

