The Role of Methylarsonic Acid Across Industry, Research, and Environmental Stewardship
Historical Development
Methylarsonic acid doesn’t turn heads in most public debates, but this compound has deep roots in agricultural history. In the early 1900s, folks leaned on inorganic arsenic compounds for pest control, especially in cotton fields where boll weevils threatened whole harvests. As toxicity and environmental issues became obvious, chemists sought arsenic formulations that wouldn't stay in the environment forever. The introduction of methylarsonic acid as a more “manageable” arsenic derivative changed the game. It brought new thinking about how to balance persistent pest problems with growing awareness of chemical residues in soil and water. This balance proved elusive, but the compound’s story shows how new science often rides on the back of both hopes and mistakes.
Product Overview
Methylarsonic acid landed on the market with promises of increased weed control and less environmental build-up compared to straight arsenic compounds. Instead of sitting as a dry laboratory curiosity, it found real work as a weedkiller in fields from cotton to turfgrass. It exists as a colorless, odorless solid, usually supplied as a technical-grade powder, and it dissolves easily in water. That property made it attractive for big spraying rigs rolling across broad fields. Its rise and eventual decline reflect changing attitudes among farmers, regulators, and scientists—not a straight path, but one full of lessons about how we weigh risks against rewards.
Physical & Chemical Properties
Methylarsonic acid walks a fine line between stability and reactivity. It shows up as a crystalline solid, fairly stable at room temperature. It dissolves well in water and forms clear solutions. In air, there’s none of that strong rotten garlic smell often linked with other organoarsenic compounds. Its chemical structure keeps an arsenic atom bound to a methyl group and three oxygen atoms, which gives it special reactivity compared to plain arsenic trioxide or arsenates. The balance between its organic and inorganic qualities shapes how it behaves in soil, water, and living organisms.
Technical Specifications & Labeling
Product labels for methylarsonic acid once filled shelves at farm supply stores. The standards focused on purity—typically over 98 percent for agricultural use. Manufacturers listed solubility, recommended application rates, and clear warnings about use around food crops. Labels paid special attention to storage and handling, since arsenic residues presented potential hazards for workers and the environment. Over the years, changes in regulations forced more detailed language, especially as concerns about groundwater contamination and worker exposure came to the fore. Honest communication on product labels became a battleground between agricultural needs and safety concerns.
Preparation Method
Synthesizing methylarsonic acid took some ingenuity. Industrial preparation generally started with arsenic trioxide, which reacted with methylating agents under controlled acidic conditions. Chemists would slowly add reagents, controlling temperature and mixing rates to prevent runaway reactions or dangerous by-products. The final solution would go through purification to remove unreacted arsenic compounds and ensure consistent product quality. This process needed close attention to waste handling, since arsenic residues could find their way into waste streams and threaten drinking water sources. There’s no clean way to handle arsenic—production left its footprint, forcing industry and regulators to face real accountability around disposal and emissions.
Chemical Reactions & Modifications
Methylarsonic acid isn’t inert. In the presence of reducing conditions, it can decompose to methylarsine, a volatile and quite toxic substance. Alkaline soils or water can convert it to pentavalent or trivalent forms, each with their own toxicological profiles. Sunlight and microorganisms in soil break it down further, sometimes producing complex organoarsenic metabolites. Scientists have dug into these reactions to trace the environmental fate of methylarsonic acid, recognizing that its breakdown products can be just as concerning as its parent compound. Modifications of the molecular structure—adding more methyl groups, for example—have helped researchers model toxicity and environmental persistence.
Synonyms & Product Names
This compound lurks under a handful of names. Methylarsonic acid, methylarsonic acid, and MMA crop up in scientific literature and regulatory documents. Trade names faded from the market after regulatory restrictions, but old bottles and research papers still mention them. Synonyms reflect changing scientific language, but every name ties back to the same trouble: arsenic in an organic suit, still wielding the same molecular punch.
Safety & Operational Standards
Safety guidelines for methylarsonic acid have tightened over decades. During its heyday, workers sometimes lacked robust protective equipment or training in arsenic handling. Nowadays, any use, storage, or disposal requires gloves, face shields, and well-ventilated areas. Regulations demand strict inventory controls and spill response measures. For anyone handling the compound, washing procedures and medical surveillance programs keep exposure in check. Wastewater from manufacturing and agricultural runoff goes through treatment to catch any dissolved arsenic before it reaches rivers. Community pressure has pushed for continued improvements in safety, forcing both companies and regulators to scrutinize every step from factory to field.
Application Area
Its application carved out a niche in fighting weeds in cotton, rice, and turfgrass. Farmers turned to methylarsonic acid after older arsenic sources left residues that built up over years. Its selective herbicidal action limited collateral damage to crops. Turf managers used it to manage stubborn grass species that resisted standard broadleaf killers. Reliance faded as new classes of synthetic herbicides—often less toxic and more biodegradable—claimed their share of the market. The lessons from widespread use highlighted the deep connections between agricultural innovation, market pressures, and environmental impacts.
Research & Development
Ongoing research carved new paths into understanding arsenic metabolism in plants and soil. Scientists used methylarsonic acid as a model to trace how crops absorb, transform, and shoot out organoarsenic chemicals. It shaped studies on how soil microorganisms handle environmental toxins, opening doors to phytoremediation—using plants to clean up contaminated ground. Analytical chemistry advanced, too, as teams developed better tools to detect trace arsenicals and map their spread through ecosystems. Despite dropping out of regular commercial rotation, methylarsonic acid finds its way into university laboratories and environmental monitoring projects, continuing to influence how we think about chemical safety and pollution.
Toxicity Research
Toxicologists don’t ignore methylarsonic acid’s checkered track record. Chronic exposure studies flagged risks for cancer, organ damage, and developmental problems. Worker health records showed higher rates of skin lesions and neural problems in communities living near arsenical herbicide-treated fields. Animal testing revealed accumulation in liver and kidneys along with dose-dependent toxicity. Regulatory agencies used these findings to justify tighter controls and bans in many countries. Even after restrictions, past use still leaves scars in some soils, where residual arsenicals leach into crops and groundwater. Tracking and addressing these residues remains a long-term challenge for public health.
Future Prospects
Most countries have phased out methylarsonic acid in favor of safer herbicides. Its lasting influence has less to do with current fields and more with managing contaminated sites and handling legacy pollution. Research teams now explore ways to break down arsenic residues using microbes, fungi, and advanced materials. The compound’s chemistry supports ongoing work in understanding environmental arsenic cycles, as well as the design of new herbicides that turn the lessons of old mistakes into fresh opportunities for safer agriculture. Moving forward, dealing with methylarsonic acid means learning from the past, keeping a close watch on contaminated soils, and teaching new generations of researchers to handle complex chemical risks with the respect they deserve.
What is Methylarsonic Acid used for?
A Brief Look at Methylarsonic Acid
Most people don’t bump into methylarsonic acid outside of a chemistry lab or a farm supply store. This chemical comes from arsenic, a naturally occurring element found in the earth’s crust. Methylarsonic acid draws the interest of both scientists and farmers because of how it’s used and the potential consequences it carries.
The Agricultural Angle
Farmers looking to protect crops from weeds have reached for arsenic-based compounds for over a century. Methylarsonic acid steps in mainly as a herbicide, helping control unwanted plants that threaten rice, cotton, and some other crops. The logic is simple: more control over weeds means better crop yields. Products like monosodium methanearsonate (MSMA) and disodium methanearsonate (DSMA) contain methylarsonic acid and have been sprayed over fields throughout the U.S. and other countries. In rice paddies, weeds like barnyardgrass destroy harvests, so growers historically used this type of chemical to tip the odds in their favor.
Concerns About Health and Environment
Talk to any farmer using these products, and you’ll hear about the tightrope walk between tackling weeds and protecting people and ecosystems. Methylarsonic acid, being arsenic-based, comes with baggage. Studies from both the EPA and independent researchers show that arsenic can move from treated fields into the water supply. Arsenic exposure does not play around — the World Health Organization counts it as a top-tier chemical hazard, linked to skin lesions, cancers, and developmental issues. This knowledge makes the use of arsenic-based herbicides a complicated decision. Some regions restrict or even ban their use because of the risk to people living nearby, the workers applying the chemicals, and the long-term consequences for soil and waterways.
Lessons from Personal Experience
I grew up around farms, and watching my relatives head out with sprayers, you see the value in having tools to handle weeds. You also start to wonder what happens years later if those tools stick around in the soil. Old-timers would talk about certain areas where nothing seemed to grow quite right, and later studies found elevated arsenic levels. That’s not just some distant danger; it’s something that shapes the way communities think about farming and safety today.
Where Science Is Headed
Researchers now have their eyes on ways to manage weeds without relying on methylarsonic acid. Newer weed control strategies blend techniques like crop rotation, cover cropping, and selective hand removal. Some schools and regulators encourage biopesticides or mechanically tackling weeds, which limit exposure to risky chemicals. For those still using methylarsonic acid, careful application and water testing play a big role in lowering risks to neighbors and nature.
Finding a Path Forward
Methylarsonic acid shows the double-edged sword of chemistry in agriculture — boosting crop output on the one hand, raising health and environmental alarms on the other. Farmers, scientists, and regulators must keep looking for safer answers that don’t sacrifice today or tomorrow. Facts about arsenic exposure can’t be ignored, and seeing the effects first-hand makes the need for thoughtful solutions all the more real.
Is Methylarsonic Acid hazardous to health?
Grasping the Problem
Methylarsonic acid. Not a name that comes up often in daily conversation, but it should raise eyebrows when it appears in the news. This chemical belongs to a family with a reputation—the arsenic compounds. If you’ve ever heard warnings about arsenic in drinking water, you already know why this topic matters. In the past, methylarsonic acid found its way into agriculture, especially as a herbicide in rice fields across parts of the southern United States and other countries. The reason for its use: control of tough weeds, which farmers struggled to manage otherwise.
Routes of Exposure
The big issue with methylarsonic acid comes down to exposure. Farmers have worked near rice paddies treated with it; nearby communities can face runoff into groundwater or surface water. Food can become contaminated when rice or other crops absorb arsenic from the soil. I’ve seen concern rise in families who learn the groundwater they once thought clean now carries traces of compounds like this one. Children, pregnant women, and the elderly don’t stand a chance against chronic exposure—their bodies can’t detoxify chemicals the same way healthy adults can.
Health Hazards: What Science Says
Research connects methylarsonic acid exposure to many of the dangers tied to arsenic. Consumption over long periods leads to skin lesions, changes in the skin’s appearance, or even cancers of the bladder, lung, and skin. Evidence from studies in Asia and the Americas backs this up. In the short term, symptoms like nausea, vomiting, abdominal pain, and diarrhea can creep in. Chronic low-dose exposure worries public health experts more, as it quietly increases risk of illness over years. Even low concentrations can disrupt normal fetal development and set the stage for lifelong health problems.
One of the nastiest features of these arsenic compounds is their ability to hide: flavorless, colorless, odorless. People don’t realize they’re drinking or eating something toxic until it’s far too late. Elevated levels persist for decades in soil and water.
Tackling Contamination
Cleaning up methylarsonic acid isn’t impossible, but it proves challenging and expensive. Soil removal, water filtration, and phytoremediation (growing certain plants to absorb the toxin) stand out as the most common tactics. Replacing contaminated groundwater takes patience and often government intervention. What’s missing in many places isn’t just technology; it’s access, funding, and long-term policy support. I’ve watched advocacy efforts struggle for traction because these solutions rarely become a political priority until the damage appears in children’s blood tests or in crops on supermarket shelves.
Moving Toward Safer Agriculture
One lesson from years of agricultural work: switching to safer alternatives takes trust, education, and ongoing support for farmers. People often choose these chemicals because older tools failed or cost too much. Bringing safer herbicide methods and providing government incentives can help. Transparency from regulators and industry lets communities make informed choices about drinking water and the food on the table. Frequent monitoring, full public access to data, and responsible use rules matter. It’s tough to overstate the importance of these basic steps in keeping families safe from hard-to-detect toxics like methylarsonic acid.
What are the storage and handling requirements for Methylarsonic Acid?
Paying Attention to Chemical Risks
Methylarsonic acid shows up mainly in weed killers and has a tough record for health and environmental hazards. Its ties to arsenic call for extra care, both in day-to-day use and long-term storage. Skimping on safety, or relying on shortcuts, might put workers in the line of fire and push communities closer to accidental exposure.
Storage: Getting the Environment Right
A steady, cool, dry spot goes a long way toward keeping methylarsonic acid stable. Direct sunlight and warmth speed up chemical changes, raising the odds of vapor release or spills. Keep it in clearly labeled, corrosion-proof containers that won’t give in after a few chemical cycles—polyethylene and glass outperform many metals, especially around acids. Storing it among food or near potable water risks mixing and accidental ingestion, so keeping a safe distance between methylarsonic acid and everyday supplies matters more than most people imagine.
Protection for People Nearby
Anyone storing or moving containers needs serious personal protective equipment. That means sturdy gloves, splash-resistant goggles, and chemical-resistant aprons. Even experienced workers get surprised by how fast exposure symptoms hit: irritation, headaches, or worse after a single splash. Strong ventilation cuts down inhalation risks, and chemical fume hoods improve indoor air safety. Skipping these basics quickly leads to contamination both inside and outside storage rooms.
Avoiding Contamination and Spills
The real headache from methylarsonic acid starts after a spill or leak. Once this compound gets loose, especially near drains or open soil, cleaning up grows complicated. Concrete floors with sealed cracks make spills easier to contain and clean. Impermeable secondary containment—the simple tray under barrels—catches leaks before they spread. In my time working around agricultural compounds, a single missed step meant calling in a hazardous materials response team, who had to suit up and follow time-consuming protocols just to mop up residue. Even small leaks stuck around in the environment longer than anyone wanted to admit.
Handling Waste and Residue
Even empty methylarsonic acid containers can’t land in the regular trash. Rinsing with water spreads contamination, so approved hazardous waste procedures should be the final stop for old barrels. Incinerators made for chemical waste or licensed hazardous waste companies remain the only real ways to close the loop without risking soil and groundwater. Skipping this step, or farming it out to fly-by-night waste handlers, poisons ecosystems, and piles up long-term cleanup bills for real people later.
Training and Awareness Save Lives
All the technical rules fail if folks don’t take training seriously. Stories from the field always come back to the importance of drills, clear labeled zones, and keeping Material Safety Data Sheets within arm’s reach. Nobody thinks about chemical leaks or exposure until alarms ring—and by then, every minute counts. It always starts with someone rushing, ignoring a label, or skipping a glove, and ends with lessons learned the hard way.
Building a Culture of Safety
A real safety culture doesn’t rely on memory or habit. Having a checklist, keeping eye-wash stations checked, and running through emergency steps takes time up front but pays back once pressure hits. It takes some upfront investment, but cutting corners here sends ripples across entire communities. Anyone handling methylarsonic acid, or any toxic chemical, owes it to themselves and their neighbors to treat this work with respect—and insist on the tools, spaces, and training to keep the worst from happening.
How should Methylarsonic Acid be disposed of safely?
What Makes Methylarsonic Acid Tricky?
Methylarsonic acid belongs to a group of organoarsenic compounds often found in older herbicides and industrial waste. Exposure carries real health risks—chronic intake links to skin and internal cancers, nerve problems, and cardiovascular issues. The environmental story isn’t much better. Arsenic doesn’t just vanish; it travels through soil and water, making its way into crops, animals, even drinking water supplies. So proper disposal truly matters.
The Paths to Safer Disposal
Over the years, industry has learned—sometimes the hard way—that you can't just dump this stuff and hope for the best. Proper disposal isn't just about following rules; it's rooted in years of watching nearby wells get poisoned and farmers suffer. Facilities dedicated to treating hazardous waste offer a proven path. High-temperature incineration stands out as a reliable option. Temperatures above 1000°C break down the bonds in methylarsonic acid and trap arsenic in solid waste for controlled storage. These air-scrubbing systems don’t just keep up appearances. They slash the risk to workers and to neighbors downwind.
Not every town has a hazardous-waste incinerator nearby, and over-the-road hauling brings its own headaches. Spills during transport have caused tragic headlines more than once, so certified transport companies and proper labeling are non-negotiable. Skipping steps lands you in court or worse.
Looking for Cleaner Alternatives
Chemical treatment offers another route, provided the process gets handled by folks who know their stuff. In regulated moisture and pH, certain oxidizing agents break methylarsonic acid down, converting the arsenic to forms that bind with iron or calcium, forming insoluble compounds. These can then be buried securely in hazardous waste landfills built with multiple layers and groundwater monitoring. It’s not a panacea, but modern landfill liners do work better than the leaky pits of past decades.
Water systems dealing with low levels sometimes use activated carbon or special resins for arsenic removal. That doesn’t really help with concentrated sources from industry, but for contaminated rinse water it’s a useful tool. Those filters and resins need careful handling after use, since they're now holding the same dangerous stuff you were trying to keep away from people in the first place.
Prevention and Public Trust
Nobody should ignore the byproducts piling up from decades of chemical agriculture. Honest conversations with local communities help rebuild trust—especially in rural areas with long memories of polluted creeks and failed government promises. Posting disposal records, sharing audit results, and welcoming outside inspectors sets a better tone than closed doors and vague handouts. The public deserves details about what gets buried and where.
For anyone stuck with leftover methylarsonic acid—whether from an old farm shed or a shuttered facility—a call to licensed hazardous waste contractors is the right move. Never pour leftovers into drains or toss them in regular trash. That shortcut always falls back on someone else, usually those with the least resources to deal with it.
Reducing Tomorrow’s Burden
The story of methylarsonic acid shows the true costs of chemical shortcuts. Production has dropped in developed countries, but stockpiles still lurk on farms and older facilities. Tighter tracking, honest reporting, and a public push for safer alternatives point the way forward. Every step toward secure disposal shrinks the messy legacy left behind for the next generation.
What is the chemical structure and formula of Methylarsonic Acid?
Understanding Methylarsonic Acid at the Molecular Level
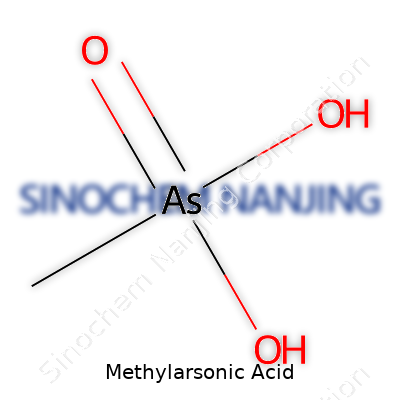
Methylarsonic acid isn’t something the average person thinks about over breakfast, but it plays an interesting role in chemistry and agriculture. Chemically, its formula is CH5AsO3. Breaking this down, it contains one carbon, five hydrogens, one arsenic, and three oxygens. This composition results in a structure where the central arsenic atom connects to a methyl group (that’s the -CH3), and directly to three oxygens, one of which holds a hydrogen, making a hydroxyl group.
This structure isn’t just a jumble of atoms. The layout matters, especially since arsenic compounds have a history—sometimes notorious, sometimes essential. With Methylarsonic acid, you have a derivative from arsenic acid, swapping out one oxygen-hydrogen for a methyl group. So instead of just arsenic and oxygen, that little addition of a carbon group changes how the molecule behaves. In practical terms, this molecule often appears as a white, crystalline solid, a common feature among many arsenic acids and their derivatives.
Why Structure Dictates Use and Risk
The huge importance of Methylarsonic acid came into focus in the twentieth century when it became a base for herbicide formulations. If you’ve ever lived near a farming region or worked in agriculture, the push for weed control feels real. Farmers used methylarsonic acid-based products to manage tough grass species, especially in rice and cotton fields. The methyl group in its structure means this particular arsenic acid derivative moves differently in soil and water compared to inorganic arsenic. It breaks down under certain conditions, but its by-products don't just disappear.
Chemical structure doesn’t just shape risk, it ties directly to how the molecule interacts with living things. Once methylarsonic acid gets absorbed by plants or leaches into the groundwater, the methyl group affects how easily living systems can process or excrete it. Researchers have shown that methylarsenicals are usually less toxic than inorganic arsenic, but prolonged exposure tells another story. In communities near treated fields, traces have ended up in water sources or rice crops. The World Health Organization and environmental watchdogs have pointed out that even lower-toxicity organoarsenicals need close scrutiny and regulation, given their persistence in soils.
Dealing With Chemical Legacy
From my days covering rural environmental issues, I saw how older fields sometimes carry a chemical legacy, with arsenic levels in soil or waterways still running high long after the last application. The persistence traces straight to the structure—organic arsenic compounds often convert slowly back into more toxic inorganic species. Scientists recommend regular soil and water testing around past application sites. Some advocate for phytoremediation—using certain plants to draw arsenic out of soils safely. I once met a farming family in Arkansas trying out hyperaccumulator ferns for their patch of stubborn creek bank, reporting slow but steady progress.
In labs, molecular analysis lets chemists visualize exactly how compounds like methylarsonic acid function, giving insight on breakdown pathways and risks. Newer strategies aim to minimize environmental stress from chemical residues, favoring integrated pest management over routine chemical spraying. The chemical formula of methylarsonic acid isn’t just a collection of elements—it ties tightly to real-world impacts: from how food grows, to what ends up in our water, to the health of future generations.
| Names | |
| Preferred IUPAC name | Methylarsonic acid |
| Other names |
Monomethylarsonic acid Methylarsonate MMA |
| Pronunciation | /ˌmɛθ.ɪl.ɑːrˈsɒn.ɪk ˈæs.ɪd/ |
| Identifiers | |
| CAS Number | 124-58-3 |
| Beilstein Reference | 87819 |
| ChEBI | CHEBI:29627 |
| ChEMBL | CHEMBL1210 |
| ChemSpider | 12173 |
| DrugBank | DB02610 |
| ECHA InfoCard | 100.036.442 |
| EC Number | 200-865-6 |
| Gmelin Reference | 7847 |
| KEGG | C19268 |
| MeSH | D015761 |
| PubChem CID | 1675 |
| RTECS number | CG3325000 |
| UNII | 2CC5T97O6L |
| UN number | UN2865 |
| Properties | |
| Chemical formula | CH5AsO3 |
| Molar mass | 138.02 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.105 g/cm3 |
| Solubility in water | soluble |
| log P | -1.17 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | 4.7 |
| Basicity (pKb) | 6.8 |
| Magnetic susceptibility (χ) | -61.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.614 |
| Viscosity | Viscosity: 1.2 cP (20 °C) |
| Dipole moment | 1.63 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 155 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -482.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -727.8 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | C01CX07 |
| Hazards | |
| Main hazards | Toxic if swallowed. Causes skin and eye irritation. Suspected of causing cancer. Environmental hazard. |
| GHS labelling | GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H301: Toxic if swallowed. H332: Harmful if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P260, P264, P270, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P308+P311, P312, P321, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-2-W |
| Flash point | 79 °C (174 °F) |
| Lethal dose or concentration | LD50 oral rat 1800 mg/kg |
| LD50 (median dose) | LD50: 700 mg/kg (rat, oral) |
| NIOSH | B0188 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Methylarsonic Acid: "0.01 mg/m³ (as As) |
| REL (Recommended) | 1 μg/L |
| IDLH (Immediate danger) | IDLH: 10 mg/m³ |
| Related compounds | |
| Related compounds |
Arsenous acid Arsenic acid Monomethylarsonic acid Methyliodic acid |

