Methylamine Nitrate: A Practical Look Into Its Development, Properties, and Role in Modern Industry
Historical Development
Factories and laboratories began working with methylamine nitrate in the mid-20th century, a time shaped by scientific exploration and expanded chemical manufacturing. Scientists wanted nitrogen-containing compounds that offered both reactivity and manageability, partly for their use in synthesis, partly for performance in industrial processes. Chemical producers saw methylamine nitrate's water solubility and energetic properties as useful in research and select commercial applications, so technical data started to appear in specialty chemical catalogs. Over the years, the material found its way into more focused research, showing up in papers about organic synthesis, fuel additives, and pyrotechnics. These applications reflect how methylamine nitrate grew from a specialty lab item to an intermediate valuable in research and experiment. Its use has always remained specialized; industrial-scale production has never rivaled more common nitrates or methylamines due to limited and often regulated demand.
Product Overview
The compound appears as a colorless to white crystalline solid. Soldiers, researchers, and chemical engineers who've worked with water-soluble nitrate salts recognize its slightly sweet, ammonia-like scent, which gives away the methylamine group. Chemically, methylamine nitrate results from the neutralization of methylamine (CH₃NH₂) with nitric acid (HNO₃), where the amine donates one proton and the nitrate acts as a stable, oxidizing counterion. Lab chemists often mention its utility in energetic material prototypes, yet outside that niche, its profile remains low-key. The product ships with warnings about powerful oxidizing action, so outside of the lab or controlled settings, handling it raises eyebrows in compliance and safety offices. In trade, methylamine nitrate sometimes travels under alias names depending on manufacturer, with synonyms like "monomethylammonium nitrate" or "N-Methylammonium nitrate" appearing in chemical databases. These alternate product names reflect how different countries and suppliers list it differently in registries.
Physical & Chemical Properties
Well-documented properties include a melting point just over 50°C, appreciable hygroscopicity in humid air, and fast solubility in cold and warm water. Anyone who has weighed or mixed this salt notices that it quickly forms a transparent aqueous solution and clumps if left exposed to atmospheric moisture. The chemical formula CH₅N₂O₃ reveals a simple structure, though strong intermolecular hydrogen bonding means the solid can cake in storage if humidity gets too high. It doesn’t stand up well to heat or physical shock, which makes clear labeling and special storage practices a must in any inventory. Direct exposure to sunlight or open flames spells real danger—thermal decomposition brings about gaseous nitrogen oxides, ammonia, and methylamine, which create both health and fire risks.
Technical Specifications & Labeling
Reliable suppliers provide methylamine nitrate in well-sealed containers, often with UN transport codes reflecting its oxidizing nature. Specifications call out purity levels (usually above 95%), detailed water content ranges, and screenings for heavy metal contaminants like lead, mercury, and cadmium. Labels list hazard information in accordance with GHS regulations: "Oxidizer," "Irritant," and "Harmful if swallowed or inhaled" statements appear prominently. Some producers also print suggestions for protective equipment (gloves, eye protection, dust masks), emergency washing instructions, and storage precautions (keep cool, use inert atmosphere for long-term storage, keep clear of reducers and combustibles). Shipping departments learn to navigate the patchwork of international regulations around ammonium, methylamine, and nitrate compounds, especially after changes in security and hazard control laws.
Preparation Method
Chemists who have handled batch preparations follow the classic acid-base neutralization approach. Water acts as the solvent. Methylamine (used as an aqueous solution or as a gas dissolved into water) goes into a reaction vessel, with cooling because the process releases heat. Nitric acid trickles in slowly, and pH meters help nail down the endpoint around 7. Over-addition of nitric acid or methylamine wastes reagent and upsets the yield. Filters remove trace impurities, and the resulting solution gets evaporated under controlled temperature, sometimes under vacuum to prevent decomposition and limit losses of volatile amine. The white crystalline salt forms as water boils off. Laboratories maintain secure fume hoods and discharge scrubbers since both methylamine and nitric acid fumes irritate the nose and throat. Technicians check each batch for unreacted starting materials and scan by ion chromatography for trace nitrate or nitrite by-products. Scaling up to pilot plant level means using explosion-proof equipment and remote handling whenever feasible.
Chemical Reactions & Modifications
Once produced, methylamine nitrate enters different chemical transformation routes. Some researchers use it in the synthesis of N-nitro derivatives or for preparing energetic coordination complexes—materials whose performance in propellants relies on nitrate reactivity combined with the methylammonium cation. Experimental organic chemists sometimes use methylamine nitrate as a mild source of methylamine in redox reactions. In solution, it reacts cleanly with reducing agents to generate methylamine gas (which must be properly vented), and with strong acids, it can liberate toxic nitrogen oxides. Rare schemes convert the salt into other methylamine derivatives by metathesis, exchanging the nitrate group for chloride or sulfate under controlled conditions. Handling these reactions requires strong ventilation, correct PPE, and systems designed to contain and treat hazardous offgassing. None of these laboratory adaptations see use in everyday chemical manufacturing; they belong in the niche world of materials development.
Synonyms & Product Names
In chemical supply catalogs, methylamine nitrate pops up under different synonyms. Most label it “Monomethylammonium nitrate,” while others use “N-Methylammonium nitrate.” CAS number 124-41-4 makes identification easier across languages or different regulatory codes. These synonyms help researchers avoid confusing it with methyl nitrate or with complex methylamine derivatives, both of which have far different safety profiles and performance characteristics. Regional regulations can affect how it’s classified—some agencies slot it under general ammonium salt controls, others treat it like an oxidizer, so product names sometimes reflect these compliance quirks. In research procurement, clear communication about synonyms and product names avoids confusion and keeps paperwork in sync with inventory.
Safety & Operational Standards
In the lab, working safely with methylamine nitrate means understanding both physical and chemical hazards. Contact with the skin or eyes stings and leaves irritation, and dust in the air irritates lungs. At scale, larger quantities become a bigger concern for fire safety and explosion risk. The oxidizing power can accelerate combustion of ordinary fuels or, in case of contamination, provoke runaway reactions. Safety data sheets point out that no one should store it near organic materials, metal powders, strong acids, or bases. In my own lab, we always double-check storage rooms for leaks and enforce mandatory PPE, including splash goggles and vapor-tight gloves, along with easily accessible eyewash stations. Anyone using even a gram weighs the product in a fume hood, and clean hands before and after work remain non-negotiable habits. Regulatory authorities hold regular spot checks, especially for research groups working with energetic materials.
Application Area
Methylamine nitrate gained most of its practical experience in the development of prototype propellants, model rocket engines, and academic energetic material programs. Some early pyrotechnics researchers examined its properties as a replacement for ammonium nitrate in specific color compositions due to its energetic output. Scientific teams also worked on using the compound to generate methylamine in situ, especially where direct handling of methylamine gas poses difficulties due to toxicity and volatility. Outside academic testing, the product’s risks and regulations sharply limit its use, so it remains almost exclusively in private research, materials science, or highly specialized industrial chemistry. Modern industry rarely selects it for bulk operations, as cost, regulation, and safer alternatives steer most users elsewhere.
Research & Development
Interest in methylamine nitrate comes up in experimental research focused on propellant chemistry, low-scale explosives development, and studies on nitrate-organic salt chemistry. Academic scientists value it as a teaching example for redox chemistry and the principles of energetic material safety and decomposition. In research circles, some look for ways to modify the cation or anion for altered sensitivity, aiming to tune properties like solubility, burn rate, or thermal stability. Researchers sometimes pair novel energetic salts with methylamine nitrate to explore energetic performance or decomposition mechanisms. Only highly specialized R&D facilities with trained personnel take on these projects, given all the necessary controls over inventory, personnel, and waste disposal. Recent publications highlight interest in green energetic materials, but most attention still tilts toward controlling sensitivity and studying fundamental reactivity, rather than launching new commercial uses.
Toxicity Research
Most toxicology studies agree that methylamine nitrate carries moderate acute toxicity, especially if inhaled or ingested. Once the body absorbs it, the nitrate portion can induce methemoglobinemia, which prevents red blood cells from carrying enough oxygen—a concern for people with compromised health or young children, and a reason why industrial exposure regulations stay strict. In animal studies, methylamine breakdown forms both methylamine and nitrate ions; high doses cause respiratory distress, neurological symptoms, and sometimes convulsions. People with preexisting breathing problems feel the effects more quickly. Cumulative risk remains modest at controlled trace exposure levels, but laboratories and companies working with this material report all spills, require well-documented material inventories, and ban eating, drinking, or smoking around preparation rooms. Wastewater from labs goes through either chemical or biological treatment to destroy nitrates and amines before disposal, reducing ecological hazards in local waterways.
Future Prospects
A few trends influence where methylamine nitrate research heads over the coming years. Interest in green chemistry and sustainable energetic materials could drive some groups to revisit its reactivity for safer, less polluting propellant designs. At the same time, stricter government oversight of energetic materials, terrorist threat mitigation, and tightening occupational exposure limits will limit its movement, particularly between countries. Engineers and chemists will keep experimenting with substitutions for ammonium or methylamine components, aiming for materials with lower toxicity and greater thermal stability. New analytical techniques could uncover modified versions suitable for sensor calibration standards or for use in ultra-sensitive analytical methods. Still, the real growth areas for methylamine nitrate probably won’t show up in the bulk chemical trade, but rather in academic papers and regulated innovation hubs with the right safety culture and compliance infrastructure.
What is Methylamine Nitrate used for?
Digging Into Industrial Purposes
Methylamine nitrate doesn’t come up in casual conversation, but it has a place in several industries. People often link chemicals to scary headlines or shady activity, yet their main roles often sit quietly in factories and labs. In my experience studying chemical supply chains for years, I’ve seen how one compound can serve legitimate and important functions.
One big use for methylamine nitrate lies in research settings. Chemists value it as a reagent, especially when they’re synthesizing other chemicals. It reacts readily, allowing scientists to manipulate molecules and test new approaches. Universities sometimes rely on it for experimentation—pretty far from anything nefarious. Chemical companies ship this compound in controlled environments, following strict safety laws.
Role in Explosive Formulations
This chemical also appears in the history of certain industrial explosives. Mining operations and demolition crews have looked at methylamine nitrate to break up rock and concrete. It works as an oxidizer, which helps explosive mixtures perform with more punch. Companies pay close attention to safety protocols when handling these recipes. Every step, from delivery to storage, follows detailed national guidelines. Trained professionals understand the serious risks, but they also know the value: safer, faster progress in places where manual labor just won’t cut it.
Control and Security Measures
No discussion about compounds like this feels right without talking about regulation. The potential for abuse means agencies such as the U.S. Drug Enforcement Administration and chemical watchdogs in Europe track every shipment of methylamine nitrate. Companies keep logs of their inventory, and governments maintain oversight. My own time working with compliance teams has taught me the paperwork can seem endless, but these records help keep dangerous chemicals out of the wrong hands.
These controls don’t just protect against illegal use. Proper regulation reduces workplace accidents, too. Strict licensing systems, equipment inspections, and regular staff training go a long way in keeping workers safe. Anyone who thinks chemical safety is just about “red tape” has never sat in a meeting after an unexpected spill. Lives depend on careful monitoring and preparation.
The Question of Illicit Use
It’s impossible to talk about methylamine nitrate without mentioning its connection to homemade explosives and even drug synthesis. News stories occasionally focus on criminals using precursor chemicals for methamphetamine or dangerous explosives. While this earns plenty of attention, the vast majority of methylamine nitrate moves through legal, tightly-regulated channels. Honest researchers and industrial workers get painted with the same brush, which frustrates many in science and engineering fields. Most just want to get their jobs done and go home safely at the end of the day.
Pushing for Smarter Regulation
I believe one solution to this tug-of-war between industry and security lies in smarter, data-driven regulation. Agencies could invest more in digital tracking, real-time reporting, and cross-border cooperation. Companies can pitch in with better staff training and transparent sourcing. When everyone—industry, regulators, and the public—understands both the risks and the value of chemicals like methylamine nitrate, it’s easier to have a conversation about innovation and safety side by side.
Methylamine nitrate might not grab the headlines like oil or gold, but its journey through factories, labs, and regulatory offices says a lot about modern industry: risk, reward, and the constant need for clear-eyed oversight.
Is Methylamine Nitrate hazardous or regulated?
Understanding Methylamine Nitrate
Methylamine nitrate’s reputation doesn’t turn many heads outside scientific or industrial circles, despite its potential risks. This chemical combines two ingredients with long histories—methylamine, widely used in chemical manufacturing, and nitrate, known well in explosives and fertilizers. Put together, the compound grabs interest in both legitimate business and law enforcement communities.
Hazards Associated with Its Use
Getting straight to the point, methylamine nitrate isn’t something you’d want in your kitchen cupboard. It’s an energetic compound. That means there’s a risk of it lighting up or exploding if you’re careless—especially around heat or a spark. In fact, history offers too many examples of dangerous accidents. The chemistry textbooks cover cases where improper storage caused massive damage, harm, or even death.
Adding to that, both methylamine and nitrate have a history in illegal drug and explosives manufacturing. That means the presence of methylamine nitrate, especially in larger quantities, brings law enforcement knocking. The Drug Enforcement Administration in the United States, for example, keeps a tight grip on methylamine because of its use in making methamphetamine. Nitrate compounds, on the other hand, send red flags through counterterrorism units because of their connection to explosives.
How Regulators Respond
Regulation isn’t theory—it comes out of years of learning from mistakes. For methylamine, many countries classify it as a List 1 Chemical (as in the US), making purchase and sale a paperwork-heavy event. Nitrates, because of their long association with explosive devices, put users under scrutiny, too. Bringing both substances under your roof requires detailed documentation, reporting, secure storage, and random inspections.
Some folks reading this may wonder if there’s an exception for research or academic labs. There are, but the bureaucracy doesn’t let up. Permits, background checks, even limits on how much chemical can sit in one place—these are all common requirements. If any of this sounds strict, it’s because real-world misuse of these chemicals has proved wreck havoc.
Environmental and Health Impacts
Leaving regulation aside for a moment, handling methylamine nitrate never comes risk-free. Exposure, even in small doses, can irritate the skin, eyes, and lungs. Inhaling dust or fumes is worse—imagine the sensation of a strong cleaner, but amplified. Runoff from accidental spills poses a threat to rivers and groundwater since nitrate pollution led to toxic algae blooms and well water issues for farm communities before.
There’s a rising call in the scientific community to develop greener, safer alternatives. That comes from experience: No matter how careful people claim to be, accidents can and do happen. In my early years working in a university lab, we drilled safety not as an afterthought but as our first step. That’s because one overlooked procedure could mean disaster—not only for those in the building, but for nearby neighborhoods too.
Looking for Solutions
Changing practices around hazardous chemicals calls for better education and tighter rules—but also smarter chemistry. Chemists have started engineering less sensitive alternatives whenever possible. Industry bodies now push mandatory safety workshops and transparent chemical inventory databases. In my own work, digital badges for up-to-date safety training built a safety net far stronger than old “read-the-manual” approaches.
As more countries tighten their export and reporting rules, and as safer replacements hit the market, the number of accidents might just start to fall. But it takes buy-in from all sides—producers, buyers, and regulators. Methylamine nitrate offers yet another example of why those in control of powerful chemistry must never slack on responsibility.
How should Methylamine Nitrate be stored?
Why Methylamine Nitrate Demands Respect
Methylamine nitrate doesn’t show up in daily conversation, but in agriculture, some labs, and emergency supply rooms, this chemical belongs front and center. Treating it lightly invites trouble. I’ve seen far too many stories about chemical mishaps that started out with just a little carelessness in storage. Even small leaks or spills can lead to health risks, legal headaches, or wasted money. For people handling it, safety means respect—there’s no shortcut around it.
Choose the Right Container and Environment
Methylamine nitrate breaks down if stored in open air or left in cheap containers. Sealed, airtight drums or heavy-duty, non-reactive plastic containers give you a fighting chance. I once watched a warehouse manager try to store similar chemicals in thin-walled barrels, only to end up losing inventory and earning an OSHA report for corroded metal and leaking product. Stainless steel or approved high-density polyethylene genuinely makes a difference.
This chemical hates heat and moisture. High humidity creates clumps and can trigger unwanted reactions, so dry, cool rooms make sense. Keep the storage area between 15 and 25 degrees Celsius. It never pays to trust “close enough” conditions—because temperature swings and high humidity slowly chip away at the material’s stability. In one memorable summer, a shipping container filled with chemicals heated up to near boiling and caused crates to burst. Once you see white powder leaking across a storage yard, you remember it.
Minimize Risk: Lock It Down
Methylamine nitrate sometimes draws attention for the wrong reasons. The same chemistry behind fertilizer can leave this chemical a target for theft or misuse. Security turns out just as important as chemistry or storage conditions. Reliable locks, cameras covering corners, and regular inspections help protect your investment. Sign-in sheets are more than paperwork—they make sure every movement of product is tracked, which can help stop trouble early.
Fire Safety Isn’t Optional
Oxidizers amp up fire risk, and mixing methylamine nitrate with anything flammable can trigger disaster. Don’t store this chemical near fuels, oil, sawdust, or even old rags. I’ve worked with people who kept “just a few things close by” and lived to regret it after a simple spark turned into a dangerous situation. Good practice looks like keeping separation walls, fire extinguishers rated for chemical fires, and exit routes clear.
Regularly reviewing safety data sheets and training new staff cannot be an afterthought. Most people remember their first real emergency drill forever. The one time I witnessed a genuine spill, what saved the day wasn’t fancy tech—it was a simple, practiced emergency plan and good teamwork.
Label Everything Clearly
Sometimes accidents happen just because a label got smudged or someone assumed the wrong drum held “the usual stuff.” Every container should carry the full name, hazard icons, and emergency contact info right on the outside. Double-check those labels after every delivery. Sharp markers and waterproof labels go a long way. Communication saves lives, and in busy shops or farms, clear labeling means no one ever has to guess.
Routine Checks Make the Difference
Checking seals, scanning for leaks, and making sure containers haven’t warped needs to become a habit. Some teams schedule monthly walkthroughs with a simple checklist. These few minutes can catch tiny problems before they grow. Documentation matters: logging each check or repair supplies accountability and proves that safety’s a daily job.
What are the safety precautions when handling Methylamine Nitrate?
Respect the Risk in Every Step
Plenty of folks have come across chemicals with big warning labels, but methylamine nitrate brings a different level of concern. Handling this compound isn’t just about ticking boxes on a safety sheet. I’ve spent years around laboratories and industrial spaces—trust gets built when you treat chemicals like methylamine nitrate with the caution they earn. If you ignore the risks, mistakes can stack up fast. Frustration doesn’t undo an accident.
Methylamine nitrate won’t always act the way you expect. It’s a strong oxidizer with real potential to react violently. Working near it, I’ve seen gloves melt from splashes and ventilation systems earn their keep. Leaving this stuff on the bench, unlabelled or uncapped, invites disaster. No one needs a story of a lab fire to know why it’s crucial to keep it secured.
Gearing Up the Right Way
Proper safety gear isn’t just a suggestion. Nitrile or butyl rubber gloves, splash goggles and a heavy-duty lab coat keep you a step ahead of trouble. Face shields never feel like overkill. If you wear polyester, think twice—synthetic fabrics catch and melt onto skin way too easily. Sturdy cotton offers peace of mind.
The lab’s air supply matters just as much as the fabric you wear. Hood fans running strong and efficient filters keep fumes away from your lungs. I’ve seen folks work without ventilation—almost always regret shows up in coughing fits or blinding headaches. Exposure builds up over time; the damage gets done before you know it. Always check the airflow before opening any bottle.
Chemical Storage Done Right
Storage cut corners cause the biggest headaches. Don’t pile up incompatible chemicals together just to save space. Acids, bases, and organics need separation from methylamine nitrate. I learned the hard way how moisture turns this compound unstable, sometimes explosive. Airtight containers, marked with clear hazard symbols, stop mix-ups and protect everyone nearby.
Temperature swings kick off dangerous reactions. Leave methylamine nitrate near a heater and you gamble with everyone’s safety. Find a cool, dark spot that stays under 25°C. Padlocked cabinets add an extra layer of control—especially in places with more foot traffic.
Planning for Mistakes
Accidents always seem far off until one lands in your lap. Training everyone to know the correct spill response isn’t just procedural. It saves lives. Absorbent materials designed for oxidizers handle spills quickly, especially when paired with non-sparking tools. Don’t let confusion stall your clean-up. Eye wash stations and chemical showers have to be reachable, not collecting dust behind cardboard boxes.
Emergency contacts shouldn’t live at the back of some binder. Post them big and bold near all workstations. Review SDS (Safety Data Sheets) at the start of every shift. I keep printed copies on hand because computer files get lost right when you need them most.
Stay a Step Ahead
Working with methylamine nitrate demands respect and preparation. Look out for your co-workers and speak up if shortcuts show up. Regular safety drills, open conversations about risks, and an understanding of the compound’s volatility protect more than just equipment. They protect lives. Ignoring the rules in the hope of getting things done faster only adds risk. Slowing down to do it right gets everyone home safe at the end of the day.
Where can I buy Methylamine Nitrate legally?
Understanding the Demand and Regulations
Methylamine nitrate grabs attention due to its role in chemical synthesis and its links to both legitimate industries and some less lawful pursuits. In the world of research or manufacturing, every substance faces a unique set of rules, and this one stands out for its scrutiny. Walk into any chemical supply conversation, and government red tape comes up right after safety concerns.
Chemical suppliers don’t add methylamine nitrate to their regular catalogs for good reason. The compound’s track record in explosives—isn’t something regulators or suppliers take lightly. Over the years, misuse in illegal drug manufacture and explosives has put it on watchlists in many regions. Federal agencies like the DEA in the United States control the sale of certain chemical precursors to prevent them from ending up where they shouldn’t.
Industry, Research, and Oversight
In my time working with research teams in both academia and industrial labs, most folks learn quickly that access to regulated chemicals means paperwork, vetting, and a clear description of use. You don’t just place an order online. The whole process usually starts with a review of who you are, the reputation of your institution, and a look at your track record with hazardous or controlled substances. Vendors—typically those with licenses and a serious compliance department—will only consider requests tied to credible institutions or registered businesses.
Universities and licensed manufacturing facilities go through background checks, follow strict inventory requirements, and regularly coordinate with local law enforcement or health and safety agencies. No one ships methylamine nitrate to a home address. Sometimes, requests trigger reporting to federal authorities like the DEA or Homeland Security. This system isn’t about making life hard for scientists or businesses; it keeps dangerous substances from fueling harm.
Public Safety Weighs Heavily
The push for public safety outweighs convenience every time. Even with a legitimate use case, only a handful of suppliers work with such substances. Names like Sigma-Aldrich, Fisher Scientific, and VWR come to mind for reputable chemicals, but they aren't going to sell someone methylamine nitrate unless all legal ducks are in a row.
It never fails—every new grad student starts out frustrated by bureaucracy. Eventually, they get why strict regulations exist; accidents or deliberate misuse impact more than just the user. Even specialized sectors like agriculture or pharmaceuticals often look for alternatives that avoid legal hassles.
Potential Solutions for Those with Legitimate Needs
Researchers or companies can still make a case for purchase. Building a relationship with vetted suppliers matters—those who know you, your track record, and application, can help guide you through permits and secure storage planning. Those working solo, outside institutional support, face obstacles built to prevent unsupervised handling.
If a researcher or business truly requires methylamine nitrate, consulting with regulatory bodies before moving forward saves time and frustration. State or national institutes post up-to-date guidance about permits, procedures, and required justifications on their official websites.
Despite the hurdles, opening up about your intentions, clarifying your end use, and staying compliant keeps access possible. Most who qualify recognize these protocols safeguard communities and support responsible research. In all cases, legal channels matter—and for those who see the value in transparency, following the rules pays off in both safety and trust.
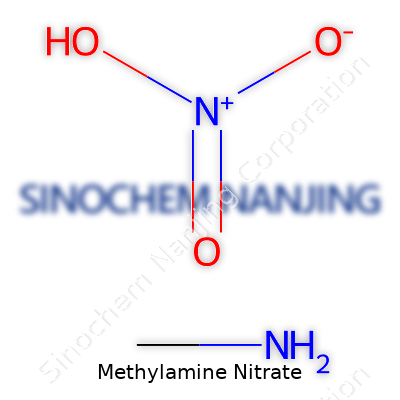
| Names | |
| Preferred IUPAC name | Methanaminium nitrate |
| Other names |
Methylammonium nitrate |
| Pronunciation | /ˈmɛθɪl.əˌmiːn ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 4218-51-7 |
| 3D model (JSmol) | `MC1=NC.O[N+](=O)[O-]` |
| Beilstein Reference | 1465050 |
| ChEBI | CHEBI:74240 |
| ChEMBL | CHEMBL1906671 |
| ChemSpider | 157403 |
| DrugBank | DB11319 |
| ECHA InfoCard | The ECHA InfoCard for Methylamine Nitrate is **"100.031.983"**. |
| EC Number | 2536-34-9 |
| Gmelin Reference | 8784 |
| KEGG | C45215 |
| MeSH | D008749 |
| PubChem CID | 164218 |
| RTECS number | UA5955000 |
| UNII | N89W8T6H0B |
| UN number | UN1061 |
| CompTox Dashboard (EPA) | DTXSID5021466 |
| Properties | |
| Chemical formula | CH5N·NO3 |
| Molar mass | 77.08 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Ammonia-like |
| Density | 1.055 g/cm³ |
| Solubility in water | Very soluble |
| log P | -3.09 |
| Acidity (pKa) | 10.64 |
| Basicity (pKb) | 10.64 |
| Magnetic susceptibility (χ) | Magnetic susceptibility (χ) of Methylamine Nitrate: -36.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.386 |
| Dipole moment | 3.82 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 218.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -80.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -365.7 kJ/mol |
| Pharmacology | |
| ATC code | V03AB37 |
| Hazards | |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H225, H290, H301, H314, H332 |
| Precautionary statements | P210, P220, P221, P261, P264, P271, P273, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P330, P337+P313, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-1 |
| Explosive limits | 4.2–41% |
| Lethal dose or concentration | Lethal dose or concentration: "LD50 (oral, rat): 75 mg/kg |
| LD50 (median dose) | LD50 (median dose): 1180 mg/kg (Oral, Rat) |
| NIOSH | UN2810 |
| PEL (Permissible) | PEL: 10 ppm (as Methylamine) |
| REL (Recommended) | REL (Recommended): 0.5 ppm (1 mg/m3) TWA |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
Methylamine Ammonium nitrate Ethylamine nitrate Dimethylamine nitrate Nitromethane |

