Methyl Tert-Butyl Ketone: Where Chemistry, Industry, and Risk Collide
Tracing the Origins of Methyl Tert-Butyl Ketone
Methyl Tert-Butyl Ketone didn't just drop into the chemical industry overnight. Looking back to the postwar boom in petrochemicals, scientists obsessed over new ways to fine-tune fuels and solvents. Big names in chemistry cookbooks noticed that tinkering with tert-butyl chains gave molecules a rare blend of volatility and solvency. MTBK, as it’s often called in labs, started cropping up in journals alongside other alkyl ketones by the 1960s. At first it lagged behind cousins like Methyl Ethyl Ketone, lurking in the background while others grabbed headlines, but folks with a nose for niche performance knew even then it had untapped promise.
What MTBK Brings to the Table
Nobody can mistake MTBK for water. This clear liquid boasts a gnarly odor that lingers on your hands even with a quick wash. With a boiling point just north of 100°C and a strong knack for dissolving oils, resins, and polymers, this ketone fits right in with its chemical brethren. Unlike some lighter solvents, MTBK trades off a smidge of volatility for a more robust ability to muscle through tougher messes. That extra carbon in its structure props up both resistance against breaking down and its niche in more challenging environments. The oxygen at its core means it participates easily in more elaborate chemistries, a fact not lost on those looking for a reactive building block for synthesis.
Reading the Fine Print: Specs, Labels, and Compliances
Industry standards don’t leave much to chance when it comes to chemicals that can spark, spill, or cause harm. MTBK’s labels flag flammability right at the top—no surprises there. High vapor pressure lands it in special storage lockers, far from hot lights or open flames. Most containers arrive with purity stamped as a priority, since even small contaminants can throw a wrench in high-stakes synthesis. Chemical handling calls for well-fitted gloves and a face shield—you don’t cut corners in labs with this kind of solvent, and anybody who's gotten even a whiff up close instantly understands why.
Baking a Batch: How MTBK Gets Made
Bringing MTBK to life takes more than kitchen chemistry. Manufacturers crank it out using a mashup of methylation and alkylation reactions, usually drawing reactants from petroleum derivatives. This often means working under pressure, watching temperatures with hawk-like care, and maintaining strict exclusion of water and oxygen to keep yields high and byproducts low. For people like me who’ve watched distillation runs go south, it’s clear that keeping everything clean and monitored makes all the difference. Industrial setups keep things moving at scale, often recycling solvents or purging unwanted chemicals to limit waste and gunk in the final product.
What Happens In the Flask: MTBK’s Chemical Playbook
MTBK reacts predictably for trained chemists. Its carbonyl group stands out as a target for nucleophilic attack, and the bulky tert-butyl group shields parts of the molecule, so not every reaction flies out of the textbook the way you’d hope. It partners well in oxidation and reduction reactions, often serving as a stepping stone toward higher-value molecules. For those looking to tweak or bulk up other organics, MTBK acts as a bridge: sticking functional groups onto its backbone, swapping pieces out, or even breaking it apart under the right set of conditions. In my own lab work, using a ketone like MTBK offered a reliable fork in the road, with more predictable handling than some funkier alternatives.
Aliases in the Wild: Synonyms and Product Monikers
MTBK may show up on an order sheet as 2-Methyl-2-Butanone, but you might stumble on it as tert-Butyl Methyl Ketone or even Methyl tert-Butanone, depending who you’re talking with. Trade names shift from region to region, and warehouse labels don’t always play by IUPAC rules. This jumble, while confusing at times, reflects the hodgepodge nature of global chemistry. People experimenting with this stuff quickly learn to verify chemical structure and batch numbers, not just names. Years of experience teaches the hard lesson not to pour from a poorly marked jug until you know for sure what’s inside.
Running a Tight Ship: Safety and Standards in Practice
Nobody with sense skips safety when MTBK pops up in the protocol. Splashing this solvent on your skin stings, and breathing its vapors turns one day’s work into a headache marathon. Modern labs keep ventilation humming, and emergency eye-wash stations earn their space. Regulatory oversight covers everything from atmospheric release to storage procedures. MTBK’s flammability draws extra attention—everyone double checks ground wires on containers and bolts down storage cabinets. Practical training and heavy supervision on new hires keep accidents scarce, since one lapse can land a whole operation on the evening news.
Why People Use It: MTBK on the Job
MTBK doesn’t headline the list of household names. It shows up in specialized applications where other ketones don’t fit quite right. Paint and coatings manufacturers eye its slow evaporation and stubborn solvency. Resin souping-up and adhesive formulations use it to get just the right flow and stickiness. Pharmaceutical startups, with one foot in risk and the other in potential, consider MTBK when milder ketones fall short. I’ve met a few stubborn old-timers in industry circles who claim it rescued a project or two where nothing else would budge gummy intermediates out of solution.
In the Trenches: Research and Development with MTBK
Researchers and industrial chemists alike have built new pathways around MTBK’s unique blend of traits. Recent studies dig into its compatibility with bio-based resins or eco-friendly polymers, as pressure mounts to green up every aspect of synthesis. Scientists chase down improved purification tricks, always hustling for higher yields or cheaper inputs. In my own experience, graduate students gravitated to it when seeking a more robust alternative to limping along with overused solvents. The chance to modify and tweak this molecule inspires innovation, even if the safety paperwork multiplies with every new tweak.
MTBK on the Scales: Toxicity and Risk Management
Toxicologists know MTBK doesn’t play as rough as some industrial nasties, but it still packs a punch if mishandled. Inhalation brings on nerve symptoms and, in chronic cases, can hammer the liver. Water solubility hovers in a middle range, so spills make their way into waterways easily enough. Environmental stewardship guidelines call for containment and scrubbers, and I’ve been in meetings where arguments over ventilation upgrades ran longer than planned experiments. Medical research teams have pushed for new insight on the subtle, long-term effects of repeated exposure, but the basic rule stands: respectful use, not casual handling.
Looking to Tomorrow: MTBK’s Future
As economies chase cleaner, safer, and more efficient chemistry, MTBK’s destiny straddles uncertainty and opportunity. Pushes for green chemistry and lower emissions prod manufacturers to invent substitutes or redesign processes, but the molecule’s resilient mix of properties keeps it in the running for advanced applications. In places where other solvents fail to deliver, MTBK remains tough to replace. If the chemical industry manages to limit exposure, recycle effectively, and adapt synthesis, I expect its reputation will grow with innovation rather than fade into the background. For anyone standing with hands in the mix between progress and risk, MTBK represents both a challenge and an invitation to do things smarter.
What is Methyl Tert-Butyl Ketone used for?
An Overlooked Player in Solvents
Methyl tert-butyl ketone, often shortened to MTBK, doesn’t show up in headlines about new technology or green breakthroughs, but it quietly supports plenty of industries most people rely on. MTBK performs as a powerful solvent. You’d find it in laboratories and production floors, helping clean, extract, and separate substances that would otherwise cling together. On a personal note, years of working with paints and coatings reveal just how much chemists count on solvents like MTBK for formulations that spread evenly and dry the way they should.
The Backbone of Coatings and Adhesives
Every time you pick up a can of spray paint or open a bottle of industrial adhesive, MTBK could have had a hand in making those products viable. Manufacturers lean on it because it dissolves certain resins and polymers quickly. Its chemical structure helps it cut through stubborn substances without reacting to them in unexpected ways. That makes production more predictable and safe. Modern furniture, automotive finishes, and even some electronics wouldn’t have the finish or durability people expect if careful solvent selection fell by the wayside.
MTBK and the Petrochemical World
Refineries and chemical plants use MTBK in processes like dewaxing and extraction, which boost the quality of fuels and lubricants. Most folks fill up their car without giving a second thought to the chain of events and materials that make smooth engine performance possible. My early days visiting oil industry sites showed just how much attention goes to these background chemicals. They play a part in making fuel burn more efficiently or oil perform under extreme conditions. If some new environmental rule forced plants to stop using MTBK overnight, much of the downstream impact would show up at the pump or in shortened engine life.
Workplace Realities and Safety
MTBK isn’t without its downsides. Wearing personal protective equipment matters. Anyone working in an environment where it vaporizes knows the risk of exposure. Prolonged inhalation or skin contact doesn’t just give minor irritation, it can impact health across the years. That’s one thing regulatory agencies cover well. In every factory I’ve walked, engineers and safety leaders teach respect for solvents—MTBK included—with real-world stories that stick well beyond the onboarding session. The chemical industry keeps pushing for better ventilation, sealed systems, and detection equipment to avoid surprises. Even responsible storage and waste handling go a long way toward protecting both people and the environment.
Environmental Questions and Cleaner Alternatives
Decades of MTBK use raise the same questions society faces about lots of chemicals: what happens after it leaves the factory? On-site spills or careless disposal can let MTBK slip into soil or water systems. Teams in charge of compliance monitor emissions, train for spill response, and carefully track solvent volumes. Researchers continue searching for greener options. Some companies blend bio-based solvents for certain tasks, but widespread replacement takes time and investment. Every step toward safer, more sustainable chemical choices involves tradeoffs—cost, performance, and availability. Still, the momentum for change is real.
Looking Ahead
MTBK will likely remain a behind-the-scenes contributor for years. Its role in paints, coatings, fuels, and chemicals connects to the products and conveniences people don’t often think about. As with many solvents used at scale, the challenge remains the same: use just enough, manage the risks, and push forward with cleaner answers when they show promise.
Is Methyl Tert-Butyl Ketone hazardous to health?
Understanding What Methyl Tert-Butyl Ketone Is
Methyl tert-butyl ketone, often known among chemists as MTBK, shows up in many labs, factories, and chemical plants. Folks use it as a solvent in manufacturing and research settings. The smell reminds me a bit of nail polish remover, and that should ring alarm bells for anyone familiar with industrial chemicals. Exposure usually comes through breathing fumes or skin contact. The substance has earned its reputation as a hard-working chemical, but questions about safety aren’t new.
The Evidence on Health Risk
Science hasn’t ignored the concerns. The U.S. Environmental Protection Agency (EPA) and European Chemicals Agency list MTBK as a material worth careful handling. Short-term exposure sometimes brings headaches, dizziness, or even nausea. My own experience working near solvents like these: you recognize the symptoms right away—throat gets scratchy, head feels cloudy, and sometimes your eyes sting long after a shift. Over the long run, repeated contact seems to affect the nerves, liver, and kidneys. Rodent studies have shown signs of organ damage after prolonged high-level exposure. These facts make it clear: MTBK isn’t just another harmless workplace ingredient.
Real-World Exposure: Who Faces the Risks?
Factory workers, chemical technicians, and those cleaning up spills meet MTBK more than most. Personal protective gear, like gloves and good ventilation, make a real difference but aren’t always perfect. Smaller companies sometimes cut corners or skip routine air monitoring. This kind of choice puts families at risk too, since clothes worn home can carry traces of solvent.
Think about the people in nearby communities. Factories rarely exist in the middle of nowhere. Chemical leaks or spills sometimes reach public water, or wind carries vapors across neighborhoods. Environmental groups pushed for tighter regulations, and rightfully so. It’s not paranoia to want groundwater and air free from hazardous chemicals.
Room for Improvement: Protecting Health
Denying the risks of MTBK only leads to more health problems. Most developed countries set occupational exposure limits. In the United States, OSHA sets an eight-hour limit lower than what makes people sick in the short term. Companies testing air regularly and giving workers proper training seem to avoid most health incidents. My own time on safety committees taught me the value of regular drills and free access to protective gear. Small steps save lives and keep workers from illness later in life.
On the environmental side, local governments and watchdog groups track emissions and spills. When chemical plants aren’t following the rules, citizens can report problems. Lawsuits and fines send a clear message: companies must take community health seriously. For people living in high-risk zones, education and consistent testing help spot issues before they escalate. People deserve to know what’s in their air and water.
There’s no perfectly safe chemical, but ignoring potential harm never works out well. Methyl tert-butyl ketone’s risks feel real to anyone who’s felt its bite in the air. Full transparency, better safety planning, and neighborhood advocacy can close the gap between industry convenience and public health.
How should Methyl Tert-Butyl Ketone be stored?
Looking at Chemical Risks First
Methyl tert-butyl ketone has power under the hood. You find it in the lab, in industry, sometimes in research settings. All that power comes with bite. This stuff evaporates quickly, acts as a strong solvent, and, if left uncared for, can cause headaches, skin problems, and fires. I’ve worked around similar liquids before: leave a canister uncapped, and the smell alone fills the whole building. Straight off, you want to keep safety right up front—no shortcuts.
Containers Make a Difference
You can’t just use any old drum or jug. Polyethylene or stainless steel containers provide protection. Thin plastics turn brittle, glass risks breaking, and unlined steel encourages rust, which can spark trouble with a chemical like this. At a former job, some younger chemists grabbed a soda bottle to store leftover solvent for cleaning. One bump, bottle split, soaked a whole bench—lesson learned. Stick to tightly sealed, chemical-grade containers. Labels should shout what’s inside, bold and waterproof. There’s no safety in mystery.
Control the Temperature
Keep this stuff cool. At higher temps, vapors shoot straight up in concentration. That means more fire risk, more fumes, and faster spoilage. I keep chemicals like this away from windows, vents, or places with sudden heat swings. If you store them near heat or in sunlight, you gamble with safety. Most storage rooms offer a cool, steady temperature—no fridges, just a climate that doesn’t shift by the hour.
Ventilation Can’t Be Ignored
Strong-smelling solvents fill a room with fumes minutes after opening. Store methyl tert-butyl ketone in an area with plenty of airflow: dedicated fume hoods, vented cabinets, or special storerooms with fans sucking air out. One whiff can surprise people unused to chemical work, and too much vapor sneaks up quickly. Years ago, I watched a coworker open a drum in a closed shed. We had to run in, open all the doors, fans blasting, clinging to our lunches. None of us forgot the lesson.
No Sparks, No Flames
With volatility comes flammability. Sparks, smoking, hot lamps, and even static electricity can light off vapors from methyl tert-butyl ketone. Store away from workshops, engines, and open circuits. A little caution goes a long way, and daily habits—like grounding all storage drums to avoid static—build that layer of protection.
Alternative Solutions Matter
Old chemicals pile up. Most facilities I’ve seen keep a running inventory, disposing of unused solvents before they become hazards. Some even store just-in-time, ordering small amounts needed for specific projects rather than stacking up dangerous stock. Less on hand reduces risk. For people at home or without formal training, proper disposal becomes a real concern. Community hazardous waste programs give a safer option than pouring chemicals down the drain or tossing them in the trash.
People and Practice Keep Us Safe
Training can’t just be a checkmark on a safety sheet. Every worker deserves hands-on learning, not just a slideshow. I've seen firsthand how walking through storage routines and discussing real incidents makes rules stick. Information from trusted sources—Occupational Safety and Health Administration (OSHA), chemical suppliers, and environmental health offices—brings facts, not just opinions. Safety grows from knowledge, habit, and a willingness to act early. Every step, from picking a canister to tossing an empty one, should reflect that mindset.
What are the safety precautions when handling Methyl Tert-Butyl Ketone?
Why Experience Matters With This Chemical
Anyone who’s spent time around industrial solvents knows that Methyl Tert-Butyl Ketone (MTBK) means business. Known for its punchy smell and strong solvency, this chemical finds use in labs, paint shops, and manufacturing plants. In my years working with different solvents, a few clear-cut habits have stuck with me — good habits make the difference between a safe shift and a dangerous event. MTBK behaves a lot like stronger cousins MEK and acetone, but it comes with its own risks.
Ventilation Can’t Be an Afterthought
I remember watching someone pour MTBK in a small room with the window closed. The air thickened quickly. Breathing got harder, and soon, everyone’s eyes stung. This substance gives off vapors that irritate your lungs and eyes before you even realize it. Open windows and exhaust fans count far more than people assume. Fume hoods play a key role in labs, but in workshops, even a pair of simple box fans can keep dangerous vapors moving outside instead of building up. OSHA sets exposure limits for a reason. Overexposure messes with your central nervous system and can leave you with a nasty headache.
The Right Gear Isn’t Optional
MTBK soaks through a lot of gloves, and regular latex doesn't cut it. Nitrile or butyl gloves stand up well. I’ve seen too many people trust thin disposable gloves, only to feel a burning tingle after a minute. Splash goggles block the vapors from stinging your eyes. In heavy-use scenarios, a half-mask respirator with organic vapor cartridges makes sense. Clothing choices matter, too. Long sleeves and closed shoes keep splashes off the skin. Regular cotton clothing helps, but aprons or lab coats give an extra layer of protection against splashes and spills.
Careful Storage and Handling
Once, I stored a jug of MTBK under a bench, right near a portable heater. The container bulged a little after a few days — heat and organic solvents don’t play well together. Store this chemical in tightly sealed containers, away from heat, sunlight, or anything that might cause sparks. One mistake and static can ignite the vapors. Ground metal drums and flow lines if you transfer large volumes. Never use glass containers with old or weakened seals, since MTBK can soften some types of plastic as well. Label every container clearly. Too many accidents start when someone grabs the wrong bottle.
Acting Fast With Spills
On a busy shop floor, a knocked-over jar can become a major hazard. Small spills demand fast action — soak them up with absorbent pads or sand, open the windows, and dispose of the waste as hazardous material. Even a damp rag out in the open can fill a room with smell. Don’t let liquid reach drains or the ground, since MTBK contaminates water and soil fast. Call in trained hazmat help for big spills. During safety drills, skipping the spill practice leads to real confusion during the main event.
Looking for Problems Before They Grow
MTBK doesn’t look threatening. But watch for dizziness, headaches, or confusion after exposure. People often ignore these early signs. I check workspaces for forgotten bottles, oily rags, or ventilation blockages. Having a buddy system can help, especially when handling large batches or during off-hours. Good communication and awareness beat overconfidence every single time.
Building a Safer Culture
Most safety problems sink in when you see a peer make a mistake or when a close call wakes you up. Sharing those moments helps everyone raise their guard. Reading the SDS sheet tells you the theory, but seeing how skin absorbs the stuff or watching how fast it flashes is unforgettable. New hires get shadowed on their first week, and checklists hang in plain view by every storage rack. Safety matters in big and small tasks. Every habit, every detail—these add up to long-term well-being around chemicals like Methyl Tert-Butyl Ketone.
What is the chemical formula of Methyl Tert-Butyl Ketone?
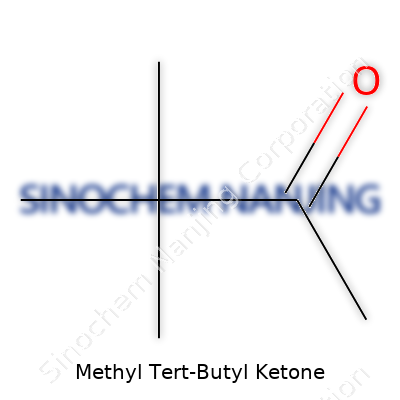
Understanding the Formula: C6H12O
Methyl tert-butyl ketone, known by its chemical formula C6H12O, isn’t just another collection of letters and numbers on a chemist’s notepad. Once you start working in an environment where different solvents and industrial chemicals pass through your hands, you get a clear sense of how formulas like this one hold more meaning than meets the eye. Every molecule tells a story—the way those six carbons, twelve hydrogens, and that single oxygen are put together explains much more than just its structure on paper.
Why the Structure Matters
In my lab days, knowing the formula saved both time and safety headaches. Back then, Methyl tert-butyl ketone (MTBK for short) came in handy, especially when I needed something strong to dissolve difficult substances. Its structure provides a solid backbone while the tert-butyl group shields the ketone, creating a distinctive pattern of reactivity. Unlike more famous relatives like acetone or methyl ethyl ketone, MTBK has features that make it survive in tough conditions, especially around heat and bases.
Where it gets even more interesting is how the formula shapes its properties. With C6H12O, you’re handling a molecule with real punch. It can dissolve both polar and non-polar compounds, so it opens doors in formulations—from adhesives to specialty coatings. A few years back, I worked with a production team trying to design a new cleaning solvent for electronics; adding MTBK into the mix let us lift residues without stripping sensitive films or plastics. There’s no substitute for seeing firsthand how a simple shift in molecular structure can fix what nothing else could.
Risks Stepping Out of the Shadows
No formula should become all about promise. Shifting to risk management, health and safety routines treat MTBK’s chemical formula with deep respect. With six carbons and a ketone group, inhalation and skin exposure present real hazards if you’re not careful. Government databases like PubChem lay it out: overexposure can cause dizziness and irritation, and no one wants that from just a few careless minutes in the fume hood. Too often, safety slips when people start treating formulas as abstract.
Reliable chemical databases and trusted industry sources show C6H12O’s use faces regulatory watch in many places. Europe and California both keep close tabs on solvents with methyl and tert-butyl groups, linking some related compounds to air and water concerns. My experience says don’t take shortcuts; waste handling and proper ventilation become personal jobs, not just checklist tasks. MTBK spills often need more than paper towels—they demand well-trained eyes and hands to act fast and keep everyone safe.
Looking Ahead: Finding Safer Alternatives
If you search for inspiration in chemistry, the formula C6H12O reminds us progress runs in both directions—solving technical challenges and reducing harm. Today, research into greener ketones inspires me most, with labs experimenting on new blends that give the same cleaning and dissolving power without the baggage. Some companies examine bio-based ketone alternatives using feedback from environmental and worker safety groups. Switching over takes time and investment, but the lesson from MTBK’s chemical formula remains: understanding composition and effect opens the way to smarter, safer choices, both in the lab and beyond.
| Names | |
| Pronunciation | /ˌmɛθɪl tɜrt ˈbjuːtaɪl ˈkiːtoʊn/ |
| Identifiers | |
| CAS Number | 569-29-7 |
| Beilstein Reference | **1851585** |
| ChEBI | CHEBI:51188 |
| ChEMBL | CHEMBL16537 |
| ChemSpider | 68125 |
| DrugBank | DB02173 |
| ECHA InfoCard | 100.118.237 |
| EC Number | 208-759-1 |
| Gmelin Reference | 82692 |
| KEGG | C08342 |
| MeSH | D008767 |
| PubChem CID | 11263 |
| RTECS number | EL5425000 |
| UNII | J9L7J8U80K |
| UN number | UN1156 |
| CompTox Dashboard (EPA) | DBLT000367 |
| Properties | |
| Chemical formula | C6H12O |
| Molar mass | 100.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Ether-like |
| Density | 0.805 g/mL at 25 °C |
| Solubility in water | Slightly soluble |
| log P | 1.97 |
| Vapor pressure | 2.8 kPa (20°C) |
| Acidity (pKa) | Enolate alpha-C-H: 20.3 |
| Basicity (pKb) | 0.81 |
| Magnetic susceptibility (χ) | −7.77×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3900 |
| Viscosity | 0.49 cP (20°C) |
| Dipole moment | 2.78 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 323.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -262.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3147 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P261, P271, P280, P301+P312, P303+P361+P353, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 16 °C |
| Autoignition temperature | 460 °C (860 °F; 733 K) |
| Explosive limits | Explosive limits: 1.0% to 8.0% |
| Lethal dose or concentration | LD50 (oral, rat): 4,000 mg/kg |
| LD50 (median dose) | LD50 (median dose): 6740 mg/kg (rat, oral) |
| NIOSH | NIOSH: SA4925000 |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 3000 ppm |
| Related compounds | |
| Related compounds |
Diisopropyl ketone Pinacolone |

