Methyl N-Hexanoate: A Deep Dive into a Key Organic Compound
Historical Development
Interest in methyl n-hexanoate dates back to early efforts in organic synthesis, where chemists searched for ways to transform naturally occurring fatty acids into useful esters. Over time, as both the flavor and fragrance industries grew, researchers valued this compound for its pleasant, fruity aroma and functional chemical structure. By the mid-20th century, improved methods for esterification helped growers and biochemists develop scalable production processes. Today, methyl n-hexanoate stands as both a product of chemical ingenuity and a building block for broader applications spanning flavors, materials science, and synthetic chemistry.
Product Overview
Methyl n-hexanoate forms through the esterification of hexanoic acid with methanol. The compound offers a clear, colorless liquid profile with a characteristic sweet odor reminiscent of pineapple or fruit. Factories who handle flavors, plastics, and specialty chemicals keep this ester in regular stock thanks to its unique balance of volatility, compatibility, and reactivity. I’ve seen it show up just as often in labs as in large-scale industrial mixing tanks, testifying to both its reliability and versatility.
Physical & Chemical Properties
With a molecular formula of C7H14O2 and a molar mass around 130.18 g/mol, methyl n-hexanoate brings predictable behavior under standard lab conditions. The liquid boils at roughly 151–155°C, and freezes near −36°C, making it stable at room temperature but easy to purify through distillation. Its density floats at about 0.87 g/cm³, just lighter than water. This ester dissolves in organic solvents—ethanol, diethyl ether, and chloroform—but resists blending with water. That’s typical for many esters, and it helps dictate where chemists can use it. The compound’s refractive index checks in at about 1.41, which helps when characterizing purity during quality control runs.
Technical Specifications & Labeling
Accurate labeling ensures sharp handling and traceability. For methyl n-hexanoate, most suppliers include CAS number 106-70-7 along with purity details—often above 98%. Labels spell out batch numbers, hazard pictograms, country of origin, shelf life, and storage guidance, setting clear standards for both safety and compliance. Product documentation frequently lists residual solvent content, water fraction, and identification methods like GC/FID or NMR. Focusing on these specifications keeps logistics tight, especially if the compound ships across borders or for sensitive use cases, such as food contact or pharmaceutical synthesis.
Preparation Method
Industries favor traditional Fischer esterification to produce methyl n-hexanoate. Hexanoic acid reacts with methanol in the presence of an acid catalyst, often concentrated sulfuric acid. The reaction proceeds with heat, typically under reflux, to drive the formation of the ester and water. After separation, drying agents remove trace moisture, and distillation yields the purified product. This route prioritizes simplicity, cost efficiency, and thorough conversion. In practice, tuning the acid-methanol ratio and optimizing reaction time can boost yield and help meet demanding quality targets. Some modern plants lean on continuous flow reactors or greener catalysts to cut waste and energy demand, reflecting broader goals in sustainable chemistry.
Chemical Reactions & Modifications
Methyl n-hexanoate keeps its stability under neutral conditions but reacts readily in targeted transformations. Hydrolysis brings it back to hexanoic acid and methanol, a process relevant in both environmental studies and food regulation. Transesterification with various alcohols exchanges the methyl group for new ester forms, giving chemists a route to tune both volatility and solubility on demand. Reduction with strong hydrides, like lithium aluminium hydride, can yield hexanol. Researchers also explore its use as an intermediate in synthesizing more complex molecules, especially in flavors and fragrances where subtle structural tweaks matter. Over time, these properties helped push methyl n-hexanoate onto the radar for advanced manufacturing and functionalized material development.
Synonyms & Product Names
Methyl n-hexanoate appears in catalogues under many aliases. Some call it methyl caproate, referencing its caproic acid origin. Other product lines list it as hexanoic acid methyl ester. These alternative names help track historical supply chains and prevent confusion in international commerce. I’ve come across a few regional brands that stylize it for niche perfumery use or pharmaceutical-grade batches, but the chemistry stays the same regardless of the label. Having a handle on naming conventions cuts down sourcing errors, especially online or through third-party vendors.
Safety & Operational Standards
Safe handling practices deserve close attention. Methyl n-hexanoate features in the GHS system, carrying flammable liquid and eye irritant pictograms under many regulations. Workers should rely on goggles, gloves, and splash-resistant lab coats during handling or transfer. Well-ventilated workspaces prevent vapor build-up and minimize inhalation risks. Material Safety Data Sheets (MSDS) highlight lower explosive limits, recommended storage temperatures, and first-aid advice. Good spill control, with absorbent pads and chemical spill kits, supports rapid cleanups if containers break or leak. Regulatory frameworks, from OSHA to REACH, demand clear labeling, record keeping, and proper storage—usually in tightly closed bottles away from open flames and direct sunlight. Regular training and up-to-date safety drills matter more than any checklist.
Application Area
Few esters cross as many disciplines as methyl n-hexanoate. The compound carves a niche in food science as a flavoring agent, lending fruitiness to baked goods, candies, beverages, and dairy imitations. Fragrance designers pull it into formulas for “green” or “fruity” notes in fine perfumes and consumer air fresheners. Plastics teams explore its use as a specialty plasticizer or additive, altering flow and stability in custom polymer blends. Synthetic chemists test it in reaction screens aiming for drug design or advanced materials. A number of agricultural researchers investigate its bioactivity, seeking natural pest deterrents or novel attractants. This spread of uses keeps the market dynamic, with each sector shaping its technical standards and purity benchmarks.
Research & Development
Active labs worldwide keep methyl n-hexanoate in their toolkit for both method development and basic mechanistic studies. Researchers fine-tune synthetic routes to lower reaction temperatures, cut catalyst waste, and recover solvents more efficiently. Teams focused on sustainable chemistry look for biocatalysts—engineered enzymes or microbes that esterify hexanoic acid in water, sidestepping harsh acids and unnecessary byproducts. Analytical chemists experiment with new detectors for faster compound identification, especially given rising requirements in quality and safety testing. In my experience, interdisciplinary work—blending organic, analytical, environmental, and engineering skillsets—brings out the full potential of this unassuming ester. Forward-thinking organizations also back partnerships between academia and industry, hoping to scale up green chemistry wins from lab-scale to factory floor.
Toxicity Research
Scientifically sound toxicity research makes all the difference. Acute toxicity of methyl n-hexanoate stays low at quantities seen in food and fragrance applications, supported by rodent tests and regulatory reviews. Inhalation and skin contact show mild irritation at high concentrations, but routine workplace exposure falls far below harmful thresholds with modern controls. Environmental assessment tracks its rapid breakdown in soil and water, thanks to fast hydrolysis and microbial action. Yet as production scales and new use cases arise, watchdog groups call for continuous monitoring. Ongoing studies tackle chronic and reproductive toxicity questions, aiming for long-term assurance. Staying transparent with data—publicly available reports, accessible toxicity endpoints—builds trust among stakeholders, from manufacturers to consumers.
Future Prospects
Demand for methyl n-hexanoate shows no sign of slackening, especially as flavor, fragrance, and specialty chemical markets diversify. Advances in bio-refining, using renewable feedstocks instead of petrochemicals, chart a greener path for large-scale synthesis. Large producers start pilot programs using enzymatic processes, nudging the sector closer to net-zero ambitions. Meanwhile, digital chemical modeling sharpens researchers’ ability to predict properties and reactions, accelerating optimization cycles and shortening time-to-market for new applications. Staying proactive in both safe handling and sustainable manufacturing unlocks greater market resilience. Companies willing to invest time and resources into next-gen production and thorough safety validation earn a distinct edge—not just in compliance, but in reputation and consumer confidence.
What is Methyl N-Hexanoate used for?
The Tooth of Chemistry
Scrolling through the endless streams of chemical compounds, most folks would probably skip right over methyl n-hexanoate. It sounds technical, wrapped in a name that gives off no hints about anything you'd find at your local market. To me, it’s a reminder of the intricate connection between the chemistry bench and things we use daily, especially when it shows up in the flavors and fragrances that bring character to both food and personal care products.
The Sweet Smell of Esters
Methyl n-hexanoate belongs to a family of chemicals called esters. If you ever bit into a ripe pineapple or opened a bag of strawberry candies, you caught a breeze of what esters can bring to the table. This particular ester carries a fruity, slightly fatty scent—far more inviting than the sterile name suggests. You’ll find it in the creation of artificial flavors—think confectionery, chewing gum, and sodas—where just a dash does the heavy lifting in transforming bland glucose into a cheerful candy store aroma.
Beyond the Kitchen—Fragrances and Personal Care
The reach of methyl n-hexanoate doesn’t stop at bubble gum or soda flavorings. Personal care companies tap its potential for fruity, creamy notes in perfumes, lotions, and soaps. Unlike heavy synthetic fragrances that often mask, this compound brings subtle sweetness that blends into the background, helping products feel lighter and less artificial on the nose. In my experience, walking through a busy drugstore aisle, that clean, fruity note is more likely to linger with people than heavier chemical scents.
Industrial Uses and the Science Behind the Scenes
Moving from the world of taste and scent, methyl n-hexanoate steps into bigger shoes as a building block in the chemical industry. Manufacturers rely on it as a solvent in various processes, and it helps produce other additives like plasticizers and lubricants. Without it, the road to safer plastics and smooth-running machines would involve more bumps and breakdowns. It’s a testament to how specialized chemical manufacturing supports everything from logistics to packaging.
Health, Safety, and Regulation
No chemical should slide into our foods or products without full transparency. Methyl n-hexanoate, like other food-grade additives, goes through safety checks and regulations. Bodies such as the US Food and Drug Administration assess purity, safe concentrations, and potential health effects. In large quantities, esters can cause irritation, so professionals wear protective equipment and keep close tabs on exposure. At the consumer level, the quantities used for flavoring and scent stay well below any risk, yet it's always a good move to support robust safety protocols in both food science labs and manufacturing floors.
Moving Forward—Green Chemistry and Sourcing
Conversation about chemical ingredients brings up another topic: sustainability. Traditional manufacturing often involves petrochemicals, which impacts the environment across extraction, processing, and waste. In recent years, more suppliers have shifted toward renewable sources—using bio-based alcohols or acids to create esters like methyl n-hexanoate. Greater attention on sourcing and greener processes promises to make everything from flavorings to solvents less taxing on the planet without sacrificing taste or utility.
The Human Side of Chemistry
Science can feel distant, but familiarity grows when you recognize the concrete ways molecules fit into everyday comfort. Methyl n-hexanoate plugs a gap most of us don’t even know exists: the difference between bland and bright flavor, the distance from musty odor to fresh air, the leap from sticky plastics to products that just work. The story of this compound proves again that chemistry isn’t just about laboratories—it's woven into the taste of our next snack and the scent on our skin.
What is the chemical formula of Methyl N-Hexanoate?
The Structure and Its Meaning
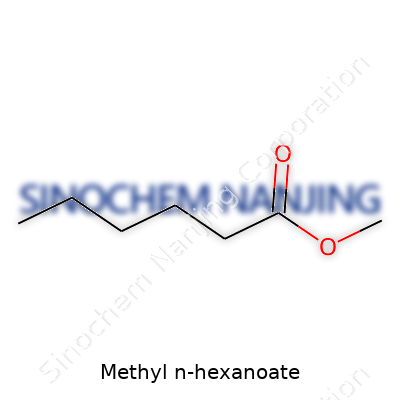
Methyl n-hexanoate lays out its structure with the formula C7H14O2. This single line gives a quick snapshot of what the compound holds. In it, you have seven carbons, fourteen hydrogens, and two oxygens. Chemists see the formula and right away, they can sketch a map in their heads: a methyl group meets a six-carbon chain that comes from hexanoic acid, pulled together by an ester bond. It’s simple oil paint for molecules, and it has real jobs in the world.
Scent and Sensibility
I still remember the surprise from the first time I opened a sample at a fragrance workshop. The soft, fruity odor lingers, a bit like pineapple mixed with apple, sometimes even coconut. That’s methyl n-hexanoate at work. The funny thing is how this tiny molecule, so easy to write as C7H14O2, has carved out a place in so many flavor and scent formulations. Look at the back of a bottle of fruit flavor concentrate or synthetic fragrance oil—there’s a solid chance this ester plays a part. Its role in these industries runs deep, all because chemists know how predictable and safe it turns out, batch after batch.
Safety and Trust
Looking up the safety data, you’ll see that it doesn’t pose a hazard under normal use. Still, like any chemical, factory workers who handle it regularly need goggles, gloves, good ventilation—the basics for chemical handling. Safety comes from respect and training, not fear. Food scientists and perfumers trust it, which brings peace of mind to consumers who care about what enters their body or touches their skin.
The Real Uses in Daily Life
People tend to ignore the tiny molecules behind their favorite experiences. This one keeps snack bars from going bland, adds a subtle note to soda, keeps shampoo smelling bright, and lifts the scent of hand lotion. Sometimes, I notice how a certain air freshener brings a memory of green apples from my childhood; there’s a good chance the same chemical sits behind that smell.
Environmental Impact and Responsible Choices
With the conversation around green chemistry getting louder every year, questions keep coming up: Where does it come from? How do we make it in a way that doesn’t harm the planet? Historically, manufacturers made esters like methyl n-hexanoate from petrochemicals. Some companies have started turning to renewable sources, using biological fermentation and plant feedstocks. That shift can help cut carbon emissions. Regulators such as the Environmental Protection Agency and the European Food Safety Authority watch these chemicals closely. Ongoing research makes sure no unwanted pollutants slip through production or disposal.
Transparency for Everyone
Information about food additives like this doesn’t hide behind a paywall. In my own work, I’ve seen customers demand straight answers and clean labeling. Sharing the chemical formula isn’t just for scientists—parents and food lovers alike look for peace of mind in what fills their plates, perfumes their homes, and brightens their skin. That level of transparency reflects the new normal.
Bridging the Gap
You don’t need a lab coat to appreciate what goes into familiar scents and tastes. Anyone benefits from understanding what C7H14O2 supplies in daily life, both in terms of function and peace of mind. The foundation of good products—and trust in them—starts with clear chemistry. That remains just as important as any catchy advertisement.
Is Methyl N-Hexanoate hazardous or toxic?
Understanding the Chemical
Methyl n-hexanoate isn’t a household name for most people, but it does show up in a handful of industries. It’s an organic compound with a faint, fruity odor, showing up sometimes in the flavors and fragrances world. Most folks who encounter it work in a lab or industrial setting. So, is it something to worry about from a safety standpoint, or can it be handled as casually as table salt?
Looking at Exposure and Health Effects
No chemical deserves absolute trust, and methyl n-hexanoate offers its own set of precautions. Experts at the European Chemicals Agency classify it with an eye to irritation. High concentrations may irritate eyes, skin, or respiratory passages, especially if someone breathes the vapors often or accidentally splashes it on themselves. My own work in a university chemistry lab hammered home that respect for even mild irritants prevents headaches—literally and metaphorically. Splashes on skin sting more than expected, and eyes react even before you know what happened.
Swallowing methyl n-hexanoate won’t typically send a person to an emergency room, but it’s far from harmless. Ingesting lab-grade chemicals introduces risks beyond irritation, including possible nausea or upset stomach. At elevated doses, animal studies sometimes show more significant effects, though large exposures do not usually happen under normal industry rules. Data shows acute toxicity figures aren't as frightening as with more aggressive solvents, yet the lack of detailed studies on chronic exposure signals that caution should remain the default approach.
Flammability Concerns
Fire hazards address the next real issue. Methyl n-hexanoate lights up more easily than water, making it a fire risk if someone gets careless or lets vapors build up near an ignition source. A friend in chemical manufacturing once shared a story about a minor lab fire caused by a tiny puddle of evaporated ester meeting a heat source—a harsh lesson on storage and labeling. The scent might seem pleasant, but it doesn’t warn of danger. Proper storage in ventilated, cool areas away from spark sources saves far bigger headaches than cleaning up after a small accident.
Environmental Impact
Spills don’t just stay in the lab. Esters sometimes drift into waterways or soil and break down less quickly than expected. Data from environmental agencies show that methyl n-hexanoate doesn’t stick around forever, but it doesn’t disappear overnight. Aquatic life sometimes reacts poorly to even moderate concentrations. I’ve walked past industrial discharge streams near chemical plants, and most folks in the environmental field would argue for vigilance with all organic pollutants—no matter how “mild” they seem. Disposal methods matter. A chemical that’s simple in one setting causes harm down the line if it ends up in the wrong place.
Safer Handling and Alternatives
Protective gear earns its keep—goggles, gloves, and lab coats aren’t just for show. They cut down the risk and keep routine exposures trivial. Fume hoods and well-labeled workspaces chip away at workplace incidents. Clear training ensures everyone knows not to treat seemingly gentle chemicals with a careless attitude. Before use at scale, risk assessments should remain part of standard procedure.
Not every application justifies using a chemical with fire and exposure risks. Alternatives sometimes deliver the same function with a lower hazard profile. In the world of fragrance chemistry or specialty synthesis, safer substitutes aren’t always available, but companies now put stronger pressure on suppliers to identify and switch when possible.
The Takeaway
Methyl n-hexanoate brings some hazard, though less than the most notorious chemicals. Respect, not fear, is the right companion. Eyes open, gear on, and care at every step keep this chemical from turning a workday sour.
What are the storage recommendations for Methyl N-Hexanoate?
Understanding Why Storage Matters
Methyl N-Hexanoate shows up on labels inside fragrance labs, flavor facilities, and even at some advanced manufacturing plants. Even before getting into technical details, anyone handling chemicals like this needs good storage habits. People make mistakes or cut corners, and that’s often when costly spills, fire risks, or ruined batches start making news.
Basic Storage Rules
The first thing that stands out with Methyl N-Hexanoate is its flammable nature. Fire is unforgiving. Store this compound in a tightly sealed container, ideally one built from glass or high-quality plastic. Keep it far from ignition sources like sparks, open flames, or heat-producing equipment. Flammable labels aren’t decorations; they point to real hazards.
I still remember an incident at a local processing site, where even a small whiff of vapor triggered a fire alarm. It wasn’t a prank—just an overlooked bottle with a loose lid sitting close to a radiator. Small things matter.
Temperature and Ventilation
Methyl N-Hexanoate can degrade in heat. Exposure to sunlight, hot plates, or even a warm windowsill can change the quality and safety profile of the entire batch. Always choose a cool, shaded area. A controlled room temperature typically works—cellars are sometimes used in older facilities, but modern storage rooms with thermostats make things easier. Let’s not underestimate the benefit of good airflow. Proper ventilation keeps vapor concentrations down, making the workspace safer for everyone involved. Invest in a ventilated cabinet if space allows—especially in labs prone to chemical traffic jams.
Avoid Water and Reactive Chemicals
This compound reacts with strong acids, bases, and oxidizers. Some folks pack chemicals tightly on one shelf to save space, but that sets up unwanted chemical contests. Separate out Methyl N-Hexanoate from anything corrosive. Moisture doesn’t belong near the storage zone; cap and seal every bottle promptly. Rusty caps mean something’s wrong and shouldn’t get ignored.
Labeling and Inventory Control
Clear labeling saves lives. Some organizations print custom hazard stickers. Handwritten labels rub off, turn unreadable, or get ignored, especially when rush jobs take over. Track everything in an up-to-date inventory. Logging batch dates helps spot aging supplies that might need disposal. One facility I toured had a lineup of alert tags for all chemicals over a year old—practical and effective.
Spill Response and Protective Equipment
Spills sound scary, but preparation works. Store personal protective equipment—like gloves and goggles—right next to the chemical supplies. Stock chemical absorbents, not kitchen towels. Run regular reminders and drills so that everyone knows where safety gear sits, and how to act fast if trouble erupts. OSHA published guidelines on spill kits and response practices; more labs should treat that as required reading, not bureaucracy.
Investing in Training and Oversight
People overlook simple things. All staff—not just managers or seasoned chemists—should walk through basic safety refreshers every six months. Facility-wide safety walks catch old leaks or expired bottles, keeping the storage area up to code and accident-resistant. Frequent visual checks work better than waiting for an audit. Documenting these steps reaffirms commitment to safety, and helps everyone—new or experienced—shape better habits.
Storing With Care Builds Trust
Good storage keeps people safe and protects investments. Anyone in the business of handling Methyl N-Hexanoate can name at least one near-miss where simple attention to detail made all the difference. Following these recommendations turns a risky activity into a responsible, repeatable process. That’s the real value—consistency, safety, and respect for everyone on the team.
What are the physical properties of Methyl N-Hexanoate?
The Basics of Methyl N-Hexanoate
Methyl N-Hexanoate stands out with a fruity, sweet odor that often reminds me of pineapple or apple-flavored candies. In a basic lab setup, the first thing you notice is its clear, colorless appearance. This ester’s physical properties give clues about its behavior, safety needs, and the way it works in real-world applications.
Boiling and Melting Point: What the Numbers Tell Us
Looking at the boiling point, you spot a reading near 151°C (around 304°F). For folks in quality control, this means the compound stays stable in room temperature formulas, but you see noticeable vapor if the process involves heat. In the winter, the melting point drops to about -70°C. That’s way below standard freezers, making the liquid state almost guaranteed in everyday use. These properties help set the rules for safe handling and storage—and keep things simple in both lab and industrial settings.
Solubility and Density: Mixing and Matching
Methyl N-Hexanoate doesn’t mix well with water. If you pour it into a beaker of water, it floats and forms droplets on the surface. That classic behavior lines up with its density, measured at about 0.86 g/cm³ at 20°C. Pour a little over ice, and you can watch it rise to the top. The low solubility makes cleanup easier since simple absorbents pick up spills.
On the other hand, throw it into alcohol or ether, and it blends easily. This makes sense with esters in general—most find good company in solvents with similar molecular character. Chemists care about those traits when they want a flavoring, a fragrance, or a solvent that won’t break down in harsh water-based environments.
Vapor Pressure and Volatility: What Goes Up, Comes Down
If you walk into a room where Methyl N-Hexanoate is used, the scent often hits fast. That tells you about its volatility. Its vapor pressure sits around 3 mmHg at 25°C, which means it evaporates steadily at room temperature but not so fast that it’s hard to control. The nose test—still a favorite for perfumers—is backed up by this fact: more pressure, more scent in the air, more need to tighten up ventilation during large-scale use.
Safety officers love how an average volatility helps avoid major inhalation risks when proper care is taken but keeps the compound effective in both small and big environments.
Practical Lessons on Handling
On my own bench, pouring Methyl N-Hexanoate means wearing gloves and goggles because the liquid can irritate skin and eyes. With solvents like this, I never take storage for granted. It belongs in a well-sealed glass or plastic container in a cool place, far from heat or open flames. Ester odor builds up in closed rooms, so a fume hood feels like insurance, not overkill.
The low flash point—about 49°C—raises fire risks if left unattended near hot equipment. Many incidents in teaching labs start with someone underestimating how fast vapors spread. Keeping labeling clear and remembering that even sweet-smelling esters demand respect help build good lab habits.
Value in Real-World Use
Bakeries, fragrance makers, and even synthetic chemists rely on these physical details. Texture, safety, and process design all connect back to basic numbers on the data sheet. Knowing the volatility, mixing ability, and thermal stability lets industry pros protect their products—and their people—by planning for both day-to-day and out-of-the-ordinary events.
From lab glass to production tank, understanding Methyl N-Hexanoate’s physical properties is never just about the numbers. It’s about working smarter and safer every day.
| Names | |
| Preferred IUPAC name | Methyl N-hexanoate |
| Other names |
Hexanoic acid, methyl ester Methyl caproate Methyl hexanoate Hexanoic acid methyl ester |
| Pronunciation | /ˈmɛθɪl ɛn hɛkˈsænoʊˌeɪt/ |
| Identifiers | |
| CAS Number | [629-27-6] |
| Beilstein Reference | **1209247** |
| ChEBI | CHEBI:21140 |
| ChEMBL | CHEMBL3180894 |
| ChemSpider | 15200 |
| DrugBank | DB14153 |
| ECHA InfoCard | 100.108.549 |
| EC Number | 214-720-9 |
| Gmelin Reference | 8829 |
| KEGG | C14385 |
| MeSH | D020942 |
| PubChem CID | 7674 |
| RTECS number | AT2620000 |
| UNII | V93FVJ44F7 |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C7H14O2 |
| Molar mass | 130.19 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.877 g/mL at 25 °C (lit.) |
| Solubility in water | Insoluble |
| log P | 2.8 |
| Vapor pressure | 0.5 mmHg (at 20 °C) |
| Acidity (pKa) | pKa ≈ 25 |
| Basicity (pKb) | 11.1 |
| Magnetic susceptibility (χ) | -6.49e-6 |
| Refractive index (nD) | 1.416 |
| Viscosity | 0.759 mPa·s (25 °C) |
| Dipole moment | 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 389.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -407.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3906.3 kJ/mol |
| Pharmacology | |
| ATC code | '' |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. H315: Causes skin irritation. H319: Causes serious eye irritation. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P273, P280, P302+P352, P305+P351+P338, P308+P313, P332+P313, P337+P313, P362+P364, P370+P378, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | > 86 °C |
| Autoignition temperature | 215 °C |
| Explosive limits | Explosive limits: 1.1–7.5% |
| Lethal dose or concentration | LD50 (oral, rat): 3200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Rat oral 5000 mg/kg |
| NIOSH | No Evidence Found |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 mg/kg |
| Related compounds | |
| Related compounds |
Methyl acetate Methyl propionate Methyl butyrate Methyl valerate Methyl heptanoate Methyl octanoate Ethyl hexanoate Hexanoic acid |

