Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate: Substance Commentary
Historical Development of Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate
Tracing the path of Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate takes you straight into the mid-20th century, when chemists set out to design pesticides that could hit fungal targets but sidestep the health burden carried by older, heavier compounds. At universities and industry labs across Germany, Switzerland, and the US, the search for a new benzimidazole derivative drove innovation. Lab and field tests shaped this molecule’s role in crop protection, especially as grains and vegetables worldwide fell to blights and molds. Researchers poured over thousands of derivatives, tweaking side chains and functional groups, zeroing in on one that mixed strong antifungal punch with manageable toxicity. This pathway reminds many of us how much progress hinges on patient lab work, years of collaborative back-and-forth, and a willingness to learn from both victories and failures in the greenhouse.
Product Overview and Key Naming
Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate—a name that’s a mouthful but familiar in scientific and agricultural circles—belongs to the benzimidazole family. Other synonyms for this substance include Carbendazim N-butyl derivative, along with historical labels such as “Fungitex NB.” On the market, product codes and trade names often shift across regions, making precise chemical identification crucial for regulators and users. Formulators choose this substance for seed treatments and post-harvest protection, thanks to a reliable mode of action that disrupts microtubule development in fungi, stalling cell division, and stopping the spread of common crop diseases.
Physical and Chemical Properties
Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate forms as a crystalline solid, presenting a white to off-white color. Lab reports reflect a melting range between 160°C and 162°C. Its structure gives rise to moderate water solubility—useful for suspension concentrates yet stubborn enough not to disappear readily in heavy rainfall. In storage, air-tight containers and stable temperature prevent degradation into less active forms, as light and humidity spark hydrolysis. Chemists value the logP and low vapor pressure for easy formulation and controlled release, allowing treatments to linger on plant surfaces. This property mix helps it stay on leaves longer than many alternatives, giving persistent protection without frequent re-application.
Technical Specifications and Label Information
Producers must standardize on purity levels, setting thresholds for impurity content, residual solvents, and moisture. At the point of sale, technical data sheets often list at least 98% purity, with clear warnings on incompatibility with strong acids or oxidizers. Labels carry directions for mixing, batch numbers, and mandatory QR codes tying to safety data sheets. Regulatory bodies like the EPA or ECHA require translated label information to guide occupational exposure and environmental risk. Rural application puts a premium on labeling that’s both readable and specific in local dialects, backing up farmer-facing extension work.
Preparation Methods and Laboratory Synthesis
Most routes start with benzimidazole, a backbone that gets functionalized through acylation and carbamoylation steps. I once helped with a pilot batch using methyl chloroformate and N-butyl isocyanate in the presence of a mild base like triethylamine. The process calls for tight temperature control, slow addition to suppress side reactions, extraction into non-polar solvent, and scrupulous filtration. After crystallization, washes clear away colored byproducts, and vacuum drying finishes off the process. The chemistry behind these steps feels familiar to anyone who’s spent hours balancing yield and purity, trying to outsmart hydrolysis or isomerization.
Chemical Reactions and Future Modifications
In the lab, the reactivity story revolves around the benzimidazolyl ring and its carbamate functionalities. Nucleophilic attack opens avenues for derivatization, helping scientists tune solubility or persistence. Mild hydrolysis, promoted by heat and humidity, splits the carbamate, sometimes reducing fungicidal strength. Looking forward, efforts steer toward attaching bulky groups that slow metabolism, cutting down on application rates and resistances. Green chemistry trends push for less hazardous synthesis, favoring water-based or mechanochemical processes and less persistent waste.
Uses and Application Areas
Farmers and agricultural specialists spray this benzimidazole on grain, fruit, and seed crops to arrest fungal rot, leaf spots, and mildew outbreaks, especially in regions that see high rainfall. Packing houses dip produce to suppress mold during shipment. In fieldwork, I’ve watched how even low concentrations prevent problems at the seedling stage, helping growers dodge catastrophic yield loss. Beyond the farm, researchers dust off methyl N-(1-N-butylcarbamoyl-2-benzimidazolyl)carbamate for cultural heritage work, stopping molds on old papers and textiles. Its reach stretches to sports turf and ornamental plant care, where healthy foliage translates to value and pride.
Safety and Operational Standards
Staying safe with this compound centers on limiting inhalation, eye exposure, and skin contact. Field guidance pushes for long sleeves, gloves, and goggles; rural workers often link accidental contact to itching and mild gastrointestinal distress, based on medical reports logged by regional clinics. National agencies demand personal protective equipment, mixing stations with fume hoods, and protocols for spill response. Storage must keep it far from feed or food prep zones, using secondary containment to catch leaks. Registered disposal routes prevent leaching into water tables, while local agriculture offices run training on proper tank cleaning. Health surveillance programs look after handlers, tracking possible allergic responses and longer-term risks.
Research, Development, and Toxicological Concerns
Much of the recent publication pipeline has pivoted toward resistance monitoring and understanding chronic exposure effects. Trials on vertebrate and aquatic life help set regulatory limits; subchronic rodent data often outline liver and reproductive endpoints, guiding permissible daily intake. Field studies measure breakdown rates in loamy versus sandy soils, offering clues for dose adjustment. Overseas, medical casework sometimes links exposure to rare toxicities, setting the stage for careful monitoring and new guidelines. Institutions push for next-generation derivatives that sidestep pathways linked to embryotoxicity or estrogenic interference, bouncing ideas off big public datasets. For those of us teaching pesticide risk, this arena matters, sharpening focus on practical, evidence-driven limits.
Future Prospects and Sustainable Directions
As climate shifts redraw fungal risk maps, this carbamate faces tougher scrutiny in terms of persistence and off-target effects. Next steps lie in smarter use—timing sprays with spore forecasting, rotating fungicides to dodge resistance, integrating biofungicides for lower chemical burden. Product re-formulation targets reduced worker exposure and smaller environmental footprints, possibly with microcapsule delivery. Biotechnology and precision agriculture sketch opportunities to track residues, not just at harvest but across the supply chain. For students and practitioners, learning this molecule’s backstory provides a window into how biochemistry, ecology, and rural economics all fit together, and why tomorrow’s success will hinge on both chemistry savvy and careful stewardship in the field.
What is the primary use of Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate?
A Closer Look at a Well-Known Fungicide
Many folks outside of farming and plant science haven’t heard of Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate. Most people call it Benomyl. People started using this compound on a large scale in the 1970s after testing showed it could knock out a tough group of plant pathogens.
Benomyl treats diseases in crops, lawns, and turf. I've seen its powder-blue residue on the leaves of fruit trees in community orchards and on putting greens at public golf courses. It zeros in on fungi that munch through crops, especially powdery mildew, blight, and other fungal invaders that hit vegetables, grains, bananas, and apples. Without treatment, these diseases slash yields and ruin harvests.
So, why has this chemical made such a difference? Benomyl blocks growth in fungus cells by shutting down their ability to divide. Fungi can't spread, giving farmers a way to break disease cycles. Before systemic fungicides like Benomyl, people relied on older chemicals like copper and sulfur that needed frequent use and still left plants vulnerable if rain came at the wrong time. Benomyl gets inside the plant, spreading throughout the tissues after a single spray, so it protects new growth from infection days or even weeks later.
Food Security and Global Trade
The importance of this compound reaches into markets and dinner tables all over the world. Bananas travel thousands of miles before they land in a fruit bowl. During that trip, fungal diseases can destroy entire shipments, which hits small farmers hardest. Countries like Costa Rica and the Philippines have relied on Benomyl to protect their harvests since the 1970s. The same goes for apple, carrot, and bean growers fighting root rot and other diseases that spoil produce before it can be sold. Studies from the Food and Agriculture Organization have shown fungicides like Benomyl help save up to 20% of world food production.
Impact on Workers and the Environment
Whenever chemical sprays enter the scene, concerns about human health and environmental impact follow. There have been well-documented cases of Benomyl linked to worker rashes and, very rarely, birth defects in lab animals. The manufacturer, DuPont, pulled the chemical from US markets over two decades ago after lawsuits and stricter regulations on chemical safety. France, the UK, and much of the EU followed. Although not legal everywhere, Benomyl still finds use in many developing countries because few alternatives offer broad, reliable protection at a low price point.
From a practical spot in the field, reducing the use of legacy pesticides takes teamwork among researchers, governments, and farmers themselves. Integrated pest management gives an option—combining resistant plant varieties, crop rotation, and safer chemicals means fewer side effects for families and wildlife. These solutions also look at soil and water health. When universities like UC Davis teach growers to spot early warning signs of disease, farmers spray less. Funding for newer, cheaper, and safer fungicides will decide how quickly the shift away from old chemistries happens.
Benomyl serves as a reminder: every chemical tool in agriculture solves a problem but leaves new questions. I’ve heard growers say they worry about the day when no simple solution sits on the shelf. Until safer replacements arrive, Benomyl holds a complicated spot in the fight for food security and plant health worldwide.
What are the safety precautions when handling this compound?
Respect the Health Risks
Chemical compounds come with real risks. Many people treat lab work or industrial sites like routine settings. Underestimating the danger can cause serious injury. Some compounds damage your lungs, others burn your skin, and a few slip through gloves you thought would protect you. If you cut corners, you gamble with your health. I’ve seen smart folks hesitate to put on protective gear because “it’s a quick task.” The reality is, accidents tend to show up during those so-called quick moments.
Gear Matters More Than You Think
Never take chances on eye or skin safety. Chemical goggles, not cheap safety glasses, should cover your eyes if there’s any risk of splashing. If the instructions call for gloves, check the glove material against what you’re handling. Nitrile doesn’t help much if a solvent seeps through it. Replace gloves with holes or stains. A lab coat and closed shoes keep splashes away from your body. When I ran my first reaction, my mentor tossed me a face shield for anything volatile — I soon realized seeing well does not help if you’ve exposed your skin.
Good Ventilation Isn’t Optional
Fume hoods exist for a reason. Breathing toxic or irritating vapors is a mistake you don’t want to make twice. If the compound gives off fumes, always use a hood or exhaust system. Cracking a window doesn’t cut it. When I worked with ammonia, the hood’s fan never got switched off, even for “just a minute.” Respect for these precautions kept everyone healthy.
Label and Store Everything
An unlabeled container causes confusion and danger. I once saw a technician pour what looked like water into a sink — turns out, it was a strong acid. That could have ended badly. Always label your bottles with the compound name, concentration, and date. If you’re storing reactive compounds, read the storage guidelines. Some chemicals catch fire in the air or react with water, so dry storage or sealed cabinets prevent a disaster.
Spill Response Needs Training
Plan for spills before they happen. Spill kits should sit somewhere easy to reach, with absorbent pads, neutralizing agents, and protective gear included. Trained staff react faster and more effectively than someone reading the kit's instructions for the first time. Where I worked, we drilled spill cleanup like fire drills. That way, panic never took over — everyone knew where to move and what gear to grab.
Stay Informed; Stay Vigilant
Safety data sheets don’t gather dust for a reason. They tell you about the hazards, symptoms of exposure, and how to handle accidental contact. Regular training sessions go a long way, especially as new employees come in and procedures change. Those who ignore safety practices usually regret it later. Staying up to date and taking precautions keeps the workday from turning into an emergency.
Looking Forward
Chemicals don’t forgive mistakes. If you skip steps or ignore warning signs, you put everyone around you at risk. Building a strong safety culture takes time but pays off every shift. Clear procedures, reliable access to the right gear, and steady training lower the chances of something going wrong. When everyone respects the risks, the lab or workplace becomes a safer, more confident place to get good work done.
Is Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate toxic to humans or animals?
Digging Into the Facts
Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate goes by a more familiar name for lots of folks working around crops and livestock—this compound forms the backbone of benomyl, a popular fungicide. Scientists and farmers alike have relied on these kinds of chemicals for decades, especially for fighting blights and fungal diseases in fruits, vegetables, and even turfgrass. If you ever walked through an orchard after a rainy spell, odds are you brushed up against fruit protected by chemicals in benomyl's family.
Concerns come up because, like most synthetic fungicides, questions swirl around benomyl's safety for farmhands, consumers, pets, and wildlife. In the late 1990s, multiple studies highlighted toxic effects on lab animals when the chemical was given in high doses. Rats and rabbits developed birth defects and liver trouble, showing that, at least at those high levels, the compound has the ability to disrupt normal functions.
What Science Says About Exposure
It’s important to think about how people and animals actually come into contact with this compound. Handling undiluted products, inhaling fine powders, or eating contaminated produce could lead to higher risk. The US Environmental Protection Agency reviewed benomyl data and, after reviewing the evidence, flagged risks for those who mix and apply the product without adequate protection—things like skin contact, eye exposure, or accidental inhalation.
For everyday consumers, residue levels on food typically remain low, but long-term or repeated exposure remains a question mark. Agencies asked food producers to keep residues below strict safety cut-offs, so in most markets, regulation helps curb acute risk.
Long-Term Effects and Environmental Issues
Long-term health risks always attract questions. In the case of benomyl, chronic exposure in animal tests caused liver damage and changes in blood chemistry. Some studies linked the compound to reproductive harm, making the stakes even higher in places where families farm or live near treated fields.
Wildlife also faces risks. Fish and aquatic invertebrates react badly to runoff, while beneficial insects get hit if sprays drift. Years back, the US banned benomyl after lawsuits over birth defects and growing environmental evidence—an example of regulators stepping in when risks outweigh the benefits.
Facing the Risks
Chemical safety improves fastest when people get clear, accurate information and act on it. Where I’ve worked in agriculture training, nothing replaces protective gear and careful mixing habits. Farmers who track weather and use precision sprayers do better at keeping chemicals in the right place. Sometimes, switching to alternative fungicides, rotating crops, or trying resistant plant varieties can all push chemical use down and lower risk for families, farm workers, and animals.
Consumers get extra security from food testing, better washing routines, and pushing for tighter safety limits. Many countries with the resources run regular residue checks on fresh produce, keeping contamination below harmful levels. Still, imported crops sometimes slip through cracked inspection nets, which means strong international cooperation matters just as much as local rules.
Looking Forward
Stories like that of methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)carbamate show the balancing act behind modern food systems. Today, constant re-evaluation and investment in safer farming methods can make the difference for communities, food buyers, pets, and ecosystems. Facts come first—then clear action to protect health. It’s not about avoiding all risk, but about making smart moves with solid information in hand.
How should Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate be stored?
Taking Chemical Storage Seriously
Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate sounds like a mouthful, but for anyone who works in agriculture or chemical labs, this compound has a practical side. It’s better known by its trade names in some pesticides and fungicides. Letting it degrade or become contaminated could mean serious risks for both users and the ground it touches.
Storing this chemical goes beyond stacking bags in a shed. Personal experience working in a rural cooperative taught me early on that shade, ventilation, and vigilance can make the difference between a usable product and a costly clean-up. One heated summer, a barrel stored in direct sunlight started to leak and ruined a whole section of garden supply. The lesson stuck: chemicals can spoil, and so can livelihoods.
Key Considerations for Handling
The manufacturer’s label pins the basics. This compound doesn’t love heat, moisture, or direct sunlight. If temperatures creep above 30°C (86°F), breakdown speeds up and fumes can build. These fumes aren’t just unpleasant—they’re dangerous. The EPA lists this active ingredient among hazardous materials, and inhalation can harm respiratory health over time.
Storing it in a dry, cool, well-ventilated spot isn’t negotiable. Locked cabinets with metal shelves, away from direct heat sources, stand up over the long run. I never leave it beside fuels or oxidizers, either. Once, a contractor in my town stacked a container near a rusted can of bleach. That story ended with a call to the hazmat team.
Labels and Record-Keeping Matter
Labels never go in the trash. Every repackaged batch needs a clear tag, not just for legal reasons, but so nobody mixes up dangerous compounds. Detailed logs of what comes in and out prove essential when something goes wrong. In my work, inspections by safety officers caught mistakes that saved both money and reputations.
Personal Safety and Local Laws
Gloves, safety glasses, and protective clothing rank above comfort. The CDC reminds workers every year: skin absorption and inhalation still send folks to the emergency room. Never open the container in the living areas or kitchens to avoid accidental contact.
Most people forget about local and federal laws. In the United States and Europe, failing to follow rules on hazardous chemical storage brings steep fines and, worse, community health risks. My local farm bureau holds yearly workshops to keep members aware, but some still think "out of sight" beats safe practices. If in doubt, the local fire department or poison control center can give solid advice.
Better Solutions and Community Knowledge
I’ve seen community-run storerooms for chemicals in small farming towns where everyone shares proper facilities. These setups, monitored by a trained manager, prevent both theft and accidental misuse. Bigger organizations install climate-controlled storage and use spill containment kits. My local school switched to digital logs for inventory, which flagged expired containers and kept things transparent.
As someone who’s learned a few hard lessons from chemical mishaps, I see value in sharing knowledge instead of hiding problems. By focusing on secure storage, honest labeling, and worker education, it’s possible to work with tough compounds like Methyl N-(1-N-Butylcarbamoyl-2-Benzimidazolyl)Carbamate without putting people or the planet in harm’s way.
What is the recommended method of disposal for this product?
Why Proper Disposal Choices Matter
Disposal often shows the gap between convenience and responsibility. Years back, I worked at a local recycling center, sorting through endless piles of leftovers from daily life. From that side of the process, it’s easy to see how choices at home ripple outward. Tossing a battery or an old phone in the regular trash doesn’t seem dramatic. Yet, hazardous bits of these devices leach into landfills, and the cumulative effect harms water, soil, and eventually people.
Consider electronics. According to the EPA, only about 15–20% of e-waste ends up recycled. A big chunk lands in dumps here or shipped overseas, where regulations grow thinner. Toxic metals like lead, cadmium, and mercury drift into the environment from there. Simply handing your old gadget to a designated drop-off—often available free at big box stores—keeps those toxins out of local streams and lets trained workers reclaim rare materials.
Community Solutions and Local Knowledge
Local governments develop different programs to handle everything from paint to prescription drugs. A city like San Francisco bans plastic bags entirely, while others offer specialty pickups for household hazardous waste. Yet many people find the right disposal site confusing—so confusion leads to shortcuts. Even after years of working in waste management, friends and family still ask the same questions: Where should expired medicine go? What about broken thermometers?
Experience shows the best path is often to check city and state web pages for updated guides. They point to pharmacies for drug take-back, detail which plastics can hit the curb, and list scheduled toxic drop-off days. If that sounds complicated, it’s because regulations try to strike a balance between cost, safety, and environmental concerns. The sticker on a product means little if people can’t easily find info at home.
The Human Side of Everyday Disposal
Teaching kids about waste turns responsibility into habit. My youngest—now in high school—spent an afternoon on a class trip touring the municipal dump. Seeing TV screens shatter and paint cans spill drove home lessons that lectures can’t. Even one household sorting batteries and compost from general garbage shapes cleaner communities.
Manufacturers share part of the burden. Some now label packaging with clear recycling symbols and reduced ink. Popular clothes retailers feature in-store bins to reclaim hangers, used jeans, or shoes. This producer involvement nudges consumers forward, but policy can push harder. Extended producer responsibility laws, in place across Europe, force companies to fund collection and recycling.
Building a Stronger Culture of Accountability
A simple act—checking product packaging or a city’s drop-off map—pushes change along. It’s less about feeling guilty for mistakes and more about seeing disposal as part of everyday stewardship. If cities put out more language-friendly guides and retailers keep drop boxes handy at every store, the right choice becomes the crowd’s habit.
Deciding where to toss something may feel small, but it stacks up. Over a life, it means cleaner rivers, less toxic residue, and neighbors growing up just a bit safer. Every product, whether a leftover paint can, an LED bulb, or the phone you upgrade every couple of years, deserves a final chapter that keeps health and environment in mind.
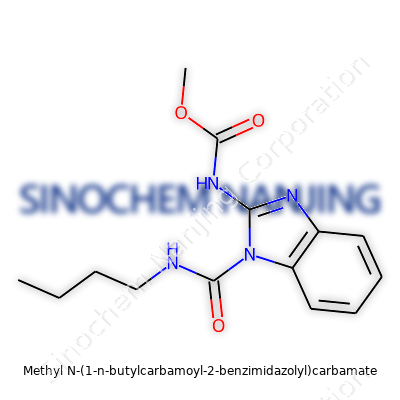
| Names | |
| Preferred IUPAC name | methyl N-{[1-(butylcarbamoyl)-1H-benzimidazol-2-yl]carbamoyl}carbamate |
| Other names |
Benomyl Benlate Fundazol Fungicide 1991 Vangard Benom Carbendazim methyl Benzimidazolyl methylcarbamate |
| Pronunciation | /ˈmɛθ.ɪl ɛn ˈwʌn n ˈbjuː.tɪlˌkɑːr.bə.mɔɪl tuː bɛnˈzɪm.ɪdˌzɔːl.il ˈkɑːr.bə.meɪt/ |
| Identifiers | |
| CAS Number | [3615-21-2] |
| 3D model (JSmol) | `3D Model (JSmol) string`: `CCCCC(=O)NC1=NC2=CC=CC=C2N1C(=O)OC` |
| Beilstein Reference | 76334 |
| ChEBI | CHEBI:8165 |
| ChEMBL | CHEMBL2106417 |
| ChemSpider | 22275 |
| DrugBank | DB00730 |
| ECHA InfoCard | 100.080.235 |
| EC Number | 3.5.1.86 |
| Gmelin Reference | 333086 |
| KEGG | C07536 |
| MeSH | D008772 |
| PubChem CID | 6918207 |
| RTECS number | EB2975000 |
| UNII | VAO9S9UX4T |
| UN number | 2811 |
| Properties | |
| Chemical formula | C13H18N4O3 |
| Molar mass | 326.38 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.31 g/cm3 |
| Solubility in water | Insoluble in water |
| log P | 0.97 |
| Vapor pressure | 1.73 x 10^-7 mmHg at 25°C |
| Acidity (pKa) | 7.8 |
| Basicity (pKb) | 3.75 |
| Magnetic susceptibility (χ) | -78.9×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.648 |
| Dipole moment | 4.06 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 577.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -399.8 kJ/mol |
| Pharmacology | |
| ATC code | P02CA02 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS06, GHS08 |
| Signal word | Warning |
| Hazard statements | H302, H319, H410 |
| Precautionary statements | P264, P270, P280, P301+P312, P305+P351+P338, P308+P311, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-1 |
| Flash point | 137°C |
| Autoignition temperature | > 410 °C (770 °F; 683 K) |
| Lethal dose or concentration | LD50 oral rat 1190 mg/kg |
| LD50 (median dose) | LD50 (median dose): 930 mg/kg (oral, rat) |
| NIOSH | SKN sensitizer |
| PEL (Permissible) | PEL (Permissible) of methyl N-(1-n-butylcarbamoyl-2-benzimidazolyl)carbamate: 5 mg/m3 (OSHA as total dust) |
| REL (Recommended) | 0.1 mg/kg |
| Related compounds | |
| Related compounds |
Carbendazim Benomyl Thiophanate-methyl Methyl benzimidazol-2-ylcarbamate N-Butylcarbamate |

