Methyl Isovalerate: Commentary on Its Historical Development and Modern Relevance
Historical Development
Methyl isovalerate has been around for well over a century, floating quietly through the chapters of organic chemical research. Chemists began to notice the ester’s pleasant fruit-like scent in early explorations of volatile plant compounds. Its discovery is a testament to the close tug-of-war between art and science—where natural aromas inspire synthetic replication. Early researchers wanted to replicate flavors and fragrances found in rare botanicals, since making them directly was costly or impractical. Drawing on basic esterification techniques, scientists started producing methyl isovalerate in larger batches by reacting isovaleric acid with methanol. Over time, availability expanded, and standards improved as chemical industries matured—turning what was once a curiosity into a commercially significant chemical.
Product Overview
This compound is mostly known as a flavor and fragrance ingredient, but it also sneaks into specialty solvents and intermediate roles for chemical synthesis. It draws the attention of perfumers and product formulators hunting for a sweet-yet-earthy undertone, while analysts use it to benchmark instrument sensitivity due to its notable volatility. From laboratories to food manufacturers, methyl isovalerate plays a supporting role that helps creators achieve specific aroma profiles or adjust volatility in custom products. Its market presence isn’t huge compared to major industrial solvents, but it fills a unique gap, especially in the niche where flavor science meets industrial chemistry.
Physical & Chemical Properties
Methyl isovalerate appears as a colorless, clear liquid with a strong, fruity odor reminiscent of ripe apples and grapes. Its boiling point hovers around 110°C under reduced pressure, and it has a low melting point near -90°C. The density falls slightly below that of water, which means spills tend to spread fast—an accident in the lab will scent the air before long. It dissolves readily in most non-polar organic solvents but shuns water due to its hydrophobic alkyl chain. The vapor pressure marks it as a fairly volatile compound, explaining why its aroma is caught on the nose so quickly.
Technical Specifications & Labeling
Careful quality control separates technical grade from food and fragrance grade batches, with GC-MS analysis revealing impurities that would affect either performance or safety. Product purity above 97% is standard for food applications, while industrial users might tolerate slightly more byproduct, especially if the ester serves as a chemical intermediate. Labels should carry hazard symbols since inhalation or skin contact leads to irritation, especially at higher concentrations common in industrial drums. Standard packaging ranges from sealed glass bottles for lab-scale work to steel drums with vented closures for bulk delivery.
Preparation Method
The classic route to methyl isovalerate uses Fischer esterification. Isovaleric acid, often derived from the oxidation of plant-based amino acids like leucine, meets methanol under a strong acid catalyst, usually sulfuric acid. Heat and vacuum strip away lingering water to drive the equilibrium to completion. Chemical companies streamline this with continuous distillation, recycling unreacted starting materials and trimming the energy footprint where possible. Research labs will sometimes swap in alternative methods, such as enzymatic catalysis, for specialty applications needing greener credentials or enhanced selectivity.
Chemical Reactions & Modifications
Methyl isovalerate resists many mild reagents, thanks to the stability of the ester functionality. Strong bases or acids can cleave it by hydrolysis, regenerating the parent acid and methanol—this reaction sits at the heart of both industrial recycling and laboratory cleanup strategies. Nucleophilic substitution on the methyl group does not proceed under normal conditions due to poor leaving group properties, but vigorous hydrogenation or oxidation will tear into the molecule, forming smaller fragments or alcohols. Creative chemists sometimes tweak the isovaleric backbone to produce analogues with related scents, extending the use of this core building block.
Synonyms & Product Names
Methyl isovalerate turns up in catalogs and research as methyl 3-methylbutanoate—a name that foregrounds its branching chain structure. Older literature or fragrance industry references might list it as methyl isopentanoate. International chemical suppliers offer it under brand names tailored to specific regions, so ingredient labels in consumer goods could list it differently depending on regulatory norms. Consistency in naming developed as global chemical databases integrated and harmonized synonym lists, making it easier for buyers and professionals to track and compare product specs.
Safety & Operational Standards
Methyl isovalerate comes with a set of risks—nothing dramatic compared to many industrial chemicals, but repeat exposure will irritate eyes, respiratory tract, and skin. The fruity odor doesn’t mask its potential for harm at higher concentrations. Good ventilation counts for a lot in labs or production lines using this ester. Regulations direct safe storage away from heat and open flames, given its low flash point and vapor-forming habits. MSDS sheets spell out spill response, usually involving absorbent materials and prompt disposal due to flammability. Eye wash stations and gloves stand as first lines of defense for teams making or using this compound.
Application Area
Flavorists lean on methyl isovalerate for its ability to mimic fruit and sweet dairy notes in candies, baked goods, and beverages. Its intensity and volatility help lift top notes in perfume blends, especially in genres echoing orchard fruits or green apple. Some detergent companies add trace amounts to balance harsh odors left behind by cleaning agents, while paint and resin chemists have turned to it as a reactive diluent or intermediate in custom polyester formulations. The ester occasionally finds work in medical research as an internal standard or calibration tool due to its reliability and detectability in trace analysis.
Research & Development
Recent innovation sees enzymatic and bio-based production methods gaining ground, both to cut down on petrochemical feedstocks and to meet consumer appetite for “natural” flavor ingredients. Academic labs publish papers exploring new catalysts for higher yields or milder conditions, chasing cost and environmental improvements. The challenge lies in matching the scalability of classic acid-catalyzed synthesis. Startups and established players alike dabble with genetic engineering of microorganisms to ferment isovaleric acid from renewable resources, closing the loop for future green chemistry applications.
Toxicity Research
Toxicological studies show moderate acute toxicity, with LD50 values (oral, rat) in the thousands of mg/kg—so accidental exposure, while unpleasant, rarely spells disaster. Chronic exposure data remain cagey, but regulatory agencies note the lack of definitive carcinogenic or mutagenic evidence in animal models. In vitro testing points to cell irritation at higher levels, with long-term inhalation flagged as a potential risk for workers in industrial settings. Consumer risk stays low due to modest concentrations in foods and perfumes, but pre-market safety assessments continue, especially as the hunger for “clean label” product grows stronger.
Future Prospects
Methyl isovalerate rides the wave of natural flavors, as regulators sting harder on artificial ingredients in food and fragrance markets. Expanded bioprocessing could reshape its production costs and green credentials. Digital marketplaces and global chemical databases lower the barrier for small companies to source specialty esters, helping niche brands break through with innovative products. Researchers keep digging into new properties and reaction pathways, while toxicologists refine safety limits in light of fresh data. The story for methyl isovalerate points to a future where targeted applications and updated safety playbooks keep it useful in both traditional and emerging sectors.
What is Methyl Isovalerate used for?
The Real Reason Fragrances Stand Out
Step into a bakery or a perfumery. The warmth in the air, that distinct fruity aroma, often traces back to more than just flour or flowers. Methyl isovalerate, a clear liquid with a sweet, apple-like scent, often makes appearances in places you’d least expect. My own first encounter with it happened during a high school chemistry project. The lab filled with the gentle scent of apples the moment our teacher uncorked the bottle.
This compound’s pleasant scent comes in handy for those who design scents and flavors. Perfume makers, for example, rely on its fruity, crisp signature to soften sharper notes or lift heavier bases. In the world of food, its use changes the experience of eating: a touch of methyl isovalerate inside a yogurt or candy delivers the essence of apples or pineapples without ever peeling actual fruit.
Digging Into the Science — And the Impact
Composed of carbon, hydrogen, and oxygen, methyl isovalerate gets produced artificially, but it also exists in nature — apples, blackberries, and even cheese carry traces. Its low toxicity and easy breakdown in the environment help ensure safety when used as directed, meeting many food safety and fragrance standards. This quality matters to me both as a consumer and as someone who’s read enough recall notices to know regulators pay attention to even trace amounts of compounds in processed food.
According to food and fragrance regulatory groups like the US Food and Drug Administration and the European Food Safety Authority, methyl isovalerate can be safely used in small amounts in foods and personal care products. Makers conduct rigorous safety tests, and plenty of independent studies back up these assessments.
Bigger Picture Uses—And Risks
The reach goes further than flavor and scent. Methyl isovalerate finds a niche in the lab as a solvent or chemical building block for plastics and other synthetic materials. Its manageable volatility and solubility—how easily it evaporates or mixes—give chemists room to experiment with new reactions and products.
Of course, overuse or careless handling creates problems. In industrial settings, spills or poor ventilation could irritate eyes, nose, or skin, so companies follow strict exposure limits. Personal experience on the factory floor taught me that the biggest risks come from ignoring the basics: proper storage, labeling, and adequate ventilation save headaches, literally and figuratively.
What’s Next for Methyl Isovalerate?
As more people demand safer, environmentally friendly flavors and scents, interest in natural sources continues to grow. Some manufacturers turn to fermentation instead of crude oil or petrochemical synthesis, swapping giant refineries for bio-based reactors. This isn’t just a marketing shift—consumer confidence in “natural” ingredients continues to rise, and the science backs up their gentler impact on local ecosystems.
Better education and clear labeling let consumers decide for themselves, so transparency in sourcing and manufacturing steps up as a key industry standard. For foodies, scent fans, and everyday folks concerned about what goes into the home or body, information matters. I appreciate knowing that a familiar apple note in my shampoo comes not just from a lab, but from a well-understood and scrutinized process.
Is Methyl Isovalerate safe to handle?
What Methyl Isovalerate Brings to the Table
Methyl isovalerate smells a bit like apples and pineapples, which is why the chemical often finds a spot in perfumes and flavorings. Many folks working in labs or food businesses bump into this compound. This naturally leads to questions: Is handling it a big deal, or is it just another harmless scent molecule?
What Science Says about Handling
This chemical belongs to a class known as esters. Most esters share similar risks—low toxicity, but they can irritate the eyes, nose, and skin. Methyl isovalerate isn’t different. Touching it without gloves can cause red, itchy skin. Breathing in a little might make your nose tingle or lead to a cough, especially in places with poor ventilation. Getting a mouthful isn’t wise, either. Swallowing chemicals can have unpredictable results; while methyl isovalerate isn’t known as a poison, it could upset the stomach or trigger nausea.
Stories from the Lab Bench
Ask anyone with time in a college chemistry lab, and they’ll tell you: Respect every chemical, even those with pretty smells. Once, during an undergraduate synthesis, the sweet scent of methyl isovalerate drifted out of a flask. A labmate leaned in for a sniff and ended up with watery eyes for the rest of the session. Most esters behave like that. It pays to treat them with caution and not just out of habit but out of a respect for long-term health.
Reliable Rules for Safe Work
Most safety data sheets (SDS) echo the basics: use gloves, goggles, and work near a fume hood or open window. In my own experience running student labs, gloves always come first. Fume hoods keep the air fresh and reduce headaches or throat tickles. Sometimes students get careless—running a quick taste or sniff “just to check”—and always regret it. Sticking to standard precautions blocks most problems before they start.
Risk Is Always Relative
Chemicals like methyl isovalerate rarely cause disaster by themselves. Trouble usually starts with shortcuts. Spills soak into skin or get into cuts. Vapors collect in small rooms, leading to headaches or more serious breathing trouble. It’s not about avoiding methyl isovalerate entirely—this substance plays a useful role in making flavors and fragrances. The problem shows up with careless handling. No one flips on a stove burner with a bottle of flammable liquid open next to it at home; the same straight thinking should apply in a workplace or lab.
What Makes a Real Difference
Information, not fear, brings safer workplaces. Training new employees—making sure they know what gloves to grab, which masks to wear, and how to clean up spills—gets real traction. Companies willing to invest a few extra minutes in training cut down on injuries and sick days. Better lab ventilation brings fresher air and less risk. PPE (personal protective equipment) might seem like a hassle until it saves someone’s eyesight or skin.
Practical Steps Forward
Provide new workers with hands-on demonstrations and quick, clear fact sheets. Make sure those safety goggles and gloves stay within reach, not locked in a back closet. Set up easy-to-read warning labels; confusion never helped anyone. Design workspaces so they’re easy to keep clean, with good airflow. If everyone learns to treat all chemicals—methyl isovalerate included—with an equal shake of caution and common sense, then problems stay small and rare.
What are the physical and chemical properties of Methyl Isovalerate?
Getting to Know Methyl Isovalerate
Methyl Isovalerate pops up in flavors and fragrances more often than many people realize. This colorless liquid brings a fruity aroma that’s sometimes compared to apples or berries. That familiar scent owes much to its chemical structure, which makes it a favorite for adding subtle sweetness in perfumes and food essences.
Physical Characteristics You Notice Right Away
You can spot Methyl Isovalerate by its light, volatile nature. It evaporates quickly, so you catch its scent even from an unopened bottle. With a low boiling point hovering near 142°C (287°F), this compound vaporizes more easily than water. It sinks in water though, thanks to a density around 0.87 grams per cubic centimeter. Sometimes, that means a leak leaves behind an oily residue, which tells you to grab gloves and a mask if you’re handling a spill.
It doesn’t blend well with water—there’s always a separate layer because of the way its molecular structure repels water molecules. On the other hand, it mixes smoothly with many common organic solvents like ethanol, which lets manufacturers use it in different product bases without much trouble.
How Chemistry Shapes Its Use
Methyl Isovalerate packs an ester group at its core. Esters are big in the world of flavors because they break down in the body to harmless byproducts and mimic natural smells. The molecule builds on isovaleric acid and methanol, which not only shapes its scent but also affects its reactivity. It turns out this ester is stable at room temperature but starts to degrade or form acids under high heat or strong acid environments.
You wouldn’t call Methyl Isovalerate highly flammable, but it does catch fire if you give it enough heat—something to remember in factories or labs. Static discharge or sparks near stored bulk material can become a big headache overnight, and safety rules cover these basics for good reason. Breathing its vapors in high amounts can irritate eyes and throat, though most people have to be in close quarters with vats of the chemical to notice. Common sense says to keep it away from open flames and wear personal protective equipment in places where it’s poured or transferred.
The Stakes in Handling and the Environment
Companies that work with Methyl Isovalerate monitor storage, not just to avoid accidents but also to keep the aroma pure. Oxygen and moisture in the air trigger chemical changes that weaken both the scent and safety, so airtight containers make a difference. Leaks also bring up environmental questions. This compound breaks down with exposure to sun and air, but in large amounts, runoff can harm water sources—fish and insects respond quickly to even tiny traces. Some labs run simulations to check breakdown rates and ensure spills won’t cause lasting trouble.
Finding a Smarter Way Forward
The fact that Methyl Isovalerate behaves as both a helpful tool and a potential hazard calls for a careful, informed approach. Training workers on the right protective measures, re-engineering ventilation, and running spill drills matter just as much as running product tests. It’s one example where knowing the specifics of a chemical pays off in safety, quality, and peace of mind for everyone from the person at the factory floor to the consumer opening a scented candle at home.
How should Methyl Isovalerate be stored?
Understanding What Methyl Isovalerate Needs
Methyl Isovalerate pops up in plenty of workplaces where flavors, fragrances, or specialty chemicals get top billing. With a fruity aroma and a tendency to evaporate fast, this liquid turns heads in labs. Handling it isn’t rocket science, but skipping the basics leads to trouble down the line.
The Real Hazards Behind the Aroma
You’ll notice the sweet, apple-like scent right away, but beneath that, methyl isovalerate brings some risks most folks overlook. It can irritate the skin and eyes, punch up headaches when vapor concentrations climb, and if stored sloppily, invites both fire danger and workplace slip-ups. The volatile nature means it wants to escape the bottle and hang in the air, which a closed, controlled storage setup helps prevent.
Finding the Right Container and Location
Experience teaches that glass or tightly sealed metal containers offer peace of mind. A leaky plastic bottle lets vapor drift into workspaces, and before you know it, complaints arrive from the folks next door. Store the container upright, label it like you care about your coworkers, and keep it where it won’t tip or get knocked over. Pick cabinets designed for flammable liquids; wood with a chemical-resistant liner or steel both stand up well.
Leave it in a cool, dry spot out of direct sunlight. Heat speeds up evaporation, and sunlight kicks off chemical changes in sensitive organics. A basement shelf might seem out of the way, but temperature swings down there lead to condensation and corrosion. Find a place away from heat sources like radiators, steam pipes, or coffee makers. Vapor build-up in a warm room goes unnoticed until it becomes a bigger problem.
Keep Away from Problem Chemicals
Flammable solvents, oxidizers, and acids spell trouble for methyl isovalerate. One slip — maybe a splash during cleanup, maybe an accidental swap of bottle caps — turns a routine day upside down. Gloves, eye protection, and a healthy respect for separation keep these headaches away. Store methyl isovalerate with other esters or compatible organic liquids, but never lump it in with strong bases, peroxides, or nitric acid.
Ventilation Stops Accidents
No one likes the sting of chemical vapors hanging in the air. Keeping storage areas well-ventilated lets any stray vapor out fast. If the building already features exhaust fans above storage cabinets, use them. If not, open a window on workdays and keep traffic around the storage site to experienced staff familiar with the risks.
Permanent Labeling and Clear Records
I’ve seen old containers lurking in dusty corners, their labels faded to a whisper. This guarantees confusion. Each bottle deserves a clear, waterproof label showing both the chemical name and the date received. Log every new delivery and each use, so no one grabs a mystery bottle a year down the road.
Responding to Leaks and Spills
Small spills mean don’t panic, but don’t delay either. Ventilate fast, throw absorbent pads or sand on the spot, and scoop everything into a sealed waste container. Floors get slick in a hurry. Large leaks? Follow the employer’s spill protocol and get help.
Treat Storage as Ongoing Work
No storage setup stays good forever. Check bottles for cracks or residue, and inspect cabinets for corrosion and broken locks. Review logs and restock absorbent materials. Keeping up these habits proves you value both your work and the people doing it alongside you.
What are the common applications of Methyl Isovalerate in industry?
A Flash of Fruity Scent—Why Methyl Isovalerate Matters
Step into any factory that churns out flavors and fragrances, and you'll catch the secret helpers long before you see them. Methyl Isovalerate gives off that crisp, fruity, slightly sweet aroma—think apples and pineapples blended together. You’ll find it at work behind the scenes, creating those food and personal care products that people enjoy every day. It’s not just there for show: it plays a real functional role and gets chosen for specific reasons that have everything to do with chemistry and consumer demand.
Flavors and Fragrances: Tiny Volumes, Big Impact
Food scientists use methyl isovalerate to simulate or sharpen the flavor profiles of candies, beverages, and dairy products. A little goes a long way. Its aroma appeals to the human nose, making artificial flavors feel much more natural. Research published in the “Journal of Food Science” has pointed out methyl isovalerate’s ability to reinforce familiar fruit notes, building up the taste experience without overwhelming or off-putting aftertastes.
Perfume and personal care producers also lean on this compound. They want fragrances that last but don’t overpower. Methyl isovalerate lends top notes—those subtle hints detected first. When pairing these with other chemicals, the final scent becomes more rounded. It’s a subtle move, adding punch and freshness to lotions, shampoos, and body sprays.
Wider Reach in Industry—Beyond Scent and Taste
Chemists looking for a solid starting point for synthesizing other compounds turn to methyl isovalerate because of its predictable behavior. It reacts smoothly with other chemicals, letting teams create esters, alcohols, and acids that eventually end up in plastics, packaging, and specialty solvents. Its use in lab settings as a flavor standard also helps researchers compare new food additives and fine-tune detection equipment.
Safety Comes First
Handling this compound calls for respect and careful management. Anyone who has worked in chemical manufacturing knows the importance of following established safety protocols. Companies make sure workers get proper training, and, as highlighted in safety datasheets reviewed by the European Chemicals Agency, standard ventilation and protective gear help keep risks low when methyl isovalerate is in play.
Growth and Future Uses
With the steady growth in natural and imitation flavors, the demand for reliable compounds increases. People want flavored goods that taste like the real thing but still fit tight shelf-life and regulatory demands. Methyl isovalerate helps deliver these outcomes, pushing manufacturers to find more uses, such as in paint solvents, specialty lubricants, or new biodegradable materials.
If companies can boost the yield of greener synthesis routes and recycle waste along the way, methyl isovalerate could keep its place in the industry without driving up environmental concerns. Researchers are already hunting for new ways to produce it from bio-based sources, aiming to shrink the environmental footprint.
As technology changes, so do the expectations of both buyers and makers. From the subtle notes in a dessert to a new, safe paint thinner, methyl isovalerate adapts—the kind of versatility industry never stops needing.
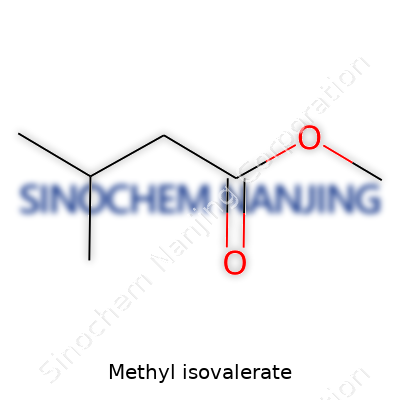
| Names | |
| Preferred IUPAC name | Methyl 3-methylbutanoate |
| Other names |
Methyl 3-methylbutanoate Methyl isopentanoate 3-Methylbutanoic acid methyl ester |
| Pronunciation | /ˈmɛθɪl aɪsəˈveɪləˌreɪt/ |
| Identifiers | |
| CAS Number | 556-24-1 |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:90768 |
| ChEMBL | CHEMBL2039829 |
| ChemSpider | 23719 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.104.232 |
| EC Number | 203-293-7 |
| Gmelin Reference | 2166 |
| KEGG | C09752 |
| MeSH | D008762 |
| PubChem CID | 8122 |
| RTECS number | OJ2450000 |
| UNII | 44W47C4098 |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | `DTXSID4070112` |
| Properties | |
| Chemical formula | C6H12O2 |
| Molar mass | 102.15 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.868 g/mL at 25 °C(lit.) |
| Solubility in water | Insoluble |
| log P | 1.960 |
| Vapor pressure | 0.8 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 25 (alpha hydrogen) |
| Basicity (pKb) | pKb ≈ 24.41 |
| Magnetic susceptibility (χ) | -7.53×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 0.611 mPa·s (20 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 278.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -481.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3183 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 64 °C |
| Autoignition temperature | 451 °C |
| Explosive limits | Explosive limits: 1.1–7.5% |
| Lethal dose or concentration | LD50 oral rat 4,350 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 3,300 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Methyl Isovalerate: Not established. |
| REL (Recommended) | 300 mg/m³ |
| IDLH (Immediate danger) | IDLH: Unknown |
| Related compounds | |
| Related compounds |
Methyl valerate Isovaleric acid Valeric acid Ethyl isovalerate Isobutyric acid Methyl butyrate |

