Methyl Isobutyrate: More Than Just a Solvent
Tracing Back: The Evolution of Methyl Isobutyrate
Many folks believe that laboratory chemistry is a modern phenomenon, fueled by high-tech gadgets and rapid-fire innovation. Methyl isobutyrate’s story suggests otherwise. Chemists explored esters long before the digital age, and by the early 20th century, they singled out methyl isobutyrate for its sweet, fruity note and practical properties. Its early years found it helping flavorists and perfumers expand their palettes. With time, the chemical’s clean evaporation and reliable stability opened more doors. Decades of incremental improvements laid groundwork for today’s safer, greener manufacturing methods. These days, anyone tracing its journey can appreciate how cross-talk between academics, flavor houses, and chemical engineers shaped the industry’s expectations for this compound.
A Look at the Product: Aromatic, Mobile, and Ready to Work
Walk into a lab, open a vial, and methyl isobutyrate greets you with something close to the smell of ripe fruit. It’s a clear, colorless liquid, rarely sticky, running thinner than water. Its ability to dissolve a range of nonpolar compounds puts it to work in coatings, perfumes, and plastics, all thanks to that balance between stability and volatility. A low freezing point means it won’t seize up in a cold storeroom. The boiling point, not far beyond the 90-degree Celsius mark, lets processors reclaim it through distillation when necessary. This isn’t just trivia—these physical traits drive practical decisions out in the field. Chemists and formulators, myself included, rely on them when troubleshooting. If a paint turns cloudy, methyl isobutyrate’s clarity and solvent action can point the way to a solution.
Physical and Chemical Properties Keep Things Practical
Getting a handle on the nuts and bolts—density, refractive index, vapor pressure—is not just an academic exercise. Methyl isobutyrate fits into everyday workflows because its physical constants hit a sweet spot. Its density, lower than water, helps emulsions settle out cleanly. The modest refractive index means it won’t overpower optical measurements in coating applications. Vapor pressure matters for those running reactors or blending flavors: too high, and you waste product to evaporation; too low, and the aroma doesn’t release when needed. Chemical resistance matches the needs of current polymer and solvent systems. From experience, I’ve found methyl isobutyrate remains reliable in the face of mild acids and bases, and blends well without kicking off troublesome side reactions.
Technical Specifications & Labeling: Beyond the Small Print
On a shipping drum, every milliliter means money and safety. Product specs for methyl isobutyrate go beyond purity numbers. Labels list hazards, exposure limits, and any residual moisture that might disrupt a reaction. Flammable warnings aren’t just regulatory box-checking—the flash point means storage near open flames spells danger. Data sheets remind users to check batch numbers for traceability, which keeps quality up and recalls rare. Accurate labeling keeps labs and factories in line with both safety officers and customer demands, something I quickly learned working alongside logistics teams. End-users expect these details to help troubleshoot suppliers and guarantee product consistency.
Making Methyl Isobutyrate: From Reactors to the Loading Dock
Chemists favor straightforward synthesis: react isobutyric acid with methanol using an acid catalyst, and out comes methyl isobutyrate along with water. Simple in theory, real-world production calls for careful control. Catalyst choice can swing batch yields by a wide margin. Processors tweak temperature, pressure, and separation techniques to reduce off-odors and keep unwanted by-products to a minimum. Water removal helps drive the reaction, so drying steps matter. Scaling up from beaker to barrel-sized runs highlights new snags, like corrosion in pipes or solvent recycle rates. These practical details, field-tested by plant operators worldwide, matter more than any textbook promise of “high yields.”
Chemical Reactions & Modifications: Versatility in Action
Methyl isobutyrate may seem simple, but its chemical backbone opens doors for modification. Under controlled hydrogenation, it switches over to alcohols valuable in other industries. Chlorination or hydrolysis steps tweak its performance in different applications. Chemists use these reactions to produce specialty intermediates for agrochemicals and pharmaceuticals. Nothing stays static here: as new catalysts and greener reagents show up in journals and trade shows, plants switch over to cut costs and meet rising environmental standards. Years in research labs and small-batch pilot plants told me that every tiny optimization on reaction pathways translates to real savings and less waste down the line.
Synonyms & Product Names Cut Through Confusion
Call it methyl isobutyrate, methyl 2-methylpropanoate, or even isobutyric acid methyl ester—the product inside the drum stays the same. Brand names hop from one supplier to the next. Synonyms and alternate product codes show up in old research, order sheets, and customs declarations. Tracking all these references is more than an administrative hassle; it prevents costly mix-ups during procurement, regulatory filings, and cross-border trade. Having spent years sorting through stubbornly inconsistent supplier catalogs, I know language matters just as much as chemistry for global business and safe handling.
Safety and Operational Oversight Matters
This ester’s sweet smell can fool the unwary into thinking it poses little risk. Flammable vapors call for explosion-proof hardware and well-maintained ventilation systems. Liquid contact causes irritation, so gloves and splash goggles have a real place on the bench. National safety agencies, from OSHA to the EU’s REACH, regulate safe limits and demand up-to-date training for workers. Spills need quick cleanup; fire departments want clear reporting on site storage volumes. I have watched plant safety drills catch problems that checklists missed, from faulty drum seals to inattentive forklift handling. Good safety programs keep workers healthy, product loss low, and communities informed.
Application Areas: Where Methyl Isobutyrate Shines
Turn over bottles of flavorings, check the contents of certain coatings, or review the process sheets for specialty intermediates, and methyl isobutyrate makes quiet appearances everywhere. Its role in food flavors and fragrances stands out, due to a profile that bridges artificial and natural tastes. Its evaporation properties make it prized in nitrocellulose lacquers for fast-drying finishes and automotive paints. It carries sunscreen actives and fixes scents in high-end perfumes, tasks where both volatility and solvent power count. Other applications branch into pharmaceuticals, agrochemicals, and laboratory reagent supply chains. From my direct experience in contract synthesis, customers focus on methyl isobutyrate for its consistent results and clean regulatory record, not just its chemistry.
Research and Development: The Forward Push Never Stops
Scientists keep pressing for better catalysts, lower waste, and cleaner by-products. Projects in university-industry partnerships aim to fine-tune production for higher yields with less waste water, fitting the needs of modern chemical manufacturing. In specialty areas, R&D teams explore new blends that cut emission from industrial coatings or stretch shelf life in food flavors. Green chemistry efforts look at biodegradable alternatives and safer reaction conditions, hoping to outpace regulatory crackdowns and shifting customer values. Across all this activity, learnings from past industrial accidents and incremental improvements drive daily decisions, more than any top-down mandate ever could.
Toxicity Research Informs Responsible Practices
Health agencies track everything that might end up in consumer products. Decades of toxicological testing show methyl isobutyrate’s acute toxicity remains low compared to more aggressive solvents; inhalation of concentrated vapor can still irritate eyes, lungs, and skin. Chronic exposure at high levels, such as in poorly-ventilated factories, risks headaches and dizziness. Animal studies set boundaries for human safety, while audits by regulators keep production plants transparent. Reviewing the literature, I’ve seen consumer advocates and chemical engineers work together to set lower workplace exposure limits, influencing both how labs operate and how safety information is shared with customers.
Outlook and Future Prospects: Keeping It Relevant
Esters as a class continue seeing renewed interest, and methyl isobutyrate stands to benefit from rising demand for sustainable chemical solutions. As coatings and flavor markets shift toward bio-based ingredients, the industry invests in renewable feedstocks and better recycling for solvents. Regulatory changes nudge manufacturers away from more toxic or persistent alternatives, giving methyl isobutyrate more room as a safer substitute in diverse markets. My contacts in the industry tell me that as external pressures force supply chain transparency and greener manufacturing, this ester’s clean profile and historical track record will help it stay relevant, attract more research dollars, and support new application areas for years to come.
What is Methyl Isobutyrate used for?
Why Methyl Isobutyrate Earns Attention
Take a stroll through any chemical plant, and you’ll spot some unsung workhorses. Methyl isobutyrate, with its sweet, fruity odor, rarely makes headlines. Yet this compound shapes what we touch, smell, and even eat by showing up behind the scenes in industries ranging from paints to flavors. Its effectiveness comes from a straightforward combination: easy evaporation and the ability to dissolve both water-loving and water-fearing substances.
Role in Solvent Systems and Coatings
Ask an industrial chemist about solvents, and the list quickly grows long. What makes methyl isobutyrate stand out is its balance — it evaporates quicker than many bulkier chemicals, yet lingers enough to make sure pigments and resins blend nicely. In my experience developing coatings, the search for smooth application and a uniform finish leads right to compounds like this. Add methyl isobutyrate to a paint mix, and watch the brush glide on clean. It pulls its weight without gumming up the formula, giving both quick drying times and improved surface appearance.
Car refinish shops are one good example. Crews don’t have hours to wait before adding another layer. They need solvents that won’t tangle with other components or fade color. Methyl isobutyrate helps them carry on with efficiency and fewer headaches, thanks to its dependable evaporation profile.
Flavors and Fragrances: More Than Just Chemistry
Step into a laboratory that creates artificial fruit scents, and you’ll find methyl isobutyrate quietly working its charm. It carries crispness in flavor formulas for products like candy, baked goods, and drinks. Its presence is neither overpowering nor shy, which means product designers can enhance the taste or bouquet without drowning out other notes. Over time, this compound has been recognized as safe by food authorities when used within set limits, giving a nod to decades of practical experience.
Perfume crafters use it for apple and pineapple tones. Blenders balance it alongside stronger esters, allowing the final product to feel rounded and lifelike. This sort of subtlety turns what could be a flat smell into something that grabs attention on the shelf.
Potential Hazards and Steps Toward Safer Use
No chemical gets a free pass. Methyl isobutyrate, while handy, brings flammability concerns. I’ve been on teams that drill safety training for storage and use, making sure ventilation keeps vapor from building up. Direct exposure can irritate skin and eyes, and breathing the vapors over time isn’t wise. Factories keep spill kits and fire extinguishers at the ready any time bulk solvents get handled. Using closed systems and fume hoods can knock risks down without adding cost or complexity.
Where We Go From Here
As research into greener solvents grows, attention shifts toward reducing dependence on compounds with volatile organic content. Even so, methyl isobutyrate’s track record speaks for itself. Industry leaders now try to source it through processes that cut down energy and waste. Switching raw materials or exploring biobased routes could make a difference. For now, careful handling — guided by both regulation and hard-learned lessons — keeps industry moving while keeping workers and neighbors safe.
Is Methyl Isobutyrate safe to handle?
Seeing Past the Name
Methyl Isobutyrate comes across as a mouthful, but in real workspaces, it stands out as a basic solvent. I’ve seen it filling shelves in research labs, carried in drums through industrial corridors, and tucked into the back rooms of small manufacturing sites. Its fruity odor might trick someone into thinking it’s harmless. That smell once fooled me, too, but experience showed that safety doesn’t sit in appearances.
The Real Hazards
You can’t spot all dangers by reading a label. A skin splash led to a rash for a colleague. He thought an hour without gloves wouldn’t matter—regret kicked in the next morning with redness and discomfort. Methyl Isobutyrate may evaporate easily, adding flammable vapor to the work area. Sparks and static can light up this vapor, and that threat hovers even in a cool lab corner. The flash point for methyl isobutyrate sits at 14°C (57°F), pretty low. One missed grounding connection, and there’s a real fire risk. That isn’t scaremongering—it’s exactly what fire marshals see after incidents when solvents like this go up.
Invisible Threats in the Air
Breathing in even a small concentration can lead to headaches or irritation. I used to think an open window meant enough ventilation. Real data tells a different story. OSHA has not set a specific exposure limit, but organizations like NIOSH recommend treating it with caution. Use a proper fume hood or at least a robust local exhaust system. Ask any veteran lab tech: most have worked with coworkers who ran into nausea or dizziness from lingering vapors.
PPE: Non-Negotiable
Personal protective equipment isn’t overkill. I always reach for gloves made of nitrile or neoprene, plus chemical splash goggles. Lab coats help, but if any chance of splashing exists, a full apron saves laundry and skin both. Clothes and shoes matter, too. Open sandals have no place around Methyl Isobutyrate. Years ago, I saw someone mop up a spill with bare feet nearby—they sprinted to a sink when the liquid crept toward them.
Practical Storage and Spill Control
Store it in tightly closed containers, far away from heat, sparks, or sunlight. I’ve watched too many well-meaning folks leave bottles near radiators or windowsills. One time, a cracked cap led to slow vapor release, and a whole room needed airing out. Absorbents and spill kits are smart backups. Even tiny leaks add up if nobody pays attention. A weekly walkthrough beats frantic cleanup every time.
Why Proper Training Matters
Real safety comes only with solid information. No one gets everything right the first week on the job, but with guidance, mistakes turn into stories—not injuries. Whenever I train new workers, I start with the safety data sheet. Simple checklists and hands-on demonstrations help folks remember habits that textbooks can’t teach. If you ever wonder why a specific rule exists, speak with someone who’s faced an accident. Their tales stick longer than any safety poster.
Making Decisions with Confidence
There’s no substitute for knowledge and preparation. Respecting chemicals like Methyl Isobutyrate keeps work environments safe and people protected. Good habits—tested and retested—beat luck every time. With care and attention, using this solvent becomes just another part of getting the job done right.
What are the storage requirements for Methyl Isobutyrate?
Safety isn’t Optional
Methyl isobutyrate isn’t one of those chemicals you can just shove in a corner and ignore. Its low flash point, somewhere around 16°C (61°F), means this liquid catches fire fast. Anyone storing it ought to keep it locked away from any spark, flame, or hot surface. It doesn’t matter if you’re in a huge industrial warehouse or the back room of a lab—treat it like it wants to ignite. Keeping it away from heat and grounding the storage containers for static build-up control turns out critical. Too many stories float around about someone carrying a drum across a concrete floor in low humidity, only for a tiny static spark to ruin everyone’s day.
Containers Matter
Standard practice says store it in tightly sealed steel drums or approved plastic containers, but not everything labeled “chemical drum” fits the bill. If that seal leaks, vapor gets out and fills the room with a sweet, fruity smell—but also a concentration that’s anything but harmless. These vapors form explosive mixtures with air, so tight-fitting lids do more than keep things tidy: they keep people alive. Never transfer it to a milk jug or non-rated container. Someone will do it to “save money” or because another drum isn’t handy—and those shortcuts end poorly.
Ventilation Keeps You Breathing
Walking into a chemical storage room and feeling dizzy or getting a headache? Poor ventilation’s probably to blame. Methyl isobutyrate’s vapors need somewhere to go besides your lungs. Open windows make a difference, but good mechanical ventilation proves more reliable, especially in warm weather. Those working in labs know it only takes a small leak to put you on your back if the air’s not moving. If you have to store larger quantities, dedicated ventilated storage—like flammable cabinets with exhaust—gives another layer of safety and peace of mind.
Away from the Wrong Chemicals
Storing methyl isobutyrate beside oxidizers like nitrates or perchlorates turns a fire risk into something much worse. A responsible operator double-checks what’s sitting next to each other on the chemical rack. Even acids and bases, if spilled together with methyl isobutyrate, cause unplanned reactions. Emergency response folks know that once these get mixed, normal fire extinguishers might not cut it. Proper segregation isn’t just a regulatory box to check—it’s what keeps small mistakes from escalating.
Label Everything, Track Everything
“Unknown” bottles turn up in any chemical store room, and nobody wants to guess whether a residue is water or something flammable. Clear, durable labels—with date received and hazard class—make sure the next person won’t pay for someone else’s carelessness. And keeping an inventory log isn’t office bureaucracy—if you know what came in and what went out, you won’t discover leaking, expired drums by accident.
Protect People, Protect Places
I’ve seen complacency take hold in facilities that run for years without incidents. But the smallest slip with methyl isobutyrate causes real harm: chemical burns, inhalation injuries, or large-scale fire. Unloading drums on a loading dock with an idling truck a few feet away? That’s how lessons get learned the hard way. Following basic storage protocols protects workers as much as property. Local fire marshals, insurance providers, and neighbors all expect those in charge to treat flammable liquids with respect. That means not cutting corners, not skipping daily checks, and never assuming a closed drum is completely safe. Safety wins out by actually doing the work, not just posting the rules on a wall.
What is the chemical formula of Methyl Isobutyrate?
The Real Story Behind C6H12O2
People ask about the formula for methyl isobutyrate. That’s C6H12O2, a simple line of letters and numbers. It shows up in textbooks and on product labels, but most only see the science on the surface and miss the everyday ways it drives progress in everything from fragrances to coatings. I remember walking through a factory and catching a whiff of a fruity note in the air, sweet and sharp. It always surprised people that something with a technical label could smell like apples or pears.
Methyl isobutyrate isn’t a shadowy figure in the background, either. It shows up in flavors, perfumes, and even paints. It’s an ester—a family of chemicals making everything from fruit esters to synthetic resins possible. Its boiling point sits just above room temperature, so it evaporates quickly, leaving only scent or chemical signature behind. Labs and industries value that kind of predictability. A chemist once told me, “You only ever notice esters when they’re missing or wrong.” That’s true in food science, too. If you’ve bitten into candy that tasted flat, it probably missed something like this.
Keeping Quality High and Risks Low
Safety and reliability stay at the front of conversations around methyl isobutyrate. Volatile organic compounds, or VOCs, like this one need careful handling. In workplaces where workers handle barrels or drums, ventilation, gloves, and eye protection become second nature. Health and environmental scientists push for tighter limits year after year. In the US, both the Environmental Protection Agency and OSHA have their eyes on it, and for good reason. Breathing too much can cause headaches, dizziness, and can even irritate the throat. As a writer, I’ve interviewed workers who remember times before modern safeguards. They talk about coughing fits now mostly gone because workplaces took chemical management seriously.
Every industry using organic solvents has faced tough choices about safety and waste. I’ve seen manufacturers switch to closed-loop systems and better packaging just to keep emissions down. This move doesn’t just come from regulation, but because lawsuits and accidents cost time and trust. Customers look for companies willing to make safety a priority, not just an afterthought. People remember when things go wrong.
Solutions and Responsible Use Going Forward
If chemistry has a future, it’s built on respect for both power and limitations. Next-gen manufacturing processes rely on innovation to reduce environmental impact. Researchers hunt for bio-based alternatives that can match the performance of petrochemical esters like methyl isobutyrate. It isn’t just about swapping one ingredient for another, but about rethinking how chemicals fit into supply chains.
Green chemistry offers a promise, not a guarantee. Some companies test fermentation-based approaches—yeast and bacteria tweaked to pump out esters efficiently. Others cut back on solvent use or recycle waste through energy recovery. These steps look small up close but add up across global operations.
The formula C6H12O2 represents more than six carbons, twelve hydrogens, and two oxygens. It highlights a challenge in balancing demand for flavor, scent, and materials with worker health and a cleaner world. It’s in the hands of both the experts behind the scenes and the everyday people counting on safer, better products.
How should I dispose of Methyl Isobutyrate?
Why This Chemical Matters
Methyl Isobutyrate isn’t as familiar as bleach or gasoline, yet it shows up in labs, manufacturing, and sometimes even in hobby chemistry circles. It's a clear liquid with a fruity smell, flammable, and can irritate skin or eyes. Tossing this stuff down the drain spells trouble for local water systems and ecosystems. Getting rid of chemicals safely isn’t just ticking a box. It has a real impact — neighbors' health, soil, fish, and even the air we breathe depend on conscious handling.
Direct Experience Drives Safe Choices
Years back, I watched a well-meaning but uninformed neighbor pour excess paint thinner into a storm drain. The stink told me it was more toxic than he realized. I learned then nobody gets a free pass with chemical disposal. Local rules often seem like a headache, but they’re there for good reason. Methyl Isobutyrate belongs to a class of organic solvents. These substances typically don’t break down easily and can end up in groundwater. Even small amounts can damage aquatic life and pollute drinking water sources. If you have leftovers, treat this task the same as handling used motor oil or antifreeze: it shouldn't touch household drains.
Rules and Resources
City or county hazardous waste facilities are built for jobs like this. They know how to process and neutralize solvents without endangering workers or the environment. Look up the next collection event, call them, or check their website for instructions. Most areas accept labeled containers. Never mix leftover chemicals together. Mixing can cause reactions, fires, or create even more toxic byproducts.
If you’re in an academic or industrial setting, the built-in safety officer or environmental manager handles this. Hobbyists and DIYers might find municipal drop-offs a lifesaver, but sometimes private companies also pick up waste for a fee. Cost aside, their responsibility matters, since they follow EPA and state guidelines that go beyond curbside recycling.
Doing It Right at Home or Work
Seal methyl isobutyrate tightly. Label the container if it isn't the original, listing the full name and a "flammable" warning. Stash it far from children, pets, or food. Transport it in a sturdy box or bucket lined with absorbent material so a spill in the car doesn’t turn an errand into an emergency.
In labs, I’ve kept a dedicated flammables bin and practiced regular inventory to avoid surprise leftovers. When possible, using up product fully or giving it to another user cuts waste, but only if safe and allowed by law. Never burn or evaporate chemicals outside; vapor inhalation can harm the lungs and attract unwanted regulation.
Thinking Beyond Disposal
A lot of us don’t realize how easy it is to switch to safer alternatives in some cases. Less toxic solvents or water-based substitutes often do the job. Buying only as much as needed means less left over. In every case, learning about safe disposal and applying it protects more than your own patch of ground. Community well-being, cleaner water, and healthier air start with what each of us does at this basic level. For Methyl Isobutyrate and relatives, respect for safety regulations goes a long way toward keeping both people and wildlife out of harm’s way.
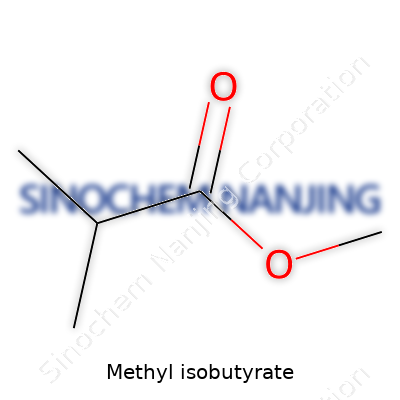
| Names | |
| Preferred IUPAC name | Methyl 2-methylpropanoate |
| Other names |
Isobutyric acid methyl ester Isobutyric acid, methyl ester Methyl 2-methylpropanoate Methyl isobutanoate |
| Pronunciation | /ˌmɛθ.ɪl aɪ.səˈbjuː.tɪ.reɪt/ |
| Identifiers | |
| CAS Number | 547-63-7 |
| 3D model (JSmol) | `CCCC(=O)OC` |
| Beilstein Reference | 1209086 |
| ChEBI | CHEBI:85307 |
| ChEMBL | CHEMBL48661 |
| ChemSpider | 60539 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.111.622 |
| EC Number | 203-693-7 |
| Gmelin Reference | 63172 |
| KEGG | C01333 |
| MeSH | D008758 |
| PubChem CID | 7823 |
| RTECS number | NU8225000 |
| UNII | 7Y9V087OK7 |
| UN number | UN1249 |
| CompTox Dashboard (EPA) | DTXSID1022541 |
| Properties | |
| Chemical formula | C5H10O2 |
| Molar mass | 102.132 g/mol |
| Appearance | Colorless liquid |
| Odor | fruity |
| Density | 0.864 g/mL at 25 °C |
| Solubility in water | Solubility in water: 1.4 g/L (20 °C) |
| log P | 1.21 |
| Vapor pressure | 13.7 mmHg (20 °C) |
| Acidity (pKa) | pKa ≈ 25 (α-hydrogen) |
| Magnetic susceptibility (χ) | -8.81×10⁻⁶ |
| Refractive index (nD) | 1.372 |
| Viscosity | 0.624 mPa·s (25 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 340.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -426.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2411 kJ/mol |
| Pharmacology | |
| ATC code | M01AB |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H336 |
| Precautionary statements | P210, P240, P241, P261, P280, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P403+P235 |
| NFPA 704 (fire diamond) | 1-3-1 |
| Flash point | 55 °F (13 °C) |
| Autoignition temperature | The autoignition temperature of Methyl Isobutyrate is **460°C**. |
| Explosive limits | Explosive limits: 1.3% - 7.6% |
| Lethal dose or concentration | LD50 Oral Rat 7400 mg/kg |
| LD50 (median dose) | LD50 (median dose): 3,400 mg/kg (rat, oral) |
| NIOSH | NIOSH: PM1735000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Methyl Isobutyrate is "250 ppm (950 mg/m3)". |
| REL (Recommended) | 150 ppm |
| IDLH (Immediate danger) | IDLH: 1,600 ppm |
| Related compounds | |
| Related compounds |
Methyl methacrylate Isobutyric acid Isobutanol Methyl acetate Ethyl isobutyrate |

