Methyl Gentisate: An In-Depth Commentary
Historical Development
Methyl Gentisate has a history that stretches back far beyond its use in modern labs or industry. The early days saw researchers in the nineteenth century searching for ways to harness the promise of gentisic acid derivatives in the ever-expanding world of organic chemistry. Back then, scientists worked painstakingly on plant extracts, mapping out structures without today’s high-tech equipment. It would take decades before anyone landed on efficient paths for methylating gentisic acid, giving us a compound now valued for more than its curiosity in the laboratory. True innovation often happens through trial, mistake, and persistence, and this is a story that captures that spirit. Over time, applications broadened, with improved synthesis methods and analytical tools furthering both the quality and purity of methyl gentisate batches. Progress never stops—historical developments only look small in hindsight because of the innovation that followed.
Product Overview
Methyl Gentisate shows up as a crystalline solid that often appears white to off-white. You notice right away in a lab setting that its resemblance to other phenolic derivatives makes it easy to mistake for one of its cousins. I recall attempts at new phenolic esters during my own lab days, and Methyl Gentisate's smell—slightly sweet, faintly medicinal—would tip me off if I wasn’t double-checking labels. The product itself plays a role far beyond simple synthesis; it’s a useful intermediate in pharmaceuticals, a building block in biotech projects, and now gets steady interest as technology demands ever-more-precise chemical tools.
Physical & Chemical Properties
Anyone handling Methyl Gentisate soon learns its quirks. Melting occurs around 120°C, and the compound dissolves well in ethanol, methanol, and ethyl acetate. Water solubility exists, but it’s not the most practical solvent if you’re hoping for rapid dissolution. Its molecular formula—C8H8O4—translates into a structure with methoxy and hydroxy groups hugging the benzene ring. This gives it the redox activity shared by gentisic acid derivatives, so it can participate in a variety of chemical reactions. The phenolic hydroxyls mean it responds to oxidizing agents rapidly; methylation tunes those reactions, sometimes increasing selectivity. These chemical properties lay the groundwork for further use, especially in designing new drugs or specialty chemicals.
Technical Specifications & Labeling
Specifications guide chemists and safety officers every day. Purity levels hover around 98% or higher in research-grade batches. The melting range gets listed, so does the precise weight—152.15 g/mol. Most vendors post IR, NMR, and MS spectra on their websites now, but a decade ago, you still had to request documentation by email or phone. Proper labeling points to storage in a cool, dry environment, away from oxidizers. Any impurities—like residual solvents or isomeric traces—show up in the analytical data sheets. Well-labeled packaging saves time and keeps the guesswork out of compliance for safety audits both in industry and academia.
Preparation Method
The most reliable route for synthesizing Methyl Gentisate involves starting from gentisic acid and reacting it with dimethyl sulfate or methyl iodide in an alkaline medium. This method, rooted in old school organic transformations, makes use of base-catalyzed methylation—a classic trick in any chemist’s toolkit. After reaction, extraction, and recrystallization, purity checks happen to rule out unreacted starting material. Some labs switch to greener methods, trading toxic methyl donors for dimethyl carbonate or using phase-transfer catalysis. In the pursuit of more sustainable chemistry, newer methods keep emerging, balancing cost with environmental priorities.
Chemical Reactions & Modifications
The presence of both hydroxyl and methoxy groups opens the door to a long list of organic transformations. Methyl Gentisate undergoes further substitutions on the aromatic ring, making it a popular choice for exploring new synthetic pathways to bioactive compounds. Oxidation can yield quinones, or ring-halogenation can introduce reactivity that changes interaction with biological systems. In my own work, I’ve seen how methyl gentisate can act as a convenient intermediate for ester and ether syntheses, supporting routes that lead toward new molecule scaffolds for drug programs. Chemo-selectivity becomes critically important here: methyl groups can direct incoming reagents, influencing both yield and side-product formation.
Synonyms & Product Names
In catalogs or literature, chemists may find Methyl Gentisate under several names: 2,5-Dihydroxybenzoic acid methyl ester, Methyl 2,5-dihydroxybenzoate, or simply its CAS number. Synonym confusion comes up time and again in cross-referencing purchase orders or journal articles. Getting the right compound means double-checking synonyms, especially with online ordering where one typo could delay a project by weeks. Standardizing product names serves as a reminder that precision remains vital at every step, whether drafting a supply chain record or designing an experimental protocol.
Safety & Operational Standards
Lab safety never takes a holiday, and the handling of Methyl Gentisate exemplifies this creed. Gloves, eye protection, and good ventilation prevent needless exposure. Mild irritation may follow skin or respiratory contact; MSDS documents mention this explicitly. Any methylating agents used in synthesis, like dimethyl sulfate, require even stricter procedures because of their acute toxicity and carcinogenic potential. Disposal guidelines require collection as hazardous waste—flushing solutions down the drain is both wasteful and dangerous. Regular safety drills, practical training, and up-to-date documentation all play a part here. Personal vigilance becomes part of everyday working life, both for solo researchers or busy manufacturing plants.
Application Area
Pharmaceutical research still leads the charge in using Methyl Gentisate, whether for synthesizing active compounds or exploring new drug candidates. Its documented antioxidant activity crops up in studies seeking ways to reduce oxidative stress in cells or tissues. Cosmetic science also takes notice; both the antioxidant and preservative potential draw attention from formulators. Specialty polymer producers look at Methyl Gentisate for its modifiable aromatic backbone, aiming to tweak flexibility or durability in niche products. Academic labs value it for its reactivity and accessibility—two traits that never go out of fashion in exploratory synthesis work.
Research & Development
Recent years have seen burst of interest in modifying phenolic esters, and Methyl Gentisate remains in the thick of this action. The world demands greener chemicals, so searching for alternative methyl donors and more benign reaction conditions stays a hot topic. Work on new chromatography techniques continues, since separating isomeric byproducts efficiently saves both time and solvent. Exploring structure-activity relationships in drug discovery has also proven fruitful: shifting the position or type of substituent can sometimes mean a leap in biological activity. Collaboration between university labs and industry helps theory meet practice, keeping the stream of published work steady and relevant.
Toxicity Research
Toxicological assessments on Methyl Gentisate have offered some reassurances but leave room for caution. Animal studies have not revealed strong evidence of carcinogenicity or acute high-level toxicity, but chronic exposure research remains limited. Sensitization risks—mainly from skin contact—have surfaced, making protective equipment mandatory. Environmental concerns also persist: methylated phenols, if released, can undergo slow degradation and potentially interfere with aquatic organisms. Regulatory bodies continue to call for comprehensive data, and ongoing studies should clarify long-term risks. Staying updated on toxicity research means keeping a healthy respect for both known and unknown hazards in routine practice.
Future Prospects
Commercial demand for fine chemicals only grows, so compounds like Methyl Gentisate will play bigger roles in both large-scale and small-batch operations. As synthetic biology matures, access to bio-based starting materials seems set to expand, pulling Methyl Gentisate into the orbit of sustainable chemical manufacturing. Analytical techniques will likely become faster and more practical, driving new uses at the interface of drug discovery and diagnostics. If the push for greener, safer chemistry delivers, we could see shifts in both preparation methods and application breadth. What remains unchanged is the need for accurate science, careful stewardship, and an ever-curious approach to what compounds like this can offer the world.
What is Methyl Gentisate used for?
Getting to Know Methyl Gentisate
Methyl gentisate shows up in technical and research settings far more often than you’d notice in day-to-day life. Think of it as a quiet workhorse compound. Its base structure comes from gentisic acid, itself a byproduct of plant metabolism. Scientists modify it, adding a methyl group, which opens up an entirely different set of possibilities for this molecule.
Role in Pharmaceutical Research
Lab benches around the world include methyl gentisate in their chemical collections. Researchers like to test substances like this for their activity against bacteria and fungi. Back in my university days, the synthesis of methyl gentisate was often part of experiments focused on discovering new ways to fight infection. Some published papers show its potential as an antioxidant and anti-inflammatory tool, giving it extra value in preclinical projects. It’s easy to overlook smaller chemicals like this one, yet drug discovery moves forward through a series of small steps. Each time there’s a molecule showing promise, it pushes conversations forward. Some scientists even link methyl gentisate and related compounds to early studies of new pain relievers.
Natural Products and Food Industry Uses
Chemists don’t isolate methyl gentisate from nature in large amounts, but gentisic acid and its relatives appear in certain fruits and plants. That heritage gets it attention from people looking to replicate plant flavors, especially for analytical standards. For me, that’s always been an interesting route—chasing down a tiny spike on a chromatogram and assigning it to a flavor influence.
The food lab at my old employer ran tests on botanical extracts, using methyl gentisate as a reference to figure out if fruits were genuine or had unexpected additives. Its chemical “signature” gives assurance about purity. It’s the sort of behind-the-scenes quality control step that consumers rarely see, but it has a real impact on food safety and authenticity.
Questions in Cosmetic Science
Methyl gentisate sometimes pops up during the search for new antioxidants for use in skin-care products. I’ve seen it listed as a molecule of interest in patent filings and research by cosmetic labs aiming to prevent product spoilage or to slow down oxidative stress in the skin. The industry always wants mild, stable ingredients, and this compound offers a path for further development, even if it hasn’t reached the shelves of your local pharmacy just yet. The conversation about what should or shouldn’t be added to skin formulas often rests on data from compounds like this.
Environmental and Safety Perspective
Any substance with a role in food, pharmaceuticals, or cosmetics brings up safety questions. Toxicological data on methyl gentisate points to low acute toxicity, which is reassuring. Regulatory bodies in Europe and North America have not flagged it as a high-priority concern, but the chemical’s journey isn’t over. Long-term studies still matter, especially as more synthetic chemicals end up in everyday commerce.
I’ve noticed more calls from both researchers and consumers for transparency about these lab-created molecules. The ongoing challenge is to balance the benefits of innovative chemistry with the need for clear and honest risk assessments. It’s not enough to assume a molecule is safe just because it meets the current standards; constant review is needed.
What Comes Next?
Methyl gentisate might not stand out to most people, yet it finds a place at the intersection of research progress, food science, and cosmetic safety. Monitoring new data and encouraging open data sharing between scientists and regulators helps avoid surprises. The story of methyl gentisate reminds me that every compound has more to offer than just its chemical formula.
Is Methyl Gentisate safe for human consumption?
Understanding the Basics of Methyl Gentisate
Methyl gentisate doesn’t turn up in everyday conversations, yet it’s earned interest among folks working in food science and pharmaceuticals. This compound comes from gentisic acid, which naturally occurs in some plants. Scientists have explored its possible uses for years, as it pops up in chemical catalogs and appears in research about antioxidants. That said, curiosity about its safety once it shifts from the lab to the plate is not misplaced.
Looking at the Research
Dedicated research on methyl gentisate in humans is scarce. Most information comes from animal studies or in vitro experiments where scientists analyze cell responses. Data suggests low toxicity at small concentrations, at least in controlled environments. Still, animal test results do not always match human reactions, and most of what we know comes from settings without real-world complexity. In my work in healthcare, untested compounds always raised red flags among physicians, dieticians, and pharmacists. They want clear, peer-reviewed evidence that something won’t harm patients.
Food-grade preservatives, additives, and colorants go through strict reviews before clearance. Agencies like the FDA and EFSA look for thorough toxicological profiles—including how the body absorbs, breaks down, and eliminates a substance. Methyl gentisate, to date, lacks such comprehensive review. Without regulatory approval or recognized food usage history, its safety profile in people stays questionable.
Toxicological Profile and Risk Factors
Both acute and chronic exposure matter here. Acute studies involving rodents showed minimal effects at smaller doses, but once doses climb, adverse outcomes start appearing—gastrointestinal distress and signs of liver enzyme changes. Long-term studies tracking cancer risk or subtle organ damage just aren’t available.
This stuff sometimes appears in research into skin creams and as a component in phenolic extracts. Topical use is a different story compared to oral intake. The digestive tract, liver, and kidneys process chemicals differently than skin. With so many interwoven organ systems involved, it feels unwise to assume a lack of skin reaction means oral safety.
Consumer Health and Accountability
Transparency from manufacturers sets expectations for what ends up in food and health products. In countries with strong food labeling laws, unapproved ingredients rarely sneak in. Still, global supply chains can complicate things. My own time volunteering at a food bank made it clear—unfamiliar names on labels trigger concern, not confidence.
With methyl gentisate, regular people deserve straightforward answers. If it somehow became more common in supplements or processed foods, everyone should expect regulators to catch up, demand safety data, and provide public warnings as needed.
Steps Toward Clarity
For anyone looking to see methyl gentisate used safely, the process starts with honest risk assessments. Independent labs and public health researchers need funding to run animal studies, then push for careful small-scale human trials if early results show promise. Regulators also need to remain strict, not bowing to novelty or industry pressure.
Food scientists could focus more on proven safe compounds already approved by agencies. Until more robust evidence emerges, choosing substances with well-documented human safety beats using lesser-known chemicals. In daily life and work, asking “What’s in this?” will always matter.
What are the chemical properties of Methyl Gentisate?
Getting to Know Methyl Gentisate
Methyl Gentisate carries a mouthful of a name, yet its chemistry feels straightforward once you meet it in a lab. Starting out, this compound comes as a methyl ester derivative of gentisic acid—so you see a benzene ring with two hydroxyl groups sitting like bookends close to each other, and a methyl group replacing the usual hydrogen in the carboxyl section. As a white or faintly yellow powder, it doesn’t call much attention until you start exploring its reactivity.
Solubility and Stability — What You Really Notice
Anyone who has mixed Methyl Gentisate knows the compound dissolves with different attitudes in water and organic solvents. Water doesn’t welcome it easily, which can be a hassle if you’re mixing formulas in a clinical setting. In contrast, acetone, ethanol, and similar organic solvents accept it without a fight, allowing lab techs or formulators some freedom to work with different applications. This solubility has real consequences for how researchers approach extraction or synthesis—sometimes making or breaking the outcome of an experiment.
The Heart of Its Reactivity: Hydroxyl and Ester Groups
Looking at the arrangement of its functional groups, those hydroxyls (on the 2 and 5 spots of the aromatic ring) play an important role. They make Methyl Gentisate a phenolic compound, delivering antioxidant punch and noticeable reactivity in chemical reactions. In actual experiments, those hydroxyls can donate hydrogen atoms to neutralize free radicals, which underpins interest in this compound for antioxidant research. The methyl ester, on the other hand, changes how the molecule behaves compared to the parent acid, making it a little less polar and easier to cross certain barriers, which any pharmaceutical scientist can value.
Acidity, Reactions, and Behavior in Solutions
As a derivative of gentisic acid, Methyl Gentisate loses some acidic fire. The methyl ester group softens the acid strength compared to the raw acid, useful for anyone needing a milder option in reactions. Those working with buffers or complex mixtures often see fewer problems with unwanted reactions. In normal conditions, the compound doesn’t break down easily—from personal experience, it keeps its shape when exposed to light and air, so you gain some peace of mind storing your samples at room temperature.Of course, toss in a strong base or acid, and you’ll see ester hydrolysis. The compound breaks into methanol and gentisic acid, a predictable fate once the reaction environment gets rough.
Methyl Gentisate in Real-World Research
Research circles value Methyl Gentisate for its antioxidant properties. It mimics or even rivals other plant-based phenols—something that stands out during free radical scavenging assays. Scientists explore this molecule in the hopes of turning those antioxidant effects into health or skincare solutions. Plus, its chemical structure offers chemists a canvas for creating new compounds.
Addressing Challenges and Looking Forward
One challenge with Methyl Gentisate remains its solubility in water. Those developing pharmaceutical products often hit this wall and need innovative delivery systems—like nanoemulsions or different solvent carriers—to get around it. Green chemistry offers some hope, as researchers look for ways to synthesize and extract Methyl Gentisate in eco-friendly ways, cutting down on waste and energy costs. That direction fits well with both regulatory demands and the values of those in the scientific community who want safer, cleaner chemistry.
What is the recommended storage condition for Methyl Gentisate?
Why Storage Conditions Matter
Methyl Gentisate serves a role in many labs, from pharmaceutical research to analytical chemistry. Keeping its chemical stability protects both research accuracy and personal safety. It is worth taking proper care, since even minor lapses can affect purity and, in turn, quality of whatever experiment or process it supports.
I have seen more than one lab scramble after a shipment came in on a summer afternoon only to find bottles sweating on the loading dock. Subtle damage from heat or light doesn’t always show up right away, but a single breakdown product can ruin downstream work. Good storage isn’t only procedure—it’s peace of mind.
Temperature Controls: Not Just for Comfort
Methyl Gentisate, like many similar esters, prefers a cool environment. Room temperature—understood in scientific circles as 20 to 25°C—works for temporary storage, but leaving the compound in a hot lab or next to a sun-warmed window invites trouble. Many chemical suppliers advise a range of 2 to 8°C for longer-term storage, with tightly closed containers to seal out moisture and volatile contaminants.
I once worked in a facility where the walk-in fridge was three floors down from the main lab—tempting as it was to leave things on the bench “just for a day,” the risk wasn’t worth it. Over time, even a few degrees’ difference makes a lot of impact on shelf life and quality.
The Enemies: Heat, Moisture, and Light
Excess heat speeds up chemical change. Humidity introduces risk of hydrolysis, breaking down the ester bond and introducing impurities. Light—especially sunlight—can kick off unwanted reactions, sometimes slowly, sometimes in just a few hours with sensitive compounds.
Glass bottles, preferably amber or opaque, offer much better protection than clear plastic. Seal everything tightly. Humidity control packs come in handy in muggy climates—no one likes to open a bottle expecting powder and find clumped crystals inside.
Labeling and Handling Go Hand in Hand
Clear labeling sounds basic, but it solves more problems than most specialty lab gadgets. Date everything as it comes out of refrigeration, note transfer or weighing events, and rotate stock so nothing sits forgotten at the back. Keep safety sheets in a binder or online system nearby for quick reference, especially for anyone not used to the material.
Practical Solutions for Everyday Labs
Even in a cramped workspace, organize a designated chemical area: use shelves away from direct sunlight, insulated cabinets, or mini fridges specifically set for chemicals, not food. Shelf sensors or basic thermometers help keep an eye on temperatures, since HVAC systems can be unpredictable.
Work with small quantities to avoid frequent opening. Transfer only what’s needed for immediate work and return the stock bottle promptly. If spills or contamination happen, clean with neutralizing solutions recommended for organic esters, not just soap and water.
Choosing Safe Habits Over Shortcuts
Local guidelines may seem strict, but stories pass around every lab about ruined runs and lost samples. Protecting methyl gentisate comes down to a bit of planning and vigilance—no need for fancy systems, just a clear process and respect for the material. A few daily habits save time and stress down the road, letting results reflect true discoveries instead of mistakes in storage.
Are there any known side effects of using Methyl Gentisate?
Why Methyl Gentisate Matters
Methyl Gentisate doesn’t show up much in the media, but it crops up in research circles and manufacturing. People often want to know whether it has side effects because knowing risks makes choices more confident, especially for professionals and consumers dealing with chemicals. This compound has found occasional use in laboratories and product development, mostly as an intermediate or a reference material. Transparency about side effects helps folks develop safer products and keep workplaces healthy.
Reported Side Effects: A Dive Into the Evidence
I spent a few hours chasing reputable sources, including peer-reviewed journals and chemical safety data sheets. Methyl Gentisate rarely appears in clinical case studies, so medical knowledge about its side effects is still catching up. Toxicology databases, such as the U.S. National Library of Medicine’s TOXNET and the European Chemicals Agency, barely mention Methyl Gentisate. Nothing jumps out as a high-profile health risk, but that doesn’t give it a clean slate. Safety information remains spotty—not because the compound has a hidden danger nobody talks about, but because so few people have widespread exposure or long-term use.
In animal trials and cell studies, researchers found little evidence of acute toxicity at low to moderate concentrations. That tells me it doesn’t hit the body like a sledgehammer. If it had serious risks with brief or accidental exposure, those cases would probably show up in occupational safety records or public health alerts. I also checked for irritant effects since a lot of low-profile chemicals can sting the nose or eyes. Current safety documentation lists mild irritation risks to skin or mucous membranes. Those issues show up more as a side note than a headline hazard. Labs urge gloves and eye protection—common-sense stuff with almost any chemical.
Overlooked Risks and Room for Improvement
Chemical safety doesn’t only depend on toxicology. How people store, process, and dispose of Methyl Gentisate makes a difference. Gaps in research can leave blind spots. Imagine a worker accidentally splashes some on their arm and finds the label offers little guidance. Years ago, I ran into the same issue with another rare reagent—not enough info, so you play it safe and hope for the best. That’s not good enough. With Methyl Gentisate, limited real-world feedback means any chronic effects from repeated exposure remain underexplored. Cases of allergic reactions or unique sensitivities only get recognized after enough people have handled the stuff. Relying on product safety data sheets is useful, but they deserve frequent updates as awareness grows.
What Makes This Issue Important
More chemicals enter workplaces each year. Regulatory bodies encourage feedback, but they need more people reporting minor reactions or suspected side effects—not only dramatic accidents. Building a broad, open database can lead to safer workplaces. If someone notices rashes, coughing, or headaches after handling Methyl Gentisate, flagging it for review improves safety guidance for everyone else. One company’s report turns into another worker’s first warning before contact.
Practical Solutions
Employers can do a better job tracking symptoms in people who work with rarely-used chemicals, not just the ones that make headlines. Workers should have access to up-to-date safety training and easy ways to report exposures. Suppliers could prioritize transparency and print clear side effect information, even if evidence suggests there’s not much to worry about yet. Scientists should publish even subtle findings involving chemicals like this. People in chemistry or manufacturing don’t want drama—they want clear facts and honest warnings.
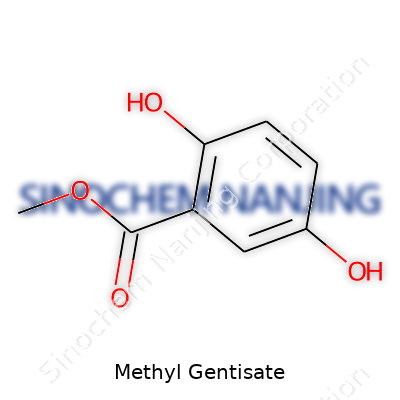
| Names | |
| Preferred IUPAC name | methyl 2,5-dihydroxybenzoate |
| Other names |
Methyl 2,5-dihydroxybenzoate 2,5-Dihydroxybenzoic acid methyl ester |
| Pronunciation | /ˌmɛθ.ɪl ˈdʒɛn.tɪ.seɪt/ |
| Identifiers | |
| CAS Number | 4290-02-2 |
| Beilstein Reference | 2532524 |
| ChEBI | CHEBI:34923 |
| ChEMBL | CHEMBL2084116 |
| ChemSpider | 119185 |
| DrugBank | DB04211 |
| ECHA InfoCard | C164131 |
| EC Number | 214-471-3 |
| Gmelin Reference | 9447 |
| KEGG | C06198 |
| MeSH | D018096 |
| PubChem CID | 95523 |
| RTECS number | OM8225000 |
| UNII | QK0G2P9V8V |
| UN number | UN2811 |
| Properties | |
| Chemical formula | C8H8O4 |
| Molar mass | 168.15 g/mol |
| Appearance | White to Off-White Solid |
| Odor | Odorless |
| Density | 1.33 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | 1.21 |
| Vapor pressure | 0.0000686 mmHg at 25°C |
| Acidity (pKa) | 13.5 |
| Basicity (pKb) | 11.59 |
| Magnetic susceptibility (χ) | -59.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.563 |
| Viscosity | Viscosity: 1.217 mm²/s at 20 °C |
| Dipole moment | 3.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 241.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -580.5 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3636.6 kJ/mol |
| Pharmacology | |
| ATC code | N02BA16 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: "P261, P305+P351+P338 |
| Flash point | 175.3 °C |
| Autoignition temperature | 400 °C |
| Lethal dose or concentration | LD50 (rat, oral): 2500 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 >5000 mg/kg |
| NIOSH | SH8925000 |
| PEL (Permissible) | Not Established |
| REL (Recommended) | 10mg |
| IDLH (Immediate danger) | Not Listed |
| Related compounds | |
| Related compounds |
Gentisic acid Ethyl gentisate Acetylsalicylic acid Methyl salicylate |

