Methyl Fluoroacetate: Lessons from a Potent Molecule
Looking Back: A History of Caution and Curiosity
The story of methyl fluoroacetate winds back to the early chapters of organic chemistry, a time when chemists pushed boundaries to uncover new compounds and reactions. Early studies flagged its relationship with sodium fluoroacetate, a notorious plant-derived toxin. Folks in the lab soon realized this wasn’t just another chemical to tinker with—its structure, a simple methyl group tagged with a fluoroacetyl backbone, married a sense of curiosity with the need for strict caution. I remember reading about fluoroacetates’ impact on ecosystems. Ranchers and researchers alike have watched grazing animals fall victim to the natural precursor in certain Australian plants. For decades, toxicologists have traced these perils and tracked accidental exposures, urging safety while exploring its far-reaching chemical behavior.
Product Overview: Understanding the Basics
Methyl fluoroacetate stands as an example of how a small change in a molecule flips its nature entirely. Just a single fluorine atom swaps the benign into something actively toxic. The compound is a clear, low-viscosity liquid under standard room conditions. The faint, ester-like odor might lull a careless observer, yet that subtlety hides its true punch. On paper, its chemical formula, CH3O2CCF, looks simple enough. In practice, it’s a lot more layered.
Physical and Chemical Properties: Nothing Ordinary Here
With a boiling point hovering just above room temperature, methyl fluoroacetate walks the line between volatility and handling risk. Water solubility is decent, so spills do not simply settle and vanish—they mix, drift, and can persist in a working space if not managed with care. Even at low concentrations, the molecule retains its breath-stealing toxicity, seeping into the air if left unchecked. This highlights the importance of proper fume extraction. Chemically, it acts with the expected reactivity of an ester. Despite this, the fear factor comes not from its reactivity, but its tragic efficiency in disrupting biological processes.
Technical Specifications and Labeling: More than a Name
In any storeroom or lab, labeling this compound demands clarity and urgency. The term “danger” isn’t hyperbole. Regulations call for stark hazard communication—skull and crossbones, strict signal words, and detailed pictograms—reflecting decades of hard-learned lessons. Handling instructions draw a line in the sand for personal protection: gloves resistant to organic solvents, goggles, and optionally, face shields for splash-prone procedures. Technical data sheets, though usually written in dry bureaucracy, serve as a vital map, updating researchers on both the health risks and the regulatory hearsay that comes with such a potent substance.
Preparation: Chemistry’s Double-Edged Sword
Most reference syntheses utilize methanol and fluoroacetyl chloride—a combination that delivers a clean conversion, but at substantial risk. Working in a professional setting, people know you don’t lean over the flask or pipette by hand. With just a hint of inattention, one error ruins more than just an experiment. There’s no getting around the facts: decades of process safety protocols have evolved from incidents during these very synthesis steps. Today, remote-handling tools, full enclosure hoods, and double-checked waste protocols have become the norm, all because of compounds like this.
Chemical Modifications and Reactions: Tread Lightly
Chemists appreciate how small tweaks in esters and related compounds shape reactivity. Methyl fluoroacetate, though, isn’t eager to play along with classic schoolroom reactions. It can move through hydrolysis fairly quickly, breaking down to the sodium salt in biological settings, which spells trouble for living systems. Attempts to mask or modify the fluorine rarely offset the underlying hazards. In fact, historical efforts to “deactivate” its toxicity for utility in synthesis almost always resulted in lost time or a worse problem down the line.
Changing Names, Shared Danger
Across the chemical world, methyl fluoroacetate pops up under a handful of synonyms, from methyl monofluoroacetate to the more clinical-sounding methyl ester of fluoroacetic acid. The jargon varies, but every label points to a single truth: take care. Even experienced research teams swap stories about near-misses, mixing up vials or missing a synonym in a safety document. It pays to be vigilant with naming conventions to avoid accidental exposure or confusion.
Operational Standards and Safety: Small Steps, Big Impact
Health and safety authorities do not tip-toe around this one. The best labs follow a zero-tolerance approach to exposure, with detection badges, spill kits, and robust incident reporting. Safety showers and eyewash stations crowd the shared workspaces. I recall a safety seminar where a seasoned chemist explained that your last line of defense comes down to simple habits: never opening a bottle outside a certified fume hood, and triple-checking every hand-off between team members. This culture of preparation saves lives, plain and simple.
Where It Makes a Difference
Field applications for methyl fluoroacetate was always limited by its toxicity. In a pinch, it saw use as a predatory mammal control agent, but backlash from the environmental sector grew as the unintended consequences showed up in field studies. Food chain persistence, impact on non-target species, and groundwater issues all heated up debates, and regulatory action clamped down. Today, only a handful of tightly controlled research sites and animal control programs try to balance its power against ecological fallout, usually erring on the side of tighter restrictions and new safeguards.
Driving Research and Innovation
Research into fluoroacetates has opened windows into enzyme inhibition and the cross-talk between simple molecules and living systems. Biochemists use methyl fluoroacetate in controlled cell studies, probing how it crashes the energy cycle through aconitase inhibition. This molecular violence delivers insight on everything from pesticide risks to antidote design. In classrooms, stories of methyl fluoroacetate’s dangers reinforce the gap between concept and practice. I’ve watched new grad students flinch as case studies lay out what can really go wrong if protocol slips.
Toxicity: The Ongoing Red Flag
The biggest black mark in methyl fluoroacetate’s dossier is its toxicity. By slipping quietly past the body’s lines of defense, it becomes the silent saboteur, shutting down cellular respiration and turning energy enzymes against their host. High-profile research teams have published work outlining the lowest observed adverse effect levels in animal testing. Results prove just how little exposure it takes to trigger disaster. Antidote options remain few; treatment leans on supportive care, and despite new research, the risks persist. Training remains the only sure bet, hammering home that one bad move means a trip to the hospital, or worse.
The Path Ahead: Tightening Gaps, Unlocking Value
Future prospects for methyl fluoroacetate will likely circle around two focal points: innovative medical research and broader environmental regulation. Efforts in drug discovery keep nudging boundaries, with some research teams testing modified esters as targeted cancer inhibitors, but always in tiniest doses and with backup plans. Regulatory shifts push the bar ever higher for storage, shipping, and destruction. There’s no way back to the days of casual handling. As regulatory frameworks grow sharper and detection technologies evolve, safe handling procedures set the gold standard, and the push for greener alternatives stays alive. My personal view: the toughest molecules show us our limits, and force us to grow. If we focus on training, vigilance, and relentless curiosity, we stand the best chance at safely unlocking real value, without repeating the missteps of the past.
What is Methyl Fluoroacetate used for?
What Exactly Is Methyl Fluoroacetate?
Methyl fluoroacetate shows up in various research labs but rarely gets any headlines. This chemical, made from fluoroacetic acid, has a simple structure and packs intense toxicity. That dangerous reputation shadows nearly every conversation scientists have about it.
Uses in the Lab and Industry
Researchers use methyl fluoroacetate while looking for ways to understand how poisons affect living things. It often acts as a tool in toxicology to study how organisms react to something that interrupts basic cellular processes. The real reason it holds a spot in laboratories comes from its remarkable ability to shut down the Krebs cycle—that core metabolic engine inside cells. This means even at the smallest dose, it can stop cells from making energy, a trait that’s hard to find outside compounds like this.
Field researchers sometimes rely on its derivatives for pest control studies, though its use faces tight restrictions. Australia, for instance, watched its cousin compound (1080) get public attention due to use in invasive species control. But governments don’t hand out permissions lightly; even small leaks pose risks for dogs, birds, and farmers themselves.
Danger to Humans and the Limits on Use
This chemical’s high toxicity makes it too dangerous for regular agricultural or household use. The body turns methyl fluoroacetate into the more familiar fluoroacetate, and just a sprinkle triggers severe reactions—nausea, heart problems, seizures. Even trained handlers must take strict precautions.
For this reason, regulatory agencies across most countries keep close tabs on who handles it and how. The risk of accidental poisoning or environmental contamination outweighs any quick benefits for most people outside science or regulated pest control.
Investigating New Solutions
Strict controls mean researchers push for safer alternatives. More targeted rodenticides appear, ones that go after pests but don’t pose the same wide-ranging hazard. Some universities trial genetic changes in crops or natural predators instead, hoping to cut reliance on blanket chemical solutions.
The science world also keeps its eyes open for treatments. In Australia, efforts run to find effective antidotes for livestock or wildlife after accidental exposure. Those efforts encourage open dialogue between universities, public agencies, and the people tending land where risks matter most.
Why Transparency Matters
Past accidents taught everyone that secrecy helps no one when it comes to toxic chemicals. Farmers, animal health workers, and community members want clear information. Open records, tight labeling, and honest warnings stay at the top of priorities, especially with chemicals that could kill in minutes.
The lessons around methyl fluoroacetate reflect the wider debate about what role such chemicals should play today. People deserve to know what enters the food supply, water, or nearby wildlife areas. Investment in safety, oversight, and long-term alternatives seem like the most reasonable steps, based on what the evidence tells us.
Building on What Works
No one in their right mind chooses risk without thinking. Methyl fluoroacetate’s history serves as a reminder: short-term solutions built on dangerous chemicals often set up bigger problems years later. As more communities focus on health, safety, and ecology, the push for smarter, less hazardous approaches carries real weight.
Is Methyl Fluoroacetate hazardous or toxic?
Understanding the Substance
Methyl fluoroacetate doesn’t show up in daily news feeds unless a serious incident occurs, but this chemical deserves a closer look because of its extreme hazards. In the chemistry lab, handling unknowns goes with the job, yet even veteran researchers grow cautious around anything with the “fluoroacetate” tag. A key fact: methyl fluoroacetate belongs to a family of chemicals related to the infamous compound found in the plant gifblaar and in pesticide formulations like 1080 (sodium fluoroacetate). Just a trace can act with frightening speed.
Toxicity—No Room for Error
A colleague once described working with it as “handling a pit viper with chopsticks”—the margin for a mistake shrinks to nothing. Animal studies and forensic cases both confirm that even tiny doses create chaos in the body. Methyl fluoroacetate, once ingested, converts into a metabolite that jams the citric acid cycle, which pushes cells into energy failure. That doesn’t just sound bad on paper: in practice, the heart, brain, and muscles all shut down fast.
No ordinary detox method fixes exposure. No standard antidote exists. The reported human lethal dose drops below 10 mg per kg. Historical lab accidents in Australia and in Eastern Europe led to death within days. Symptoms creep up: nausea, twitching, confusion, seizures. For livestock and wildlife, this same mechanism wipes out entire populations exposed through contaminated food.
Environmental Concerns
The issues extend beyond acute human poisoning. Once in the environment, methyl fluoroacetate works like a hidden snare. Birds, small mammals, pets—none show obvious warning signs before it’s too late. In New Zealand, using related chemicals to control pests set off cycles of controversy. Carcasses of poisoned animals put scavengers and raptors at risk, endangering already threatened species.
Ignoring methyl fluoroacetate’s risk profile doesn’t make it go away. The chemical persists unless specialists neutralize it. Some reports claim it breaks down in water, but soil and cooler climates appear more stubborn. Wildlife managers regularly debate safer alternatives, knowing that just a minor spill threatens food chains for months or years.
Oversight and Practical Solutions
Lax handling would mean putting lives in danger. Only trained chemists, backed by full protective gear and rigorous protocols, dare measure or transfer methyl fluoroacetate. Storage includes lock-and-key cabinets, double-sealed containers, and regular inventory checks. Disposal almost always demands high-temperature incineration under government oversight, which comes from painful experience: the aftermath of accidental exposures lives on for decades.
Moving away from such high-risk toxins falls within reach. Australia and New Zealand now regulate and restrict use, thanks to public outcry and evidence-based campaigns. Synthetic alternatives continue to emerge, lowering the odds of runaway poisoning events and protecting ecosystems.
I learned cautious respect for chemicals like methyl fluoroacetate through safety briefings and old research papers. Reading between the lines, there’s a clear message—ignore the dangers, and the consequences turn tragic fast. Only a tight community of experts should ever interact with compounds whose risks stack up this high, especially where antidotes remain out of reach.
How should Methyl Fluoroacetate be stored?
The Real Hazards Behind Methyl Fluoroacetate
The chemical world holds some real heavy hitters when it comes to risk, and methyl fluoroacetate easily falls into that category. I’ve seen up close how just a small mistake in handling these kinds of toxic materials can change lives or careers in a flash. Direct exposure isn’t just inconvenient—it’s often deadly. The margin for error sits razor-thin. Folks who manage these compounds don’t have to guess or hope; they need rock-solid systems at their back. It’s not only about personal safety. Methyl fluoroacetate slips past the skin or gets inhaled, and then the story turns tragic. Coughing, sweating, heart failure: the symptoms escalate before you know it. I’ve run drills where one small oversight nearly led to disaster. That’s all the reason anybody needs to get this right every time.
Why Ordinary Storage Fails
Too many times, corners get cut around storage. You run out of lab-grade space and think, “It’ll be fine for a day or two.” Suddenly that storeroom becomes a bigger problem than any complex science you manage. Plain cabinets or basic shelves provide no help during an emergency. Ordinary containers crack, corrode, or leak fumes, and you find yourself wishing you weren’t on cleanup duty. I saw a bottle left under a sink because someone thought a locked cupboard sufficed. The fumes almost knocked out the cleaning crew the next morning. This stuff isn’t bleach or acetone—this is a chemical assassin.
The Setup that Actually Works
I’ve adopted habits from chemists who have survived decades in the lab. That means getting specific, not casual. Methyl fluoroacetate sits best in a tough, sealed glass bottle with a screw cap—no exceptions. High-density polyethylene resists the chemical, so no glass shortage justifies swapping in soft plastics or anything translucent. Containers face clear labels, not scribbles or codes that only one technician remembers. Every bottle deserves a hazard symbol and emergency instructions taped within sight. Anything less lays a trap for someone down the road.
Placement inside a lab tells its own story. My old supervisor insisted on using explosion-proof refrigerators for temperature control since room temperature only increases the risk of volatile fumes. You lock that fridge every time, and you track the key. It’s tempting to place everything on an accessible shelf, but these poisons find their spot low and in secondary containment trays. Spills drip into those trays and not onto your hands, shoes, or worse. For a few dollars, those plastic bins saved hours of panic after a shelf collapse one lab over.
Beyond the Storage Room
I have lost count of the times routine checks saved our skin. Schedule inventory days—don’t just hope everyone knows what sits where. Buddy checks keep everyone accountable. If you see any crust, cloudiness, or rusty cap, log it, and get professional disposal help before the material escapes its bottle. It’s tempting to make your own call, but government agencies write safety standards for a reason. Reach out to poison control lines or certified waste handlers whenever in doubt.
Every worker from student to senior researcher deserves proper protective gear. Gloves, goggles, and a tight-fitting mask create a second line of defense, not a guarantee. Fume hoods—well ventilated, never cluttered—provide the breathing room needed to work safely. I remember that feeling of security standing behind a good barrier, knowing the air pulled those invisible threats away before they could do any harm.
Building a Safer Culture
Sensible safety isn’t just a checklist. It’s a mindset, grown from respect for chemicals and concern for everyone who walks through the door. Managers show it by funding lockable cabinets, reinforced bottles, and walkthroughs that catch overlooked risks. Colleagues protect one another with honest communication about hazards and errors. Nothing replaces experience, but nobody should have to learn these lessons through tragedy. Leadership means owning mistakes and demanding better, not cheaper.
Every hazard label, every double-check, every minute spent reviewing proper storage buys a little more peace of mind. Knowledge charts the path forward. After you’ve moved a few bottles and patched up small leaks, you realize dignity at work doesn’t just come from paychecks or titles. It comes from knowing everyone goes home as healthy as they arrived.
What are the safety precautions when handling Methyl Fluoroacetate?
Danger in a Bottle: What Methyl Fluoroacetate Really Means
Methyl fluoroacetate may not ring bells unless someone handles chemicals for a living. Still, this compound earned infamy for good reason. Fluoroacetate blocks a key step in cell metabolism, making it easy to cause accidental or deadly poisoning. There’s little room for mistakes. I remember walking into a storage room once, catching a strong chemical whiff, and feeling a chill—sometimes just the wrong breath or fumble with these toxins puts you in real trouble.
No Shortcut: Personal Protection Means Everything
Most labs train staff to suit up properly, but people sometimes cut corners out of comfort. With methyl fluoroacetate, that’s a gamble nobody can afford. Gloves that resist permeation, thick lab coats, full splash goggles, plus face shields at the bench—these set the baseline. Specialized gloves, such as those made with laminated film materials, stand up better to serious toxins, and switching gloves between tasks takes a few seconds that could avoid hours in the emergency room.
Breathing in the dust or vapors can cripple a person just as quickly as skin contact. For powder handling, I’ve seen colleagues work inside fume hoods so strong the air whistles past. Respiratory protection, like properly fitted masks with acid gas cartridges, reads more like a warning than a guideline in these scenarios. Don’t trust a regular dust mask; only equipment intended for real chemical defense offers safety.
Planning for the Worst Before Touching a Drop
Every spill plan should start with knowing the cure—knowing exactly which decontamination solution counters the spill. With methyl fluoroacetate, things escalate fast, so labs keep calcium gluconate gel close by and trained staff ready to use it. Informing every team member of the symptoms—lightheadedness, nausea, irregular heartbeat—turns coworkers into early warning systems.
No one wants to play hero during cleanup, so spill kits built for this chemical stay on hand. Absorbent pads, calcium-based neutralizers, sealed bags for disposal—these make up the real shield. Containers always get labeled in bold letters, not stickers that fade or peel after a week. Sharps disposal boxes and chemical-safe waste containers help avoid cross-contamination, which often sneaks up in labs, especially when people get too comfortable or in a rush to leave.
Training and Systems Back People Up
Even with protective gear and emergency gear lined up, routine training makes the difference. Reviewing safety data sheets, running drills, and knowing the poison control number by heart turn best intentions into habits that save lives. Stories circulate about researchers who skipped just one training session, then found themselves facing a medical nightmare no textbook could fix.
Using a buddy system helps too, which means never working alone—at least one other pair of eyes in the room provides real-time backup. Having routine checks on storage containers and ventilation systems only takes a few minutes and helps catch cracks or leaks before a crisis starts.
Raising the Bar on Chemical Culture
Handling methyl fluoroacetate pulls no punches. Every layer of safety exists for a reason: lives depend on attention to detail. Whether it’s double-checking the lock on the storage fridge or insisting on that uncomfortable face shield, cutting corners vanishes when the dangers are spelled out clearly. The right culture in one lab often becomes the model others follow. If someone cares enough to insist on full safety steps, it’s never wasted effort—it’s all about going home healthy at the end of the day.
What is the chemical structure of Methyl Fluoroacetate?
The Structure in Plain Terms
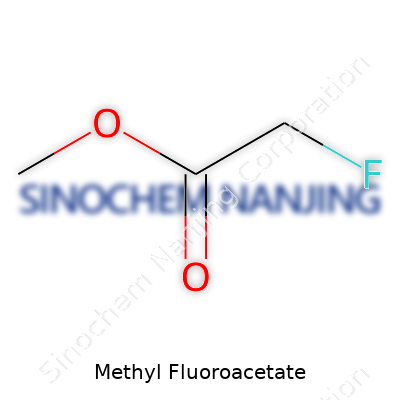
Methyl fluoroacetate stands out among organic chemicals, and not because the name rolls off the tongue. Its structure is simple but packs a punch. The molecule consists of a fluoroacetate group joined to a methyl group through an ester bond. For anyone following along with a mental picture: there’s a two-carbon backbone, with one carbon holding a fluorine atom and an oxygen. The methyl piece attaches through that oxygen, creating the ester linkage. Chemically, you see it written as CH2FCOOCH3.
Imagine the basic skeleton of acetic acid, swap out one of the hydrogens for a fluorine, then replace the acidic hydrogen with a CH3 group—there you have it. This small change, inserting a fluorine atom where you’d usually expect a hydrogen, makes a world of difference in the way the molecule behaves.
Why Structure Matters
I remember sitting in an undergraduate organic chemistry lab, marveling at how swapping a single atom could flip a substance from mild to dangerously toxic. That’s exactly what happens in methyl fluoroacetate. The fluorine atom makes it react with the enzymes inside many organisms in a way that plain old methyl acetate never will. The structure defines this action.
In nature, structural tweaks like this mean survival advantages for plants and animals using these compounds as a defense. In the laboratory or out in the environment, it spells caution—and, sometimes, tragedy.
Real-World Impact and Toxicity
Methyl fluoroacetate caught the attention of chemists and toxicologists for good reason. Its fame—and notoriety—comes from being far more toxic than many simple organic esters. Why? Because the body breaks it down to fluoroacetate, which shuts down a critical process in energy metabolism. Toxicologists learned to respect this molecule, and farmers in Australia learned some hard lessons about the dangers of so-called “1080” poisons (which are based on sodium fluoroacetate, a close chemical cousin).
The chemical’s structure drives this unique danger. A single fluorine changes enzyme activity so dramatically that even low doses become life-threatening for animals that lack natural resistance. Sheep, cattle, and pets have all ended up in vet clinics because of unwanted exposure. In practice, understanding this structure is about more than just memorizing formulas—it means knowing which chemicals can be safely handled, and which can’t.
Need for Awareness and Safe Practices
This molecule keeps showing up in scientific papers, environmental studies, and safety seminars because people keep stumbling onto its risks the hard way. I once had a conversation with a researcher who confessed that mishandling a bottle of methyl fluoroacetate left him hospitalized for days. The lesson here: chemical structure can seem like academic trivia, yet real-world safety depends on grasping these details.
Handling chemicals like methyl fluoroacetate doesn’t belong only to those with postgraduate degrees. Training, clear labeling, and up-to-date material safety data sheets make the difference between a safe day at work and an emergency. The stakes are high, and the responsibility falls on everyone from lab techs to warehouse workers. No one gets a pass when the margin for error is this slim.
Solutions for Preventing Accidents
Clear education goes a long way. In my experience, chemistry students and professionals need real-life case studies highlighting the effect of minor changes in a molecule’s structure. Planting this awareness early sets workers up to spot dangers before they’re close. Regular safety drills, modern labeling systems, and easy access to protective gear help people remember the less-obvious hazards.
Methyl fluoroacetate’s story boils down to a lesson in humility. The smallest shifts at the atomic level can upend biological systems, posing risks that last well outside the lab. Real science isn’t just about formulas and theories—it’s about recognizing hazards and building habits that protect people, livestock, and communities.
| Names | |
| Preferred IUPAC name | Methyl 2-fluoroacetate |
| Other names |
Fluoroacetic acid methyl ester Methyl fluoroethanoate Methyl monofluoroacetate |
| Pronunciation | /ˈmɛθɪl flʊəroʊˈæsɪˌteɪt/ |
| Identifiers | |
| CAS Number | Its CAS Number is '453-18-9'. |
| 3D model (JSmol) | `COC(=O)CF` |
| Beilstein Reference | 1131751 |
| ChEBI | CHEBI:42088 |
| ChEMBL | CHEMBL3322031 |
| ChemSpider | 167396 |
| DrugBank | DB11370 |
| ECHA InfoCard | 100.007.882 |
| EC Number | 205-296-0 |
| Gmelin Reference | 8289 |
| KEGG | C19136 |
| MeSH | D008772 |
| PubChem CID | 65858 |
| RTECS number | WN9625000 |
| UNII | 95XP70B2D8 |
| UN number | UN2015 |
| CompTox Dashboard (EPA) | DJ9NUN4W2O |
| Properties | |
| Chemical formula | C3H5FO2 |
| Molar mass | 106.07 g/mol |
| Appearance | Colorless liquid |
| Odor | pleasant, fruity |
| Density | D=1.263 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.3 |
| Vapor pressure | 14 mmHg (20 °C) |
| Acidity (pKa) | 21.44 |
| Magnetic susceptibility (χ) | -32.5e-6 cm³/mol |
| Refractive index (nD) | 1.3400 |
| Viscosity | 0.455 cP (25°C) |
| Dipole moment | 2.16 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -686.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -825 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | Methyl Fluoroacetate does not have an ATC code. |
| Hazards | |
| Main hazards | Highly toxic by ingestion, inhalation, or skin absorption; may be fatal; causes central nervous system and heart effects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301: Toxic if swallowed. H311: Toxic in contact with skin. H331: Toxic if inhaled. |
| Precautionary statements | P210, P260, P262, P264, P270, P271, P280, P301+P310, P302+P350, P304+P340, P308+P311, P314, P330, P363, P405, P501 |
| NFPA 704 (fire diamond) | 3-3-2 with W |
| Flash point | 110 °F (43 °C) - closed cup |
| Autoignition temperature | 260 °C (500 °F) |
| Lethal dose or concentration | LD50 oral rat 10 mg/kg |
| LD50 (median dose) | LD50 (median dose): 10 mg/kg (oral, rat) |
| NIOSH | PB1238000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Methyl Fluoroacetate: No OSHA PEL established |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | 0.025 ppm |
| Related compounds | |
| Related compounds |
Fluoroacetic acid Methyl chloroacetate Ethyl fluoroacetate Sodium fluoroacetate Chloroacetic acid |

