Methyl 3-Isopropylphenylcarbamate: A Close Look at History, Properties, and Impact
History: The Road from Discovery to Market
Walk through the last seventy years of organic chemistry, and carbamates show up everywhere—pharmaceuticals, pesticides, industrial intermediates. Methyl 3-Isopropylphenylcarbamate doesn’t sit apart from these trends. Researchers first chased down its synthesis to answer demands for safer pesticides and less toxic intermediates in the 1960s, when awareness of environmental and human health risks forced a shift away from old-school, highly persistent organochlorines. The structure made sense: by using a methyl group and an isopropylphenyl backbone, chemists could tailor reactivity, solubility, and, crucially, biological action. Scientists learned plenty from failed analogs: balancing the hydrophobic backbone, tweaking the methyl substitution, and discovering which arrangements kept potency without nasty side effects. Early synthesis papers tell stories of endless glassware, distillation rigs, and the patience required for column chromatography, all set to the hopeful pace of industrial expansion and agricultural change.
Product Overview: Practical Realities of a Modern Compound
Methyl 3-Isopropylphenylcarbamate ends up as a pale yellow to almost colorless crystalline powder. I've seen it stored in high-density polyethylene bottles, with careful labeling that stresses sensitivity to moisture and light. The compound often arrives in batches ranging from ten grams to several kilograms, straight from contract manufacturers or dedicated fine chemical companies. Standard packaging doesn't just follow regulatory norms—it responds to practical experience: double-bagging prevents clumping, and a silica gel packet keeps it dry. Labs using this carbamate take pride in the batch-to-batch consistency, which allows tight dosing and predictable outcomes in testing assays. High purity—often 98 percent or greater—turns up as both a selling point and a necessity; in my own experience, the smallest impurity can skew test results or cause unwanted coloration in final products.
Physical and Chemical Properties
Beneath a microscope, Methyl 3-Isopropylphenylcarbamate reveals itself as needle-like crystals, with a melting point set around 54-58°C. The substance dissolves best in organic solvents such as ethanol, acetone, and ethyl acetate, but holds up poorly in water, reflecting its aromatic and carbamate backbone. Vapor pressure stays low at room temperature, an advantage for storage and reduced inhalation risk. Chemically, the compound carries a near-neutral pH in ethanol-water solutions, and shows only moderate reactivity with mild acids or bases under ambient conditions. The N-H group of the carbamate stands out as a handle for further modification, and the isopropyl group offers some steric hindrance that influences both chemical stability and biological target engagement. Analytical labs track degradation with HPLC, often relying on a UV detector set to the phenyl ring's absorbance peak, ensuring end-users spot breakdown products before they use contaminated material.
Technical Specifications & Labeling Standards
Labels tell stories beyond a name and a CAS number. They list batch number, synthesis date, and percent purity. Safety pictograms, GHS statements, and handling guidelines crowd every bottle I've handled. Technicians value clarity—labels need to mention not just the hazardous components but specific storage guidelines: cool, dry, out of direct light. Some pharmaceutical partners demand Certificates of Analysis, tracing each raw ingredient and solvent lot, and requiring confirmation from both TLC and proton NMR. These aren’t just for compliance; they build trust, especially as the chemical finds its way into trial batches for crop protection or experimental phases of drug screening. A missing storage note on the label once triggered a waste of half a batch in my previous lab—temperature swings broke down the carbamate ring, leaving behind chalky residues no one dared to use.
Preparation Methods
Synthetic routes for Methyl 3-Isopropylphenylcarbamate generally start from 3-isopropylaniline. Chemists treat this precursor with methyl chloroformate and a suitable base, like triethylamine, using a dry solvent such as dichloromethane. Reaction conditions demand careful temperature control, usually between 0-10°C, to tamp down side reactions and color formation. Work-up stages typically involve extraction with brine, drying over anhydrous sodium sulfate, and slow rotary evaporation to prevent heat-induced decomposition. Instead of relying on complicated purification, most labs use flash chromatography, with silica gel columns and a mixture of hexane and ethyl acetate. The process shines when scale-up happens: remote monitoring, batch tracking, and real-time GC/MS spot-checks all cut down on wasted effort and product loss. Some specialty suppliers tweak the procedure—for example, switching out methyl chloroformate for safer, less volatile methyl carbonate sources—to improve workplace safety and lessen environmental impact.
Chemical Reactions & Modifications
In synthetic chemistry, Methyl 3-Isopropylphenylcarbamate serves as both a tool and a scaffold. Its aromatic ring undergoes classic electrophilic substitutions—like nitration or halogenation—giving rise to a variety of downstream intermediates. The carbamate group reacts under mild acid or base, yielding free amines, especially valuable in multi-step syntheses where timing and selectivity count. Some research teams use this molecule to protect amines in heterocycle construction, later recovering the underlying amine via standard deprotection steps. Cross-coupling reactions, notably Suzuki or Heck types, also benefit from the stable yet reactive core of this carbamate. Old synthetic manuals describe methods for converting it into sulfonated or fluorinated analogs, aiming for new pharmacological profiles or fine-tuned electronic properties.
Synonyms & Product Names
Chemical catalogs list Methyl 3-Isopropylphenylcarbamate under names like Carbamic acid, methyl ester, 3-(1-methylethyl)phenyl ester, and the more systematized N-Methylcarbamate of 3-isopropylaniline. Supplier listings may abbreviate to MIPC, especially in communication-heavy fields like pesticide formulation. Synonym confusion has landed more than a few orders in the wrong stockroom or on the wrong HPLC run, so I always double-check CAS registrations. Translators in global firms even prepare lists of trade names to compare regional differences, cutting down on costly errors.
Safety & Operational Standards
Industry standards for handling carbamates run strict, based on evidence from occupational health studies and regulatory actions in the past three decades. Gloves, goggles, and fume hoods come standard for handling even small quantities. Safety Data Sheets for Methyl 3-Isopropylphenylcarbamate list skin, eye, and respiratory tract irritation as risks. Emergency procedures demand speedy rinsing and immediate medical follow-up, not only from a compliance standpoint but because I've watched a spill lead to lasting dermatitis on an ungloved chemist in the next lab over. Disposal means designated hazardous waste, and European REACH regulations require tracking every shipment, with some batches crossing borders under strict controls. Training isn’t a formality—labs incorporate direct demonstrations, fit-checks for respirators, and ongoing conversations about accident response.
Application Area: Agriculture, Industry, and Beyond
Farmers and pest control companies turn to carbamates for selective action and speed of breakdown in soil, hoping to sidestep the legacy issues of older persistent chemicals. Methyl 3-Isopropylphenylcarbamate fits in as a pre-emergent herbicide in some regions, targeting weed seeds and young shoots without lasting environmental burden. Fine chemical firms use the compound’s dual reactivity for synthesizing urethane foams, specialty coatings, and pharma intermediates—anywhere robust linkers and fine-tuned kinetics give an edge. Research labs reach for it when exploring new CNS agents or enzyme inhibitors, since carbamate-based structures often act as reversible acetylcholinesterase blockers, giving insight on therapies for neurodegenerative diseases or pesticide poisoning antidotes.
Research & Development Efforts
Pharmaceutical research groups continue to probe the structure-activity relationships of carbamates. Teams modify the isopropyl or phenyl region, hunting for candidates with higher selectivity for certain enzymes, or altered metabolic stability. Agrochemical firms tinker with adjuvants and formulation partners, aiming to increase rainfastness or reduce drift. Collaborative alliances test combinations with biopesticides or genetically modified crops, hoping for cleaner results and less input overall. Analytical chemists experiment with derivatization—attaching isotopic tags to Methyl 3-Isopropylphenylcarbamate molecules for fate and transport studies. Regulatory scientists run chronic exposure tests, gather field data, and document residues in crops, moving policymaking out of the realm of theory and into the field.
Toxicity Research: Human Health and the Environment
Researchers track toxicity using in vitro assays, rodent models, and long-term ecological surveys. Acute toxicity numbers for Methyl 3-Isopropylphenylcarbamate fall well below those of legacy organophosphates, but outcomes vary by exposure route. Oral LD50 values hover in the several hundred mg/kg range for rats—high enough to allow careful handling, but not so high to eliminate oversight. Field biologists test runoff and persistence in soil and water, searching for sublethal effects on beneficial insects, aquatic larvae, and small mammals. In one university study I followed, repeated low-level exposure reduced pollen foraging in bees, suggesting that even short-lived carbamates have subtle but real ecological footprints. Medical researchers look hard at cholinesterase recovery times in exposed workers, concluding that recovery happens within days once exposure ends, but warning of potential vulnerabilities in children, elders, or those with preexisting neurological disease.
Future Prospects: Safer Systems, Cleaner Chemistry
Regulators and industry voices push for carbamates with enhanced selectivity, lower non-target impact, and greener synthesis. Companies bank on new catalysts, solvent swaps, and process intensification to cut waste and energy consumption. Some startups develop biodegradable versions or blend botanically sourced modifiers, blending tradition with modern safety practices. Drug research branches employ in-silico screening to identify analogs with better pharmacokinetics and fewer off-target effects. Field-scale deployment relies on real-world data, not just lab-bench hopes. My own hunch—few compounds disappear entirely, but instead carve out niche applications, thanks to smart stewardship and continued transparency between researchers, users, and regulators.
What is Methyl 3-Isopropylphenylcarbamate used for?
Understanding Where Methyl 3-Isopropylphenylcarbamate Shows Up
Methyl 3-Isopropylphenylcarbamate sounds like a mouthful, but it's better known by those who deal with agriculture and pest control. Many of us might not spot it at the grocery store, but its role links to the food we eat, the gardens we plant, and the pests we try to control. This compound’s main use lies in its function as an active ingredient in herbicides, particularly for crops like rice and sugarcane.
How It Works in the Field
This chemical steps into action as a pre-emergent herbicide. That means it stops weeds before they break through the soil surface. When farmers treat their fields with this compound, they protect their rice and sugarcane seedlings from weeds that would compete for sunlight and nutrients. Studies have shown that controlling weeds early can help boost crop yield by preventing losses that usually go unnoticed until harvest time.
Many chemical herbicides rely on a similar approach: target the problem before it becomes visible. Weeds often germinate right alongside crops, and a chemical like Methyl 3-Isopropylphenylcarbamate helps tip the balance in favor of the food plants. This efficiency lets farmers rely less on repeated manual weeding, which can be costly and time-consuming on large plots. In places where labor is expensive or scarce, a single application can keep a field productive and weed-free for weeks.
Concerns and Safety
No discussion about agricultural chemicals feels complete without touching on safety and environmental impact. Decades of observation taught me that farmers want solutions that work with the least risk. Strict regulations now govern how and where this herbicide gets used, from maximum residue limits in food to guidelines on timing and application rates. Studies by regulators and independent scientists suggest that, used by the label, the compound poses minimal danger to humans and non-target animals.
Problems can arise if applicators ignore instructions, but in my experience, most growers understand what’s at stake. The real worry surfaces with resistance — those weeds that manage to survive and set seed despite chemical treatment. Overreliance on any one herbicide, including Methyl 3-Isopropylphenylcarbamate, can set up a situation where pests outsmart the chemical, pushing farmers to use more or switch to something stronger.
Exploring Better Ways to Use It
I’ve seen clever approaches in places where farmers rotate their herbicides, mix them, or pair them with non-chemical methods like crop rotation or mechanical weeding. Combining strategies makes it harder for weeds to adapt and reduces the need for repeated chemical use. Collaboration between researchers, farmers, and regulators helps keep these practices moving forward, all while protecting the soil and water around us.
Modern agriculture depends on innovation. Chemicals like Methyl 3-Isopropylphenylcarbamate, when used with skill and care, help feed millions. But the bigger goal remains: safeguarding people and the land while providing enough food. We get better at this by learning from every season and every field, and by trusting those who balance yields against real-world safety and sustainability.
What are the safety precautions when handling Methyl 3-Isopropylphenylcarbamate?
Understanding the Risks
Most people who work around industrial chemicals know that every bottle and bag comes with a story. Methyl 3-Isopropylphenylcarbamate brings its own list of hazards to the table. You inhale its dust or let it touch your skin, and things can get tricky. This chemical can irritate your lungs, eyes, or skin, and I’ve seen more than one experienced operator get caught off guard just because they thought routine tasks didn’t demand extra protection.
The biggest mistake is treating this substance like common table salt or baking soda. Even after handling thousands of pounds of less reactive compounds, anyone working with this carbamate needs to pause and focus, as its potential to cause harm depends on proper habits, not just the label on the drum.
Personal Protective Equipment Isn’t Optional
Handling Methyl 3-Isopropylphenylcarbamate calls for putting all the PPE to work. Nitrile gloves keep hands safe from spills, while splash goggles protect your eyes. A laboratory coat or full apron shields your clothing and skin. Reaching for a dust mask or, even better, a properly fitted half-face respirator helps keep fine particles out of your lungs. Once, during a hot summer job, a technician I knew skipped the coat and regretted it—chemical irritation set in after sleeves rolled up, and that discomfort lingered long past the clean-up.
Consistent habits save time and suffering. I make a point of watching coworkers for missed steps—sometimes fatigue tempts us to cut corners, and a gentle reminder goes a long way.
Workplace Controls Matter
Trying to handle powdered or granular chemicals in a cramped or poorly ventilated room adds a whole layer of risk. In one facility, a new hood vent made all the difference. Without it, the dust would hang in the air after each pour; with it, lenses stayed clear, and breathing felt easier. Checking for proper airflow before starting a job never feels like wasted time.
Never eat or drink anywhere near chemicals. Keeping food and personal items out of production areas not only keeps the workplace safe but also keeps lab mates from unexpected stomachaches or worse.
Emergency Response: Plans in Place
Spills happen, and having a plan makes the difference between a smooth recovery and a disaster. Absorbent materials, chemical-specific spill kits, and knowing where the eyewash station sits lets anyone react fast. I remember an afternoon when a container cracked open, and because the right supplies were close, the cleanup ran smoothly. Without them, exposure could have been far worse.
Everyone on site should feel comfortable asking about protocols or alerting others if they have safety concerns. Open communication builds a team that trusts each other in every situation.
Training: The Real Foundation
Relying on past experience isn’t enough. Regular training keeps everyone sharp. Refreshers on new data sheets, demonstrations on removing contaminated gloves, and hands-on spill exercises build confidence. I’ve watched rookies and pros benefit from going through the steps together, making it less likely anyone misses critical details down the line.
Treating every batch and bottle of Methyl 3-Isopropylphenylcarbamate with respect means going beyond just reading safety sheets—real safety comes from teamwork, attention, and action.
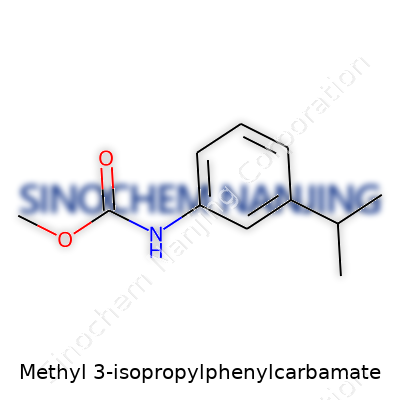
What is the chemical structure and formula of Methyl 3-Isopropylphenylcarbamate?
Digging Into the Structure
Methyl 3-isopropylphenylcarbamate brings together a few recognizable chemical building blocks. Picture a benzene ring at the center — that familiar six-carbon ring with a circle in the middle, almost like the workhorse in many medicines and agricultural chemicals. At the third carbon spot, there’s an isopropyl group sticking out. That’s just a short branch — two methyl groups bonded to a central carbon — giving the molecule a touch of bulk and shape.
The other significant end features a carbamate group. Here, a carbamate is essentially the combination of a carbonyl group (C=O) linked to both a nitrogen and an oxygen. Most folks in chemistry will spot a carbamate and think of ureas and certain insecticides. On this molecule, the carbamate’s nitrogen connects directly to the benzene ring, while the oxygen comes linked with a methyl group. Methylting anything in organic chemistry sticks a single carbon and three hydrogens onto an atom, nudging the molecule in both structure and solubility.
Chemical Formula and Details
The formula for Methyl 3-isopropylphenylcarbamate is C11H15NO2. If you want to lay it out:
- Eleven carbon atoms
- Fifteen hydrogen atoms
- One nitrogen atom
- Two oxygen atoms
The structure houses three main features: the benzene ring, the isopropyl side chain, and the methylcarbamate moiety.
Why Small Chemical Tweaks Matter
Tweaking a molecule by dropping on an isopropyl group or inserting a methyl group chances its whole personality. A few years in a university lab taught me that even a single tiny chemical tail such as isopropyl flips a compound’s behavior inside living things. Isopropyl groups often bump up fat solubility, helping molecules slip into cell membranes. That means a chemical like this has a better shot at getting inside bugs or weeds, which is probably why some carbamate relatives show up in pest control.
The methyl group, meanwhile, plays a role in steering how the molecule reacts. Methylation sometimes blocks a molecule from breaking down too fast inside water-based fluids. That changes storage life and affects how long a compound lingers in soil or inside a plant.
Importance in Real-World Uses
Structures like this get a lot of attention in areas such as crop protection or public health because of how they interact with enzymes. Carbamates often block acetylcholinesterase, an enzyme critical for nerve function in insects and humans alike. Designing these compounds always boils down to finding that razor-thin line where a chemical zaps only the bugs and not the crops — or people.
Some countries keep a watchful eye on how chemicals like methyl 3-isopropylphenylcarbamate are used, especially as their effects on the environment gain more attention. Stories from rural communities remind me that overspraying, or not respecting safe waiting times, risks river water and the critters downstream.
Safer Paths Forward
Continued research into molecular tweaks holds the key to safer chemicals. Swapping a single group — isopropyl for something less bioactive, for instance — can lower toxicity to mammals while still knocking back unwanted insects. Rigorous independent testing, transparent labeling, and clear communication with those applying these substances go a long way toward keeping food and water clean. Data-sharing among scientists and regulators helps spot patterns before they become problems.
Chemicals like methyl 3-isopropylphenylcarbamate play a role in society’s push to protect crops and fight nuisance insects, but every tweak to their structure carries consequences worth close attention.
How should Methyl 3-Isopropylphenylcarbamate be stored?
Handling Chemicals With Care
Working in research labs and industrial spaces taught me that every chemical compound wants you to put your guard down. Methyl 3-Isopropylphenylcarbamate sits among those substances that look ordinary on paper but call for real street-smarts if you plan to store them for the long haul. Accidents happen in storage rooms, not just in experiments, and a little attention here can keep disasters in check.
Why Storage Details Count
Nobody wants fire, spills, or toxic fumes in their workspace. If you ever watched the news after a chemical leak, you know that safe storage is far more than a box-ticking exercise. Keeping chemicals stable isn’t about wasting money on extra gear—it's about protecting health and the community.
Lessons From the Safety Data Sheet
The company's safety data sheet says this compound needs cool and dry conditions. It feels routine, but stories from experienced lab techs keep popping up in my mind—improper storage led to damaged labels, ruined stock, and once, a corroded shelf. Moisture loves to find its way in, especially during humid months, slowly wrecking packaging and risking a reaction no one wants to see. Heat is another silent agent, stoking decomposition or releasing fumes. So, picking a storage location away from steam or sunlight saves more than just product value; it keeps everyone breathing easier.
Keep It Tight, Keep It Right
Methyl 3-Isopropylphenylcarbamate goes in tightly sealed containers, away from food, direct sun, and heat sources. Time spent wiping down containers, checking lids, and dating the arrival of each batch always pays off. Steel cabinets resist corrosion, and plastic bins falter in the face of solvents, so choosing compatible materials counts.
Isolation sounds strict, but keeping this chemical away from oxidizers, acids, and alkalis often avoids chain reactions. I learned this after seeing an innocent-looking storage shelf become a clean-up project, all because incompatible bottles crowded each other. This chemical’s label won’t warn you about its neighbors, but cross-contamination can lead to big problems.
Clear Labeling and Record Keeping
Labels fade and peel if ignored. I spent hours relabeling misplaced stock during inspections. A bold, legible warning and a date let anyone know what’s inside and how old it is. Records serve more than audits; they guide staff in rotating stock, disposing of expired material, and quickly tracing issues if something turns up missing.
Ventilation and Emergency Measures
Rooms storing even small quantities of chemicals need good airflow. I’ve seen the difference a proper exhaust system brings—no headaches, no weird smells lingering long after you close the door. Spill kits and protective gear nearby cut response time if something tips, giving staff firm ground in a crisis.
Training and Oversight
Most mishaps happen on shifts with newer staff or tired hands. Ongoing training sticks better than a single session, especially for recognizing decaying packaging or spotting leaky lids. A culture where workers feel encouraged to report near-misses builds more trust than locked doors.
Supporting Safe Science and Industry
Safe storage always begins at the loading dock and ends with careful disposal. Nobody should learn these lessons the hard way. Storing Methyl 3-Isopropylphenylcarbamate in a cool, dry place, in the right container, far from reactive chemicals, with commitment to labels and safety checks—these details protect everyone under the same roof.
What are the possible health effects of exposure to Methyl 3-Isopropylphenylcarbamate?
Everyday Safety in Chemical Environments
Most people won’t recognize the name, but chemicals like Methyl 3-Isopropylphenylcarbamate show up in industrial sites and agricultural fields more often than most realize. Day-to-day, workers come into close contact with this compound, especially in manufacturing or farming jobs. The reality is, without clear communication about its potential risks, people put themselves at unnecessary risk—mainly because the long names often hide real-world dangers.
Health Effects: Immediate and Long-Term
Direct exposure can bring on symptoms that workers know all too well: skin irritation, red eyes, and difficulty breathing. Reports from the workplace say employees exposed to dust or vapors sometimes struggle with coughing or wheezing by the end of a shift. Fact sheets from safety authorities highlight that higher levels of exposure make the outdoor job much harder, especially under summer heat or limited ventilation. In smaller operations where folks know each other’s names, people sometimes brush off these problems, chalking them up to “just part of the job.” But a few rashes or headaches can add up to lasting trouble.
Studies linking longer-term exposure to carbamate chemicals—including this one—show evidence for nervous system effects. Workers in pesticide production share stories about tingling in their fingers or fatigue that doesn’t let up at home. Regulatory agencies in the U.S. and Europe agree that chronic contact increases the risk for neurological symptoms, which match what’s often seen in rural clinics or among long-time plant staff. The worry isn’t just for the people who apply it—warehouse and maintenance teams, and even cleaning crews, deal with the aftermath.
Leaning on Experience and Reliable Data
Anyone who has worked in a dusty plant knows the temptation to skip a pair of gloves or a mask, especially when there’s pressure to finish the job fast. Years ago, an older co-worker told me, “That stuff just dries out your hands. Wash up and you’ll be fine.” As evidence grew, I saw folks pay the price with hospital visits or lost days on the line. Mixing real workplace knowledge with research helps people see how today’s headaches might echo as tomorrow’s health problems.
Carbamate pesticides like this one have a well-documented history of causing health issues if inhaled or absorbed through the skin. Peer-reviewed studies, such as those in journals like Environmental Health Perspectives, lay out risks in plain terms: repeated exposure wears on the body, with nervous system and liver effects among the most serious.
Finding a Safer Path Forward
Information and training offer better protection than hope. Companies with low incident rates make safety a routine, not an afterthought. They use respirators, gloves, and good ventilation. Speaking up when something feels wrong—or when someone gets a rash that doesn’t go away—brings real problems into focus before they spiral. Medical checkups help catch symptoms early, which matters most for those who depend on their body for a living.
Better labeling, plain-language warnings, and open reporting all strengthen trust on the shop floor and in the fields. No chemical—even one with a long technical name—deserves a blank check on safety. Looking after yourself and your co-workers isn’t just about rules; it’s about the kind of work life everyone deserves.
| Names | |
| Preferred IUPAC name | Methyl N-(3-propan-2-ylphenyl)carbamate |
| Other names |
Carbamic acid, (3-isopropylphenyl) methyl ester Methyl (3-isopropylphenyl)carbamate |
| Pronunciation | /ˈmɛθ.ɪl ˈaɪ.səˌproʊ.pɪl ˈfiː.nɪl kɑːrˈbæm.eɪt/ |
| Identifiers | |
| CAS Number | 63134-38-3 |
| 3D model (JSmol) | `/data/c6e4dae9c4e88a5d/mjsmol/jmol.php?model=CC(C)C1=CC(=CC=C1)NC(=O)OC` |
| Beilstein Reference | 2081426 |
| ChEBI | CHEBI:77809 |
| ChEMBL | CHEMBL455637 |
| ChemSpider | 21588401 |
| DrugBank | DB07715 |
| ECHA InfoCard | 100.094.286 |
| EC Number | EC 252-518-4 |
| Gmelin Reference | 135942 |
| KEGG | C18707 |
| MeSH | D008795 |
| PubChem CID | 10042333 |
| RTECS number | SZ8575000 |
| UNII | NV1ZB6M02H |
| UN number | UN3266 |
| CompTox Dashboard (EPA) | DTXSID8042215 |
| Properties | |
| Chemical formula | C11H15NO2 |
| Molar mass | Molar mass: 207.26 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 1.08 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 2.7 |
| Vapor pressure | 0.000073 hPa (25°C) |
| Acidity (pKa) | 14.8 |
| Basicity (pKb) | 10.02 |
| Magnetic susceptibility (χ) | -60.94e-6 cm³/mol |
| Refractive index (nD) | 1.525 |
| Viscosity | Viscosity: 1.19 mPa·s (at 20 °C) |
| Dipole moment | 2.99 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 399.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -322.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6474.6 kJ/mol |
| Pharmacology | |
| ATC code | N05BA11 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P403+P233, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 1, Instability: 0, Special: - |
| Flash point | 110 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 3700 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 250 mg/kg |
| NIOSH | RC2625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Carbaryl Propoxur Bendiocarb Fenobucarb Isoprocarb |

