Methyl 2-Hydroxypropionate: A Down-to-Earth Look at Its Place in Science and Industry
Historical Development
Exploring chemicals like methyl 2-hydroxypropionate sends me back to the stories of organic chemistry’s steady march forward. This compound, known to many researchers as methyl lactate, has been part of laboratories and factories since the late 19th century. I remember reading about its early synthesis from sour milk—where lactic acid gets isolated, then transformed for industrial use. As the textile and cosmetic markets grew, so did the demand for these mild organic esters, and that push shaped the paths of chemists through the last century. The easy availability of biomass and interest in greener chemistry in the past two decades has increased attention to methyl lactate again, as renewable methods shift from niche to necessary.
Product Overview
Methyl 2-hydroxypropionate stands out in the world of esters for one main reason: a gentle solvent presence paired with biodegradable traits. Its molecular structure, a methyl group joined with the lactate backbone, enables this chemical to blend natural function with industrial purpose. Not everyone realizes that substances like these bring together the needs of formulators seeking mildness and biochemists who care about environmental footprints. I see it playing a role not just because it's 'mild,' but because it fits regulatory shifts and consumer awareness about what goes into products on our shelves.
Physical & Chemical Properties
This ester has a light, almost unnoticeable scent, bringing back memories of work in brightly lit labs with clean glassware. Its boiling point falls in a moderate range, which changes how we use it in different settings. In liquid form at room temperature, methyl lactate offers an excellent alternative to traditional solvents that carry odor or volatility problems. The molecule holds both polar and non-polar characteristics, due to its methyl and hydroxypropionyl groups. This balance lets it dissolve a variety of compounds, from paint residues to stubborn grease. The small, two-carbon chain grants it water solubility, something often lacking in traditional solvents. On the environmentally friendly side, its ready biodegradability means breakdown in soil and water occurs swiftly—no buildup across years.
Technical Specifications & Labeling
Reading product labels, I've always appreciated clarity. For methyl 2-hydroxypropionate, purity above 98 percent stands as the norm in industry use, with water content kept low to avoid processing problems. Color stays as light as possible—yellows cast a bad light over many chemicals, and buyers notice. The usual labeling leans on names like methyl lactate, with hazard details about irritation to skin and eyes where necessary. Clear identification helps avoid mix-ups, especially in multi-product facilities, and meets the legal expectations set by both local safety rules and international agencies. Users want to see real, straightforward details so they feel confident about safety and handling.
Preparation Method
The main process for making methyl lactate sticks to time-tested chemical transformations. Lactic acid reacts with methanol using either acidic or enzymatic catalysis, shedding water in a reaction that traces back to classic organic textbooks. The switch to enzymatic routes, which took off in the last couple of decades, has always impressed me for its lower energy needs and smaller pile of unwanted byproducts. These methods draw in companies eager to shrink their carbon footprint and drive up purity. Access to greener pathways matters—a lot of new research dollars chase improvements here, with fermentation feedstocks coming from corn, sugarcane, and even cheese industry waste.
Chemical Reactions & Modifications
Chemists love testing the limits of small molecules. Methyl 2-hydroxypropionate holds up to a variety of transformations. Its ester can undergo hydrolysis to swing back to lactic acid, which is handy for closed-loop biochemical processes. Conversion to acrylic acid or methacrylic derivatives opens doors to plastic and polymer manufacturing. The molecule’s structure supports substitution or reduction reactions, giving rise to new building blocks for specialty resins and pharmaceutical intermediates. Watching these modifications play out has always reinforced how chemistry shapes the trajectory of modern material science.
Synonyms & Product Names
In most catalogs, methyl 2-hydroxypropionate appears as methyl lactate, but names like lactic acid methyl ester or lactic acid, methyl ester also surface. These labels can cause confusion, especially for professionals new to the industry. In my own experience, clear documentation makes a big difference—nothing derails a project faster than using the wrong form of a substance due to name overlap. For major uses and safety data, sticking with the IUPAC or the classic methyl lactate remains the surest path to clear communication.
Safety & Operational Standards
While mild in nature, methyl lactate isn’t completely free of risk. Handling guidelines warn about skin and eye irritation, and decent ventilation goes a long way. In busy plant settings, workers benefit from splash-resistant gloves and eyewear because even so-called gentle chemicals sting without warning. Storage calls for dry, cool spots, away from acids and bases that could trigger unwanted reactions or speed up decomposition. In fire safety drills, professionals focus on its low to moderate flash point, reminding all that a gentle solvent can still fuel accidental fires. Following up-to-date safety practices and maintaining respect for the product matter more than any label can convey.
Application Area
I’ve spotted methyl lactate across a wide spectrum of industries. Paint strippers and degreasers rely on it for a safer profile than heavy-duty petroleum solvents. In the pharmaceutical field, it stars as an intermediate for active ingredients and for cleaning sensitive equipment. Natural cosmetics wouldn’t look or feel the same without this ester supporting fragrance blends, gentle cleansers, and biodegradable lotions. Some food processing chains employ it to extract delicate aromas and flavors, though regulations keep a close eye on purity. The printing sector replaces old-school solvents with methyl lactate to lower emissions and meet green targets without sacrificing print quality. Across the board, this chemical’s mild profile meets the push for sustainability and tighter workplace safety rules.
Research & Development
The push for renewable chemicals propels much of today’s R&D into methyl 2-hydroxypropionate. Research teams focus on scaling enzymatic routes, cutting down on fossil fuel inputs, and raising yields from fermentation. New catalysts and engineered bacteria open up ways to squeeze more methyl lactate from agricultural waste or process side streams. Lifecycle analysis helps pin down the real gains in carbon reduction, proving that the greener path isn’t just wishful thinking. In the application sphere, scientists look at combining methyl lactate with other organic solvents to deliver better cleaning, paint removal, or resins for automotive and electronics use. Facing tough legislative barriers in Europe, North America, and Asia, producers know R&D doesn’t stop at good enough—it presses forward to stay ahead on both environmental and technical fronts.
Toxicity Research
Reading toxicology studies on methyl lactate helps me keep risk in perspective. Most evidence says mild to moderate irritation is the main concern, usually tied to its ester nature. Short-term inhalation in busy workplaces can cause coughing or discomfort, but it doesn’t build up in the body or trigger chronic harm in healthy adults. That profile brings some comfort, yet long-term studies on aquatic life show even mildly toxic organics pose threats when misused or over-applied. Waste management practices must reflect these findings, preventing untreated rinses or spills from slipping into waterways and harming sensitive species. Toxicity research isn’t about supporting bans or panic, but about finding sound boundaries for safe use and disposal.
Future Prospects
The market signals for methyl 2-hydroxypropionate have been clear for years. Demand rises with every new push for safer solvents, stricter workplace controls, and greener supply chains. Producers face challenges in raw material sourcing and keeping prices controlled against more established, petrochemical competitors. Public policy shifts can send shockwaves, suddenly driving up interest for bio-based esters when regulations turned against old solvents. In my view, technical advances in microbial engineering and catalytic processes will cement methyl lactate as a mainstay in sustainable chemical practices. Products with lifecycle transparency, clear labeling, and proven safety will attract buyers and regulators alike. Offering stability and flexibility in sourcing—alongside rigorous adherence to environmental and toxicity standards—will separate the winners from the rest in a world where old chemistry gives way to new responsibility.
What is Methyl 2-Hydroxypropionate used for?
Straightforward Uses in Everyday Products
Methyl 2-hydroxypropionate doesn’t sound like something you’d want anywhere near your home or body, yet it plays a quiet but steady role in things just about everyone uses. Take a walk in a grocery or home improvement store, and you’ll spot dozens of products benefitting from its unique properties. It crops up in the world of solvents and cleaners. Companies lean on it for dissolving certain substances that water can’t handle, so you’ll find it in paint removers and degreasing agents. This chemical melts away grime where plain soap and water fall short. From my own attempts at DIY, I’ve learned that paints and finishes only come off cleanly with the right solvent. Methyl 2-hydroxypropionate gets picked because it has the muscle but doesn’t kick up the harsh fumes that tougher solvents like acetone bring to the table.
Backbone in Polymer and Resin Production
Industry watches this chemical because it plays a major role in the making of plastics and resins. It’s a building block, forming the foundation for various polymers. These substances pop up in packaging, coatings, adhesives, and even medical devices. Over the years, I’ve noticed how plastic packaging can look and feel different depending on its source. Shifts in plastic quality often trace back to the raw chemicals used during manufacturing. The presence of methyl 2-hydroxypropionate delivers a certain flexibility or clarity that companies chase in their finished products. This affects not just the look but the shelf life and safety of foods, medicines, and everyday goods.
A Friendlier Option for the Environment and the User
Concerns about chemical exposure and environmental impact won’t go away. Methyl 2-hydroxypropionate steps up compared to more toxic rivals. Its lower toxicity and easier biodegradability put it ahead in the push for safer workplaces and cleaner air, especially as authorities keep tightening rules around volatile organic compounds. Cleaner processes reduce risk for people who spend all day on a shop floor. Anyone who’s felt lightheaded after using a common cleaner or stripping paint in a cramped room knows why this matters. Replacing more aggressive chemicals with less hazardous ones like this reduces accidents and sick days.
Challenges and Room to Improve
Wide adoption brings pressure. Even a chemical with a cleaner record can leave a mark. Production on an industrial scale uses energy and generates byproducts. Regulators keep their eyes on downstream effects, trying to balance industrial needs with environmental care. This calls for continued work on waste management, recycling, and cleaner synthesis routes. Progress comes from both innovators and watchdogs. As someone who’s lost track of countless recycling rules, I appreciate when companies carry their share of the load, seeking out ways to cut emissions and recover used materials before they land in a landfill or water source.
Building Trust with Reliable Information
Misinformation and chemical names make it easy to worry about safety. Clear, evidence-based guidelines simplify choices for anyone shopping for home repair, cleaning, or food storage. It matters that data about methyl 2-hydroxypropionate gets checked, shared, and updated. My trust in a product climbs when I see that the manufacturer follows regulations and supports their claims with studies. The same expectation should guide everyone making or buying products that rely on specialty chemicals. Open data, stronger oversight, and honest labeling help keep confidence high—and risk low.
Is Methyl 2-Hydroxypropionate safe to handle?
What Is Methyl 2-Hydroxypropionate?
Methyl 2-hydroxypropionate, known in some labs as methyl lactate, pops up as a solvent and chemical building block in many manufacturing environments. It comes from lactic acid, so the chemistry behind it feels pretty familiar to anyone with a background in biochem or industrial production. I’ve seen this stuff used for specialty cleaning, certain flavors and fragrances, and plastic production.
Possible Health Hazards
Spending time in labs and handling chemicals, it’s hard not to notice which substances demand a little more respect. Methyl 2-hydroxypropionate behaves much like other solvents. If you splash it on your skin, irritation isn’t rare. Breathe in the vapors for an extended time, and soon enough, headaches, dizziness, or throat irritation will creep up. Even though methyl lactate has a relatively benign reputation among solvents—it’s less aggressive than toluene or xylene—people shouldn’t treat it as completely harmless. Chronic exposure and poor ventilation raise risks. Sensible lab safety rules say gloves and goggles aren’t optional. I've seen a few colleagues think it's "safer" and come away with red, itchy skin after a spill.
Environmental Impact
Disposal sometimes gets ignored until the drum is full. Methyl 2-hydroxypropionate breaks down quickly in nature, but large releases will harm aquatic life and mess with the balance in waterways. I learned early on that pouring leftovers down the drain causes headaches for municipal water treatment—not to mention the fines that come with poor disposal practices.
Real-World Lab Safety
In every place I’ve worked, the material safety data sheets (MSDS) line the walls. Most point out the same things: keep it off your skin, wear goggles, avoid breathing high concentrations, don’t eat or drink during use. I keep a box of nitrile gloves on the bench and make sure spill kits stay stocked.
A big lesson from experience: don’t trust the “safer solvent” label too much. That term just means safer compared to harsher chemicals, not safe in every situation. One slipup with a leaky bottle on a crowded bench gets ugly fast—volunteer clean-up crews hate dealing with sticky, strong-smelling messes. Surface residues build up on benchtops, too, if folks wipe things down with bare hands.
Regulation and Best Practices
Regulations don’t rank methyl 2-hydroxypropionate as the most tightly controlled chemical, but most safety guidelines mirror the treatment given to more hazardous compounds. This approach works because unexpected exposures in any quantity lead to health complaints. Best practices keep the odds of accidents pretty low: ventilate workspaces, use closed containers, and train everyone to recognize symptoms of exposure. Emergency eyewash stations shouldn’t collect dust.
What Actually Works?
Practical solutions matter here. Ventilation removes vapors before they cause trouble. Good gloves and eye protection beat skin issues. Labeled containers and common-sense storage routines reduce mystery spills. I find that safety talks, quick clean-up routines, and a culture of speaking up when safety shortcuts show up do more than an extra sign on the door ever does.
Staying healthy with chemicals, including methyl 2-hydroxypropionate, takes more than a piece of paper taped up in the breakroom. Daily practice and respect for the stuff make the real difference.
What are the storage requirements for Methyl 2-Hydroxypropionate?
Understanding the Chemical
Methyl 2-hydroxypropionate shows up in labs and industries where solvents, coatings, or specialty chemicals play a role. Every time someone asks about storing this compound, they’re tapping into more than textbook chemistry—they’re asking about keeping people safe and product quality high.
The Role of Temperature
Cooling liquids gets plenty of attention in chemical storage. From my time working in small-scale research, it’s clear that this compound holds steady at room temperature, but only if the surrounding area stays well-ventilated and shaded from direct sunlight. Exposure to heat speeds up evaporation and can trigger chemical changes. If left next to heating equipment or in a sunlit workspace, vapor pressure creeps up, raising risks of leaks or worse.
Moisture Makes Trouble
Keeping water out of the storage area is non-negotiable. Even basic condensation on bottles starts off a chain reaction: dilution, unwanted hydrolysis, and the potential for an acidic mix no one wants around. In practice, using a tight-sealing, moisture-proof container makes sense. Silica gel packets or simple drying agents work if humidity threatens to sneak in through handling or a slightly cracked lid.
Avoiding Reaction with Other Chemicals
In my early days in a shared lab, carelessness with chemical segregation caused more than a few headaches—especially when organics like methyl 2-hydroxypropionate sat next to strong acids, bases, or oxidizers. Accidental spills or fumes from nearby containers prompt unexpected reactions. Storing this chemical on its own shelf, well labeled, far from potential reactants, helps sidestep trouble before it starts.
Material of the Container Matters
Not all plastics or rubbers handle solvents the same way. I’ve watched a cheap container get brittle and leak after a few months’ exposure. Glass does the job, as do high-density polyethylene and some fluoropolymer plastics. Anything permeable or easily degraded shouldn’t hold this chemical long-term. Tight screw-cap lids, ideally lined with PTFE, cut down the chances of escape or contamination.
Ventilation and Safe Air Quality
Proper ventilation ranks high in any storage plan. Vapors build up in closed rooms or cabinets, turning a minor spill into a headache-inducing hazard. Fume hoods and local exhaust points lower the risk, as does storing the chemical away from cramped or poorly-aired spaces. Even in an average room, crack windows or keep a mechanical exhaust running if large quantities get stored.
Labeling, Spills, and Accident Response
Good labeling helps everyone—especially in busy spaces. I’ve found that clearly marked shelf locations, easy-to-read hazard symbols, and a detailed inventory list reduce confusion on rushed days. For accidents, everyday safety kits with nitrile gloves, absorbent pads, and simple spill-neutralizing powders give peace of mind. No matter how careful or experienced, accidents rarely offer warning, so preparation pays off.
Disposal and Legal Responsibility
Handling leftovers shows respect for both health and the law. Treating methyl 2-hydroxypropionate as hazardous waste means turning to certified chemical disposal services. Pouring it down the drain spells disaster for water quality—and may carry legal consequences. Documentation—logbooks, pick-up receipts, signed manifests—ensures transparency and meets regulatory expectations.
Better Habits for Safer Storage
A good routine solves most problems before they appear. Pair frequent checks on storage spots with proper housekeeping, and the odds of an unfortunate event shrink. Ask questions, share observations, and encourage everyone in the workspace to flag small issues early. Safe storage ends up being less about the chemical and more about the people handling it day in, day out.
What is the chemical formula of Methyl 2-Hydroxypropionate?
Digging Into the Science
Methyl 2-hydroxypropionate comes with the formula C4H8O3. If you look at its structure, you’ll find a basic framework familiar to anyone who’s spent time in a chemistry lab or brewing beer—carbon, hydrogen, and oxygen, holding hands in a pattern that lets the compound take on a long list of jobs in everyday life. Picture a methyl group joined to 2-hydroxypropionic acid, and you have the backbone of this molecule. It’s easy to think of chemical formulas as strings of letters and numbers, but for folks in industry, research, and even medicine, this seemingly simple code unlocks a whole world of applications and responsibilities.
Everyday Connections: How This Molecule Shows Up
Manufacturers use methyl 2-hydroxypropionate in plenty of products. Paints, coatings, adhesives, and detergents all tap into its solvent powers. As the world inches toward greener chemistry, this compound gets more attention because it can be sourced from renewable materials such as lactic acid made by fermenting corn sugar. This links it directly with sustainable practices. In an era when climate news regularly fills headlines, anything that swaps petroleum for plants deserves real attention.
Handling Risks in the Lab
Reading chemical formulas and mixing compounds goes far beyond memorizing classroom rules. Safety comes first, since exposure can cause skin and eye irritation. I remember working in the lab and watching new students splash more carelessly than they realized—goggles fogged, gloves sometimes forgotten. One day, a colleague ignored a tiny spill, assuming a “benign” substance couldn’t pack much punch. She learned quick that all organic solvents demand respect. Understanding C4H8O3’s structure isn’t enough unless folks also get trained in proper handling. Industry regulations require clear labeling, suitable personal protective equipment, and appropriate ventilation. These aren’t just checkboxes; they keep real people out of clinics and emergency rooms.
Environmental Impact and Responsibility
The formula tells only part of the story. Waste management and emissions shape whether methyl 2-hydroxypropionate becomes a stepping stone for greener chemistry or ends up harming ecosystems. Wastewater from factories sometimes leaks trace amounts into rivers, which ought to raise concern. Scientists have shown that smaller molecules, especially organic acids and their esters, can linger and challenge wastewater treatment plants. There’s a real need for monitoring and investing in treatment technology so these compounds don’t spread beyond the factory fence.
Moving Toward Safer, Smarter Chemical Use
I’ve seen companies start to rethink ingredient choices in response to consumer pressure and new rules. Switching to less hazardous solvents, recycling waste, and adopting safer processing methods can make a big dent in occupational hazards and pollution at the same time. Chemists and policymakers have to work together, looking at both the formula and the footprint. Schools should put more focus on practical chemical literacy so the next generation can spot risks right away and avoid repeating past mistakes. The right formula, handled with care, can help build a safer world both inside and outside the lab.
What is the shelf life of Methyl 2-Hydroxypropionate?
Understanding the Basics
Methyl 2-Hydroxypropionate keeps showing up across labs, workshops, and even in a few home-brewed projects. Better known to some as methyl lactate, this compound serves as a solvent, a building block for chemical synthesis, and sometimes in flavor or fragrance formulation. The big question I hear is: How long does this stuff actually last once you get your hands on it?
What Affects How Long it Keeps
It’s easy to assume a bottle of chemical has an endless life if stored on a shelf, but air, moisture, and sunlight all put their own stamp on its timeline. In my own experience in a university lab, a bottle of methyl 2-hydroxypropionate kept tightly sealed in a cool, dark cupboard consistently held up for two years and sometimes beyond. That said, a clumsy cap, a sunny window sill, or a humid storage room can take months off useful life. Most manufacturers stamp two or three years as the shelf life, but only if it stays unopened and protected from extremes.
Why Purity Matters
Purity plays a bigger role than folks think. Once the bottle sees the light—or more importantly, the air—water vapor sneaks in and begins to work. Methyl 2-hydroxypropionate can draw in moisture, start to break down, or even polymerize. Little changes in the liquid can hurt its performance in a chemical reaction or make it unsuitable for more sensitive uses, like working with pharmaceuticals or food flavorings.
It’s worth pointing out that once you open the bottle, you’re on the clock. Even with a clean pipette, each dip increases the risk of contamination. In my lab, some students made the mistake of using a “quick and dirty” approach and the leftover stock, once clear, turned yellow after six months. That’s a sure sign its chemistry has shifted. From there, nobody wants to trust the results or the safety of what’s inside.
How to Make It Last Longer
Here’s what works: stash bottles in a cool, dry closet away from direct sunlight, keep caps tight, and avoid plastic droppers or scoops that pull in air. I’ve seen teams stretch the life up to three years just by splitting bulk orders into smaller amber bottles, only opening what they need. Keeping a supply chain flowing, so small amounts get used up before they go bad, proves smarter than chasing big savings on a giant drum that sits around absorbing dust and humidity.
Quality Over Convenience
Cutting corners with storage or handling never pays off. Search for packaging with a recent production date, avoid stock past its best-before year, and document lot numbers if traceability matters. A regular check—sniffing for sour or musty odors, watching for cloudiness or yellowing—keeps mistakes from slipping into production or experiments. I’ve learned to trust my nose, eyes, and records as much as any batch certificate.
Better Habits, Less Waste
Relying on methyl 2-hydroxypropionate with a solid shelf life saves time, frustration, and cash. By learning from a few preventable mistakes and taking storage seriously, labs and manufacturers hold on to quality and avoid those scrambling last-minute replacements. Simple changes in how we buy, store, and check this compound go a long way toward better projects and safer outcomes.
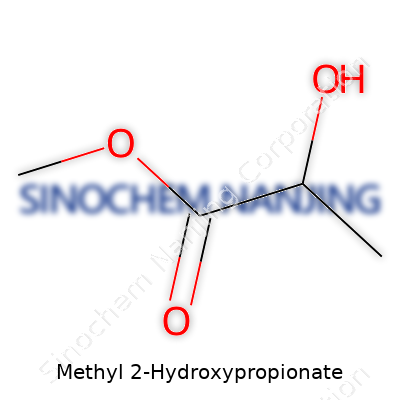
| Names | |
| Preferred IUPAC name | Methyl 2-hydroxypropanoate |
| Other names |
Methyl lactate Lactic acid methyl ester Methyl 2-hydroxypropanoate |
| Pronunciation | /ˈmɛθ.ɪl tuː haɪˌdrɒk.si proʊˈpiː.əˌneɪt/ |
| Identifiers | |
| CAS Number | HN: 920-36-5 |
| Beilstein Reference | Beilstein Reference: 1720239 |
| ChEBI | CHEBI:18373 |
| ChEMBL | CHEMBL16326 |
| ChemSpider | 11679 |
| DrugBank | DB03793 |
| ECHA InfoCard | DTXSID1039234 |
| EC Number | 2918-66-5 |
| Gmelin Reference | 120415 |
| KEGG | C01008 |
| MeSH | D001322 |
| PubChem CID | 10739 |
| RTECS number | OJ1875000 |
| UNII | 7A2J965L5X |
| UN number | UN2497 |
| Properties | |
| Chemical formula | C4H8O3 |
| Molar mass | 90.08 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | mild |
| Density | 1.18 g/cm3 |
| Solubility in water | miscible |
| log P | -0.41 |
| Vapor pressure | 0.11 mmHg (20°C) |
| Acidity (pKa) | 13.0 |
| Basicity (pKb) | 15.09 |
| Magnetic susceptibility (χ) | -7.44 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.414 |
| Viscosity | 2.12 mPa·s (20 °C) |
| Dipole moment | 2.53 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -726.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1,263.7 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | A16AX06 |
| Hazards | |
| Main hazards | Causes skin irritation. Causes serious eye irritation. May cause respiratory irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H315, H319 |
| Precautionary statements | Precautionary statements: P261, P264, P271, P280, P301+P312, P305+P351+P338, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-2-1-W |
| Flash point | 83 °C |
| Autoignition temperature | 430 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): 5,840 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 5020 mg/kg |
| NIOSH | RX8750000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 50 mg/L |
| Related compounds | |
| Related compounds |
Ethyl lactate Propylene glycol Lactic acid Methyl lactate Methyl pyruvate |

