Methoxyethylmercury Acetate: Historical Background, Science, Safety, and the Road Ahead
Historical Development
Throughout the early and mid-20th century, organomercury compounds found their way into laboratories and industrial setups across the world. The search for specialized reagents capable of site-specific modification in organic synthesis sparked curiosity around compounds like Methoxyethylmercury Acetate. Researchers didn’t stumble into using this substance overnight. Documented work from academic groups, particularly those interested in mercury’s peculiar chemistry, uncovered that substituting traditional groups with methoxyethyl chains often led to more reactive—and more hazardous—intermediates. The recording of these developments reflects both excitement for new tools and a difficult legacy of toxicity. Experience has shown that progress sometimes outpaces a clear understanding of risk, and in the case of organomercury chemistry, that fact shaped regulations and perceptions for decades.
Product Overview
Methoxyethylmercury Acetate bears significance in specialized synthesis. The molecule combines an acetate group with an organomercury backbone, and a methoxyethyl chain that sets it apart from simpler mercury compounds such as methylmercury or phenylmercury derivatives. Over the years, its main customers have been research institutions and chemical manufacturers. On the shelf, one finds it as a pale solid or sometimes a faintly colored powder. While it has never enjoyed the broad use seen with industrial mercury salts, its niche nature doesn’t diminish the necessity for strict handling and regulatory clarity.
Physical & Chemical Properties
This compound stands out for its relatively high molecular weight, pronounced density, and low volatility. The addition of a methoxyethyl group increases its solubility in organic solvents like chloroform or ether, and the acetate enhances its reactivity in nucleophilic substitution. At room temperature, Methoxyethylmercury Acetate remains a solid, tipping towards instability if stored under hot or damp conditions. Long-term storage demands non-reactive containers—glass lined with Teflon fares better compared to common plastics, as mercury-based agents often degrade common polymers. Chemists working with this material need to recognize its potential to decompose and release vapors that carry even greater risks.
Technical Specifications & Labeling
Quality comes defined by purity, with reputable suppliers providing certificates covering assay percentage, moisture content, and trace contaminant levels. Labels carry not just the chemical name but also vital hazard statements: “Very toxic by inhalation, ingestion, or skin contact.” Syringe-filling and weighing stations involve closed, ventilated hoods, and clear notations highlight correct personal protective equipment: thick gloves, polymer sleeves, and disposable aprons. Each batch links to precise lot numbers and storage conditions—often under dry nitrogen and at sub-ambient temperatures. Those little details may seem excessive, but in my work, ignoring them proved costly, occasionally bringing research to a halt until we restocked or proved compliance.
Preparation Method
Manufacturing Methoxyethylmercury Acetate usually starts with the reaction of mercuric acetate with methoxyethanol under controlled synthetic protocols that limit exposure and waste. Batch reactors use tight seals, and process chemists keep water and moisture to an absolute minimum due to hydrolysis risks. The process typically concludes with repeated recrystallization, and final steps demand careful filtration to avoid mercury particle contamination. Modern labs opt for sealed glassware with in-line exhaust traps, reflecting lessons drawn from past exposures and environmental mishaps. The final product undergoes rigorous testing for mercury content and acetate purity to prevent downstream complications.
Chemical Reactions & Modifications
Methoxyethylmercury Acetate finds use as a transfer agent for the methoxyethyl group, particularly in organic synthesis tasks requiring selective functionalization. Process chemists appreciate how the mercury center directs group addition or allows for creation of carbon–mercury bonds amenable to later transformations. Side reactions happen, usually involving hydrolysis or unwanted transmetalation. Key modifications include replacement of the methoxyethyl chain or swapping the acetate for other carboxylates, fine-tuning reactivity with different target molecules. That flexibility remains the main reason it exists at all in the modern chemical toolkit.
Synonyms & Product Names
Methoxyethylmercury Acetate goes by various synonyms in catalogs and scientific literature, including 2-Methoxyethylmercury Acetate or Acetic Acid, 2-methoxyethylmercury(II) salt. European suppliers might use alternate naming conventions, such as EINECS identifiers or CAS listings. These variations, though minor, sometimes lead to confusion when importing or exporting. I've run into procurement issues where a shipment used a trade name rather than the systematic one, causing delays and regulatory bottlenecks. Getting to know these naming quirks ahead of time can save more paperwork than one might expect.
Safety & Operational Standards
Strict protocol isn't an option—it's a mandate. Any exposure, even at low concentrations, can throw health into turmoil: mercury compounds cross the skin barrier, accumulate in nervous tissue, and exert toxic effects that remain decades after exposure. Risk assessments recommend not just gloves and goggles, but barriers that cover every exposed area. Spill kits need more than basic absorbents; specialized mercury-vapor traps are essential. Disposing of waste challenges even well-equipped sites: incineration remains forbidden, and instead licensed hazardous waste firms take charge. Workplace mercury monitoring and blood tests for personnel add layers of reassurance and, frankly, accountability. Over years in the lab, a healthy respect for organomercury compounds became second nature, driven not just by paperwork but by the hard reality of their consequences.
Application Area
Pure research sees most of the action. Methoxyethylmercury Acetate rarely shows up in mass-production settings or broad commercial applications, largely because of its toxicity and environmental persistence. Instead, small-scale synthetic projects, especially those working up unique carbon frameworks or evaluating heavy-atom effects in catalysis, call for this compound. Environmental scientists sometimes employ trace amounts to model mercury movement, though many now reach for less hazardous analogs. When a project demands utmost selectivity and reactivity, the risk-to-reward ratio sometimes justifies turning to a specialized compound like this.
Research & Development
Ongoing work has shifted from new synthetic uses to safer analogs and alternatives. Academic publications from the last ten years reveal a drop in direct experimentation. Regulatory pushback and stricter occupational health standards convinced most groups to look elsewhere unless Methoxyethylmercury Acetate seemed truly indispensable. Collaboration between academia, regulatory bodies, and industry produced digital tools to model reactivity, lowering the necessity for real-world exposure. Laboratories investing in this area now focus equally on containment, detection, and safe disposal as on reactivity profiles.
Toxicity Research
Long-term studies confirmed what early case reports suggested: exposure to organomercury compounds pushes the boundaries of safe occupational practice. Animal studies document neurological and renal damage at exposures far below acute toxicity limits. Strip away the technical jargon, and the story boils down to irreversible nerve damage, tremors, loss of coordination, and, in worst cases, fatality. Modern toxicity research stresses the value of blunting exposures using engineering controls, not personal heroism. Regulations stemming from these studies shaped entire workflows, with nations instituting threshold limit values approaching zero. My time spent handling these compounds led directly to mandatory in-person hazard training and annual re-certification, far outpacing the routine protocols other agents required.
Future Prospects
With each year, the demand for Methoxyethylmercury Acetate wanes as safer and greener alternatives crowd the specialty reagent shelves. Still, for those manipulating complex targets or pioneering new mechanisms, its precise reactivity and unique properties keep it relevant. Green chemistry movements and policy changes push manufacturers to lean into less persistent alternatives, and investment in detoxification technologies grows. The path forward seems set: balancing the drive for discovery with ironclad protections for both chemists and the environment. The legacy of Methoxyethylmercury Acetate reminds everyone that innovation without responsibility can cast a shadow for generations.
What is Methoxyethylmercury Acetate used for?
The Basics Behind Methoxyethylmercury Acetate
Methoxyethylmercury acetate doesn’t pop up in daily life but plays a niche role in labs and specialized industries. This compound belongs to the family of organomercury chemicals, which means it carries mercury linked to carbon. The use of mercury-based compounds might raise alarms—deservedly so, given the heavy metal’s toxic legacy—but each one fits a particular slot in the world of research and manufacturing.
Where Methoxyethylmercury Acetate Flows: Application and Use Cases
Labs and some chemical manufacturers use methoxyethylmercury acetate as a reagent. Like other organomercury compounds, it steps in where precise control over chemical reactions matters. Researchers lean on its unique properties for making other chemicals or testing analytical methods that track mercury in environmental samples.
Methoxyethylmercury acetate’s knack for binding with sulfur groups gives it a spot in specific organic synthesis work. Chemists might pick this compound when aiming to introduce methylmercury groups into new molecules. Its selectivity can shape research outcomes, especially for projects that map how mercury behaves in plants or animals. University researchers, in my experience, use it to help mimic contamination scenarios in controlled settings, so they can better understand how mercury travels through ecosystems.
Health and Environmental Concerns
Mercury leaves a heavy footprint, and organic forms like methoxyethylmercury acetate ramp up the risk. Even small amounts pose hazards. Workers must wear gloves and use fume hoods, with protocols tightening every few years as more health impacts come into focus. Inhalation or skin contact could lead to mercury poisoning, which brings neurological symptoms and possible organ damage. The toxic legacy of mercury in compounds like this one shapes tighter regulations and expensive disposal processes.
I recall news stories about industrial mercury spills that left rivers and fish unsafe for years—reminders of just how persistent these substances stay in the environment. Regulatory agencies, including the EPA and OSHA, keep a close watch on chemicals like methoxyethylmercury acetate. These rules force everyone involved to take proper precautions and keep detailed logs.
Balancing Use with Responsibility
The need for methoxyethylmercury acetate comes down to its chemistry. There’s no easy swap for every application, but its dangers push chemists to look for alternatives whenever possible. Some labs substitute mercury-based chemicals for safer reagents, sacrificing a bit of reliability to keep scientists healthier and ecosystems cleaner.
Waste management plays a huge role. Responsible labs and manufacturers never pour it down the drain or toss samples in trash bins. Licensed hazardous disposal companies pick up the waste. Training new researchers to understand why safety matters—beyond just ticking boxes—forms a real defense against mercury’s risks.
Solutions and Future Directions
There’s a trend toward greener chemistry. Funding agencies ask researchers to justify every purchase and to pick safer chemicals when they can. Universities redesign courses and labs to reduce mercury exposure, nudging the next generation away from these older practices. Collaborations between researchers and regulatory agencies spark new ways of testing and tracing mercury, cutting reliance on methoxyethylmercury acetate but improving our picture of mercury pollution.
Every bit of progress here—safer alternatives, better training, stricter regulation—shrinks the space methoxyethylmercury acetate occupies. The path isn’t fast, but it’s clearer today than ever.
What are the health risks associated with Methoxyethylmercury Acetate?
Mercury Compounds Don't Play Around
Most folks know mercury means trouble. Methoxyethylmercury acetate fits right into that picture, packing a punch both for those working with chemicals in the lab and people who might bump into it by accident. Even tiny doses go straight for the nervous system. Feeling numbness, tingling, headaches or trouble concentrating? That’s your body telling you it’s not built to handle this stuff.
Real-World Contacts and Hidden Pathways
I spent years working in a facility where bottles of mercury sat on shelves, some labeled in scribbled handwriting. All it takes is careless storage or clumsy handling for vapors to seep out. Methoxyethylmercury acetate, in particular, doesn’t always show its teeth right away. The danger becomes clear after it’s entered the body, either from breathing in dust, letting it touch bare skin, or swallowing residues. Mercury isn’t the sort of metal you wash off easily—once it's in, it sticks around, especially in the nervous system.
Kicking the Immune System While It’s Down
Autoimmunity turns the body against itself, and chemicals like this compound can throw fuel on that fire. Studies have tracked increased autoimmune diseases in communities exposed to mercury compounds. The more the body tries to get rid of these molecules, the more the immune system finds itself in a losing fight, leading to chronic illness. You can’t always see this happening, but the consequences build up year after year.
Reproductive and Developmental Threats
Women of childbearing age and kids face the sharpest edge here. Methoxyethylmercury crosses the placenta without much resistance. Pregnant women exposed to even small amounts risk fetal brain damage. Mercury exposure in young kids holds back brain development, causing speech delays, poor coordination, and learning struggles. In plain terms: once mercury gets in, it takes away potential, not just health.
Looking for Answers
Getting rid of mercury exposure means thinking about every step in the process. Workers need proper gloves, masks, and regular training to spot risky practices—no exceptions. Places selling these chemicals should track them tightly. I’ve seen labs where the right protocols dropped exposure rates to almost nothing, proving that careful stewardship works.
Communities need to hear the specifics about mercury, not just generic warnings. Doctors should ask the right questions when seeing symptoms that could look like anything from ADHD to tremors, not dismiss things as stress or normal aging. Schools and local agencies can teach folks how to spot hazards in the first place.
Disposal counts, too. Dumping mercury down drains or tossing scrap in regular trash only passes the risk onward. Secure, high-temperature incineration and proper waste management actually stop the circle. There’s no substitute for doing it right the first time, because once mercury spreads, clawing it back gets nearly impossible.
Drawing on Experience to Build Safer Habits
I’ve watched old habits die hard—people convincing themselves small exposures add up to nothing. Science keeps proving the opposite. The risks from methoxyethylmercury acetate go well beyond chemical charts; they walk around in the real world, hitting brains, bodies, and the people least able to fight back.
How should Methoxyethylmercury Acetate be stored and handled safely?
Understanding the Risks
Methoxyethylmercury acetate has a reputation for a reason. Not every chemical brings a real fear factor, but mercury compounds do. With any kind of organomercury material, skin absorption, inhalation, and contamination sit high on the risk ladder. Exposure can hit the nervous system and development in ways you don't want to test first-hand. In factories and labs, routines often try to cover mercury risks, but that’s not enough. Real safety needs stronger habits.
Smart Storage Protects Health
This chemical can’t sit on just any shelf. Dry and cool storage cuts down vapor build-up. Direct sunlight brings heat and can cause decomposition or raise vapor levels, so lockers or ventilated cabinets make more sense. Flammable solvents go in a flammables cabinet. Mercury compounds demand a separate, clearly labeled, tight-sealing container, away from anything that could react, especially strong acids and oxidizers. All containers must survive leaks and remain tightly closed. No one wants to find a cracked plastic container oozing something this toxic.
Personal Safety Means Respecting the Limits
Every place I’ve worked with mercury, the best protection came from simple, direct habits. Gloves rated for chemical protection, splash goggles, and well-covered clothes make a difference. A dust mask doesn’t cut it — you need a proper respirator when there’s a risk of vapor or dust. If a spill lands on bare skin or in the eyes, immediate washing is the only option. Safety showers and eyewash stations have to be nearby and unlocked. No one can predict every spill, but reacting fast changes outcomes.
Accidents Happen: Prepare for the Worst
People think, “that won’t happen here,” right up till it does. Mercury compounds can volatilize, so even a small spill becomes a big deal. Special mercury spill kits with amalgamation powder or pads matter. Never use a regular vacuum cleaner. That only spreads vapors. Disposal means hazardous waste, not the trash or standard drain. Emergency contact sheets and training go a long way but get ignored until reality hits.
Rules Keep Everyone Safer
OSHA and EPA both carry strict guidelines for mercury storage and handling. Following their steps doesn’t show obedience to a faceless regulation — it cuts risk to real people. Local authorities add layers because a small mistake spreads to the whole neighborhood’s air and water. Medical surveillance and regular air testing keep secrets from building up. On paper, rules seem like a burden, but anyone who’s seen the aftermath of a mercury spill knows they come from real pain and loss.
The Human Side of Chemical Safety
I remember an old lab where people thought they’d cleaned up a tiny mercury spill years back. Later, a new team measured the workbench with a better meter. The contamination remained. Young and experienced staff alike trusted old habits, but mercury doesn’t care how long you’ve worked. Overconfidence lets exposure creep in and go unnoticed.
In the end, strong habits, constant attention, and treating each job as having no margin for error actually save lives. Methoxyethylmercury acetate deserves more respect than casual use ever provides — and every safe day in the lab is proof that paying attention works.
What should I do in case of accidental exposure to Methoxyethylmercury Acetate?
Understanding a Real Health Threat
Methoxyethylmercury acetate isn’t just another chemical with a tongue-twister of a name. Sitting somewhere between industrial labs and specialty manufacturing, this compound packs a real punch. It’s got mercury in its bones, and mercury always draws a red flag. Lives have been changed by accidental exposure to mercury compounds, and not in good ways. Folks have lost memory, motor skills, even the strength to pick up a coffee mug. Ask anyone who has worked with mercury in a lab for years. Accidents haunt people long after the spill is cleaned up.
What Happens on Contact?
Contact with skin doesn’t always trigger immediate pain. That’s where things get sneaky. Strangely enough, it can sit there and seep in, starting damage long before the first headache or tremor makes you worry. Mercury plays rough with kidneys, nerves, and the brain. If slips or splashes reach the eyes or mouth, this chemical doesn’t hold back. Lab technicians share stories about rashes that won’t quit and nausea stalking them for hours after forgotten spills. That experience sticks with you —and warns others—because too many folks ignore the early symptoms.
What Can You Actually Do If It Happens?
Panic doesn’t help. Quick action does. Strip off contaminated clothing, toss it in a sealed bag, and don’t try to save any of it. There’s no washing away poison that bonds with fabric. Hit the nearest source of running water, and rinse the exposed area thoroughly for at least fifteen minutes. Scrubbing or using solvents serves only to rub the stuff in deeper, which doesn’t end well. Mercury’s not something you can scrub away like mud.
If the chemical lands in your eyes, every second matters. Flush with cool water and keep the eyelids wide open. Don’t rub. Rubbing hurts more than helps. Swallowing even a little bit calls for urgent emergency attention—run, don’t walk, to a hospital or call your emergency number, and bring the container or label with you, if possible. Doctors won’t waste time guessing; they’ll get straight to treating possible poisoning symptoms with chelation agents.
Why Prevention Is Better Than Scrambling After the Fact
Those who work around Methoxyethylmercury acetate keep personal protective equipment handy for a reason. Gloves, goggles, lab coats, and even fume hoods form a frontline defense worth every ounce of hassle. Too many accidents start with a shortcut—no goggles during a “quick” transfer, or gloves tossed aside to scribble a note. It pays to respect the rules, even on busy days. I’ve watched a fellow researcher spend months in recovery after a spill that started with an untied gown. A few seconds of protection would have saved him years of health problems.
What Needs Fixing?
Mistakes thrive in silence. Training must focus on exactly what this compound can do, why cutting corners isn’t worth it, and how to act without hesitation if anything goes wrong. Real stories of those who recovered—and those who didn’t—stick better than dry instructions. Organizations should keep spill kits stocked with mercury-absorbing materials, not just a dusty broom. Emergency procedures need practice. Hospitals must stay ready to spot and treat mercury exposure fast.
Community support matters, too. Reporting a spill shouldn’t carry stigma or risk. Open, honest communication inside teams helps spot hazards before they become tragedies. Folks who work with dangerous chemicals deserve more than a laminated safety poster—protecting health means putting those policies into real action.
Is Methoxyethylmercury Acetate regulated or restricted by any authorities?
Why Mercury Compounds Demand Attention
Mercury grabs headlines for the wrong reasons. Over my years following chemical safety news, I keep seeing communities bear the brunt of poor regulations. The stuff -- in its different forms -- creeps into water, soil, food. Parents worry, scientists debate, and doctors see the side effects in real time. Methoxyethylmercury acetate sounds obscure, but it belongs to the family of organomercury compounds, which have a track record of causing trouble both for ecosystems and human health.
Patchwork of Regulations — Or the Lack Thereof
Some chemicals never escape the margins of scientific literature, but methoxyethylmercury acetate is no lightweight. Very few countries treat organomercury compounds as harmless these days. In the United States, the Environmental Protection Agency (EPA) puts mercury and its compounds under the microscope because they can provoke neurological and developmental harm, disrupt reproduction, and linger in the environment for generations. Methoxyethylmercury acetate often falls under broad “alkyl mercury” or “organic mercury” labels in federal rulebooks. Other names might shield its presence in products or processes, making tracking trickier for officials. In the European Union, REACH legislation brings most mercury compounds under strict monitoring. Still, regulatory loopholes surface because authorities can’t stay ahead of every derivative cooked up in a lab.
Blind Spots in Chemical Controls
Watching family members get sick from well water in my own rural community taught me that toxicity has a way of sneaking into daily life before anyone realizes. The slow grind of science doesn’t help if decision-makers move slower. Fact: many mercury-based pesticides, cosmetics, and lab reagents have fallen out of favor or face outright bans. Methoxyethylmercury acetate rarely turns up on the open market these days, likely due to pressure from international treaties like the Minamata Convention. Still, nothing guarantees full prohibition or transparency. If a researcher walks into a chemistry storeroom, there’s no blinking red light to warn that a bottle of methoxyethylmercury acetate brings as much risk as the big-name toxins. Countries without strong chemical oversight might unwittingly host the next contamination crisis.
Forging Smarter Chemical Policy
People deserve clear warning labels and up-to-date lists of regulated chemicals—especially when research points to lasting impacts on kids and wildlife. I’ve talked to environmental professionals who want more than just after-the-fact cleanup. They want tough pre-market screening and public inventories. If chems like methoxyethylmercury acetate disappear from regular trade, that’s good: But out-of-date rules or spotty enforcement keep loopholes alive. Closing the gap calls for governments to update their banned and restricted chemical lists, to keep universities and companies honest, and to empower whistleblowers who spot rogue sales or dumping. If chemical safety training becomes the norm from high school onward, fewer folks will unwittingly take chances with mercury derivatives.
The Price of Complacency
Mercury’s history in industry and medicine sends a clear warning. Ignoring “niche” chemicals like methoxyethylmercury acetate creates future headlines nobody wants to read. I’ve seen community groups force change by raising their voices about contamination. Regulators, industry, and the public must keep turning up the pressure so these hidden dangers don’t slip through the cracks. Smart rules, watchdogs with teeth, and better education about what’s in chemical storerooms—these steps protect not only individual towns, but the wider world their runoff and emissions reach. Every overlooked chemical brings fresh risk; waiting for disaster never pays off.
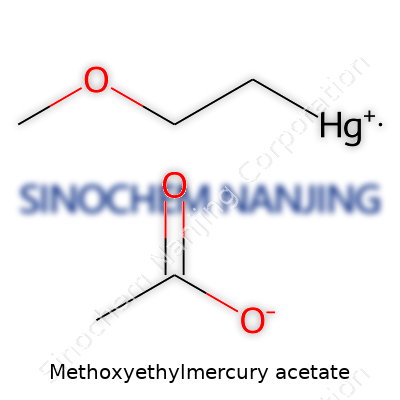
| Names | |
| Preferred IUPAC name | 2-methoxyethylmercury acetate |
| Other names |
Acetic acid, (2-methoxyethyl)mercury(II) salt Mercury, (acetato-O)bis(2-methoxyethyl)- 2-Methoxyethylmercury acetate |
| Pronunciation | /ˌmɛθ.ɒk.siˌiː.θɪlˈmɜː.kjʊr.i əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 6939-97-7 |
| Beilstein Reference | 88729 |
| ChEBI | CHEBI:131209 |
| ChEMBL | CHEMBL2107609 |
| ChemSpider | 15660740 |
| DrugBank | DB02683 |
| ECHA InfoCard | 03db947d-bb40-4857-b153-e028c4b87df0 |
| EC Number | 218-243-5 |
| Gmelin Reference | 8656 |
| KEGG | C18753 |
| MeSH | D008759 |
| PubChem CID | 6917903 |
| RTECS number | OV9625000 |
| UNII | 7QU8PX8I9P |
| UN number | UN2761 |
| Properties | |
| Chemical formula | C5H10HgO3 |
| Molar mass | 364.78 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.52 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.2 |
| Vapor pressure | 2.6×10⁻⁷ mmHg |
| Acidity (pKa) | 8.2 |
| Basicity (pKb) | 5.25 |
| Magnetic susceptibility (χ) | -96.0e-6 cm³/mol |
| Refractive index (nD) | 1.463 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 269.6 J K⁻¹ mol⁻¹ |
| Pharmacology | |
| ATC code | T01AC03 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled or in contact with skin; causes damage to organs; very toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. H373: May cause damage to organs through prolonged or repeated exposure. |
| Precautionary statements | P261, P272, P273, P280, P302+P352, P314, P321, P363, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2 |
| Lethal dose or concentration | LD50 oral rat 36 mg/kg |
| LD50 (median dose) | 56 mg/kg (rat, oral) |
| NIOSH | WA4500000 |
| PEL (Permissible) | 0.01 mg(Hg)/m3 |
| REL (Recommended) | 0.01 mg-Hg/m3 |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Methylmercury Ethylmercury Phenylmercury acetate Thiomersal Mercuric acetate |

