Methanesulfonyl Fluoride: A Candid Look at Its Place in Chemistry and Industry
The Historical Footprint
Few specialty chemicals have shown the strange persistence of methanesulfonyl fluoride, known in some corners by names like mesyl fluoride or methylsulfonyl fluoride. Its emergence traces back to the curiosity-driven postwar boom in organosulfur chemistry, with researchers fascinated by the electro-withdrawing punch of the sulfonyl group. By the late 1950s, folks were digging hard into sulfur-based fluorides because of their ability to throw a wrench into enzyme systems. This wasn’t just lab curiosity—scientists were hunting for new acetylcholinesterase inhibitors among other pharmaceutical tools. Methanesulfonyl fluoride grabbed attention for being strong enough to matter, steady enough to handle, and reactive enough to do something interesting.
Physical and Chemical Character
Anyone who’s handled methanesulfonyl fluoride can tell right away it stands apart simply by nature of its smell and volatility. This stuff condenses into a clear, almost ghostly liquid at room temperatures, but don’t let that fool you. Its vapor likes to climb unless kept corked up tight, and even a little stray whiff can sting the nose with a sharp, almost acrid quality. The chemical itself, built around a methyl group tied to a sulfonyl fluoride, brings together stability with a surprising reactivity, which explains why chemists keep coming back to it. It doesn’t set off at the drop of a hat, which makes storage possible, but reacts quickly when given the right nudge.
Technical Details and Labels
Labels on bottles of methanesulfonyl fluoride try to warn you without painting the whole picture—flammable, reactive, toxic if inhaled or absorbed. Standard concentration bottles usually come in tightly sealed containers with built-in vapor barriers. In any workroom, this compound gets respect: corrosion-proof labels, fume hood handling, safety glasses tight to the head. Established safety data sets list precise boiling and melting points, densities, and vapor pressures, serving less as trivia and more as marching orders for anyone set to use the stuff.
How It’s Made
Making methanesulfonyl fluoride doesn’t call for arcane alchemy, but it does demand solid chemistry. Most routes take a methyl-containing precursor—often methanesulfonyl chloride—and push it into a reaction with a solid fluoride donor. Nasty byproducts can pop up, so cleanup steps count for a lot. Generations of synthetic chemists have tweaked these conditions to avoid harsh temperatures or uncontrolled release of toxic fumes, favoring methods that balance yield with worker safety. Since physical contact with raw ingredients or incomplete product can turn dangerous quickly, strict control over reaction time and workstation setup never gets ignored in seasoned labs.
Chemical Reactions and Tweaks
Methanesulfonyl fluoride’s true value shows in its behavior during nucleophilic substitution and enzyme inhibition. Chemists lean on this reactivity both as a blocking group and as a chemical scalpel in organic synthesis. In biochemistry, it’s an efficient acetylcholinesterase inhibitor—an ability that opened new pathways in neurobiology research. Tinkering the molecule itself—say, swapping in other alkyl groups or attaching functional handles—has generated a suite of analogs. These tweaks allow researchers to fine-tune inhibition strength, reaction timing, or environmental persistence, giving scientists the flexibility to study enzyme systems without relying on old, less selective inhibitors.
Synonyms and Street Names Among Chemists
Depending on the lab, methanesulfonyl fluoride goes by mesyl fluoride, MSF, or sometimes just methylsulfonyl fluoride. Among medicinal chemists it might catch another nickname depending on its role as an inhibitor or precursor. The naming conventions don’t just change with geography, but also with end-use and familiarity—novices stick to the full IUPAC, while old hands in neuropharmacology call it MSF and move on.
Safety from Experience, Not Just a Sheet
Respect for methanesulfonyl fluoride comes less from reading the data sheet and more from time in the lab. Gloves, proper ventilation, and full-seal goggles are not suggestions—old stories from grad students remind us what a drop in the wrong place can do. Absorption risk through the skin or mucous membranes sits at the top of every safety list. Proper storage facilities minimize temperature swings, with chemical-resistant trays standing guard underneath bottles. Incidents from poor fume hood maintenance or carelessness continue as grim reminders that even for skilled workers, a single lapse can turn nasty.
Real-World Application
The medical and biochemical scenes steal the spotlight for methanesulfonyl fluoride, largely because of its precision as an acetylcholinesterase inhibitor. Research teams have used it to slow enzyme activity in animal and cell experiments, teasing out the mechanics of neurotransmission and neurodegeneration. Drug developers saw its selectivity and began exploring related molecules for clinical applications, hoping to build safer enzyme inhibitors for treating diseases like Alzheimer’s. Outside the clinic, the compound has cropped up in organic synthetic labs as a tool for installing sulfonyl groups, often crucial for advanced material synthesis. Reports from fine chemical manufacturers show steady, if specialized, demand among research and development programs.
Pursuits at the Research Frontier
Recent research stretches the potential of methanesulfonyl fluoride even further. Machine-learning driven modeling now guides the crafting of new analogs, allowing for more predictable fine-tuning of inhibitory action. In enzyme research, this means uncovering ever finer details about protein mechanics and drug interactions. Parallel work in process chemistry continues, chasing after safer, greener, less toxic pathways for producing both the molecule and its derivatives. Published studies detail strategies for trapping or neutralizing off-gas, and collaborative groups seek catalysts that might lower energy use or reduce waste. For many groups, the biggest prize isn’t just in new discovery, but in making the whole process safer and less punishing for both workers and the environment.
Toxicity: Risks in Plain Language
Plenty of studies paint an honest picture about the risks tied to methanesulfonyl fluoride. Accidental exposure—through inhalation, ingestion, or direct contact—can hit the central nervous system, disrupt breathing, or bring on headaches and nausea long before larger troubles show. This is not a compound for careless storage or dimly lit home labs. Chronic exposure studies in animal models flag heightened risk for organ damage, especially when basic safety barriers get skipped. Risk isn’t abstract—older researchers and safety officers tell real stories about leaks, spills, and rare but severe accidents. The best solution has never been a looser hand or lowered vigilance. Instead, teams push for safety training, tough-as-nails protocols, better engineering controls, and—where possible—substitution with less hazardous alternatives.
Setting Sights on the Future
Looking beyond present day, the fate of methanesulfonyl fluoride rides on our ability to use it smartly and handle it safely. The effort to modernize and make greener synthesis pathways cuts across major research institutions. For years, calls for tighter regulation lingered, aiming to make sure use cases align with proven science and health standards. The next wave likely brings further refinements in analog development, possibly unlocking new treatments for diseases marked by enzyme dysregulation. At the same time, pressure mounts to minimize occupational risks, limit environmental release, and manage lifecycle impacts from cradle to disposal. Knowing where the compound does the most good and the most harm will guide responsible policies in science and commerce. Methanesulfonyl fluoride holds a peculiar niche—never likely to be mass market, always commanding focus on safety, and promising new science as long as we don’t cut corners on care or curiosity.
What are the primary uses of Methanesulfonyl Fluoride?
What Makes Methanesulfonyl Fluoride Useful?
Methanesulfonyl fluoride, often shortened to MSF, has built a reputation in both research labs and industry for its strong reactivity. Having worked in labs where chemical selectivity matters, I have seen firsthand how even a small mistake in handling tools like MSF can change the outcome of a project. Its power lies in the way it reacts with certain proteins and enzymes, so it earns attention from scientists hunting for new medical therapies and tools for drug discovery.
Role in Medicine and Biology
In my experience with neurobiology research, MSF shows up as a key compound for those studying the enzyme acetylcholinesterase. This enzyme helps break down neurotransmitters in our brains. By blocking or ‘inhibiting’ this enzyme, MSF helps scientists learn what happens when acetylcholine—the messenger—can’t get cleared out as quickly. This effect looks a lot like what happens in Alzheimer’s disease and other cognitive problems, so MSF serves as a way to build animal models for studying memory failure and possible treatments.
There’s genuine value in how reliably MSF can act as an inhibitor, and researchers keep reaching for it because it stands up to strong acids and heat during experiments. Unlike some biochemistry tools that lose their punch quickly, MSF sticks around long enough for reliable results. It’s easy to overlook how careful scientists must be during preparation: inhaling this stuff or letting it touch your skin is a serious danger. Special gloves, sealed hoods, and even face shields become non-negotiable in labs using it.
Chemical Synthesis and Industry
Beyond biomedicine, MSF pulls its weight in synthetic chemistry. If you ever had to build complex molecules, you’d know that specific chemical bonds are hard to make and break without messing up everything else. MSF acts as a selective ‘sulfonylating’ agent, giving chemists a way to attach the methanesulfonyl group to other compounds. Many medications, pesticides, and specialty polymers got their start from these reactions—some trace back to steps involving MSF.
I’ve watched teams choose MSF for its clean reactions. Compared to other chemicals with similar strengths, it leaves behind fewer useless leftovers. That matters both for the environment and for workers’ safety. Any reduction in hazardous waste brings sighs of relief in crowded chemical plants. Yet, MSF demands respect: even tiny spills can cause trouble, so experienced staff must watch for leaks and know how to handle exposure right away.
Potential Solutions and Forward Thinking
MSF, like many specialized chemicals, sparks strong debate around safe use and waste management. Governments and safety boards have been tightening the rules in recent years, calling for better training and monitoring wherever these kinds of reagents show up. Stronger ventilation, regular inspections, and improved waste neutralization methods have already brought down accident rates.
More research could uncover safer alternatives with similar effectiveness. For now, MSF holds a crucial spot in enzyme research and the chemical manufacturing chain. Balanced handling, increased staff awareness, and dedicated facility upgrades promise to cut down on risks without sacrificing scientific progress. As someone who’s seen the good and the bad of using MSF, I believe its careful application will keep opening new doors in both medicine and industry.
What is the chemical formula and molecular weight of Methanesulfonyl Fluoride?
Looking at the Science
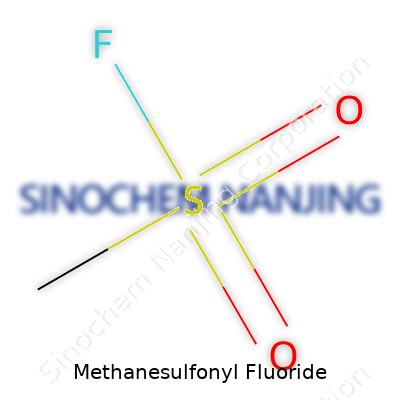
Methanesulfonyl fluoride, or MSF, carries the chemical formula CH3SO2F. The structure pulls together a methyl group, a sulfonyl group, and a fluorine atom. It counts up to a molecular weight of 114.09 g/mol. These numbers carry more meaning than simple facts on a label — the arrangement and weight affect how the molecule reacts, how it gets stored, and what safety steps folks in the lab need to take.
Real-World Impact
I’ve watched researchers handle methanesulfonyl fluoride with a level of respect that borders on caution. It’s not some curiosity buried at the back of the stockroom. This compound blocks cholinesterase, the body’s enzyme for breaking down acetylcholine. That effect holds real weight in both science and medicine, where it finds a spot as a tool for studying nerve agents or conditions involving disrupted neurotransmission. I’ve seen papers that rely on MSF to freeze brain slices in a certain biochemical state. These aren’t just theoretical uses — every decision builds on those core facts about its formula and weight.
Handling and Safety Concerns
This molecule doesn’t fit comfortably in a casual work environment. The fluorine atom can increase toxicity, and MSF itself gets classified as an organofluorine compound — a category known for toughness and persistence. Gloves and eye protection aren’t negotiable. Just reading the formula gives a clue about its strength as a sulfonyl fluoride, which lines up with its action on enzymes that use active-site serine. Forgetting this risk can lead to nerve or lung issues. Stories from seasoned chemists always come back to the residue a small spill can leave, and how rapid cleanup matters much more than with less reactive organics.
Environmental Issues
The sturdy bonds inside CH3SO2F mean it sticks around. Once released, it hardly breaks down. That persistence shapes decisions in waste management. Universities and industry both face regulations for handling effluents. Years ago, I watched an environmental safety specialist train a group on this—one errant rinse or unmarked disposal can turn a routine day into a scramble for containment. Factoring in the molecular weight, even a gram can pack a punch. The core lesson: knowing the formula isn’t academic trivia. It shapes choices from storage to long-term monitoring at disposal sites.
Paths to Safer Use and Disposal
Few chemicals with the power of methanesulfonyl fluoride make their way into research labs without a clear safety playbook. Experienced staff set up clear labeling and strict inventory controls. Fume hoods run constantly where MSF gets measured or transferred. Companies and universities often invest in specialized training for those handling agents like this. Disposal routes tie back to secure high-temperature incineration and documented transfer to certified hazardous waste facilities, making sure that CH3SO2F doesn’t become a silent contaminant in groundwater or air.
Scientific Progress Runs on Facts
The details in chemistry matter. Molecular formula and weight look simple at a glance, but they steer every part of how people use dangerous chemicals. Methanesulfonyl fluoride’s reputation for both risk and value comes directly from its structure and size. Through proper respect and constant learning, researchers and safety professionals keep its promise from turning into peril.
What safety precautions should be taken when handling Methanesulfonyl Fluoride?
Respecting the Hazards in the Lab
Methanesulfonyl fluoride isn’t a household name. In the real world of chemistry labs, folks recognize it for what it is: a potent, volatile, and toxic chemical. Having worked with organosulfur compounds for years, I can say nobody should view this reagent lightly. It serves specialized applications in chemistry—especially in making certain pharmaceuticals and chemical probes—but safety mistakes with this substance lead to real consequences.
Personal Protection Comes First
Goggles, gloves, and lab coats sound like obvious gear, but too many skip steps when in a rush. Methanesulfonyl fluoride is an irritant for both eyes and skin. Direct contact can cause burns. Splash-proof eye protection and a properly buttoned lab coat shield you against spills and unpredictable splatters. Standard nitrile gloves resist organic chemicals, but double-gloving cuts risk. Keep the skin covered, including arms, and tie back any loose hair or items that might brush over the bench.
Ventilation and Exposure Controls
Strong chemical fumes threaten not only the person at the bench but everyone in the lab. Methanesulfonyl fluoride gives off a pungent, stifling vapor that causes coughing, headaches, and worse if inhaled. Open bottles or reactions beneath a working fume hood. I’ve seen the difference a proper hood makes: even a small leak vanishes into exhaust, sparing everyone from harsh air. Never pipette by mouth—that relic from the past has no place with modern safety standards.
Handling and Storage Practices
Experienced hands never leave reactive chemicals out on a crowded bench. Clearly label every bottle, including hazard information, and keep storage closed tight when not measured out. Store this fluoride in a cool, dry spot, separated from acids, bases, or water, since accidental mixing can trigger dangerous reactions. Shortcuts, like open bottles or messy bench space, create problems. Use dedicated glassware and measure out only what you need. After finishing, double-check that containers are sealed and stashed securely.
What to Do if Something Goes Wrong
Spills or splashes mean prompt action. In my own experience, quick rinsing under the safety shower saves skin from severe burns—don’t waste time thinking about it. Eyes need a solid fifteen-minute flush at the eyewash as a minimum. Call for help; don’t try to tough it out. Update your workplace or building management about any spills, no matter how minor. For airborne exposure, evacuate the lab to fresh air right away. Keep the emergency number posted nearby and know routes to medical support.
Building a Culture of Safety
Mistakes happen, especially with new team members. Sharing proper training and posting clear safety sheets on wall boards goes a long way. Veterans in the lab who spot unsafe habits should speak up—not as scolds, but to avoid injuries that spark regret. Regular drills and refreshers make emergency steps second nature instead of guesswork. A safety-minded team and good habits turn even a risky chemical into a tool instead of a threat.
Science Demands Responsibility
Every reagent brings its own challenges, but methanesulfonyl fluoride tops the list for nastier mistakes. Taking personal safety seriously, respecting others in shared spaces, and treating procedures as non-negotiable creates healthy, productive labs. Over time, these habits build confidence and help young chemists learn from experienced hands. The respect paid to dangerous chemicals pays off with fewer accidents and smoother research. That’s what real lab experience has taught me: no shortcut beats a safety-first mindset.
How should Methanesulfonyl Fluoride be stored to ensure stability?
Why Storage Matters for Methanesulfonyl Fluoride
Methanesulfonyl fluoride stands out as a strong sulfonylating agent, but anyone who’s ever worked with it knows stability turns into a concern quickly. If you’ve ever seen a reagent degrade faster than you expected, frustration hits hard—chemical waste, lab downtime, and sometimes safety risks appear out of nowhere. Methanesulfonyl fluoride reacts swiftly with water, spilling over into hazardous byproducts or even ruining valuable batches. That’s not just wasted money; sometimes, you’re looking at extra disposal headaches, too.
Keeping Reactivity Under Control
What storage conditions steer clear of those problems? Dryness tops the list—any persistent trace of humidity can eat away at your methanesulfonyl fluoride over time. Use a tightly sealed glass bottle, ideally with a PTFE-lined cap, and double-check seals after each use. Toss in some silica gel or another desiccant inside the storage container for a bit of insurance. I’ve seen labs skip that step and end up regretting it—reagent turns yellow, or the whole bottle just off-gases before its time.
Room temperature sometimes sounds like a fine idea, but a cool environment outperforms. Chemical suppliers recommend refrigeration, usually at 2–8°C. A dedicated chemical refrigerator, not the same one as your lunch, keeps temperature swings from introducing condensation. That one trick alone can add months of shelf life. It’s not just about chemistry, either—cold slows down everything, and that low-energy environment helps methanesulfonyl fluoride sit tight.
Watch Out for Water and Air
Contact with moisture means trouble, and air isn’t far behind. Methanesulfonyl fluoride doesn’t spark up with oxygen, but indirect exposure through atmospheric water vapor can cause slow hydrolysis. Even flipping a bottle top open for just a few seconds lets a bit of moist air in. I stick to the glovebox or dry nitrogen atmosphere whenever possible, especially when transferring larger volumes or drawing multiple samples from the same stock container.
Plastics sometimes look tempting for storage, especially because they’re light and cheap. Methanesulfonyl fluoride does nasty things to some polymers, especially if small molecules seep out and start corroding the container in slow motion. Glass shows more reliability. For labs that use small amounts occasionally, storing aliquots in ampoules, flame-sealed under inert gas, offers higher confidence. That extra step might sound like hassle, but anyone who’s lost a full bottle knows peace of mind comes from a bit more preparation.
Training and Labeling Build Safety
Anyone handling methanesulfonyl fluoride should understand what the chemical can do if mishandled. I’ve had younger colleagues treat every lab bottle like sugar water—proper orientation comes from mentoring as much as manuals. Labels must clearly state contents, date of receipt, and hazard information. Safety data sheets stay nearby for emergencies. Check expiry dates regularly, because sometimes chemicals look fine but deliver poor results as they break down.
The reality is that good chemical storage never happens by accident. Facilities that standardize storage procedures and invest in training avoid most surprises. Small investments in dry storage, regular checks, and clear labeling pay off long term. Keeping methanesulfonyl fluoride stable isn’t just a technical box to tick—it protects people and budgets, all while supporting good science.
What are the typical packaging options for Methanesulfonyl Fluoride?
Understanding the Real Risks
Methanesulfonyl fluoride walks into the lab looking plain but packs a punch. The first thing people mention about this chemical isn’t what it can do, but what it can do to you if you drop your guard. Highly toxic and reactive, one careless spill spells trouble. So, anyone moving or handling this material cares about packaging, maybe even more than the chemistry itself.
Why Bulk or Drums Aren’t Always the Right Fit
Picture a warehouse stacked with huge drums. Great for solvents and oils—terrible for something like methanesulfonyl fluoride. This compound doesn’t play nice with moisture or open air. It goes off quickly, releasing gases you do not want to breathe in. That’s why suppliers lean toward tough, tight-sealing containers that won’t corrode and won’t let the fumy contents leach out. A lot of folks reading safety data sheets early on in their careers learn this lesson before they even see a bottle in person.
Glass Bottles: The Small Batch Workhorse
In most research labs, you’ll find small amber or clear glass bottles—usually between 25 milliliters and 500 milliliters. Sealed under inert gas, each bottle comes with a PTFE-lined (polytetrafluoroethylene) cap and heat-shrunk security band. Glass resists attack, and labels stick well even through cold storage or the panic of a small-scale fume hood cleanup. One advantage for glass: seeing any clouding right away.
Specialty Metal and Composite Containers
Scale up past a few dozen grams, and things shift. Stainless steel or nickel-lined small cans, sometimes called “Sample Cans,” hit the supply list. Manufacturers include tamper-evident seals and pressure relief systems, knowing customers don’t appreciate surprises. Composite materials, like fiber drums with lined interiors, show up for larger repeat customers—though everyone agrees nothing beats a sealed metal can if you value sleep at night.
Pain Points in Storage and Shipping
Losses from leaks or cracked containers get expensive in dollars and reputation—ask anyone whose order arrived with a whiff of something sharp in the box. The push toward DOT and IATA-certified packages isn’t just red tape. Shippers have to meet strict rules about absorbent cushioning, dual containment, and chemical compatibility. Out in the wild, different climate zones push bottles and steel cans in ways lab techs never see behind four walls and fluorescent lights.
Solutions on the Horizon
Some packagers look for greener, safer plastics but keep hitting the same two walls: chemical resistance and keeping the closure actually tight. There’s still faith in classic glass, especially paired with modern cap liners. Smart labeling, including QR codes for instant access to safety data, is another practical fix. Simple upgrades, but they earn their keep by lowering the risk of mistakes in the warehouse or at the receiving end.
Making Choices with Safety in Mind
My own days in shared storage rooms taught me this: packaging isn’t an afterthought. Every container speaks to lessons learned by people who handled tough stuff before us. As new solutions roll out, the focus stays the same—stop leaks, crush accidents before they start, and treat every bottle like there’s a team on the other side counting on you.
| Names | |
| Preferred IUPAC name | Methanesulfonyl fluoride |
| Other names |
MSF Methanesulphonyl fluoride Methylsulfonyl fluoride Sulfuryl fluoride, methyl- Methanesulfonofluoridic acid |
| Pronunciation | /ˌmɛθ.eɪn.sʌlˈfoʊ.nɪl ˈflʊr.aɪd/ |
| Identifiers | |
| CAS Number | 558-25-8 |
| Beilstein Reference | 1209240 |
| ChEBI | CHEBI:41446 |
| ChEMBL | CHEMBL2205681 |
| ChemSpider | 157378 |
| DrugBank | DB02137 |
| ECHA InfoCard | 100.012.232 |
| EC Number | 220-864-4 |
| Gmelin Reference | 67687 |
| KEGG | C14318 |
| MeSH | D008746 |
| PubChem CID | 70196 |
| RTECS number | WS6475000 |
| UNII | 66X61L4G9P |
| UN number | UN3272 |
| Properties | |
| Chemical formula | CH3SO2F |
| Molar mass | 102.09 g/mol |
| Appearance | Colorless liquid |
| Odor | Pungent |
| Density | 1.422 g/cm³ |
| Solubility in water | soluble |
| log P | -0.19 |
| Vapor pressure | 21.6 hPa (20 °C) |
| Acidity (pKa) | Acidity (pKa) of Methanesulfonyl Fluoride: -1.5 |
| Basicity (pKb) | −6.5 |
| Magnetic susceptibility (χ) | -44.0 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.335 |
| Viscosity | 0.85 cP (20°C) |
| Dipole moment | 1.95 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 89.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -566.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -867.5 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | N05CM21 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled. Causes severe skin burns and eye damage. |
| GHS labelling | GHS02, GHS05, GHS06 |
| Pictograms | GHS06,GHS05 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P305+P351+P338, P304+P340, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-1-W |
| Flash point | 80 °C |
| Autoignition temperature | 213 °C |
| Explosive limits | 6–21% |
| Lethal dose or concentration | LD50 Oral Rat 200 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 210 mg/kg |
| NIOSH | JN9275000 |
| REL (Recommended) | 0.01 mg/m³ |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Methanesulfonyl chloride Methanesulfonic acid Trifluoromethanesulfonyl fluoride |

