Mercury Thiocyanate: A Chemical With a Storied Past and a Cautionary Present
Historical Development
Mercury thiocyanate has left a curious mark on the history of chemical experiments and scientific spectacle. Dating back to the nineteenth century, chemists discovered this crystalline powder and quickly found out just how dramatic its decomposition could be. Classic chemical demonstrations in schools and early public exhibitions have relied on it for its “Pharaoh’s Serpent” effect—a small pile of powder that, when ignited, erupts into long, sinuous ashes that twist and grow before the audience’s eyes. The fascination with mercury thiocyanate’s behavior reflects a broader human urge to probe and play with the secrets of matter, sometimes to our peril. Even in books from the Victorian era, there’s mention of this peculiar chemical, with warnings about its poisonous nature running alongside instructions for backyard stunts. Long before terms such as ‘haz-mat’ or ‘occupational health’ existed, mercury thiocyanate was awakening folks to the thrills and risks lurking in all corners of chemistry.
Product Overview
This compound stands out for its singular role as a curiosity and teaching aid. Sold as off-white to yellowish crystals, mercury thiocyanate doesn’t see much use in large-scale industry or consumer goods. Its main draw has always been spectacle—part firework, part warning—demonstrating chemical transformation in ways few other substances can match. Kids and adults alike have gawked at the “snakes” this powder creates, while educators highlight its properties to students who are just starting their journey with science. But over the decades, growing awareness of mercury’s impact on health and the environment sent this compound to the back shelf in many classrooms and public spaces. The change reflects wider shifts in our relationship to risk, especially with chemicals that embed themselves in air, water, and our own bodies.
Physical & Chemical Properties
Handling mercury thiocyanate always brings a sense of wariness. It looks fairly nondescript—no telltale color, no strong odor—but its chemical makeup demands respect. In solid form, the powder remains stable under normal storage, but the fun starts once it meets flame. Upon gentle ignition, it breaks down, producing a tangled mess of black and gray ash shaped into snakes many times its original size. The reaction unleashes a cocktail of byproducts, including carbon disulfide and the highly toxic mercury vapor. The transformation is dazzling but short-lived, leaving behind material you don’t want on your hands, clothes, or workspace. As with most mercury compounds, the risk is not always about the moment but the aftermath; residues stick around, potentially contaminating surfaces and presenting an exposure route long after the main event.
Technical Specifications & Labeling
Official guidance on labeling and handling has grown much stricter in recent times. Regulations make plain that mercury thiocyanate belongs in the category of substances that warrant not just gloves and eye protection, but a thoughtful approach to storage and use. Accurate labeling means flagging its toxic nature, unimpressive appearance notwithstanding. There’s more to a chemical label than a name or formula—hazard statements warn of its dangers to kidneys, the nervous system, and anyone downwind of where it gets burned. Safety data emphasizes both personal protection and broader environmental stewardship. Chemists who once could buy this substance from mail-order catalogues without much fuss now face a complex array of rules designed to limit accidents and pollution. The paradigm shifted, valuing safety over spectacle.
Preparation Method
Production of mercury thiocyanate relies on combining mercury salts like mercuric nitrate with an alkali metal thiocyanate, such as potassium thiocyanate, in an aqueous solution. The reaction yields a precipitate that gets washed and dried to form the final product. In my own experience, the steps may sound almost routine in the hands of a trained chemist, but the reality holds more risk and mess than most modern labs are willing to shoulder anymore. There’s a lingering smell of sulfur and an edge of danger whenever elemental mercury comes out, even in sealed flasks. Not long ago, the ease of making this substance tempted more experimenters into the act, overlooking the long-term threats that traces of mercury leave behind. These days, labs view such procedures through a stricter lens, outweighing the chemistry with strong checks and airtight safety measures.
Chemical Reactions & Modifications
While most people associate mercury thiocyanate only with its fiery decomposition, its chemistry runs deeper than a party trick. It acts as a weak acid and can react with strong bases or other mercury compounds. On ignition, complex chemical changes unravel the bonds, pumping out mercury vapor, carbon disulfide, nitrogen gases, and a tangled polymeric ash. Modifications center on limiting danger—proposing buffered mixtures that reduce toxic vapor output or experimenting with alternative “serpent” demonstrations using less hazardous ingredients. Progress in chemical safety means seeking out substitutes, not just tinkering at the edges. Whenever someone asks for a more impressive demonstration, the conversation circles back to the unseen costs these reactions bring to people and the world outside the lab window.
Synonyms & Product Names
Over time, mercury thiocyanate picked up an assortment of formal and informal titles: mercuric thiocyanate, mercury(II) thiocyanate, and even “Pharaoh’s Snake powder” in some old retailers’ catalogs. From a chemical standpoint, the synonyms make tracking regulations and historical usage more complicated. In the tangled world of science writing and supply chains, those alternative names sometimes mask hazards and confuse buyers, especially in a market that spans languages and continents. The lesson carries weight: whether reading an old recipe or a modern safety sheet, names matter, and their confusion can carry real consequences.
Safety & Operational Standards
The rise of occupational health has shaped not just what substances show up in science classrooms but how professionals prioritize risk. Mercury thiocyanate basically serves as a case study in why chemical showmanship now comes with strict ground rules. Regulators in many countries demand locked storage, use of fume hoods, detailed logs, and formal training. Cleaning up spills means not just sweeping up and tossing powder in the trash but adhering to hazardous waste disposal procedures. The rules sometimes receive pushback for appearing too stringent, yet my own experiences suggest there’s no such thing as overkill with mercury compounds. Stories abound of lingering contamination in old classrooms, accidental poisonings, and long-term health effects that follow people out the door. Building real expertise means learning from hard lessons and valuing protective measures no matter how inconvenient.
Application Area
Once upon a time, mercury thiocyanate earned its keep in chemistry demonstrations and a scattering of old-school fireworks. Its practical applications outside these narrow confines have always been limited. Industrial chemists have largely left it behind, wary of cleanup headaches and regulatory crosshairs. The rare appearance of this compound in specialized testing or analytical procedures now comes with extensive safeguards. The public taste for chemical wizardry shifted toward safer alternatives and computer simulations that offer wonder without the risk. These shifts do not mark a loss so much as a sign of growing knowledge—replacing spectacle with models and methods that spare viewers from invisible harms. Even so, the memory of its wonders lingers in documentaries, science fairs, and the recollections of teachers who once watched ash snakes rise from modest piles of powder.
Research & Development
Funding and attention for mercury thiocyanate now flows mostly to studying environmental fate and detoxification rather than new uses. Old patents and articles describe efforts to stabilize the compound, reduce its runoff, or analyze its absorption in soils and water. Researchers looking to trace mercury pollution sometimes use it as a kind of surrogate or model for tracking where and how the element travels in the environment. More often today, energy goes to finding or developing alternatives for chemical demonstrations—new materials that can dazzle students without the shadow of toxicity. The measurable progress here doesn’t feel dramatic at first, but it keeps labs and classrooms safer while still honoring curiosity. In my own reading, the best research asks not just how to handle mercury thiocyanate with extra care, but what can replace it altogether.
Toxicity Research
Few chemicals receive as much scrutiny for long-term effects as compounds containing mercury. Toxicologists track how mercury thiocyanate’s decomposition releases vapors that find their way into lungs, settle on furniture, and persist in dust. The body doesn’t easily rid itself of mercury, and chronic exposure—especially for children—links to neurological damage, developmental problems, and a list of other health nightmares too grim to ignore. Safety agencies worldwide classify it among the most hazardous substances still found in labs. Even decades-old exposure can emerge in blood tests of people who had early, casual contact. Researchers expand this knowledge, not just by tracing acute poisoning, but by mapping all the slow, subtle ways mercury seeps from places we once thought safe into the lives of people who never touched the stuff. It’s a stark reminder that chemical progress sometimes comes with a bill due generations later.
Future Prospects
The future of mercury thiocyanate seems to trend toward the margins. More schools swap out dangerous science demonstrations for multimedia, simulations, or safe chemical stand-ins. Environmental policies keep pushing for phased bans on mercury compounds, denying them not just in classroom kits, but in consumer products and manufacturing. There will always be a handful of researchers who need to wrangle with historical materials or investigate environmental cleanups, but the everyday use case is fading fast. Some museums still display the snakes produced by this reaction behind sealed glass—a relic of times when the line between learning and risk felt thinner. The story of mercury thiocyanate matters not for what we can do with it today, but for what it teaches about progress, responsibility, and all the invisible work it takes to keep curiosity from tripping over its own shoelaces.
What is Mercury Thiocyanate used for?
An Unusual Chemical with a Fiery Reputation
Most people only hear about mercury thiocyanate in the context of old science kits or those mesmerizing YouTube videos where it twists and grows into smoky serpents when set on fire. That "Pharaoh’s serpent" trick became a staple in science demonstrations because of how strangely beautiful and alarming it looks as the chemical contorts itself out of a tiny white pile into a snaking, smoldering column. I caught my first glimpse of it during a high school chemistry class, feeling a mix of awe and mild concern as my teacher rushed to open the windows.
From Fireworks to Early Photography
People used mercury thiocyanate far beyond parlor tricks. Back in the nineteenth and twentieth centuries, this compound served as a crucial piece of the puzzle in fireworks displays and photographic development. Its most dramatic use stayed in the world of pyrotechnics. Chemists blended it with oxidizers to help create unique effects, such as the expanding "serpent" or "dragon" shapes that entertain onlookers during festivals. This effect drew on the chemical’s decomposition; when heated, it quickly turns into mercury vapor, carbon nitride, and a few other byproducts, leading to those strange, wormlike shapes.
Early photographers also experimented with chemicals like mercury thiocyanate. Here, it played a less glorious role, helping sensitize plates in processes like wet collodion photography. Modern digital sensors have pushed those chemical methods aside for safety and precision, but it’s striking to see just how experimental people got with the materials they could get their hands on. Mercury thiocyanate cropped up in analytical chemistry, too, used in certain color tests to identify or measure other substances.
The Heavyweight Downside: Toxicity Stakes
Any story about mercury-based compounds calls for honesty about safety. My teacher wasn’t reaching for those open windows because mercury thiocyanate sets off a simple smoke alarm—the stuff is downright hazardous. Decomposition creates not only mercury vapor, which directly affects the nervous system, but also cyanide compounds. Accidentally breathing in even a little can create lasting health problems. Today’s safety regulations, grounded in years of experience and tough lessons, have basically pushed this chemical out of classrooms and public science shows. The European Union and the United States both restrict its sale and use, except under strictly controlled scientific conditions.
Lessons in Legacy: Safer Science for All
Growing up, kids marveled at those mesmerizing "serpent" effects and occasionally tried to replicate them at home, learning some hard lessons about why safety goggles and supervision aren’t just bureaucratic red tape. Cautionary examples like mercury thiocyanate have helped shape stricter laboratory training, teacher education, and regulation. Countries with strong science education systems now teach budding experimenters to appreciate the beauty of science without risking health.
What Moves Us Forward
Modern chemistry has plenty of ways to inspire awe without putting anyone in harm’s way. Technology and rigorous safety rules give teachers safer alternatives to demonstrate chemical change. Substances with much lower toxicity can show similar expanding effects, letting excitement for science bloom without risking irreversible harm. This shift shows the best kind of progress—using our knowledge not just to dazzle, but to protect. Mercury thiocyanate holds a place in the chemical hall of fame as a reminder that scientific wonder should travel hand-in-hand with responsibility.
Is Mercury Thiocyanate hazardous to health?
Understanding the Risks
Mercury thiocyanate grabs attention in chemistry classrooms and online science demonstrations. The "Pharaoh’s Serpent" effect—fiery, twisting columns that seem to come alive—makes a great show. Dig beneath the spectacle and the real story begins. Mercury thiocyanate contains mercury, a well-known toxic element. Inhaling, ingesting, or even touching it without protection introduces direct health dangers. Mercury’s reputation for causing nerve damage, kidney problems, and developmental delays in children didn’t come from rumors. It came from years of people getting sick, lab mishaps, and health studies tracing mercury poisoning to surprising sources.
Direct Exposure Dangers
Handling the powder or watching its fiery reaction carries risks even in a controlled setting. Lighting mercury thiocyanate releases mercury vapor and other poisonous chemicals, including carbon disulfide and sulfur dioxide. Breathing these in, even briefly, can set off headaches, breathing trouble, and nausea. Stories exist of students and hobbyists working in rooms that seemed safe, only to wind up with symptoms that sent them to the emergency room. I remember a college chemistry lab where strict gloves-and-goggles rules sounded dramatic until someone spilled a small vial of mercury compound. Getting a safety lecture didn’t seem so boring after watching the clean-up crew suit up and deal with everything.
The Long-Term Effects
Short-term symptoms—nausea, headaches, lightheadedness—pass with fresh air and time. Mercury lingers far longer. It settles in soft tissues and nerves and takes years to leave the body. Long-term mercury exposure links to tremors, memory problems, and mood swings. Children get hit hardest; brains still growing are far more vulnerable. The CDC reminds us that no safe level of mercury exposure exists for developing minds. That message always lands with weight for parents and teachers, since curiosity often pushes kids closest to old chemistry tricks like the Pharaoh’s Serpent.
Environmental Hazards
Mercury compounds didn’t just trouble classrooms. Dumping leftover mercury thiocyanate, even just a small spill, spreads contaminants into soil and water. Mercury persists in the environment. Whenever it enters the food chain, it rises up into larger fish and then into people who eat them. The EPA lists mercury as a priority pollutant for exactly this reason. A memory comes back: a science fair had to cancel its fireworks demonstration years ago when state officials realized the soil around the demo area tested high for mercury contamination—three years after the event. The cleanup took the better part of a summer.
Safety Steps and Alternatives
Modern chemistry sets leave out mercury compounds for these reasons. School safety guidelines prohibit use in student experiments. Teachers and professionals reach for safer alternatives. For those running demonstrations, keeping reactions in fume hoods, wearing gloves, and using only tiny amounts became the minimum standard. Public education campaigns remind hobbyists: if you find a bottle of this chemical in an old kit, it doesn’t belong in the trash or down the drain. Local hazardous materials programs know how to handle these relics safely.
The decorative, mesmerizing effect of mercury thiocyanate reactions belongs in history books and guarded museums, not classrooms and YouTube channels. Health isn’t worth risking for a cool-looking snake of foam.
How should Mercury Thiocyanate be stored?
The Real Hazards Lurk in the Details
Mercury thiocyanate's claim to fame usually sits with “Pharaoh's Serpent” science tricks, but there’s a darker reality to this substance. Both mercury and cyanide spell trouble for anyone who skips over safety basics. One whiff of its decomposition fumes can ruin health, especially when heat or moisture let the chemical break down. Most folks, including experienced people in schools or labs, don't weigh this risk heavily enough until they see the results up close. I worked with a high school science teacher once who brooded over forgotten bottles in a back cabinet—those orange stains from leaky containers served as a stark warning. That lesson never left me.
Bad Storage Choices Lead to Pricey Cleanups
Poor storage puts more than just people at risk. Mercury vapor and thiocyanate dust linger in unexpected places—HVAC ducts, ceiling tiles, even the inside of cardboard boxes. Many facilities spend thousands decontaminating entire rooms, all because the original container cracked or moisture seeped in. You don’t just face a lab shutdown, but also the possibility of expensive hazardous waste removal and red tape with environmental agencies. These headaches dwarf the simple effort of storing chemicals carefully from day one.
Smart Storage Starts with Containment
Locking up hazardous powders doesn’t call for high-tech equipment, but it always demands a solid approach. Airtight, chemical-resistant containers—think glass or sturdy polyethylene with fitted lids—make a world of difference. Never trust the bag or bottle Mercury thiocyanate ships in; double containment cuts the risk of spills or cross-contamination. Once, a university saved thousands just by switching weak snap lids for screw-cap jars inside steel bins. There’s no room to compromise here—moisture and vapors move faster than people notice.
Keep Chemicals Where Heat Stays Away
Even a small mistake, like sitting Mercury thiocyanate near a sunny window or in a room with temperature swings, invites disaster. Heat speeds up decomposition. Warmth makes the stuff break down and release mercury vapor—no alarm, no warning smell. My own close call happened during a summer storage room audit: a jar sweating inside a metal locker, right under a leaky roof vent. That batch didn’t just ruin shelving; it triggered a building evacuation. Only storage in cool, dry spots gives real peace of mind.
Access and Accountability Trump Lock-and-Forget
Many let hazardous samples gather dust in dark cabinets, with labels faded and inventory lists forgotten. One solid rule: keep a log, check on old stocks, and never let unknowns pile up. Only trained folks should handle or even touch Mercury thiocyanate. Secure shelving, locked cabinets, and keys limited to qualified staff will help prevent both mishandling and theft. A monthly review habit keeps surprises to a minimum, and regular reminders make rules stick for good.
Clear Rules Don’t Ask for Perfection—Just Consistency
Mercury thiocyanate brings enough risk without extra complications from careless storage. Routine acts matter: dry, cool shelving, airtight containers, logged access, and clear labeling. Labs and schools fight fewer fires—figurative and literal—when everyone treats old chemicals like their health really depends on it. Safety culture isn’t a slogan here—it’s the only way to make sure the past doesn’t haunt the next audit or, worse, the next generation.
What happens when Mercury Thiocyanate is burned?
The Allure of the Pharaoh’s Serpent
Lighting a pile of mercury thiocyanate brings out a scene that sticks with anyone who values science and spectacle. Flames lick the small, white pile, then a writhing black mass pushes out, twisting and growing like a slow-motion monster. The reaction isn’t just for show; it has fascinated people for well over a century. Folks in chemistry labs and backyard experimenters see those sulfurous tentacles and want to know more: what drives this transformation, and why does it draw such mixed reactions among scientists and regulators alike?
Chemistry at Work
The stuff on display is called the Pharaoh’s Serpent. When burned, mercury thiocyanate produces mercury vapor, carbon nitride, and a handful of other byproducts, along with that growing black “snake.” The reaction splits the original compound into smaller pieces, recombining them into dramatic forms and releasing energy and gases. It’s easy to see why people want to show it off at science fairs—combining chemistry, art, and performance creates a sense of wonder.
The Human Side of Curiosity
Hands-on experience often drives home what a textbook can’t. During high school, I saw this reaction up close with my science club. We gathered around our teacher’s demonstration, not really grasping the dangers in front of us. Later, I learned about mercury vapor and cyanide compounds—how even a single unsafe inhalation can threaten your nervous system or leave lasting harm. Seeing our teacher handle the powder with heavy gloves and a fume hood, then immediately removing it afterward, made a strong impression.
The Danger Behind the Drama
The risks go far beyond temporary discomfort. Mercury compounds have poisoned people through careless handling. The World Health Organization calls mercury a global threat, highlighting its links to developmental damage and kidney failure. Mercury vapor, invisible and without real odor, travels quickly in the air and gets into the bloodstream fast. Thiocyanate byproducts bring their own risks, irritating the eyes and lungs, and in large doses can attack the thyroid.
Regulations restrict public sale of mercury thiocyanate in many countries. Lab supply firms call for protective equipment, ventilation, and solid waste disposal plans. The popular “Snake Kit” from long ago disappeared for good reason—schools and homes became safer the day those boxes vanished from store shelves.
Finding Safer Paths
The urge to see real chemical reactions doesn’t need to clash with safety. Modern educators teach science using safer compounds that mimic dramatic effects without risking mercury poisoning. Those foam “elephant toothpaste” reactions and simple baking soda-vinegar volcanoes deliver fun and learning together, without back-end health risks. The memorable lessons come from clear eyes and healthy lungs, not from weeks spent dealing with headaches or trips to the doctor.
Responsible Curiosity Helps Us All
Questions and curiosity drive us. Science grows stronger when people ask why and how. There’s value in knowing and respecting what chemicals can do, both good and bad. When thinking about burning mercury thiocyanate, I remember that the price of a fantastic show sometimes runs higher than anyone expects. My experiences remind me to dig deeper, talk to experts, and never rush into experiments that promise more risk than reward. The real magic lies in keeping the wonder alive—and everybody safe enough to enjoy it again tomorrow.
Are there safer alternatives to Mercury Thiocyanate for experiments?
The Classic ‘Pharaoh’s Serpent’ and Its Risks
Watching the ‘Pharaoh’s Serpent’ reaction live feels almost magical. Solid mercury thiocyanate, heated, bursts to life with towering, writhing pillars of ash snaking out of nowhere. Many educators and hobbyists remember this trick from science fairs or lab days. But beneath the spectacle sits a major health concern: mercury poisoning. Long after the reaction, toxic vapors linger in the air, and the aftermath leaves hazardous waste behind. The fallout isn’t limited to a stained beaker. It touches everyone in the room, long after the oohs and ahhs die down.
Studying Science Without Health Hazards
I’ve helped organize hands-on science demos for neighborhood kids. Parents get nervous about lab materials, and rightfully so. Mercury’s toxic legacy raises alarms even decades after it stopped showing up in thermometers. Tiny mistakes—spilled powder, inhaled fumes—can lead to lifelong issues. There’s no good reason to risk it, especially with young learners. We’ve seen the hazards add up most in places with poor ventilation or loose safety oversight. Safety data from agencies like the CDC show clear links between mercury exposure and chronic neurological issues in children. Even adults can end up with health problems that disrupt careers and families.
Better Choices for Curious Minds
Fortunately, chemistry isn’t short on dramatic, safe reactions. Sodium bicarbonate and acetic acid never fail to impress; vinegar and baking soda can launch a ‘volcano’ or fill balloons with carbon dioxide. For a modern twist on chemical snakes, burning sugar with a little baking soda forms impressive, non-toxic carbon foam. The cleanup stays simple, with only some harmless residue left behind. Teachers at public schools and community centers have used this experiment as a direct substitute, reporting fewer safety incidents and cleanup headaches.
Chemists looking for vivid colors or impressive firework-style effects turn to compounds like ammonium dichromate—though it’s not perfect and carries its risks—or potassium nitrate with sugar for smoke bombs. Both hold their own hazards, but neither matches mercury’s legacy of environmental devastation. What matters is clear communication. Before starting any demonstration, science leaders must check chemical safety sheets and prepare plans for every scenario, from spills to allergic reactions. In my own lessons, students always get gloves and goggles, but they also get the story behind choice of chemicals—why we avoid one, why we choose another.
Making Informed Choices and Building Trust
Classrooms, after-school programs, and home kits shape how kids see science. Trusting the adults sharing knowledge comes from small details: What checks went in before the demo started? What happens if something spills? Local science teachers often swap safety tips and favorite alternatives, building a living library of safe, effective experiments. Organizations like the American Chemical Society regularly update guidelines to reflect the latest research—aligning with E-E-A-T standards, prioritizing real experience and up-to-date evidence over tradition.
Bringing Excitement, Not Danger, Into Science Education
Chemistry should inspire, not threaten. Even the most dramatic effect means little if it brings long-term harm. Competent science leaders keep up with alternatives, discuss their thinking with students, and help the next generation associate curiosity with care. Safe chemistry opens doors for everyone, turning risky displays of the past into memorable, risk-free adventures.
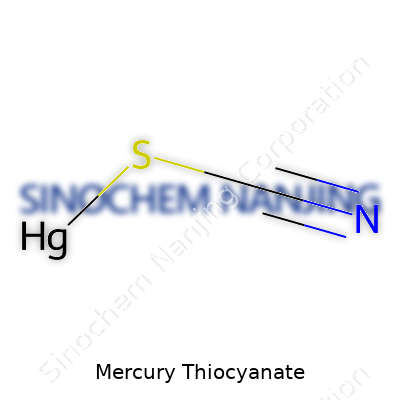
| Names | |
| Preferred IUPAC name | mercury(II) thiocyanate |
| Other names |
Mercuric thiocyanate Mercury(II) thiocyanate Hg(SCN)₂ |
| Pronunciation | /ˈmɜːrkjʊri θaɪ.oʊˈkaɪəneɪt/ |
| Identifiers | |
| CAS Number | 592-85-8 |
| Beilstein Reference | 14611 |
| ChEBI | CHEBI:39051 |
| ChEMBL | CHEMBL1241713 |
| ChemSpider | 76897 |
| DrugBank | DB13155 |
| ECHA InfoCard | 100.962.595 |
| EC Number | 205-725-8 |
| Gmelin Reference | Gmelin Reference: 20967 |
| KEGG | C01896 |
| MeSH | D008629 |
| PubChem CID | 10113 |
| RTECS number | WM4900000 |
| UNII | LQ8296L77S |
| UN number | UN1587 |
| Properties | |
| Chemical formula | Hg(SCN)₂ |
| Molar mass | 252.73 g/mol |
| Appearance | White to gray solid |
| Odor | odorless |
| Density | 3.271 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -0.549 |
| Vapor pressure | Negligible |
| Basicity (pKb) | -4.38 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.709 |
| Viscosity | Viscous solid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 143.6 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -116.4 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -173 kJ/mol |
| Pharmacology | |
| ATC code | V09CX02 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H302: Harmful if swallowed. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P260, P262, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P310, P314, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Autoignition temperature | 210 °C (410 °F; 483 K) |
| Lethal dose or concentration | LD50 (oral, rat): 60 mg/kg |
| LD50 (median dose) | 46 mg/kg (oral, rat) |
| NIOSH | SN1225000 |
| PEL (Permissible) | 0.1 mg/m³ |
| REL (Recommended) | Not recommended |
| IDLH (Immediate danger) | IDLH: 5 mg/m³ |
| Related compounds | |
| Related compounds |
Calcium thiocyanate Barium thiocyanate Mercury(II) cyanide Mercury(II) chloride |

