Mercury Salicylate: History, Properties, Uses, and Prospects
Historical Development
Mercury salicylate stands out as a compound with a long and checkered history, reaching back to the early 20th century. Chemists at the time sought ways to combine organic acids with heavy metals, hunting for new drugs and preservatives. Salicylic acid had already gained fame for pain relief and anti-inflammatory properties, while mercury compounds attracted notice for their broad-spectrum antimicrobial effects. Mixing the two built a chemical with both medicinal potential and real risk. Doctors prescribed mercury salicylate decades ago for conditions like rheumatism and syphilis before the scope of mercury toxicity became fully known. The development of antibiotics gradually edged mercury salicylate out of mainstream medicine. Still, its story reflects society’s ongoing efforts to balance medical innovation with hard-earned lessons around safety and unintended consequences.
Product Overview
Mercury salicylate, often labeled as mercuric salicylate in scientific circles, formed from the reaction between mercuric oxide and salicylic acid. The resulting compound: a white or faintly yellow powder. Historically, it was sold in crystalline or powdered form in sealed glass bottles to limit degradation from air, light, and moisture. Pharmaceutical catalogs from the 1930s and 1940s often listed mercury salicylate among their medicinal compounds, touting its supposed benefits for arthritis and skin infections. The compound also appeared in microbiology labs as a preservative or reagent, prized for its longevity and effectiveness in preventing bacterial contamination.
Physical & Chemical Properties
Mercury salicylate carries the structural formula C7H5HgO3. It registers a relatively high melting point above 200°C and does not dissolve easily in water, but organic solvents like ethanol or chloroform break it down efficiently. Its chemical stability results from the strength of the bond between mercury and salicylate ions. Given mercury’s density and toxic nature, the powder feels heavy in the hand and gives off a faint, almost metallic scent. Direct handling without protection leaves a bitter metallic taste and shouldn’t be attempted; mercury residues can easily pass through skin or enter the respiratory system.
Technical Specifications & Labeling
Chemical suppliers label mercury salicylate with exhaustive hazard warnings, including skull and crossbones symbols and urgent phrases regarding potential for acute toxicity through inhalation, ingestion, or skin contact. Detailed sheets list exact mercury content (nearly 45% by weight), melting temperature, and recommended storage conditions: cool, dry, protected from light. Samples destined for research or chemical synthesis bear precise batch numbers and certificate-of-analysis documentation, so researchers can trace their origins. All shipments require robust containment to prevent contaminating the environment and exposing handlers.
Preparation Method
Laboratories prepare mercury salicylate by gradually adding a cold solution of mercuric chloride or mercuric oxide to a stirred solution of salicylic acid in ethanol. The mixture reacts, and the insoluble mercury salicylate separates as a precipitate. Technicians filter, wash, and dry the residue before grinding it into a fine powder. Precision matters; incorrect stoichiometry or contamination leads to impure or unstable product. Handling the precursor chemicals—both toxic in their own right—requires well-maintained fume hoods, gloves, and strict adherence to chemical hygiene standards.
Chemical Reactions & Modifications
Mercury salicylate reacts with strong acids to regenerate salicylic acid and soluble mercury salts, many of which are even more toxic. Heat or exposure to ultraviolet light gradually decomposes the compound, liberating mercury vapor and other harmful byproducts. Researchers sometimes work to create derivatives of mercury salicylate by altering or substituting the organic acid portion. Such modifications aim to tune the antimicrobial effectiveness or reduce toxicity, but the challenge remains: replacing a heavy metal core rarely dodges the central problem of mercury’s biological reactivity.
Synonyms & Product Names
Through the years, this compound has picked up a variety of names: mercuric salicylate, mercury(II) salicylate, C.I. 76210, and mercurius salicylas in older pharmaceutical texts. Each label reflects a particular era of chemical nomenclature or a particular field. A few pharmaceutical products in the mid-20th century combined mercury salicylate with other ingredients, sold under proprietary names, most of which have long disappeared due to regulatory bans and liability concerns.
Safety & Operational Standards
Dealing with mercury salicylate requires deep respect for its hazards. Mercury compounds cross the blood-brain barrier and accumulate in organs, causing damage that scientists have documented in case after case: tremors, memory loss, kidney failure, birth defects. Lab workers learn to work with sealed containers and handle spills with specialized mercury “sponges” or powders that bind the element. Medical emergencies after mercury salicylate exposure demand chelation therapy, strict monitoring, and a swift switch away from exposure sources. Detailed safety protocols fill training manuals, and disposal routes lead only to licensed hazardous waste facilities—not the ordinary trash or drain.
Application Area
Mercury salicylate’s direct use in medicine faded out after safer antibiotics and anti-inflammatory agents came along. Its legacy lingers mostly in reference standards and chemical research. Some legacy industrial products—niche leather treatments or antifouling coatings—once included mercury salicylate to fight microbial growth. Modern environmental laws in Europe, North America, and other regions ban these uses outright. Most current application stories involve academic chemistry labs probing heavy metal-organic interactions or exploring new ways of tracking mercury contamination in natural systems.
Research & Development
Researchers occasionally revisit mercury salicylate and similar compounds, not for fresh applications but for the lessons they offer in toxicology, drug development, and materials science. Studies might focus on how different organic ligands change the mobility and environmental persistence of mercury. Academic groups sometimes explore ways to break down or neutralize mercury-organic complexes safely. Newer methods in analytical chemistry—and especially in environmental monitoring—use mercury-organic standards to calibrate instruments and validate protocols for detecting trace contamination.
Toxicity Research
Toxicity data for mercury salicylate emerged from both animal experiments and hard-earned clinical experience. Early reports noted acute kidney and brain damage following ingestion, as well as chronic effects from repeated low-level exposure. Researchers have shown that mercury salicylate breaks down in the body to release inorganic mercury ions, which wander through tissues, bind to enzymes, and disrupt normal metabolism. There’s no evidence that any dose level stays “safe”—exposure always carries risk, from skin rashes to irreversible neurological injuries. Modern toxicological research tracks how mercury leaves the body, how quickly it’s excreted, and which therapies—usually some form of chelation—speed up detoxification. Long-term studies on populations exposed to mercury-contaminated products show increased rates of developmental disorders and organ dysfunction, adding weight to regulatory calls for near-total elimination from consumer and medical markets.
Future Prospects
Given its known risks, mercury salicylate is unlikely to stage a comeback in medicine or consumer industries. Regulatory pressure mounts to root out mercury from every product category. Ongoing interest in mercury salicylate now centers on two areas: tracking historical contamination and creating best-in-class analytical standards. As environmental chemists push for better ways to detect and neutralize mercury, the compound still pops up in comparison studies, method validation, and archival reviews of mercury’s role in pharmaceutical history. If any future exists for mercury salicylate, it lies in educational texts, chemical database records, and as a cautionary tale of how a promising compound could have unintended, long-lasting impacts on health and environment. People who work with or study it today do so with an acute awareness that some lessons from the past carry an enormous cost, and scientific curiosity does not erase responsibility.
What is Mercury Salicylate used for?
Understanding Mercury Salicylate’s Place in Medicine
Mercury salicylate, a combination of mercury and the salicylate compound found in aspirin, once showed up in pharmacy cabinets more regularly than today. Over the years, medical science has shifted its perspective on compounds like this. Concerns about heavy metal exposure and safer alternatives have pushed mercury salicylate to the sidelines. Still, its story says a lot about both the evolution of medicine and how we weigh risk versus benefit in healthcare.
Where Mercury Salicylate Fit In
Doctors used mercury salicylate mostly for its antibacterial and antifungal properties. Decades ago, the medical world leaned heavily on chemicals like these to treat infections of the skin, mouth, and sometimes the throat. You might have found it in oral antiseptics, topical ointments for fungal infections, and even some ear drops. The way it worked didn’t have much mystery: mercury killed many types of bacteria and fungi, while salicylates helped lower inflammation and pain.
Growing up, I saw jars of antiseptic creams with long, intimidating names in my grandmother's medicine cabinet. Back then, parents trusted anything that said “antibacterial.” The scars and bumps on scraped knees all got slathered with ointments containing all sorts of ingredients now seen as less than ideal. At the time, the danger seemed distant, and infections were much scarier than chemical exposure.
Risks and Why Medicine Moved On
By the late twentieth century, mercury-based treatments started drawing much more scrutiny. Researchers piled up data showing toxic effects, especially with repeated use. Mercury builds up in the body and targets the nervous system and kidneys. Even low doses can have lasting effects, especially in children. Stories came out about tremors, memory loss, and kidney damage linked to certain medications and even some vaccines that used similar mercury-containing preservatives.
Today's guidelines reflect these dangers. Health authorities in the United States, Europe, and many other places now advise against mercury-based medications except under rare circumstances. This change didn’t just come out of fear — it followed real reports of people harmed by overuse and long-term exposure. Countries also introduced tough rules around manufacturing and labeling. Where safer options exist, such as modern antibiotics and antiseptics, health professionals recommend switching to those.
Lessons Learned and Safer Choices
Looking at the big picture, mercury salicylate’s history reminds us how science moves forward. It also proves why peer-reviewed research and regulatory oversight matter so much in healthcare. Today, you’re more likely to see alternatives like bacitracin, neomycin, or plain old hydrogen peroxide — options with much lower toxicity and proven records. Doctors keep an eye on underlying causes of infection now, rather than just knocking out symptoms. Many people pay closer attention to ingredient labels before using over-the-counter products. That awareness marks real progress.
The story of mercury salicylate shows that chemistry, medicine, and public health never stand still. As a patient and as someone who still keeps a first-aid kit at home, I feel a lot more comfortable knowing that we have both smarter regulations and more effective, safer products on hand. Trust in medicine relies not just on what worked yesterday, but on our willingness to keep learning and to let go of what no longer serves us well.
What are the side effects of Mercury Salicylate?
Mercury Salicylate: A Closer Look
Mercury salicylate combines two powerful substances, each carrying its own risks. Mercury stands out for its toxic reputation in medicine—most people know this metal from warnings about old thermometers or industrial spills. Salicylates, meanwhile, are best known as the foundation of aspirin and other pain relievers. Putting these two together might seem like an old-fashioned solution, but the mix can pack a nasty punch if not handled with care.
Recognizing Toxicity Risks
Your body treats mercury as a threat. It doesn’t matter whether it comes in a shiny bead rolling on the ground or hidden inside a chemical salt. When absorbed, especially over time, mercury builds up in organs. Some people develop tremors, trouble concentrating, and memory loss—mercury can hit the nervous system hard. In my years exploring toxic exposures with patient advocacy groups, I've seen community members struggle through changes in mood, sleeplessness, or depression, all unknowingly tied back to mercury.
The kidneys often suffer most. People exposed to mercury salicylate may notice changes in urination or rising blood pressure. There’s a link between mercury and kidney inflammation, sometimes even kidney failure in extreme cases. These are not the kind of health surprises anyone wants from a treatment.
Salicylate Side Effects Don’t Fall Behind
Salicylates alone can upset the stomach, cause ringing in the ears, or even trigger dangerous bleeding in higher amounts. For those with asthma, a whiff of salicylate irritation can tighten airways. Not every person encounters all these troubles, but no one really wants to take that risk, especially when safer options crowd today’s pharmacy shelves.
Older relatives sometimes mention medications from their childhood that did double duty—pain relief plus some “mercury” for skin conditions or infections. My grandmother remembered using ointments with odd names and strong smells. She considered herself lucky to have strong kidneys and a cast-iron stomach, but doctors today wouldn’t take that bet with anyone.
Why Monitoring Matters
The FDA and other regulators ban or strictly control mercury-containing products for good reason. Mercury salicylate hasn’t passed modern safety standards, and there's no reason to expect it will. Even small doses absorbed through the skin or mucus membranes can build up over time, setting off effects long after the medication stops.
Doctors now look for early signs of toxicity if there’s any chance someone has come into contact. They order blood and urine tests, check for protein changes, listen for telltale neurological complaints. No one wants to miss a subtle sign that could lead to something more serious.
Better Alternatives and Safer Choices
Doctors, pharmacists, and patients have moved on from mercury salicylate because the risks don't balance with benefits. There are safer pain relievers, antiseptics, and anti-inflammatory options now—none of which rely on heavy metals. Reviewing your medicine cabinet, it’s wise to check for any outdated jars or tubes with mystery ingredients. If you find something with mercury or salicylate that doesn’t come from a reputable pharmacy, it belongs in hazardous waste, not in home health care.
Public health efforts matter here. Teaching families about these old compounds helps keep accidental poisonings to a minimum. A conversation with your pharmacist makes a difference if uncertain ingredients turn up, and medical professionals won’t hesitate to help sort out safer choices. Keeping up with the science saves lives and protects health, one practical step at a time.
How should Mercury Salicylate be administered?
Understanding the Role in Medicine
Mercury salicylate doesn’t come up in discussions as often as other medications, but it has lingered in the background of pharmaceutical history. Back in school, we learned just how easily certain compounds stick around in human tissue. Mercury salts fall into that group. Many folks worry about mercury exposure, for good reason, because too much of it causes more harm than good.
Why Dosing and Method Matter
Doctors think twice before using any mercury-based compound because the risks are clear. Acute toxicity shows up as tremors, mouth sores, mood changes, and, with prolonged exposure, kidney or brain damage. The World Health Organization raised alarms for decades, warning people not to take mercury lightly. So, anyone handling this drug at any level must show care at every step.
No cutting corners—administering mercury salicylate requires precision. Someone in a hospital or a controlled research setting, under direct medical supervision, tends to deliver the dose. Oral use almost never happens now. Older papers describe topical agents, but standards shifted with better options available. Injection isn’t advised outside of experimental settings due to the toxic profile.
Let’s be frank: There are safer drugs for treating pain and inflammation. Aspirin and ibuprofen share the same general effect as the salicylate component, with none of the mercury risk. This is a key reason hardly anyone writes prescriptions for mercury salicylate anymore. If someone sees the name crop up, especially in an online forum or an over-the-counter import, they should double-check what they’re actually buying.
Preparing for Safe Use
If a specialized physician did decide mercury salicylate offered unique benefits for a rare case, strict precautions would follow. A compounding pharmacist, trained to deal with toxic compounds, would prepare it. Gloves, masks, and disposable tools would come out, reducing the risk of contamination. Traces of mercury left behind can make people sick, especially children or pregnant women. Local and national laws demand safe disposal, and it gets treated like a hazardous chemical waste product.
I’ve seen research teams double-bag everything, even paperwork, after handling mercury. Hand washing isn’t enough. Labs rely on chelating agents to mop up spills. This kind of care doesn’t happen in most clinics. There’s a strong argument for keeping mercury salicylate locked up or replaced with newer drugs.
Better Options and Informed Choices
Talking with physicians about medication choices helps clear up any confusion. Most pharmacists no longer see mercury salicylate prescriptions and won’t special order it unless forced by a regulation or rare case. FDA guidelines point doctors and patients toward safer treatments. Anyone who hears about mercury salicylate should ask for other options before agreeing to treatment.
If someone finds themselves responsible for old bottles at home or in a workplace, they ought to hand it over to a toxic waste disposal site. This protects families, neighbors, and the environment. Mistakes with mercury don’t just affect one person—they ripple through communities. Trustworthy health care comes from up-to-date science, not nostalgia or convenience.
Is Mercury Salicylate safe during pregnancy or breastfeeding?
Looking at the Risks Without Sugarcoating
Pregnancy turns a spotlight on every ingredient. People really want straightforward answers: can I safely use this? Mercury salicylate rarely pops up in everyday conversation, but it sometimes hides in topical pain relievers, ointments, and some specialty medicines. The name “mercury” alone is enough to make most people uneasy. That feeling isn’t misplaced.
Understanding Mercury Salicylate’s Lurking Dangers
Most folks know mercury used to be in thermometers or old vaccines. Science now flags mercury as a real threat to human health, even at low exposures over time. It’s especially rough on developing brains and nervous systems, which is exactly what you want to protect during pregnancy and while breastfeeding. Mercury salicylate combines salicylic acid—the chemical cousin of aspirin—with mercury, which carries its own risks.
Health experts keep repeating the same points for a reason: The World Health Organization, Centers for Disease Control and Prevention, and numerous toxicologists say mercury exposure during pregnancy can harm a developing fetus. Damage might not show up at birth, but can crop up later as speech delays, behavioral struggles, or learning disabilities. Mercury even passes through breast milk, potentially affecting infants after delivery.
How the Body Absorbs and Reacts
Most people don’t realize how well skin can soak up chemicals. Mercury compounds, including mercury salicylate, can slip through the skin, enter the bloodstream, and end up reaching the baby. Think about the reality: even a small dose every day adds up. Scientists estimate that regular topical exposure can equal ingesting it, especially if the skin is broken or irritated.
Salicylic acid itself can also cause problems in pregnancy. Enough of it in the bloodstream may harm fetal circulation or pose risks during breastfeeding. Salicylates pop up as a warning in most pharmaceutical guidelines for pregnant or nursing parents.
Why Some Are Still Curious About Its Use
Some healthcare providers encounter special cases where mercury salicylate was once used for certain types of pain or skin disorders. People sometimes dig through old home remedies or receive advice from older relatives. Years ago, drug ingredient safety wasn’t as clear, and people didn’t have access to today’s research resources. That kind of legacy use can crop up in family stories, but that doesn’t mean the ingredient is safe for everyone.
Straight Talk From Lived Experience
It’s natural to look for pain relief during pregnancy. I’ve seen friends and patients—sometimes even in my own family—scan lists of medications, cross-checking every ingredient. Every time, we circle back to the same truth: no level of mercury exposure is considered safe while pregnant or nursing. Even low-level, chronic contact can be enough to justify looking for safer alternatives. Pain relief shouldn’t put your health or your child’s development on the line.
Practical, Safer Alternatives
Every parent deserves up-to-date facts. Over-the-counter pain relievers such as acetaminophen carry much less risk. Non-pharmaceutical methods—heat packs, gentle massage, physical therapy—offer comfort without the chemical risk. For itch or skin concerns, look for products labeled safe in pregnancy, or ask the pharmacist for guidance. Healthcare professionals are just a call away and appreciate the chance to clarify ingredients before any harm happens.
Mercury salicylate should stay off the list for anyone pregnant, breastfeeding, or planning to become pregnant. It’s not worth the gamble. The safest option is to choose modern medications or therapies that carry clear safety records and to lean on healthcare teams for advice.
What are the precautions and warnings for using Mercury Salicylate?
Recognizing the Dangers
Mercury Salicylate sounds like something you’d only see in a science lab, but it has popped up across a few older topical products. Combining mercury and a salicylate, this compound has the potential to treat certain skin conditions. That sounds good until you realize how much trouble both mercury and salicylate can cause in the wrong hands. Mercury isn’t just a concern in old thermometers. This metal builds up in the body and messes with nerves, kidneys, and even unborn babies. Salicylates are related to aspirin, which can cause stomach bleeding, ringing in the ears, and complications for kids and folks with certain health problems.
Health Risks You Can’t Ignore
Every person I’ve known who dealt with mercury exposure ended up feeling worse than when they started. Brain fog, shaking hands, digestive problems—mercury just keeps causing harm long after it sneaks into your system. Mercury Salicylate, even when applied to skin, can get inside the body, especially through broken or diseased areas. Folks with chronic skin issues face real danger. It also sticks around if used repeatedly, so the risk climbs as days go by.
Think about kids for a second. Their skin is thinner and absorbs more. Mercury exposure in children can lead to developmental delays and permanent nervous system damage. Pregnant women face even greater risks. The developing brain of a fetus reacts badly to mercury, sometimes with lifetime consequences.
Interactions and Medical Conditions
Salicylates add another layer of worry. People with asthma, liver disease, kidney problems, or a history of ulcers stand to lose the most. Salicylates can increase the risk of stomach bleeding and worsen breathing issues. Folks already taking blood thinners, certain antibiotics, or diabetes medication run into trouble if mercury salicylate has a chance to interfere. Medicines like aspirin, warfarin, and methotrexate start interacting in ways that spill into serious health crises.
Doctors have mostly turned away from using mercury compounds because better and safer options exist. The U.S. Food and Drug Administration and similar agencies around the world warn against mercury use in skin products. You deserve to know what goes into or onto your body.
Solutions: What’s the Smarter Move?
Look for non-mercury options for any condition that used to call for mercury salicylate. Ask your doctor for alternatives—they work better and do a lot less harm. Check every label, especially for creams or ointments from sketchy online sources or old home remedies. Don’t mess with imported cosmetics or folk medicines labeled as “antimicrobial” or “whitening,” especially if they don’t list all the ingredients.
If something contains mercury salicylate, just leave it alone. Companies using safer preservatives and other active ingredients means most modern pharmacies already avoid these risks. Bringing concerns straight to a healthcare professional prevents a world of hurt in the long run. Protecting yourself, your family, and your community starts with staying informed and demanding safer products.
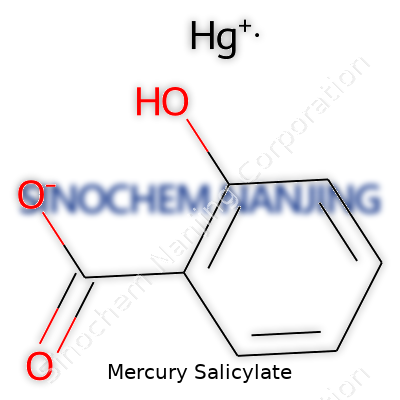
| Names | |
| Preferred IUPAC name | 2-hydroxybenzoic acid mercury(2+) salt |
| Other names |
Mercurosal Mercuric salicylate |
| Pronunciation | /ˈmɜː.kjʊr.i səˈlɪs.ɪ.leɪt/ |
| Identifiers | |
| CAS Number | [623-43-8] |
| Beilstein Reference | 607707 |
| ChEBI | CHEBI:52465 |
| ChEMBL | CHEMBL1629728 |
| ChemSpider | 21717 |
| DrugBank | DB01322 |
| ECHA InfoCard | 07b6be07-e988-4f59-9df1-1521cacd6bb8 |
| EC Number | 205-478-3 |
| Gmelin Reference | 7869 |
| KEGG | C18702 |
| MeSH | D008628 |
| PubChem CID | 24876 |
| RTECS number | OW4950000 |
| UNII | 7LXI3M3306 |
| UN number | UN1641 |
| CompTox Dashboard (EPA) | DJ6B587GET |
| Properties | |
| Chemical formula | C7H5HgO3 |
| Molar mass | 418.78 g/mol |
| Appearance | White or yellowish-white powder |
| Odor | Odorless |
| Density | 4.418 g/cm3 |
| Solubility in water | slightly soluble |
| log P | 2.54 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 7.6 |
| Basicity (pKb) | 10.8 |
| Magnetic susceptibility (χ) | -20.0e-6 cm³/mol |
| Refractive index (nD) | 1.75 |
| Viscosity | Viscous liquid |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 216.0 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | S01AB03 |
| Hazards | |
| Main hazards | Toxic if swallowed, causes skin and eye irritation, may cause allergic skin reaction, suspected of damaging fertility or the unborn child, very toxic to aquatic life. |
| GHS labelling | GHS02, GHS06, GHS08 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | Keep container tightly closed. Handle and store under inert gas. Protect from moisture. Avoid contact with skin and eyes. Avoid formation of dust and aerosols. Use personal protective equipment. Do not breathe dust. |
| Autoignition temperature | > 540°C |
| Lethal dose or concentration | LD50 oral rat 210 mg/kg |
| LD50 (median dose) | 590 mg/kg (oral, rat) |
| NIOSH | MN1050000 |
| PEL (Permissible) | 0.01 mg/m³ |
| REL (Recommended) | 0.6 mg/kg |
| IDLH (Immediate danger) | IDHL: "5 mg/m3 |
| Related compounds | |
| Related compounds |
Sodium salicylate Mercury(II) chloride Mercury(II) acetate Aspirin Phenylmercuric acetate |

