Mercurous Sulfate in Focus: Insights from Research, Industry, and Laboratory Practice
Tracing the Path: From Discovery to Technical Material
Chemists have been drawn to mercurous sulfate for over a hundred years. This salt, known to those of us in the lab as Hg2SO4, rarely appears in headlines yet its story weaves in and out of chemistry textbooks and industrial patent files. French chemists first documented crystalline mercurous sulfate in the 19th century while playing with the unique reactivity of mercury compounds. Earlier, alchemists did not separate mercurous and mercuric forms, so real progress only started when chemical analysis sharpened up. Over time, industries noticed its reliable behavior, especially its steady response as an electrode material. Laboratories latched onto it for use in electrochemical cells, while patent offices filled up with its name.
Getting to Know the Compound
Look on a typical lab bench, and the powdery, yellowish-white form of mercurous sulfate pops up in a tightly sealed container. Those who have handled it remember its density and the almost silky look that belies real risk. Slightly soluble in water, the compound stays stable as long as it keeps away from light and heat. Its decomposition can release toxic mercury vapors; so if you know anyone who worked with mercury compounds in the past, stories of headaches or stricter protocols probably come to mind. Many chemists admire how it maintains a fixed composition (Hg2SO4); its predictable crystal structure shows up often under the microscope, and the sulfate group keeps the mercury in the rare +1 oxidation state.
Technical Labels and Standards Worth Watching
I’ve seen more than one technician get frustrated parsing through technical labels. Bottle labels show the expected purity—usually above 99 percent—plus CAS number 7783-36-0 and safety warnings that jump out in bold type. Supplementary labels add transport hazard codes and limits for trace impurities, reinforcing the need for rigorous sourcing and handling. Chemical suppliers put out thorough spec sheets, but the bulk of safe handling actually happens between colleagues in the lab, reviewing PPE requirements, secondary containment, and up-to-date safety training.
Crafting Mercurous Sulfate: A Classic Preparation
I’ve spent hours preparing mercurous sulfate with students—a classic teaching moment in redox chemistry. Most protocols start with elemental mercury and strong sulfuric acid, plus a little patience. The two react at room temperature, forming a heavy precipitate of mercurous sulfate and hydrogen gas bubbles. Some methods use a small charge of mercury(I) nitrate to help control impurities. Filtration and delicate drying come next; one misstep, and you either lose product or expose the lab to toxic fumes. Chemical manufacturers use much larger reactors and keep everything closed and automated, but at the bench, you feel every step’s risks.
Chemistry in Action: Reactivity and Modifications
If you watch mercurous sulfate react, you notice it keeps its act together in cool, controlled settings but changes character quickly with heat or aggressive reagents. Drop it into a chloride-rich solution, and it turns to calomel (mercurous chloride), shedding the sulfate group entirely—a reaction I’ve seen turn beakers cloudy in seconds. Expose it to an oxidizer or leave it in open air, and it flips over to the higher mercury(+2) compounds. Each of these reactions points to both opportunity and risk: analytical chemists value mercurous sulfate’s stable, low-voltage electrode properties, but anyone managing laboratory or industrial waste has to remain vigilant.
What’s in a Name?
Speaking with older chemists, you’ll hear “mercury(I) sulfate,” “yellow mercury sulfate,” and “hydrogensulfate of mercury” tossed around. The consensus sticks with mercurous sulfate, since the “ous” refers to the +1 mercury state, a distinction that matters for analytical chemists. In journals, I’ve seen Mercury Sulfate (I) or a flurry of trade names from chemical supply houses, some of which are historical holdovers. Everyone in the lab quickly learns to clarify the oxidation state to avoid costly errors, since confusing mercuric with mercurous compounds can spoil entire batches of product or produce unexpected hazards.
Navigating Hazards: What Safety Looks Like in Real Life
Few compounds put lab safety culture to the test like mercurous sulfate. A quick look at the safety data sheet brings home the ban on skin contact, the need for everything from gloves to splash goggles, and the demand for properly vented hoods. Mercury’s long-standing persona as both a useful and dangerous element means waste disposal receives extra attention. Locally, our own department phased out many routine uses, keeping mercurous sulfate only for historical research or tightly controlled calibration roles. Lab neighbors often share notes on mercury spills, disposal procedures, and unexpected headaches—mercury’s health effects lurk in the background, reinforcing why training never becomes a formality.
Where Industry and Research Find Value
Much of mercurous sulfate’s continued relevance comes from its niche as a reference electrode. Every potentiometric measurement made with the mercurous sulfate electrode depends on the salt’s unshakable consistency. Analytical chemists rely on it for measurements in harsh chemical environments where silver chloride or calomel don’t hold up. In battery research, it provided insights into mercury’s redox properties—less common now, given environmental pressure, but still present in textbooks. Some chemical syntheses make use of its low solubility, particularly for specialized catalysis. Across all these uses, ongoing debate surrounds whether greener alternatives can finally step in, but the stability and predictability of mercurous sulfate remain a tough act to follow in key reference cell work.
Digging into Toxicity Research
Mercury in any form raises serious questions. Long-term studies tie exposure to nervous system disorders and symptoms ranging from tremors to memory issues. In the past decade, toxicologists and regulatory bodies have zeroed in on inorganic mercury compounds, including mercurous sulfate. Air and water quality monitors seek out even trace releases, pushing for more stringent exposure limits. Analytical chemists face regular biomonitoring and participate in occupational health reviews. New protocols call for secondary containment and regular spill drills. Despite strict controls, reports still pop up of accidental exposures, highlighting the stubborn challenge mercury compounds pose for both labs and waste handlers. The environmental legacy of mercury compounds—lingering in waterways, entering food chains—amplifies calls for alternatives wherever possible.
Chasing the Next Step: Future Prospects and Responsible Use
Researchers keep working to replace every one of mercury's essential uses with something safer, but what defines mercurous sulfate—a stable, low-voltage reference electrode material—still has no perfect substitute. Advanced labs experiment with new electrode materials, but reliability lags. In countries where environmental regulation tightened up, projects pivot away unless no viable alternative exists, shifting mercurous sulfate toward controlled research rather than routine industry use. Its story continues, less as a staple and more as a benchmark for technical reliability and cautionary tales about risk management. The challenge for tomorrow’s chemists will be to design processes and tools that preserve the benefits without inheriting the burdens—learning from what worked, what didn't, and what the mercury legacy still means for the practice of science.
What is the chemical formula of Mercurous Sulfate?
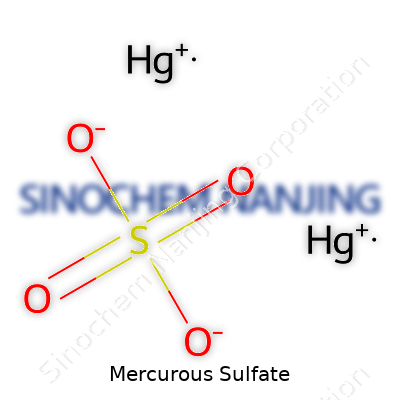
Understanding the Formula: Hg2SO4
Chemistry textbooks list the chemical formula for mercurous sulfate as Hg2SO4. That equation isn’t just facts and numbers—it actually helps many fields get the basics right. People might think of chemical formulas as dry or distant from daily life, but in my experience around labs and classrooms, the formula stops confusion before it starts. Mercurous compounds use the Hg22+ ion. That tiny “2” on mercury reveals there are two mercury atoms in the mercurous ion, which changes everything about how the compound reacts and how scientists can use it. Mistaking it for HgSO4 would lead down the wrong path. It’s not just academic—getting the details right means better experiments and safer results.
Why the Formula Really Matters in Practice
Mercurous sulfate finds its way into reference electrodes, quality control for batteries, and sometimes comes up in environmental testing. The formula keeps the science on track by indicating how elements combine, what byproducts might show up, and how toxic something could get. I’ve seen students grab the wrong mercury salt by mistake, thinking all mercury compounds act the same in their chemical reactions. But Hg2SO4 splits into Hg22+ and SO42−; its chemistry shifts compared to mercuric sulfate, which has the plain Hg2+ ion—completely different reactions, and sometimes much different risk profiles. Mercury’s hazards aren’t a joke, so you want the exact info when training new scientists or watching out for safety in a manufacturing plant.
Safety and Environmental Concerns Aren’t Optional
Understanding formulas isn’t only for passing tests. Mercury compounds, including mercurous sulfate, call for careful handling. A small slip in formula means trouble scales up in a hurry. If someone handled or disposed of mercurous sulfate as if it were a less toxic sulfate, the consequences might put coworkers or the environment at risk. Reports have linked improper mercury disposal to pollution in local waterways and long-term damage to ecosystems. The facts here underline why even small details in a product’s chemical name and makeup impact real-world safety. People managing chemical inventories or supervising labs need solid training and easy-to-access chemical information, not just for law compliance but for the health of their team and neighbors. Double-checking the formula, understanding its structure, and keeping an up-to-date safety data sheet around all cut down accidental exposure.
Building Solutions—Start With Strong Education and Accountability
Better science starts early. I remember chemistry lab mistakes from classmates who didn’t recognize the difference between “mercurous” and “mercuric.” Schools owe it to students to push clear teaching about names, formulas, and hazards, so they head into jobs ready. On the job, workplaces get a lot out of regular training—not just a dusty sign-off, but real practice in reading labels and understanding storage requirements. Chemical manufacturers should print formulas bold on packaging, making mistakes less likely. On the community side, giving clear guidance for cleanup or disposal keeps people and nature safer. All these steps build a culture where facts matter, mistakes shrink, and safety becomes second nature. The formula Hg2SO4 might look small, but it carries a big message: get the science right, and the benefits follow for everyone involved.
What are the common uses of Mercurous Sulfate?
What Makes Mercurous Sulfate Stand Out
Mercurous sulfate often goes unnoticed outside laboratories, but anyone who works in science, engineering, or electronics will eventually cross paths with it. It looks a bit like a grayish-white powder, not the flashiest on the shelf, but few substances play a more reliable role in keeping certain systems running smoothly. As someone who’s sat hunched over a lab bench for hours, it grabs attention because of what it does in electrochemical setups.
Electrochemical Reference Electrodes
People who build precise voltmeters and reference cells know how tricky it is to keep electrical measurements honest. Mercurous sulfate helps tackle this challenge by serving as a stable component in reference electrodes. Battery makers, electrochemists studying corrosion, and water quality inspectors all rely on these reference cells to monitor real-world systems—old pipes in city water lines, experimental fuel cells, even groundwater near industrial sites. By brushing a paste of mercurous sulfate onto an electrode, you get a lasting and steady point of reference for measuring voltages and chemical reactions. This gives confidence when comparing results in the field or across research projects.
Battery Manufacturing
Silver-zinc and mercuric oxide batteries built for military and space equipment sometimes feature mercurous sulfate in separators or electrolyte additives. It helps reduce unwanted chemical noise and improves energy output, especially during high-demand moments. This chemical helps prevent the buildup of gases that could lead to pressure failures or short circuits, which anyone who has worked around emergency equipment knows can be a lifesaver—sometimes literally. Hospitals and rescue teams rely on backup gear that has to work first time, every time. Mercurous sulfate gives an added layer of protection against failure.
Chemistry Experiments and Teaching
High school and college teachers sometimes show students how reference electrodes work by using mercurous sulfate. Nothing gets attention like seeing a reaction shift depending on the reference cell. These lessons stick with students; they drive home the impact of carefully selected chemicals in real scientific work. People new to lab research pick up the basics of electrochemistry with mercurous sulfate, often moving on to more advanced setups after seeing it in action.
Environmental Testing
Monitoring heavy metal pollution and acid rain in lakes can’t happen without accurate measurement tools. Environmental scientists use instruments calibrated with mercurous sulfate reference electrodes to detect changes in pH and metal ion concentrations. It lets teams catch early signs of contamination long before anyone notices dead fish or unsafe drinking water. Spotting trouble early helps make cleanups faster and less expensive, which matters to every community that depends on a safe water supply.
Handling and Safety Concerns
Mercurous sulfate comes with warnings, too. Mercury compounds are toxic; anyone using the material wears gloves, works with good ventilation, and stores it away from sinks so mercury doesn’t end up in the landfill or water system. Safety training is the norm for labs and factories that keep it on hand. Some researchers now look for substitute materials in response to growing rules around mercury use. Zinc-based or silver-based alternatives exist, but they sometimes fall short, especially for measurements requiring pinpoint accuracy year after year.
Ways to Reduce Mercury Risks
People asking about greener alternatives spark a bigger conversation about chemical safety in science and industry. Streamlined procedures for collecting, recycling, or neutralizing mercury waste keep harmful byproducts out of the environment. Research into new electrode materials could shrink the demand for mercurous sulfate, too. Until solid alternatives catch up, the chemical keeps its spot where accuracy and reliability matter—so long as people stay informed and cautious about handling it.
Is Mercurous Sulfate hazardous to health or the environment?
Understanding Mercurous Sulfate
Mercurous sulfate comes up in conversations about lab chemicals and certain industrial processes. It’s a white, odorless powder used in reference electrodes and sometimes in the manufacture of batteries. Most folks don’t cross paths with it outside a science classroom or specialty shop. The main concern boils down to mercury. Any compound containing mercury raises eyebrows for good reason.
Risks to Health
Health problems from mercurous sulfate usually tie back to its mercury content. Mercury likes to stick around in the body after exposure. It can damage kidneys, affect the nervous system, and hurt the lungs if inhaled as dust. Working in labs for a few years, I’ve seen strict rules set for mercury-based compounds—even for spills or small accidental contact. Handling even small amounts without gloves or proper protection can lead to trouble over time. Medical literature documents tremors, mood changes, and memory issues in people who take in too much mercury. Chronic exposure brings out those symptoms in ways you don’t forget. Children face even bigger risks, as their nervous systems develop.
Accidents at workstations or improper storage can spread invisible risks. Touching surfaces contaminated with mercurous sulfate, or inhaling particles from an open jar, brings real-world impacts. In labs, safety data sheets make it plain: gloves, eye protection, and careful cleanup. It’s not fear-mongering—just sound advice backed by years of workplace studies and case reports. Direct contact with skin or accidental inhalation can be more than a minor issue. Ingestion, even at small levels, brings up every warning bell because mercury doesn’t leave the body quickly.
Environmental Concerns
Mercurous sulfate doesn’t just stay put. If it enters soil or waterways, mercury starts cycling through plants, animals, and water supplies. Sometimes, mercury shifts into more toxic forms, like methylmercury, which shows up in fish and moves up food chains. Local communities near chemical dumps or industries using mercury compounds have documented higher mercury levels in air and water. Living through some of those public health scares nearby, I’ve seen firsthand how long it takes to fix things once mercury pollution starts. Methylmercury crowds headlines for its role in brain damage and birth defects. Fish advisories around contaminated lakes are there for a reason. They come from studies showing cumulative exposure over time harms whole regions. Once in the environment, mercury doesn’t quietly break down; it lingers and becomes part of everyday risks if ignored.
What Can Help?
Safe work habits protect people who handle mercurous sulfate. Regular training, solid ventilation, labeled storage, and spill plans cut down on accidental exposure. Switching to alternatives in classrooms and industry, when possible, avoids the problem entirely. Government agencies like the EPA and OSHA have set clear benchmarks for handling mercury compounds. These aren’t just paperwork—they come from evidence about long-lasting damage. Cleanup and containment after spills or leaks should follow strict guidelines, not cut corners. Reporting any health symptoms early gives doctors a chance to help before the damage sticks. On a bigger scale, phasing out mercury in processes and pushing for non-mercury batteries or electrodes reduces the long-term environmental load. Communities near waste sites need regular testing and prompt cleanup so the next generation doesn’t walk into the same mess.
How should Mercurous Sulfate be stored and handled?
Trustworthiness Starts in the Storage Room
Mercurous sulfate belongs in labs and industries working with specialized batteries and reference electrodes. Silver-white crystals bring benefits, but they come with risks. Mercury means trouble for people and ecosystems if handled the wrong way. I’ve spent enough time in chemical stockrooms to know blind spots show up when teams grow comfortable with their routines. This stuff won’t forgive sloppy habits.
Secure, Dry, and Out of Sunlight
A tight, labeled container lives at the top of my rules. Glass or hard plastic, tightly sealed, blocks spills and contamination. Moisture triggers chemical changes—mercurous sulfate reacts with water and goes from useful material to something hazardous and unreliable. I’ve seen lids left ajar, and the next user always pays. Direct sunlight shortens shelf life, so shelves or cabinets that keep things dark serve you better. Every chemical supplier prints these instructions in their safety data sheets, but paper trails mean nothing without practice.
Handling: Respect the Risks
No one wins a prize for bare-handing this stuff. Lab coats, gloves made of nitrile or neoprene, goggles—these are the basics. Mercury finds its way through latex faster than many expect. I’ve seen people skip these steps after years of routine, only to regret it. Fume hoods aren’t optional: any dust or accidental heating – even at low levels – puts you at risk for inhalation. When the label warns about toxic dust, it’s not exaggerating.
Never Work Alone, Always Know Your Plan
Accidents don’t schedule appointments. Mercury exposure symptoms creep in quietly—tremors, headaches, mood changes. Solo work with volatile chemicals puts people in danger; if someone collapses, nobody will help. There’s no shame in demanding a partner or spotter. The best teams refresh training sometimes every few months. It only takes one story of hospital visits to show stubborn coworkers why.
Proper Disposal Ain’t an Afterthought
Stuffing leftover mercurous sulfate down a drain or tossing it into regular trash spreads pollution. Mercury never disappears—it poisons water, soil, and eventually, food. Any waste goes in sealed, labeled containers, picked up by licensed hazardous waste handlers. My early days included nights cataloging every chemical and filling out disposal forms. It’s tedious, but nothing feels as awful as reading press reports about a spill traced back to lazy handling.
Fixing Gaps: Training and Accountability
The best solutions rarely involve special products or expensive gear. Groups that keep clean log sheets on chemical use and enforce regular safety drills cut down on mishaps. Open conversations matter more than rules taped to walls. If leaders model careful behavior and answer stupid questions with real attention, staff follow suit. Rewarding diligence beats threatening punishment. Building a safety culture never finishes—every newcomer needs mentoring, and old hands need honesty about shortcuts.
Closing the Loop
Safety around mercurous sulfate isn’t just about following a sheet of do’s and don’ts. It’s about discipline, respect for each other, and recognizing the cost of mistakes. No shortcut offers enough time-savings to outweigh a hospital bill or a hazardous leak. I keep these lessons close, and I hope others never need a harsh reminder.
What is the appearance and physical properties of Mercurous Sulfate?
An Unusual Sight in the Lab
Mercurous sulfate doesn’t attract much attention until you see its pale yellow powder up close. The color isn’t bright like lemon, more muted, taking on a sort of buttery tint with a subtle reddish outline if the powder gathers up. Touching it, the powder’s texture feels fine—almost chalklike, with a dry and crumbly consistency. It doesn’t cake like clay or flow like table salt. You might notice crystals shining a little under light, but there’s no gloss or shimmer that stands out.
Stability and Odor
The compound carries almost no smell. In the sciences, a substance that keeps quiet in the nose often points to stability—which mercurous sulfate does deliver at room temperature. It keeps to itself, not reacting with the air unless things warm up or get moist. Humidity leads it to darken, a familiar risk with some of mercury’s less stable friends. If moisture lingers, the yellow can slide into grayish tones, and black specks may show up as the mercury breaks away, forming elemental droplets. That transformation spells risk, since elemental mercury is no friend to human health, or the thermometer in your lungs.
Weight and Solubility
This compound packs a punch in weight, thanks to two mercury atoms for each sulfate. It sits on the scale at about 561 grams per mole, making it noticeably heavier than common table chemicals. Even with the heft, drop mercurous sulfate into water and it barely dissolves—only a tiny trace goes in. That insolubility helps keep it stable but can turn cleanup into a headache if a spill happens in a school or industrial lab. If diluted acids are around, especially sulfuric acid, the material stays content and doesn’t decompose. Add a stronger acid or alkali, the story changes—decomposition sets in, with fumes and hazardous byproducts showing up for any unprepared handler.
Toxicity and Handling
It’s impossible to handle mercurous sulfate without thinking about safety. Mercury compounds bring an undisputed reputation for toxicity. Even touching the powder, inhaling its dust, or accidentally letting it slip near food, ramps up the risk. Short term, acute poisoning can lead to trembling, headaches, or even kidney damage. Over time, chronic exposure gets even nastier. These truths make me cautious—gloves go on every single time, and I always keep the bottle closed, labeled, and locked away from direct sunlight or moisture. Most labs use glass containers with tight seals to avoid any contact with wooden or porous surfaces where lingering residue could put people at risk.
Why Physical Properties Matter
Mercurous sulfate’s stubborn insolubility and bulk make it useful in specialty batteries and electrochemical cells, especially old-school reference electrodes. Having a solid that doesn’t break down or easily leak mercury ions keeps those instruments accurate longer. The color and stability flag trouble early—yellow turning to black isn’t just a visual cue, it’s a warning that mercury metal is loose and someone needs to intervene. For me, vigilance matters as much as chemistry knowledge. Relying on visual indicators, keeping the workbench tidy, and ensuring air stays free of invisible mercury vapor aren’t just academic—they keep people safe.
Improving Safety and Education
Tighter control over storage and disposal protects both workers and the environment. Labels, proper air flow, and stricter training cut down on surprise exposures. Introducing mercury-free alternatives in batteries or electrodes—like silver-based compounds—takes time, but that shift keeps labs on the path to safer science and cleaner ecosystems. Sharing real-world handling tips helps new techs and young scientists build the habits that prevent accidents before they start.
| Names | |
| Preferred IUPAC name | Disulfur Mercury(II) Tetraoxide |
| Other names |
Mercury(I) sulfate Mercurous sulphate |
| Pronunciation | /ˈmɜːrkjʊrəs ˈsʌlfeɪt/ |
| Identifiers | |
| CAS Number | `7783-35-9` |
| Beilstein Reference | 0179905 |
| ChEBI | CHEBI:75834 |
| ChEMBL | CHEMBL1201651 |
| ChemSpider | 23406 |
| DrugBank | DB14636 |
| ECHA InfoCard | 100.029.222 |
| EC Number | 231-892-1 |
| Gmelin Reference | 85754 |
| KEGG | C18798 |
| MeSH | D008630 |
| PubChem CID | 24557 |
| RTECS number | OW4550000 |
| UNII | LT3C5706DJ |
| UN number | UN1645 |
| CompTox Dashboard (EPA) | DTXSID0022758 |
| Properties | |
| Chemical formula | Hg₂SO₄ |
| Molar mass | 472.14 g/mol |
| Appearance | White or yellowish powder |
| Odor | Odorless |
| Density | 6.47 g/cm³ |
| Solubility in water | Insoluble |
| log P | -4.94 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 6.94 |
| Magnetic susceptibility (χ) | −47.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 2.055 |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -766.4 kJ/mol |
| Pharmacology | |
| ATC code | V09AA04 |
| Hazards | |
| Main hazards | May be toxic by inhalation, ingestion, or skin absorption; causes irritation to skin, eyes, and respiratory tract; may cause mercury poisoning; hazardous to the environment. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Warning |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P260, P262, P264, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P314, P321, P330, P361, P362, P405, P501 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Lethal dose or concentration | LD50 (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat LD50: 57 mg/kg |
| NIOSH | MW5600000 |
| PEL (Permissible) | 15 mg/m3 |
| REL (Recommended) | 0.01 mg/m³ |
| Related compounds | |
| Related compounds |
Mercury(II) sulfate Mercurous chloride Mercury(I) nitrate Mercurous oxide Mercury(II) oxide |

