Mercurous Nitrate: A Thorough Commentary
Historical Development of Mercurous Nitrate
Mercurous nitrate has deep historical roots in both science and industry. Early chemists isolated and utilized it as part of the exploration into mercury compounds, often in the hope of producing new medicines or extracting precious metals. Alchemists, unaware of the dangers, valued substances involving mercury for their unique reactions and vibrant colors, not to mention mercury's bizarre liquid state at room temperature. Laboratories in the nineteenth century started using mercurous nitrate for analytical purposes, especially in detecting chlorides thanks to its reactivity. Tanners adopted it in leather finishing, largely because the salt altered the physical characteristics of leather fibers, making the material more workable and, for a time, highly prized in glove making. The development of its use reflected both curiosity and demand from craftspeople, but people steadily realized the dark side of mercury toxicity, which pushed the chemical into more specialized niches.
Product Overview
Mercurous nitrate, known for its white or yellowish crystalline form, bridges historical utility and scientific scrutiny. Manufacturers have produced it mainly to supply laboratories and manufacturing operations in need of reliable mercury sources or reagents. Product grades vary based on purity, and a little contamination can spoil results, so quality control remains tight. In the past few decades, demand has tapered off due to stricter environmental and workplace rules. Nonetheless, this compound keeps a presence in select research fields thanks to its distinctive chemical profile. Most chemical distributors mark it as hazardous, flagged both for mercury risks and for the acid present in typical samples.
Physical and Chemical Properties
The key thing about mercurous nitrate is its sensitivity and tendency to shift into mercuric or elemental mercury states under the wrong conditions. Its chemical formula is Hg2(NO3)2·2H2O (dihydrate form) or Hg2(NO3)2 as anhydrous. This compound dissolves readily in water or dilute nitric acid, but not in alcohol. It appears white to grayish or yellow, forming prismatic crystals or powder, sometimes clumping with moisture. The solid begins to decompose in air and releases toxic vapors even at room temperature. Exposure to light or warmth speeds up that breakdown, so laboratories have to keep it sealed tight, away from direct sunlight. Its molecular weight and water content shift depending on how it was prepared and how long it’s been exposed. Heating risks turning the solid into a mixture of mercury oxides and nitrogen oxides—both hazardous in closed spaces.
Technical Specifications and Labeling
Most suppliers classify mercurous nitrate as a high-risk chemical. Labeling follows strict regulatory guidelines, usually highlighting the need for personal protective equipment, environmental controls, and waste disposal steps. The UN Number (1625) and CAS Number (10415-75-5) show up on every drum or bottle. Standard chemical labels reference acute toxicity (GHS06 skull-and-crossbones), environmental hazards (GHS09 aquatic symbol), and corrosive risks. Safety Data Sheets (SDS) provide guidance for storage, accidental release procedures, and medical steps for exposure. Those technical sheets spell out conformance with local and international rules; in the US, the EPA, OSHA, and the DOT all have a say. Laboratories ordering mercurous nitrate now often must show controlled use certifications, and there’s a strong push to keep stocks to the bare minimum.
Preparation Method
Chemists synthesize mercurous nitrate through precise additions of elemental mercury to dilute nitric acid, keeping temperatures and concentrations low. Pouring pure mercury into cold, dilute nitric acid sets up a reaction that yields mercurous nitrate and nitrogen oxides, mostly as colorless gases. Extra care is taken to limit heat and acid strength, since strong acid or high temperature pushes the reaction toward mercuric nitrate instead. Skilled technicians use glass, not metal tools, because mercury can amalgamate with many metals and contaminate the product. The reaction must occur in a well-ventilated fume hood or with air control systems to scrub out nitrogen dioxide—this gas has a sharp smell and stains everything yellow. Filtering and recrystallizing under cool conditions produces the workable product, usually in damp crystal form. Drying and grinding take careful management; even small losses can contaminate workspaces and expose workers to serious risks.
Chemical Reactions and Modifications
Mercurous nitrate takes part in unique reactions, especially those that showcase mercury’s ability to flip between oxidation states. One classic use taught in university labs is the formation of “Millon’s base,” which points to presence of phenolic compounds. The nitrate anion interacts strongly with bases and other halides, creating cloudy precipitates—useful for analytical chemistry demonstrations. Besides that, chemists prize its ability to form double salts or react with reducing agents to liberate metallic mercury, which appears as silvery beads. The compound’s sensitivity makes it valuable for teaching and demonstration but also marks it as risky compared to other more stable lab reagents. Over the years, professionals have worked to swap it out for safer alternatives in most routine applications, but its niche role persists in certain metal extraction studies and instrument calibrations.
Synonyms and Product Names
Mercurous nitrate finds listing in chemical catalogs under several names, such as mercurous nitrate monohydrate, mercury(I) nitrate, or di-mercury nitrate. European sources sometimes call it nitrate of sublimate, a holdover from older chemical texts. Names may vary based on the hydrate level—dihydrate, monohydrate, or anhydrous—so buyers must pay close attention. Historic texts often refer to “mercury nitrate, basic,” but that phrase covers a range of compounds. Pharmaceutical and industrial guides highlight “HgNO3·xH2O” as shorthand, though most modern inventory lists use the IUPAC name for accuracy.
Safety and Operational Standards
Working with mercurous nitrate demands strong discipline and focus, since inhaling dust or vapors or skin contact carries a real risk of mercury poisoning. Laboratories put thick gloves, splash goggles, and lab coats between people and the chemical. Engineering controls such as chemical fume hoods act as the main line of defense, along with sealed containers and proper ventilation. Regular blood checks and environmental monitoring for mercury vapor help spot exposures long before symptoms start. Disposal ties up a lot of staff time, since waste has to go to specialized mercury handling facilities, avoiding drains at all costs. Regulatory auditors sometimes drop in unannounced to check logs and make sure no shortcuts happen. The move to phase out mercury reagents altogether has grown strong in academic teaching and in routine labs, tied to global agreements like the Minamata Convention that aim to limit new mercury pollution. Even so, for a handful of procedures, nothing has yet matched mercurous nitrate’s particular behavior, so the chemical survives in tightly controlled circles.
Application Area
Mercurous nitrate’s application range stands narrow but influential. Its main jobs show up in the synthesis of other mercury compounds, staining reagents in microscopy, and longstanding but now declining use in tanning and gilding. In analytical chemistry, folks rely on its interactions with organic and inorganic ions to reveal trace contaminants in samples. Electroplaters once counted on it for preparing metal surfaces for intricate gold leafing or silver inlay, but safer systems have taken over. Some specialty glass and ceramics shops still use it to form novel glazes or color transitions. The medical sector abandoned most mercurous nitrate uses early in the twentieth century, after people recognized the direct connection between handling the salt and dangerous chronic poisoning—no amount of modern fume hoods can truly undo that risk for mass uses.
Research and Development
Ongoing research involving mercurous nitrate focuses on understanding the chemical’s fine details and hunting for replacements in legacy chemical processes. Scientists studying heavy metal interactions see it as a window into how mercury moves between environmental reservoirs or binds to organic matter in water and soil. Some research teams keep it in stock to compare its reactions with newer, less-hazardous chemicals that might offer similar performance in catalysis, analysis, or synthesis. Universities take a cautious approach, often restricting access to graduate-level or supervised research, especially in environmental chemistry or forensic science. Publishing new findings demands proof of precautions and careful waste tracking, which slows down new advances but pushes for solutions that respect health and future environmental needs.
Toxicity Research
Mercurous nitrate presents a real hazard for acute and chronic mercury exposure. Poisoning can come from inhalation, ingestion, or skin absorption, resulting in effects that range from tremors and memory loss to kidney failure and death. Studies have documented subtle effects on brain function long before blood mercury levels turn obviously high, and no organ system escapes risk. Animal work confirms that the compound’s low solubility doesn’t make it safe; repeated contact causes the metal to build up relentlessly. Some occupational health studies from the early and mid-1900s tell harrowing stories of glove makers and lab workers, but by the 1970s, stricter rules almost entirely pushed the salt out of daily use. More recently, toxicologists have been able to trace mercury’s journey from wastes into rivers and ultimately into human diets via fish. Mercurous nitrate, thanks to its ease of converting to vapor or soluble mercury compounds, remains banned or tightly restricted in open factory settings. To this day, decades-long health studies keep confirming strict bans protect both workers and the wider public.
Future Prospects
Looking ahead, mercurous nitrate faces shrinking demand and tighter rules—people no longer look to mercury chemistry unless absolutely necessary. Most industries have shifted to alternative compounds, especially where safety or environmental stewardship matter. That said, a few critical applications do not let go so easily, since mercury’s reactivity cannot be copied perfectly with other metals. R&D continues to push for greener, more manageable chemicals, and people lead with questions about circular economy and long-term safety, not just process yield. As nations enforce new pollution standards and boost funding for toxicology research, scientists can expect further restrictions, better monitoring, and likely a full retreat to controlled laboratory spaces. For chemistry students, mercurous nitrate will probably end up a footnote or case study rather than a tool of everyday work. Meanwhile, regulators, scientists, and industry leaders need to build new frameworks for handling hazardous compounds responsibly, using lessons from the past century’s mercury mistakes.
What is Mercurous Nitrate used for?
A Chemical with a Tainted Legacy
Walk into any serious discussion about old-school chemistry and a few names keep popping up. Mercurous nitrate fits right in, famously connecting science classrooms, early photography labs, and the mining industry. In those days, it played a big part—most notably as a reagent or a catalyst. Not many people outside these circles think about it now. The reason is simple: health and safety weighed in and shifted the conversation.
Why Industries Reached for Mercurous Nitrate
In the past, textile and hat-making shops leaned heavily on mercurous nitrate. Hungry for efficiency, hatmakers used this compound to treat animal furs. It made felting easier, which anyone who’s tried to make a hat by hand can appreciate. The improved felting meant faster work and better products—at a cost. What stands out from those days isn’t only the hats they made; it’s the workers who got “mad hatter” disease because of mercury poisoning.
Older textbooks detail its use as a chemical reagent. In analytical chemistry, mercurous nitrate stepped in when folks needed to detect chlorides through precipitation reactions. Chemists saw clean, identifiable results and kept bottles of it on their benches for years. Its effectiveness in these tasks made it a mainstay in laboratories, until the dangers became too obvious to ignore.
From Photography to Mining
Photographers once used mercurous nitrate during the development process to create images that lasted. Miners took it underground to aid in extracting precious gold and silver. Exposure rules weren’t as strict. In both cases, the quick results seemed worth the risk, though hindsight tells a different story.
The Clear and Present Health Hazards
Science caught up with experience. Reports, health surveys, and personal stories stacked up around mercury exposure—tremors, memory problems, and permanent damage to the nervous system. The felt hat industry became infamous for its workers’ poor health. In schools, chemistry instructors started to question whether that textbook demonstration was really worth bringing out an old bottle of mercurous nitrate.
The presence of mercury in the compound carries serious risks. Even brief exposure can lead to harm if inhaled or absorbed through the skin. Environmental concerns grew louder, too. Mercury doesn't stay put. If it gets into the soil or water, it sticks around and cycles back into plants, animals, even our dinner plates.
Learning—and Responding—From Experience
Looking at the facts, it’s tough to argue for mercurous nitrate in classrooms, factories, or mines these days. Safer alternatives for analytical chemistry and photographic development work well and don’t threaten workers or the environment. Regulations followed the research—not just in the United States, but across the globe. Schools ban the use, the EPA enforces strict guidelines for industrial mercury, and most industries have left mercurous nitrate behind.
The story of this compound means something to anybody who thinks about workplace safety, environmental health, or public policy. We see what happens when a useful tool also brings harm, and it reminds us to keep safety at the center of any work with chemicals. A lesson learned at some cost—one we shouldn’t forget next time we consider resurrecting old methods.
What are the safety precautions when handling Mercurous Nitrate?
Why Mercurous Nitrate Demands Respect
Mercurous nitrate, with its silvery-white appearance, can look rather harmless to the untrained eye. I remember the first time I saw someone pour out a bit under the fume hood—it could’ve passed for table salt. Looks deceive. This stuff packs a punch for the body and the environment. You’d think chemicals you can’t easily buy at big box stores would offer some basic safety, but it’s the hidden dangers that burn a memory. It’s toxic. Mercury exposure ruins kidneys, messes with the nervous system, and produces symptoms people miss until it’s late. Most people never stop to consider that inhaling even small particles or letting it touch bare skin might set off a spiral of health issues.
Practical Steps for Safe Handling
No shortcut ever made handling mercurous nitrate easier—or safer. Gloves are only the start. Nitrile or neoprene work better than latex, since mercury gets right through some materials. I always double up in case a glove tears. Skip bare hands—the risk doesn't justify laziness. Eye protection isn’t for show, either. Splashing this compound into your eyes because you leaned too close while measuring—there’s no recovering from that mistake.
Ventilation feels like a basic request, but not every workspace gets it right. Fume hoods aren’t just a box with a fan. Don’t work outside of them, even for quick steps. Mercury vapor doesn’t ask permission before drifting out. Even if it’s a tiny amount, tight quarters and poor airflow increase exposure fast. If the smell hits your nose, clean air left you behind.
Storage and Spills: Where Experience Teaches the Hardest Lessons
Glass containers with tight-fitting lids soak up hazardous material scares easily. Never use metal cans—mercury corrodes many metals and sets off chemical reactions that surprise even old pros. Don’t store bottles near acids or bases; reactions in cramped chemical cabinets end with alarms and cleanups, not lessons learned. Label everything, even if it looks obvious. I once saw a clear jar left on a bench with only a faded paper scrap for identification; nobody wanted to move it before finding out what was inside.
Spills feel terrifying. Don’t grab paper towels or rags. Special mercury spill kits exist for a reason—bottles of sulfur or specific powders help absorb, while vacuuming spreads vapor. The key is to limit the area, cordon it off, and call for help without shame. The wrong move spreads contamination. I once had to throw out shoes after walking through a missed spot—not fun explaining that to the supervisor, or to health and safety.
Health and Environmental Impact
Chronic exposure to compounds containing mercury turns lab work into an occupational hazard. It’s not paranoia; it’s history. Too many stories float around about colleagues with shaky hands, foggy thinking, and frequent trips to the doctor. Washing up afterward isn’t a perfunctory step. Even after gloves, check for residue on wrists and under fingernails.
Waste disposal takes patience, not shortcuts. Never pour it down the drain or throw it in common trash. Local regulations mandate hazardous waste collection for a reason. In 2017, one missed bottle in municipal garbage led to thousands of dollars in cleanup bills and a hard conversation with residents worried about water quality.
Building a Culture that Keeps People Safe
Training beats out manuals every time. Watching a seasoned chemist handle mercurous nitrate sticks with you longer than reading the SDS. Open discussion, clear signage, and strong policies keep focus sharp, even on sluggish days. Labs and schools that maintain a culture of double-checking, not skipping steps, see fewer accidents. Safety comes from routine, yes, but also from the stories and lessons passed down by those who’ve been there and learned the hard way.
What is the chemical formula of Mercurous Nitrate?
Understanding Mercurous Nitrate
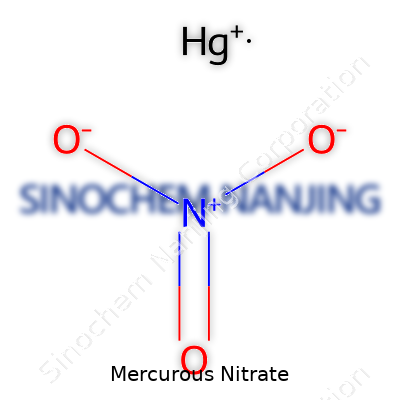
Chemicals don’t just live in textbooks. They cross paths with our daily lives in ways many of us don’t notice. Take mercurous nitrate, for example. Its chemical formula, Hg2(NO3)2, might not roll off the tongue, but you’ll find it turning up in some very hands-on places. This compound has played a role in chemistry for well over a hundred years.
Mercury and Its Double Life
Mercury loves to surprise. In mercurous nitrate, mercury forms a dimer—two mercury atoms linked together, sharing a single positive charge each. That’s what the “Hg2” part means. Chemists learned early that mercurous ions don’t just drift around solo, but always move in pairs. Why does this matter? Because the structure tells you plenty about how the compound behaves.
Nitrates have a familiar script in the world of chemistry—soluble, versatile, a bit reactive under the right conditions. Pairing them with two linked mercury atoms gives mercurous nitrate unique abilities. That quirky bonding pattern means this salt has characteristics different from its cousin, mercuric nitrate (Hg(NO3)2). Understanding the formula gives a window into why old chemistry sets once used mercurous nitrate for qualitative analysis, spotting things like chloride or bromide ions.
Safety and Impact
My first chemistry teacher always respected mercury. He’d say, “Know what’s in the bottle.” With mercurous nitrate, there’s good reason. Mercury compounds build up in living tissue. Nitrates, in large amounts, can harm both health and waterways. Today, many labs have moved away from mercury compounds altogether—but knowledge of its chemistry matters. Spill one of these salts and you’re not just cleaning a mess; you’re protecting people and the wider environment.
You still find this chemical in special analytical work or research. Handle it without proper care, though, and you risk exposure. That’s one lesson science classrooms hammer home. Policies today demand strict guidelines for storage and disposal. Checking local and national regulations often means looking up the specifics on chemicals like Hg2(NO3)2.
Connecting Knowledge and Practice
Learning the formula isn’t some dusty fact for quiz night. It helps predict behavior. If you see two mercury atoms bonded together, you know this stuff won’t behave like its single-mercury siblings. Understanding the nitrate's presence tells you to expect high solubility in water—which makes a spill that much harder to contain.
That kind of knowledge arms you to act safely. I’ve seen students and junior lab workers spot a problem by remembering these fundamentals. They knew what signs to watch for—because someone taught them to start with what’s on the label. In industry, that same attention governs how chemicals move from supplier to lab bench to waste facility.
Building Better Habits
Chemicals like mercurous nitrate serve as reminders that science connects to daily care and responsibility. The formula matters because it brings you closer to the heart of chemical safety, environmental protection, and precision in the lab. If you’re mixing, disposing, or studying, always start by understanding what’s in your flask—and respect its legacy in both science and safety.
How should Mercurous Nitrate be stored?
The Risks Nobody Can Ignore
Mercurous nitrate isn’t a chemical most people run into at the grocery store. In labs and industry, though, it pops up often enough to cause concern. This isn’t just another powder or salt — breathing fumes or touching mercurous nitrate could cause trouble in a hurry, both for humans and for the environment. Exposure may affect the kidneys, nervous system, and even result in mercury poisoning. In my view, casual treatment isn’t just careless; it’s risky and obsolete.
Good Storage Starts With A Solid Container
Glass easily absorbs some chemicals, but for nitrate salts, glass or high-density plastic containers work best. Direct sunlight kicks off reactions that nobody wants on their workbench. So, storerooms ought to keep these chemicals in sturdy, sealed bottles, well away from any windows. Years back, I saw a dusty old glass bottle crack open in an overheated cupboard — mercury fumes are tough to forget, especially when you’re trying not to breathe.
Temperature And Stability: Friends For Safe Storage
Mercurous nitrate needs a steady climate. No extremes, no dampness. Hot rooms and shifting temperatures spark chemical changes or pressure build-up that stresses even the best containers. Cooler, dry areas help prevent accidental spills or leaks. Nobody wants a container rupture, especially since mercury compounds are notoriously hard to clean up safely. In smaller labs, this often means dedicating a shelf on the lowest rack or using a well-ventilated, climate-controlled cabinet.
Avoid Mixing And Accidental Contact
Mercurous nitrate can react if it hits organic material, reducing agents, or even strong acids. In my experience, keeping it on its own—labeled and locked up—beats any workaround for avoiding dangerous mix-ups. Shelves in chemical storage often get cluttered over time. It takes constant effort to keep incompatible bottles from drifting together. Double-checking storage plans every few weeks isn’t just cautious—it’s common sense in a place where mistakes are costly.
Personal Safety Measures: Not Optional
The best chemical locker won’t fix a careless habit. Good gloves, lab coats, and safety goggles stay close at hand every time someone reaches for high-risk chemicals. Most places I’ve worked also keep powdered sulfur and spill kits ready for quick action if a container slips or cracks. Running water, eye wash stations, and clear instructions need to stay close. Mercury cleanup demands special procedures—ordinary spill kits fall short. Quick action keeps incidents from spiraling.
Community And Environmental Responsibility
Disposing of mercurous nitrate doesn’t mean dumping it down a drain or tossing it in the trash. That’s not drama—it’s the law, and for good reason. Even trace mercury can linger for decades in water or soil. Local hazardous waste programs know how to dispose of mercury compounds safely. Many universities and labs now use tracked inventory forms. These habit changes mean fewer forgotten bottles and fewer accidents. In my own hometown, the old high school cleaned out a forgotten storeroom, only to discover a long-abandoned bottle — it took days and outside professionals to handle, all because nobody checked inventory for years.
Better Habits, Safer Communities
Safe storage demands more than just rules — it calls for a culture of respect and responsibility. Mark containers clearly. Use dedicated cabinets and check them often. Push for regular safety audits. Share habits and stories, because real-world memories make safety feel less like a checklist and more like second nature. By acting on those lessons, every lab protects not only its workers but its neighbors too.
What are the health hazards associated with Mercurous Nitrate?
Why Mercurous Nitrate Raises Red Flags
Sometimes chemistry brings big benefits, sometimes it comes with heavy trade-offs. Mercurous nitrate, once used in medicine and industry, lands firmly in the “trade-offs” category these days. It’s made from mercury and nitric acid, so right away, red flags go up. My time in labs has taught me to respect warnings, especially when dealing with old-school chemicals. You feel the weight of responsibility when handling anything mercury-based.
What Makes Mercurous Nitrate Dangerous?
Mercury isn’t just another element you sweep up after a spill. This compound contains mercury in its +1 oxidation state, and exposure brings real-world risks. I learned early the dangers weren’t just theoretical. Inhalation or skin contact is enough to cause trouble. The body absorbs this stuff faster than most realize. Mercurous nitrate doesn’t just stay on the skin; it can sneak through and mess with your nervous system.
Researchers have shown that mercury can cause tremors, memory loss, and personality changes. Skin turns red and peels. Mouth ulcers show up. Even old hat makers suffered from mercury poisoning – the “mad hatter” effect. We don’t need more reminders of those grim days.
Short and Long-Term Health Effects
Acute exposure brings headaches, dizziness, and sometimes nausea. Working in places using this chemical, some workers have shared stories of trembling hands or sudden memory problems. Even those who felt healthy sometimes ended up in the ER after weeks on the job. If exposure keeps happening, the risk ratchets up. Mercury accumulates in organs. Long-term exposure leads to kidney damage and trouble with fine motor skills.
I once heard a story from a chemical plant worker who ignored an early rash and chalked it up to allergies. That’s often how mercury gets its chance – people don’t expect a little rash to signal something much worse. By the time serious symptoms appear, the body has already absorbed a dangerous load.
Mercurous Nitrate in Water and Soil
Beyond direct health effects, this compound has a knack for getting where it shouldn’t. If it leaks into water or sinks into the ground, it doesn’t just disappear. Mercury builds up in fish and then in people and animals who eat them. Public health experts agree: there’s no safe level of mercury in drinking water. Mercury pollution in rivers has shut down fisheries and sparked evacuations in some communities.
What Can Be Done?
Getting rid of unnecessary use is a must. Plenty of safer chemicals now accomplish the same industrial tasks. People working in older labs or industries benefit from clear training and strict handling rules – gloves, fume hoods, air monitoring. Any spill or exposure should bring a quick and skilled cleanup crew. Workers ought to see a doctor at the first sign of trouble. Regulators have tightened rules, but enforcement means staying alert, not waiting for bad news.
Disposing of old bottles and waste can’t be left for “later.” Some communities set up hazardous waste events to collect unused chemicals from homes and schools, keeping potential hazards far from children and drinking water. A lot of my fellow scientists now favor green chemistry, steering far away from toxic metals where possible.
Mercurous nitrate came with a heavy downside few realized at first. Careful handling, smarter substitutions, and honest communication protect people and the places they live. Ignoring the health hazards means repeating old mistakes.
| Names | |
| Preferred IUPAC name | dioxido(nitrito)mercury |
| Other names |
Nitric acid mercurous salt Mercurous nitrate monohydrate Mercury(I) nitrate Mercury nitrate Mercury(I) nitrate monohydrate |
| Pronunciation | /ˈmɜːr.kjʊ.rəs ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 10415-75-5 |
| Beilstein Reference | 358415 |
| ChEBI | CHEBI:31603 |
| ChEMBL | CHEMBL1231717 |
| ChemSpider | 79519 |
| DrugBank | DB14663 |
| ECHA InfoCard | ECHA InfoCard: 027-004-00-7 |
| EC Number | 231-819-1 |
| Gmelin Reference | 7788 |
| KEGG | C14191 |
| MeSH | D008588 |
| PubChem CID | 24774 |
| RTECS number | OV4550000 |
| UNII | NM5MW3GR7A |
| UN number | UN1625 |
| CompTox Dashboard (EPA) | DTXSID3040021 |
| Properties | |
| Chemical formula | Hg₂(NO₃)₂ |
| Molar mass | 437.209 g/mol |
| Appearance | White or yellowish-white crystalline solid |
| Odor | Odorless |
| Density | 7.2 g/cm³ |
| Solubility in water | soluble |
| log P | -2.18 |
| Vapor pressure | Negligible |
| Basicity (pKb) | pKb: 6.64 |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.63 |
| Dipole moment | 0 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -152.8 kJ/mol |
| Pharmacology | |
| ATC code | S01AX03 |
| Hazards | |
| Main hazards | Toxic by ingestion, inhalation, and skin absorption; causes irritation to skin, eyes, and respiratory tract; may cause kidney and central nervous system damage; mercury compound—cumulative poison. |
| GHS labelling | GHS02, GHS06, GHS08, GHS09 |
| Pictograms | GHS06,GHS09 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or inhaled. H373: May cause damage to organs through prolonged or repeated exposure. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P210, P220, P221, P260, P273, P280, P284, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P311, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 26 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral-rat 260 mg/kg |
| NIOSH | WW3675000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Mercurous Nitrate is "0.05 mg/m³ (as Hg), TWA". |
| REL (Recommended) | GLP |
| IDLH (Immediate danger) | 10 mg/m3 |
| Related compounds | |
| Related compounds |
Mercury(I) chloride Mercury(II) nitrate Mercury(II) oxide Mercury(I) sulfate |

