Mercurous Acetate: A Deep Dive into History, Science, and Application
Historical Development
Mercurous acetate carries a long story that stretches back to the early days of chemical exploration. Chemists began to isolate and experiment with mercury compounds in the eighteenth and nineteenth centuries, always searching for new uses in medicine and industry. It’s hard to forget how early pharmacies stocked up on various mercurials, and this specific salt found its way into classrooms as chemists studied double decomposition and redox chemistry. Natural curiosity and the search for better lab reagents cemented its place as a useful compound for both research and small-scale manufacturing even before anyone worried much about toxicity or modern standards.
Product Overview
This compound, with the formula Hg2(CH3COO)2, stands as a white, sometimes faintly yellow, powder. It showcases as a crystalline solid, easy to recognize for those who know their way around an inorganic lab. Mercurous acetate found its main role as a laboratory reagent, particularly because it sits comfortably between the realms of organic and inorganic chemistry. While industrial demand never pushed it into mass production like other compounds, its peculiar reactivity gives it a kind of niche utility, particularly in synthesis and analysis.
Physical & Chemical Properties
Mercurous acetate’s physical look tells you a lot about its molecular structure. Its faint yellow-white color and powdery or crystalline texture come from the arrangement of mercury ions paired up in the +1 oxidation state, set off against acetate anions. With a molecular weight just over 402 g/mol, it’s heavier than it looks on the spatula. You can barely dissolve it in water—its solubility stands at about 0.7 grams per 100 mL at room temperature—yet it vanishes pretty well in dilute acids. It starts to break down over 200°C and throws off sharp, acrid fumes when it decomposes. Mercury’s unique chemistry helps this compound act in redox reactions, especially those that involve swapping out acetoxy groups for halides or organic radicals.
Technical Specifications & Labeling
Bottles of mercurous acetate from suppliers carry a UN number (UN 1629), a clean white hazard label with the “toxic” and “environmental danger” diamonds, and a list of handling precautions. Purity usually hovers at or above 98%, with strict limits on elemental contamination. Good suppliers batch test their material, because trace amounts of mercuric contamination would affect both reactivity and regulatory status. Lot numbers and certificates accompany every sale, so quality control chemists and regulatory teams can track impurities, origin, and compliance data. Labels pull no punches, spelling out the need for lab coats, fume hoods, protective eyewear, and gloves with splash protection.
Preparation Method
Lab techs make mercurous acetate by shaking up a solution of mercurous nitrate with glacial acetic acid, where a mild double displacement edges out nitrate ions and pops in acetates. Filtration and careful drying round out the process. If done right, no elemental mercury or mercuric (Hg2+) residues hang around—both would raise serious flags during analysis or application. Some seasoned chemists remember the way the reaction needs a steady hand, because sudden changes in pH or excess oxygen push mercury toward the wrong oxidative state. Once filtered and dried under gentle vacuum, the powder takes on a dense feel, almost greasy between gloved fingers.
Chemical Reactions & Modifications
What grabs attention with mercurous acetate is its tendency to swap out acetoxy groups in reactions, especially in organometallic chemistry. Add a halide source, and you get mercurous halides plus acetic acid. Pair it with some alkyl iodides and expect the acetate to step out while mercury pairs off with halide partners. Sometimes it grabs a radical organic group, helping to forge carbon–mercury bonds in one pot synthetics. It’s also handy for making mercury amalgams, and in a pinch, it helps detect reducing agents by getting reduced itself—sometimes leaving that telltale black or gray mercury film on glass or filter paper.
Synonyms & Product Names
Mercurous acetate circles through scientific catalogs under names like “mercury(I) acetate,” “acetic acid, mercurous salt,” or “diacetoxymercury”. Some older literature refers to it as “ethanoic acid mercurous salt” or simply “white precipitate.” These variations show up more in the old books than in current regulatory sheets, but knowing the aliases comes in handy when cross-checking references or sifting through research from different decades. Each name ties back to its two-fold mercury atom, always in the lower (+1) oxidation state, never quite drifting into the higher (+2) state where toxicity spikes and reactions change course.
Safety & Operational Standards
Operating with mercurous acetate pushes health and safety right to the front of the bench. Mercury compounds walk a thin line between being great reagents and notorious poisons. Chronic exposure can silently build up in the body, hitting kidneys and nervous systems before symptoms flag the problem. Airtight vials, solid PPE, and strict fume hood usage offer the only real protection in a working lab. Spill containment plans must come with every protocol. Good laboratory practice means chelating waste and sending out hazardous waste instead of risking the drain. Regular health screenings offer some reassurance, but the best guarantee comes from reducing exposure in the first place and training new chemists about contamination routes and response actions.
Application Area
Today, you spot mercurous acetate most often in synthetic organic chemistry, where redox transformations or selective halogen substitutions call for careful control. It’s also known as a precipitant in analytical chemistry, especially for separating halides. Older texts and pharmaceutical treatises touch on its role as a mild anti-infective, though modern standards doom any medicinal mercury compound to the dustbin. Still, its unique chemistry keeps it on shortlists for protocols where no substitute gives the same reaction pathway. Teaching labs use it sparingly because of the risk, but advanced courses sometimes bring it out to teach about redox balancing or trace detection.
Research & Development
Mercurous acetate’s research life now looks different than in its heyday, when chemists pushed mercury chemistry in every direction. New focus centers on understanding the unique coordination structures it forms, and how those can inspire less toxic analogs for both organic and inorganic processes. Analytical chemists test its value for halide detection at trace levels, and synthetic teams monitor its ability to mediate carbon–metal attachments. Instrumental analysis and environmental toxicity studies get as much attention as synthetic applications. Ongoing studies search for ways to recycle spent mercurious salts to limit environmental impact, and funding increasingly favors R&D on decontamination protocols.
Toxicity Research
Mercury in every form brings worries in lab research, and mercurous acetate is no different. Studies in lab rodents and in vitro models show clear nephrotoxic effects after repeated exposures, and the chemical can cross cell membranes in certain forms, interfering with protein folding and enzyme function. Environmental scientists track bioaccumulation of mercury species in aquatic systems: even minor spills from past decades raised mercury levels in sediments, building a need for better monitoring and remediation. Current occupational health guidelines draw a low bar for permissible exposure, and regulatory agencies class this compound among top-priority hazardous waste. Chelators like dimercaprol stand by in hospital settings for potential poisoning, though prevention—not post-exposure treatment—gets most ethical and practical support.
Future Prospects
Use of mercurous acetate isn’t likely to surge, at least not in areas outside rigidly controlled labs. Growing restrictions on mercury waste push researchers to invent greener alternatives or redesign synthetic routes around less toxic reagents. Simultaneously, there’s a certain stubborn persistence—no one has yet found a reagent that always gives the same selectivity in organomercury syntheses, so the compound hangs on at the edges of specialty research. Environmental chemists tie much of their work to monitoring and picking apart leftover mercury compounds, trying to crack the code of remediation. Looking forward, serious investment targets both new containment strategies and novel detection methods that pinpoint even minute residues, closing the loop between discovery and long-term safety. Ultimately, the history and ongoing management of compounds like mercurous acetate force researchers and industry leaders to weigh progress against health and environmental responsibility every time a reagent gets pulled from the shelf.
What is Mercurous Acetate used for?
The Basics of Mercurous Acetate
Mercurous acetate stands out due to its ties to mercury. It isn’t something you’ll find stocked at a local pharmacy or hardware store for everyday use. It appears in scientific circles, most often inside research labs or niche industrial processes. Its pale yellow color might look harmless, but it carries a reputation for risk and careful handling. Strict safety rules go along with its use, and regulations shape who gets to work with it.
Chemistry and Reactions
Researchers value mercurous acetate for how it helps drive chemical changes. This compound often turns up in organic synthesis labs, where chemists use it to introduce mercury into molecules. Some types of chemical reactions simply won’t happen without its touch. For example, mercurous acetate can hand off an acetoxy group to alkenes or acetylenes, opening doors for further complex steps in drug and dye research. Chemists also lean on mercurous acetate when studying how certain types of double bonds react with heavy metals, a topic that still brings surprises in research journals.
I remember my college days, struggling through advanced organic chemistry. Those days shed some light on why certain chemicals matter. We watched as mercurous acetate transformed simple molecules in minutes, a process that textbooks taught but seeing it happen brought real understanding. The toxic nature meant most classes used simulations or videos, but that highlighted why specialists stick to well-ventilated and sealed lab benches.
Role in Analytical Chemistry
Besides making new molecules, mercurous acetate has another job — helping identify and separate chemicals from a mix. In analytical chemistry, it acts as a reagent, letting scientists detect or test for certain ions, mostly because of the way it reacts with other substances to create easily observed changes. In older water analysis methods, it helped pinpoint the presence of chloride ions or other halides by forming characteristic precipitates.
Though many labs have shifted to safer and more automated tests, older journals still mention mercurous acetate as a classic tool. Some methods, especially in countries with fewer resources, still rely on it, mainly because it brings reliable accuracy when done with care.
Toxicity and Environmental Concerns
No talk about mercurous acetate can ignore the shadow of mercury. Even small spills or casual exposure can lead to lasting harm. Mercury poisoning doesn’t always show up at once; symptoms can sneak up, leaving behind trouble for nerves, memory, and even basic movement. This makes waste disposal a major concern. Researchers now keep strict logs, ship waste to special facilities, and use only the smallest amounts possible.
At universities, new chemists learn about the past cases of mercury mishandling — damaged health, polluted water systems, and lawsuits that lasted for years. These real stories underline why we need strong rules, real training, and better substitutes wherever possible. Labs today look for greener reagents, but sometimes the chemistry demands what only mercurous acetate can do.
Looking to Future Solutions
There’s a steady push to develop alternatives that leave mercury out of the process. Some advances in catalysis and organic synthesis now promise similar results without the need for risky compounds. it takes time for these options to become standard. Until new methods take over, education, careful storage, sealed systems, and well-trained hands can make all the difference. The history of mercurous acetate serves as a reminder: progress in chemistry hinges as much on safety as it does on results.
What is the chemical formula and structure of Mercurous Acetate?
Understanding Mercurous Acetate
Mercurous acetate draws interest from students, chemists, and hobbyists who like exploring lesser-known corners of inorganic chemistry. Known by its chemical formula Hg2(CH3COO)2, this compound stands out because of the unique way mercury atoms stick together. Instead of a single mercury atom linked to two acetate groups, mercurous acetate connects two mercury ions, forming a dimer. This makes both the chemistry and safe handling different compared to more familiar compounds like sodium acetate.
Chemical Formula and Structure
The composition of mercurous acetate traces directly to how mercury prefers to bond. You see Hg22+ showing up as a pair of mercury atoms, not just one. Each dimer pairs with two acetate anions (CH3COO-). Put together, you get Hg2(CH3COO)2.
The structure tells its own story. Picture two mercury atoms side by side in a single unit, each donating a positive charge, and then two acetate groups swing in to balance the whole thing. This bridging sets mercurous (Hg+) compounds apart from the more common mercuric (Hg2+) versions. Chemically, the bond between those two mercury atoms matters — it's not just a formality on paper. Researchers have confirmed this connection by analyzing crystals and looking at the distance between atoms using X-ray diffraction. That close bond affects how the compound reacts, degrades, and interacts with other chemicals.
Why Care About Mercurous Acetate?
I remember studying mercury chemistry in college. Most people hear “mercury” and think of toxic liquid in thermometers or dangerous waste, but mercury’s dimeric forms like Hg22+ show off a different side of this metal. Mercurous acetate acts as a mild oxidizer, and chemists use it in specific organic reactions, such as demethylation or in the synthesis of certain coordination complexes. These reactions depend on the unique properties of that mercury-mercury bond you just don’t find in simpler salts.
Safety raises real concerns. Organic chemists must pay close attention to storage, ventilation, and disposal. The Environmental Protection Agency and OSHA both list mercury compounds as hazardous, and for good reason. Long-term exposure to even trace amounts could cause neurological and organ damage. In research labs, handling mercurous acetate always involves gloves, eye protection, and fume hoods. Strict rules make sense here; I have seen friends develop sensitivities just from working with heavy metals day in and day out.
Potential Solutions and Responsible Use
Institutions can reduce risk with rigorous safety training and regular monitoring of air and surfaces. Substitution with less hazardous reagents whenever possible protects both researchers and the environment. Digital tracking of mercury inventories, careful labeling, and required disposal protocols help prevent contamination and accidental exposure. Beyond labs, recycling and waste management companies need to keep mercury out of groundwater and the food chain.
Mercurous acetate demonstrates how one unusual chemical structure can drive scientific discovery while also demanding a healthy dose of caution. Those differences in chemical bonding end up affecting everything — from lab work to healthcare policy. For anyone handling this compound or teaching about it, practical knowledge, awareness, and thoughtful stewardship always matter far more than memorizing a formula.
Is Mercurous Acetate hazardous or toxic to handle?
Understanding the Risks Linked to Mercurous Acetate
Mercurous acetate has never earned much trust in science classrooms or research labs. This chemical contains mercury, and if you’ve ever read about mercury, you know it sparks a lot of worry for good reason. Exposure to even small amounts can damage health over time. The mercury in mercurous acetate doesn’t just vanish after touching it. Instead, it sneaks into the body through the skin, lungs, or mouth. Everyone has seen those shocking stories about mercury poisoning, often linked to broken thermometers or tainted seafood. Lab experience tells a bigger story—solid forms of mercury chemicals like mercurous acetate can be just as sneaky.
The hazardous reputation comes from its toxicity. Handling mercurous acetate without gloves or a fume hood adds unnecessary danger. Skin contact may not seem like much, but mercury builds up with repeated exposure. Lab safety sheets warn that inhaling its dust or vapors can leave someone facing headaches, dizziness, nausea, and trouble concentrating. Touching eyes or mouth after exposure raises the risk. A few years ago, a colleague forgot gloves while measuring out a small amount. The clean-up that followed took much longer than the actual experiment and raised anxiety about invisible contamination.
Why Workers and Hobbyists Must Respect This Chemical
People who study chemistry remember advice from mentors: treat every mercury compound with respect. Over time, mercury eats away at nerve cells and can damage kidneys. Medical textbooks hold case after case linking occupational exposure to long-term health problems. Chronic exposure sometimes leads to mood changes, memory loss, and shaking hands. If mercurous acetate lands on skin, it can get absorbed. Swallowing even a tiny bit by accident may cause vomiting or abdominal cramps, an experience no one forgets in a hurry.
This risk doesn’t stop at the lab. Improper disposal grows into a larger community problem. Mercurous acetate washes into waterways and lingers in soil. Once there, it breaks down and releases toxic mercury ions. Fish and shellfish soak it up, entering the food chain. It’s not just about the safety of the person holding the chemical—the consequences spread far beyond the workbench.
Safer Handling and Smarter Disposal
Smart handling means always working with mercurous acetate in a fume hood. Gloves, goggles, and a lab coat do more than make someone look official. They add layers of protection. Accidents happen, so spill kits need to be close by. Cleanup shouldn’t wait until the end of the day—immediate attention stops trouble in its tracks. In my experience, labs that get lazy with safety rules often wind up with stress and paperwork when things go wrong.
Disposal deserves special attention. Pouring this chemical down the sink counts as dangerous and irresponsible. Instead, place all waste and used gloves into sealed, labeled containers. Certified hazardous waste handlers know how to process these materials without putting people at risk. Careful labeling and record keeping helps track waste and avoid mix-ups. Years ago, a neighboring lab lost weeks of work due to contaminated equipment that traced back to poor mercury disposal.
The Bigger Picture
Everything points to treating mercurous acetate as a genuine health hazard. The science backs up the horror stories. Gloves, hoods, and safe habits aren’t just formalities. They turn what could be a troublesome substance into something manageable. Sticking to smart safety practices and responsible disposal means this chemical loses some of its bite, and no one needs to become a cautionary tale.
How should Mercurous Acetate be stored safely?
Why Proper Storage Makes a Difference
Most people rarely come across mercurous acetate unless they work in a chemistry lab or a specialty research setting. It’s a chemical that doesn’t get much press, but safe storage can decide whether it sits quietly on a shelf or becomes a hazard. Mercurous acetate contains mercury, and anybody who’s tackled spills or accidents with mercury knows how those can play out. From my years in academic labs, careless handling of even small mercury compounds turns into a headache fast. There’s a real risk to health and to the environment—you can’t just wipe up some spilt powder with a paper towel and call it a day.
Choosing the Right Container
Leave mercurous acetate in its original container as long as possible. Manufacturers go the extra mile to provide bottles that help keep moisture away and reduce the chance of leaks. Glass with tight-sealing lids works best. Don’t use metal or containers that react to acids, since corrosion or leaks may follow. Label everything clearly. One look at a big shelf of unmarked jars in the back of an old storeroom should remind anyone: a missing label is just trouble waiting to happen. For those short on time, writing the date received on the label gives an extra sense of accountability without much fuss.
Temperature and Humidity
Long experience teaches that chemicals stay safest at room temperature, away from extremes. Heat or direct sunlight can break down compounds and speed up reactions. A cool, dark cupboard that doesn’t get humid works best. If the storage area gets damp, clumping and chemical changes may follow. A decent humidity gauge on the wall and silica gel packets nearby will help. Rushed storage, like shoving bottles under a sink or leaving them on a sunny windowsill, makes everything riskier—and in labs I’ve worked at, staff training frequently focuses on using only approved chemical storage areas to stop such habits from forming.
Avoiding Incompatible Neighbors
Shelving choices matter. Don’t stack mercurous acetate with acids, bases, or organic solvents. Each of these can trigger dangerous reactions, and I’ve seen puddling and corrosion happen fast where shelves mix up incompatible chemicals. Grouping chemicals by hazard class instead of alphabetizing them looks untidy, but it prevents nasty surprises. Use spill trays under the jars so any leak gets trapped. For shared spaces, a clear sheet taped to the inside of the cupboard door lists the chemicals stored there. Everybody wins: newcomers, old hands, even the next safety inspector who walks through the door.
Think Beyond Four Walls
Secure shelves matter more than most realize. In buildings prone to vibration or mild earthquakes, a shelf needs to be bolted so jars can’t wiggle loose. Loose glass marked “toxic” jostling onto the floor causes instant problems and calls to the hazmat team. Regulations can seem like a pain, but after handling chemical accidents and filling out stacks of forms, I can say that following even one or two extra precautions saves time, money, and worry.
Know When to Call for Backup
If a jar starts looking cloudy, if something leaks or smells off, or if a bottle gets chipped, don’t try to fix it solo. The right personal protective gear should come out first—gloves, goggles, lab coat. Many labs set up simple protocols, so nobody has to guess who deals with questionable containers. In emergencies, specialists with the training and gear get called in because they know how to handle cleanup without harm to themselves or others. Quick action beats hesitation every time.
What are the physical properties of Mercurous Acetate?
Understanding the Basics
Mercurous acetate doesn't show up on the average household supplies list. For those who’ve handled it in a lab setting, it stands out as a silvery-white solid. Its chemical formula, Hg2(C2H3O2)2, tells you this compound features mercury in the rare +1 oxidation state. Mercury's behavior here changes how the material acts physically.
The Look and Feel
Walk into a chemical storeroom and spot a bottle of mercurous acetate — it's likely a soft, white powder or sometimes shows as colorless crystals. It takes on a faint, vinegar-like odor if you get your nose close enough. Touching it isn't something I suggest; lab instructors always stressed gloves, since mercury is nothing to mess with. Its density comes in at about 5.22 g/cm³ — pretty heavy for a powder, thanks to the mercury packed into its structure.
Handling and Stability
This solid holds up in a dry bottle, but humidity triggers slow decomposition. If you ever split a bit of powder onto wet paper, you’ll see it darken — a sign that the compound is breaking down, releasing acetic acid and elemental mercury. It dissolves in water, but not entirely; you’ll see some crystals stubbornly sitting at the bottom unless you stir and keep it at the right temperature. At about 170 °C, the solid begins to break apart, turning into mercuric oxide, mercury vapor, and acetic acid gas. Anyone working around it learns to stick with low heat and keep the lid tight.
Chemical Reactivity
Mercurous acetate reacts with halide ions very quickly, forming different mercury compounds. This makes it useful for lab tests, where chemists look for traces of other chemicals. It doesn’t mix well with strong acids or bases, and exposure to bright light can speed up its breakdown. Over time, especially in sunlight, you’ll notice it turning gray as the mercury slowly separates out.
Safety Notes from the Bench
Aside from its basic appearance, handling mercurous acetate always brings up safety. Every chemistry student learns early on that mercury compounds are toxic. A whiff of acetic acid escaping from an open bottle tells you to check that the container stays sealed. If a spill happens, everyone clears out, and cleanup gets serious. Labs usually have mercury spill kits on hand just for compounds like this. Chronic exposure, even at low levels, can lead to mercury poisoning — tremors, memory loss, and organ failure. So, while the powder might look harmless, trust that it requires care, awareness, and good ventilation.
Facing Environmental Responsibility
The legacy of mercury use reaches far beyond the laboratory. Disposing of mercurous acetate demands proper procedures. Sending any mercury-based waste down the drain turns into an environmental nightmare. Waste collection companies collect mercury compounds in sealed containers — every bottle labeled and tracked. Many countries have strict rules about reducing mercury release, and schools try to switch away from mercury-based reagents whenever possible. Finding ways to phase out dangerous chemicals, or choosing less toxic alternatives for tests and research, cuts down on risks for students, workers, and the environment alike.
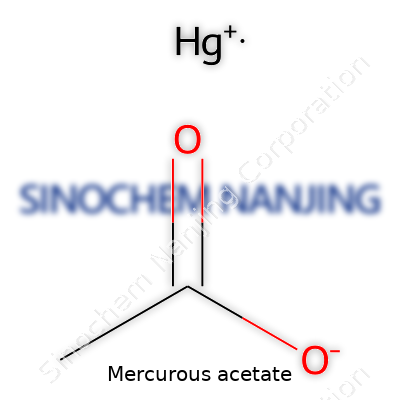
| Names | |
| Preferred IUPAC name | diacetatodimercury |
| Other names |
Acetic acid, mercurous salt Mercury(I) acetate |
| Pronunciation | /ˈmɜːr.kjʊ.rəs əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | *“1600-27-7”* |
| Beilstein Reference | 385968 |
| ChEBI | CHEBI:32222 |
| ChEMBL | CHEMBL1380041 |
| ChemSpider | 50376 |
| DrugBank | DB14642 |
| ECHA InfoCard | 100.029.140 |
| EC Number | 200-912-2 |
| Gmelin Reference | 2940 |
| KEGG | C18628 |
| MeSH | D008625 |
| PubChem CID | 66247 |
| RTECS number | OV8925000 |
| UNII | 6U8T65MGC5 |
| UN number | UN1649 |
| Properties | |
| Chemical formula | Hg2(C2H3O2)2 |
| Molar mass | Molar mass of Mercurous Acetate: "398.78 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 3.46 g/cm³ |
| Solubility in water | Slightly soluble |
| log P | -2.64 |
| Vapor pressure | 0.001 mmHg (20°C) |
| Magnetic susceptibility (χ) | `-46.0e-6 cm^3/mol` |
| Refractive index (nD) | 1.744 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 186.0 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -382.5 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AB32 |
| Hazards | |
| Main hazards | May be harmful if swallowed, inhaled, or absorbed through skin; causes irritation to skin, eyes, and respiratory tract; may cause mercury poisoning. |
| GHS labelling | GHS05, GHS07, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Warning |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. |
| Precautionary statements | P234, P260, P264, P270, P271, P273, P301+P310, P302+P352, P304+P340, P308+P311, P314, P330, P391, P405, P501 |
| Lethal dose or concentration | LD50 (oral, rat): 220 mg/kg |
| LD50 (median dose) | LD50 (median dose): 210 mg/kg (oral, mouse) |
| NIOSH | MX5250000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 5 mg/m³ |
| IDLH (Immediate danger) | 5 mg/m3 |
| Related compounds | |
| Related compounds |
Mercury(II) acetate Mercury(I) chloride Mercury(II) nitrate Silver acetate |

